Introduction
As economic status has improved, growing interest in health and well-being has resulted in increased intake of natural and health foods.1 So, food industries are facing challenges for developing value added health promoting foods as well as phytochemical rich new raw material sources. Health professionals are motivating the consumers to avail beneficial effects of probiotic food for good health. The probiotic foods currently produced are dairy products and has limitations due to milk allergies and cholesterol content.2 So non- dairy alternatives may be used to substitute dairy probiotics and can provide other health benefits also. Probiotic food formulations are leading to development of dietary supplementation with nutraceuticals and prebiotics.3 Fruits and vegetables are highly valued food for their high carbohydrate, minerals, vitamins, dietary fibers, and antioxidants which will help in buildup, repair and maintain alkaline reserve of body4 and also do not possess any dairy allergens.2 India is leading producer of fruits and vegetables but unfortunately a major proportion is spoiled due to improper processing and poor postharvest management.5 Fruits and vegetable are inherently healthy, refreshing, pleasing taste profiles and bundle of beneficial nutrients, so these can be used as suitable substrate for nondairy probiotics. Fermentation can enhance nutritional, digestibility, shelf life, safety and sensory attributes of vegetables and also nutrients are preserved which are otherwise destroyed by thermal food processing.
Beetroot contains numerous nutrients including sodium, magnesium, potassium, vitamin C, betanin and antioxidants.6,7 The beet root contained phenolic compound, carotenoids, betalain, vitamins and minerals which are major important bio compound and micronutrients and it is the 10th most powerful vegetables with antioxidant properties.8 Betalins are present mainly in two forms i.e. betacyanin (red-violet Colour) and betaxanthin (yellow-orange colour). Betalain is water-soluble and nitrogen containing natural pigments which have a high coloring capacity9 along with antiviral, antioxidant, anti-inflammatory properties10 anti-cancerous11 with no side effect.12 Both sub classes of betalins: betacyanin (red-violet Colour) and betaxanthin (yellow-orange colour), are present in red beet root in high concentrations. An effort was made to utilize all these health potential of beetroot in the form of a drink. The present study was planned to prepare a beetroot based probiotic drink.
Materials and Methods
Sample procurement and preparation
The fresh raw material beetroot was sorted from local market, cleaned and stored at 40 C for further use. Juice was extracted by juicer in a food processor. The extracted filtered juice was pasteurized at 800 C for 10 min. Pasteurized juice was further cooled to room temperature for microbial inoculation.
Inoculum Preparation
Probiotic microorganisms Lactobacillus rhamnnosus, Lactobacillus plantarum and Lactobacillus delbrueckii sb were obtained from IMTECH, Chandhigarh. For inoculum, glycerol stock culture tube of Lactobacillus plantarum, Lactobacillus rhamnnosus and Lactobacillus delbrueckii sb. was transferred in 250 ml Erlenmeyer flask having 100 ml MRS broth. Broth was incubated for cell growth at 370C. Growth was observed using spectrophotometer (590 nm). Incubation was carried out till the cell density reached 0.600 value corresponding to 9.00 Log CFU/ml, scale designed by McFarland.13
Inoculation and Fermentation
The optimum fermentation conditions were optimised using central composite rotated experimental design (CCRD) at pH range 4-7 at temperature of 37 ºC. These conditions were selected because Lactobacillus can grow at this pH and temperature condition. The pH of clarified beetroot juice was adjusted with HCL (0.1 N). Then, pre-determined concentration of inoculum was added to beetroot juice as recommended for probiotic foods14 i.e., 7.00 log CFU/ml (1 ml of MRS broth containing 9.00 CFU/ml of Lactobacillus plantarum, Lactobacillus rhamnnosus and Lactobacillus delbrueckii sb.). Fermentation was performed statically in an incubator for 24 hr adjusted at 370C temperature.
Sample Analysis
The fresh and probiotic beetroot juice analyzed for protein, acidity content, pH, TSS and betalain content by using different methods. The bile and acid tolerance were analyzed before addition of lactic acid bacteria.15
Growth of probiotic strains
Utilizing the optimized factor value (i.e., temp 37°C and pH 6.5), culture were developed in pasteurized beetroot juice (700 mL in 1 L plastic flask) adding inoculum of lactic acid bacteria 2.5%, 5%, 7% (V/V) and incubating at temperature 37°C for 24 hrs. Tests for measuring the growth and viable cells counts16 were taken each two hour. Microbial growth was assessed on the basis of pH, acidity content and sugar utilization. pH was estimated by pH meter and acidity was measured by titration method. Sugar content was observed by DNS (dinitrosalicylic acid) method.
Sensory Evaluation
Sensory analysis was carried out by using semi-trained panelists using nine point hedonic scale from liked extremely (9) to disliked extremely (1) as described by Larmond.17 Sample was evaluated on the basis of appearance, aroma, consistency, taste, mouth feel and overall acceptability.
Statistical Analysis
The result for physico-chemical, microbiological and sensory attributes of papaya based whey RTS were analysed with the help of Graph Pad Prism (La Jolla, CA, USA) (version 5.01) software. 2 way ANOVA was conducted for statistical significance for mean differences. The significance level was set at 5% (P<0.05) for all calculations.18
Results and Discussion
The three species of lactic acid bacteria (Lactobacillus plantarum, Lactobacillus rhamnnosus and Lactobacillus delbrueckii sb.) were found to be capable of growing well on pasteurized beetroot juice without any specific nutrient requirement. Viability of three strains on different pH level was observed (Table 1) and it was concluded that at pH 6.5, microbial count was more than 300 for all three strains. So, pH 6.5 was selected for further product development at 37 ºC.
Table 1: Viability of lactobacillus strain at variable pH level after 24 hrs
|
pH |
L. rhamnnosus | L. plantarum | L. delbruecki |
| 4 | 100 | 80 |
90 |
|
5 |
200 |
150 |
190 |
|
6 |
250 |
200 |
240 |
|
6.5 |
>300 |
>300 |
>300 |
|
7 |
<250 |
<200 |
<230 |
Fermentation of pasteurized beetroot juice was carried out by inoculating with culture of mixed starins (Lactobacillus plantarum, Lactobacillus rhamnnosus and Lactobacillus delbrueckii sb.). To evaluate growth kinetics, change in pH due to acid production and sugar utilization was observed. The probiotic culture suitability was observed on the basis of their acid and bile tolerance and study revealed that microorganisms were resistant to such conditions (Fig. 1).
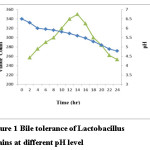 |
Figure 1: Bile tolerance of Lactobacillus strains at different pH level Click here to View figure |
The initial pH of inoculated juice was 6.5±0.03 and it gradually decreased to 4.75±0.03 in 24 hr (Fig 1). During fermentation, lactobacillus produces lactic acid which accounted for the decline in pH level. The initial acidity of the beetroot juice was 0.49 ±0.02 and was inclined to 1.02±0.01. Similar observations for pH decline and acidity incline were reported by other researchers Devi et.al.,19 Shukla et.al.,20 and Rafiq et al.,21
Change in sugar concentration with time
Lactobacillus requires sugar for growth and as the fermentation continues, sugar content of sample was declining gradually. Initially sugar (%) in juice was 9.97±0.09 which was decreased with time. Fig 2 clearly depicted that with microbial fermentation, sugar level decreased with time.
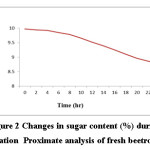 |
Figure 2: Changes in sugar content (%) duringfermentation Proximate analysis of fresh beetroot juice Click here to View figure |
Fresh beetroot juice and probiotic drink were evaluated for proximate composition (Table 2) and antioxidant activity. Slight increment in protein value was observed from 3.74±0.008 to 3.77±0.009, this might be due to presence of probiotic microorganisms and their metabolites. Acidity of sample was increased from 0.49±0.02 to 0.78±0.01 (Table 2). There was increase in antioxidant activity, total phenols and flavonoid content was observed. The increase in total antioxidants occurred due to fermentation leading to increase in phenolic and flavonoids compounds due to microbial hydrolysis reaction. Fermentation also results in structural disintegration of cell walls leading to either liberation or synthesis of different antioxidant compounds. As antioxidants possess free radical scavenging capacity and health promoting benefits, so the probiotic drink is highly valuable. Similar results have been reported by Kazimierczak et al., 2016.22
Table 2: Nutritional composition of fresh and probiotic drink
| Sample | Acidity (%) | pH | Protein (%) | Sugar (%) | Antioxidant acitivity (%) | Total Phenol(%) | Flavonoid content (%) |
| Fresh juice | 0.49±0.02b | 6.5±0.03a | 3.74±0.08b | 9.97±0.09a | 75.8±0.05b | 22.12±0.02b | 3.61±0.01b |
| Probiotic drink | 0.78±0.01a | 4.75±0.03b | 3.77±0.09 a | 8.66 ±0.09 b | 78.1±0.05 a | 24.32±0.04 a | 3.85±0.02 a |
*Mean ± SD (n=3)
a* Different upper case superscripts in the same column indicate the significant difference (p<0.05)
Sensory Analysis
The probiotic drink was less accepted (7±0.27) as compared to the fresh one (7.5±0.42), but the benefit of probiotic cultures in beetroot juice adds value in terms of health aspects. The highest mean appearance, aroma, consistency, taste, mouth feel was observed in fresh beetroot juice than the probiotic one (Table 3). Results depicted that microbial fermentation may be used as suitable technology in formulation of health promoting foods.
Table 3: Sensory analysis of fresh and probiotic juice
| Sample | Appearance | Aroma | Consistency | Taste | Mouth feel | Overall acceptability |
| Fresh juice | 7±0.54a | 8±0.28a | 8±0.36a | 7±0.42b | 8±0.39a | 7.5±0.42a |
| Probiotic drink | 7±0.51a | 7±0.32b | 7±0.42b | 7.5±0.28a | 7±0.19b | 7±0.27b |
*Mean ± SD (n=3)
a* Different upper case superscripts in the same column indicate the significant difference (p<0.05)
Conclusion and future scope
Mixed culture of Lactobacillus plantarum, Lactobacillus rhamnnosus and Lactobacillus delbrueckii sb. carried out fermentation and survived in beetroot juice at optimum conditions. Strains possessed good viability in beetroot juice without any specific nutrient supplementation. There was non-significant increase in proximate composition of fresh beetroot juice with formulation of probiotic drink. Probiotic drink was found good in antioxidants, total phenols and flavonoids content. The study depicted that beetroot probiotic drink may act as good alternative to serve lactose intolerant and the persons unable to consume probiotic dairy products due to allergic reactions.23 Though lot of further research may be conducted regarding sensory improvement, storage stability and commercialization of probiotic drink.
Acknowledgements
This research was supported by Lovely Professional University, Punjab. We thank our colleagues from Lovely Professional University and Chaudhary Devi Lal University, Haryana who provided insight and expertise that greatly assisted the research.
References
- Panghal A, Sharma K, Setia S, Ray A. Studies on utilization of whey for preparation of ready to serve (RTS) beverages. Beverage and Food World. 2007;34(8):77-79.
- Tissier H. The treatment of intestinal infections by the method of transformation of bacterial intestinal flora. CR Social Biology. 1906;60:359-61.
- Azadnia PK, Khan Nazer AH. Identification of lactic acid bacteria isolated from traditional drinking yoghurt in tribes of Fars province. Iranian Journal of Veterinary Research. 2009;10(3):235-40.
- Yadav DN, Sharma M, Chikara N, Anand T, Bansal S. Quality characteristics of vegetable-blended wheat–pearl millet composite pasta. Agricultural Research. 2014;3(3):263-70.
CrossRef - Panghal A, Dhull N, Navnidhi, Khatkar B.S. Whey Based Strawberry Ready to Serve (RTS) Beverage. Beverage and Food World. 2009;36(4):28-30.
- Georgiev VG, Weber J, Kneschke EM, Denev PN, Bley T, Pavlov AI. Antioxidant activity and phenolic content of betalain extracts from intact plants and hairy root cultures of the red beetroot Beta vulgaris cv. Detroit dark red. Plant Foods for Human Nutrition. 2010;65(2):105-11.
CrossRef - Wootton-Beard PC, Moran A, Ryan L. Stability of the total antioxidant capacity and total polyphenol content of 23 commercially available vegetable juices before and after in vitro digestion measured by FRAP, DPPH, ABTS and Folin–Ciocalteu methods. Food Research International. 2011;44(1):217-24.
CrossRef - Kushwaha R, Kumar V, Vyas G, Kaur J. Optimization of Different Variable for Eco-friendly Extraction of Betalains and Phytochemicals from Beetroot Pomace. Waste and Biomass Valorization. 2017;1-10. https://doi.org/10.1007/s12649-017-9953-6
CrossRef - Attia GY, Moussa ME, Sheashea ER. Characterization of red pigments extracted from red beet (Beta vulgaris, l.) and its potential uses as antioxidant and natural food colorants. Egypt. Journal of Agriculture Research. 91(3):1095-110.
- Aro A, Amaral E, Kesteloot H, Rimestad A, Thamm M, Van Poppel G. Transfatty acids in French fries, soups, and snacks from 14 European countries: the TRANSFAIR study. Journal of Food Composition and Analysis. 1998;11(2):170-7.
- Collins R, Peto R, MacMahon S, Godwin J, Qizilbash N, Hebert P, Eberlein KA, Taylor JO, Hennekens CH, Fiebach NH. Blood pressure, stroke, and coronary heart disease: part 2, short-term reductions in blood pressure: overview of randomised drug trials in their epidemiological context. The Lancet. 335(8693):827-38.
CrossRef - Jiang W, Zhang D, Bursac N, Zhang Y. WNT3 is a biomarker capable of predicting the definitive endoderm differentiation potential of hESCs. Stem cell reports. 2013;1(1):46-52.
CrossRef - Kangler O, Weiss N. Genus Lactobacillus beijerinck 1901, 212AL.In Sneath PHA, Mair NS, Sharpe ME, Holt JG (eds.) Bergey’s Manual of Systematic Bacteriology. Baltimore, Williams and Wilkin Publishers. 1986.
- Tsai CC, Lin PP, Hsieh YM. Three Lactobacillus strains from healthy infant stool inhibit enterotoxigenic Escherichia coli grown in vitro. Anaerobe. 2008;14(2):61-67.
CrossRef - Succi M, Tremonte P, Reale A, Sorrentino E, Grazia L, Pacifico S, Coppola R. Bile salt and acid tolerance of Lactobacillus rhamnosus strains isolated from Parmigiano Reggiano cheese. FEMS Microbiology Letters. 244(1):129-37.
CrossRef - Pereira AL, Maciel TC, Rodrigues S. Probiotic beverage from cashew apple juice fermented with Lactobacillus casei. Food Research International. 44(5):1276-83.
CrossRef - Elizabeth Larmond. Laboratory methods for sensory evaluation of food. Agriculture Canada. 1977.
- Saliganti, Vamshi, Kapila R, Kapila S.Consumption of probiotic Lactobacillus rhamnosus (MTCC: 5897) fermented milk plays a key role on newborn mice immune system development during suckling-weaning transition: Probiotics for early immune maturation. Microbiology and Immunology. 2015;114(8):1168-79.
- Devi MS, Banumathi P, Jothi N. Studies on development and evaluation of whey based fruit beverages. Beverage Food World. 2004;1:44-6.
- Shukla M, Jha YK, Admassu S. Development of probiotic beverage from whey and pineapple juice. Journal of Food Processing Technology. 2013;4(2):1-4.
- Rafiq S, Sharma V, Nazir A, Rashid R, Sofi SA. Development of Probiotic Carrot Juice. Journal of Nutrition Food Science. 2016;6(534):2.
CrossRef - Kazimierczak R, Hallmann E, Lipowski J, Drela N, Kowalik A, Püssa T, Matt D, Luik A, Gozdowski D, Rembiałkowska E. Beetroot (Beta vulgaris L.) and naturally fermented beetroot juices from organic and conventional production: metabolomics, antioxidant levels and anticancer activity. Journal of the Science of Food and Agriculture. 2014;94(13):2618-29.
CrossRef - Panghal A, Kumar V, Dhull S. B, Gat Y, Chhikara N. Utilization of Dairy Industry Waste-Whey in Formulation of Papaya RTS Beverage. Current Research in Nutrition and Food Science; 2017;5(2):168-174. doi : http://dx.doi.org/10.12944/CRNFSJ.5.2.14
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.






