Introduction
Whole grain products are considered a key component of a healthy diet. Recommendations from the Nutritional Guideline for Filipinos advocate settling for healthy food choices.1 In addition, the U.S. Department of Agriculture prescribes at least three servings of whole grain foods should be consumed daily in replacement of refined grain foods.2 According to different epidemiological studies, whole grain products provide fiber and phytochemicals that can reduce chronic diseases such as lowering of blood pressure,3 type 2 diabetes, cardiovascular disease, weight gain,4 and risks of several types of cancer.5
Wheat bread is a whole grain product that is ubiquitously consumed. Sourdough, made from flour, water, and leftover dough fermented from a previous batch, undergoes a long fermentation period to make the final bread product appealing and palatable.6 Sourdough fermentation has many advantages and beneficial purposes. First are the mechanisms wherein sourdough fermentation influences the nutritional quality of bread through changed levels and/or bioaccessibility of phenolics, sterols, and vitamins; enhanced mineral bioavailability of Ca, Fe, K, Mg, P, and Zn; solubilization of dietary fiber; gluten degradation and reduction of starch digestibility.7 Second is to extend the shelf-life of bread by harnessing the natural antagonistic properties of lactic acid bacteria, present in sourdough, against pathogenic microorganisms.8,9 Last is the improvement of the organoleptic properties of bread, which is based on the appearance, texture, and most importantly odor and taste – imparting flavor to the bread. The great demand for wheat bread with exceptional quality, shelf-life, and aroma, in addition to the aforementioned sourdough benefits, justifies the evaluation of the aroma volatile compounds and their evolution during sourdough fermentation since these compounds are precursors for the aroma compounds formed during baking in the final bread product.
Several studies have been done on the determination of aroma volatile compounds present in wheat bread crumb and crust through dynamic headspace extraction and analysis through GC/MS,10 sourdough bread containing kefir grains11 and chestnut flour-based sourdough12 through headspace solid phase microextraction (HS-SPME) and GC/MS analysis. However, information on what is happening during sourdough fermentation, especially the evolution of aroma volatile compounds at the early stages of sourdough preparation, is limited and not given full attention in other studies. The determination and evolution of these important precursor compounds, formed during sourdough fermentation, is important because they influence the aroma compounds of the final bread product.
The objectives of this study were to determine the major aroma volatile compounds present in sourdough made from whole wheat and all-purpose sourdough and monitor the evolution of such compounds through static headspace gas chromatography coupled to mass spectrometry (SHS-GC/MS). Comparison of the different aroma volatile compounds was also discussed.
Materials and Methods
Sourdough preparation
Two types of flour, 100% stone ground whole wheat (Bob’s Red Mill) and white all-purpose (Bob’s Red Mill), purchased from a local natural product food store, were used in the preparation of sourdoughs. Whole wheat flour contained 0.5g total fat, 5g fiber, 6g protein, 2% calcium, and 8% iron. White all-purpose flour contained 0.5g total fat, 1g fiber, 4g protein, 20% folate, and 10% iron. Nutritional contents were based on 38g and 34g per serving of whole wheat and all-purpose flour, respectively.
Sourdoughs were prepared in a 1:1 ratio of flour to water (mass to volume) and fermented spontaneously at room temperature. Whole wheat flour or all-purpose flour, weighing 20g, and 20mL distilled water was thoroughly mixed in a small propylene cup until lumps of flour were no longer present. Coffee filter was used as the lid. After 24 hours of fermentation, half of the sourdough was discarded and another 20g flour and 20mL distilled water were added. The process is known as “feeding”, which is done to make the sourdough manageable.6 Feeding was repeated, every day, for 28 days.
Static headspace extraction
The gas phase above the liquid extracts when heated in a sealed vial is called the headspace.32 The volatile fractions of whole wheat and all-purpose sourdough were extracted through static headspace sampling. For each static headspace (SHS) analysis, approximately 6mL of sourdough taken at 24, 168, 336, 504, and 672 hours of fermentation were placed in a 20mL headspace vial sealed with a rubber septum. The vial was placed in a hot water bath at 60±3 °C, a 50µL gas tight syringe (Vici Series A2) was inserted into the vial, and equilibrated for 30 minutes to allow the volatile compounds to establish equilibrium between the gas and liquid phases.32 After equilibration, the syringe was removed from the vial and injected into the GC/MS injector port for analysis. A previous study was also carried out using SHS on the determination of volatile compounds during production of fruit vinegars.33 It was shown that SHS method provides a simple, fast, sensitive, reproducible data, and had a good degree of accuracy. Thus, SHS was the method chosen for this study.
GC/MS analysis
GC/MS analysis was carried out through an Agilent Technologies 7890A gas chromatograph coupled to an Agilent Technologies 5977A MSD mass spectrometer equipped with a capillary 30m x 250µm x 0.25µm capillary column (HP-5ms Ultra Inert). The gas carrier was helium with a flow rate of 1mL/min and manual injections were done in splitless mode. MSD transfer line and inlet temperatures were kept at 280 and 250 °C, respectively. Initial oven temperature program was 40 °C, followed by an increase to 50 °C at a rate of 5°C/min., and a final increase to 230 °C at a rate of 5.5 °C/min. All temperature increases, including initial temperature, had a hold time of 5 minutes. Identification of volatile compounds was achieved by comparing their mass spectra against the equipment’s NIST library.
Results and Discussion
Sourdoughs contains bacteria and wild yeasts that convert sugars present in the flour through fermentation resulting to a variety of end products that give distinct flavors to bread.6 Lactic acid bacteria (LAB) and yeasts, capable of producing carbon dioxide, are responsible for the leavening of bread dough.13 In general, aldehydes and alcohols are formed inside the yeast cell from the degradation of flour amino acids.14 while LAB is responsible for the acidification of sourdough.13
Bread aroma is mainly produced upon baking. However, the importance of fermentation should not be neglected since fermentation is a source of several precursor aroma volatile compounds.13 Whole wheat and all-purpose sourdough samples were fermented for a total of 672 hours at room temperature and analyzed through SHS-GC/MS to determine and evaluate the evolution of aroma volatile compounds.
A total of 62 volatile compounds were found in the headspace of whole wheat sourdough during the whole period of fermentation. Table 1 lists all the aroma volatile compounds according to their chemical classes. Hydrocarbons (15) were the most abundant followed by esters (7), alcohols (7), ketones (7), aldehydes (6), heterocycles (5), sulfur compounds (3), an acid, and 11 miscellaneous compounds.
 |
Table 1: Aroma volatile compounds identified in the headspace of whole wheat sourdough through SHS-GC/MS Click here to View table |
All-purpose sourdough had a different aroma volatile compound composition compared to whole wheat sourdough. The majority were aldehydes (13), ketones (6), hydrocarbons (6), alcohols (4), acids (3), esters (3), a sulfur compound as well as 9 miscellaneous compounds. Table 2 shows the total aroma volatile compounds (45) found in the headspace of all-purpose sourdough.
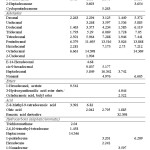 |
Table 2: Aroma volatile compounds identified in the headspace of all-purpose sourdough through SHS-GC/MS. Click here to View table |
Aroma volatile compounds during sourdough fermentation originate mainly from microbial and yeast metabolism, enzymatic or autoxidation of flour lipids, and Maillard reaction.14 Hydrocarbons were the most abundant aroma volatile compound in whole wheat sourdough15, whereas only 6 for all-purpose sourdough. Origins of hydrocarbons are still not clearly identified in the literature but occurrence is more common in studies that employed headspace extraction15,16,17,18 compared to solvent extraction.19,20,21,22
Whole wheat sourdough was found to contain 2-methyl-1-butanol (active amyl alcohol) that imparts a malty flavor to bread crumb and crust.23 Unfortunately, ethanol, a primary product of fermentation, was not found in whole wheat and all-purpose sourdough. Alcohol formation is attributed to activities of lactic acid bacteria and the Ehrlich pathway, which involves the degradation of flour amino acids inside the yeast cell. The steps involve the initial transamination of an amino acid producing an α-keto acid, irreversible decarboxylation of the acid to an aldehyde, and reduction of alcohol dehydrogenase to fusel alcohol.14
The substantial number of aldehydes, in whole wheat and especially all-purpose sourdough, are caused by enzymes specifically lipoxygenase and hydroperoxide lyase present in the wheat endosperm, germ, or bran.24 In the case of all-purpose sourdough, most of the aldehydes were aliphatic in nature like nonanal, decanal, and undecanal. Linear chain aldehydes result from degradation of unsaturated fatty acids mainly linoleic and linolenic acid.14,23 Nonanal, found in whole wheat and all-purpose sourdough, is the main oxidation product of oleic acid.25 The aromatic aldehyde, benzaldehyde, in whole wheat sourdough results from the autoxidation of 2,4-decadienal or aromatic amino acid degradation.25 Aldehydes are characterized as off-odors in bread and often related to the oxidative status of food, in general.14,23
A sizable number of ketones were represented in whole wheat (7) and all-purpose sourdough (6). Benzophenone and 2-pentadecanone were detected in both sourdoughs, and are associated with a rosy26 and spicy aroma27, respectively. Acetoin or 3-hydroxy-2-butanone, exclusively found in whole wheat sourdough, had a total peak area percent of 36.764% combined from 24 and 168 hours of fermentation. Acetoin is characterized with a butter or popcorn like aroma.14,23 Since most of the ketones were present during the early stages of fermentation of whole wheat sourdough, it is likely that ketones are a good indicator of sourdough and bread freshness.
Esters in sourdough and bread are characterized by pleasant and fruity aromas. However, a low odor activity value (OAV) and flavor dilution (FD) factor render ketones as non-major odorants in the final bread product, thus esters do not affect the flavor of bread.14 Ethyl esters such as ethyl acetate, ethyl hexanoate, ethyl octanoate, ethyl nonanoate, and ethyl decanoate originate from the reaction of alcohols and acetyl coenzyme A derivatives of fatty acids.23 Fatty acid esters, present in whole wheat and all-purpose sourdough, may be a product of free fatty acids, from further oxidation of aldehydes, reacting with certain alcohols.
Few volatile acids were found in whole wheat (1) and all-purpose sourdough (3). Acids are derived mainly from lactic acid bacteria that serve as a biopreservative, to extend bread shelf-life, and give a tart and tangy flavor to bread.8,9 Surprisingly, acetic acid was not detected in both sourdoughs; acetic acid is the most commonly cited volatile acid in the literature.14,23
Lactic acid, a conversion product from hexoses produced by homofermentative lactic acid bacteria13, was also not detected probably due to its nonvolatile nature.
Sulfur compounds were also identified in whole wheat and all-purpose sourdough. Common to both sourdoughs was benzothiazole with an intense roasted aroma.26 Volatile heterocyclic compounds such as furans (2-propyl furan, 3-methyl-2-(2-oxopropyl) furan, and tetrahydro-2-methyl furan), pyrrole (1H-pyrrole-2,5-dione, 3-ethyl-4-methyl-), and pyrazole (pyrazolidinetrione, phenyl-,4-(phenylhydrazine)) were present only in whole wheat sourdough. The pyrrole had the highest combined peak area percent of 39.261%. Heterocycles are attributed to the Maillard reaction.28 However, this cannot be the case in the present study since Castle and Crews (2005) reported that the temperature range of 40 to 60 °C had no significant effect on furan formation during headspace incubation. Take note that headspace extraction for this study was 60 °C. Furthermore, model systems for Maillard reaction products found that the reaction was active at temperatures ranging from 80 to 120 °C.30,31
Assessment of evolution of aroma volatile compounds as fermentation time progressed clearly showed a reduction, in terms of peak area percent, of aldehydes, ketones, and heterocycles for whole wheat sourdough, and of esters and sulfur compounds for all-purpose sourdough. An opposite trend was observed in the case of hydrocarbons, and aldehydes as well as acids, present in whole wheat and all-purpose sourdough, respectively. Meanwhile, fluctuating levels of alcohols, acid, esters, and sulfur compounds were found in whole wheat sourdough. Similarly, this was the case for alcohols, ketones, and hydrocarbons in all-purpose sourdough. Figures 1 and 2 exhibit the changes of the dominant aroma volatile compounds during fermentation of whole wheat and all-purpose sourdough, respectively.
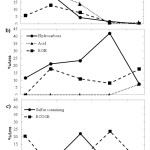 |
Figure 1: Changes in peak area percent of aroma volatile compoundsin whole wheat sourdough during 672 hours of fermentation Click here to View figure |
 |
Figure 2: Changes in peak area percent of aroma volatilecompounds in all-purpose sourdough during 672 hours of fermentation Click here to View figure |
Comparison of the two sourdoughs evaluated showed that whole wheat sourdough had a more complex aroma volatile compound composition as opposed to all-purpose sourdough. There were three key groups of volatile compounds that can differentiate whole wheat and all-purpose sourdough especially in terms of aroma. First, whole wheat sourdough was dominated by hydrocarbons, whereas all-purpose sourdough was inundated with aldehydes. Second, whole wheat sourdough contained heterocyclic compounds, such as furans, pyrrole, and pyrazole while all-purpose sourdough had none. Third, more compounds reported in the literature are associated with whole wheat sourdough namely 2-methyl-1-butanol, 3-hydroxy-2-butanone, 2-pentadecanone, benzophenone, benzaldehyde, 2,4-decadienal, nonanal, benzene, and benzothiazole. On the contrary, all-purpose sourdough aroma volatile compounds were benzophenone, 2-pentadecanone, nonanal, and benzothiazole. The differences and similarities between whole wheat and all-purpose sourdough are summarized in figure 3.
 |
Figure 3: Comparison of the aroma volatile compounds in whole wheat and all-purpose sourdough Click here to View figure |
The most cited volatile organic compounds found in bread crust and sourdough were: isoamyl and isobutyl alcohol, ethanol, hexanal, benzaldehyde, 3-methylbutanal, ethyl acetate, ethyl octanoate, acetic acid and butanoic acid.14,23 The sources of these compounds were from the fermentation process, lipid oxidation, and Maillard reactions. It has also been observed that the fermentation time could greatly affect the presence of various volatile organic compounds. We believe that our results could give insights on the control of aroma compounds in sourdough and bread prepared from whole wheat flour and all-purpose flour by varying the fermentation time. Fermentation temperature is another possible factor that can affect the fermentation process and may also be controlled to adjust the aroma compounds that will be present in the sourdough and bread.
Conclusions
The major aroma volatile compounds of whole wheat sourdough were hydrocarbons, esters, alcohols, ketones, aldehydes, and heterocycles (furan, pyrrole, and pyrazole). On the other hand, the major volatile compounds of all-purpose sourdough were aldehydes. The major sources of these compounds are fermentation due to bacteria and yeasts as well as lipid oxidation. As fermentation time increased, peak area percent of aldehydes, ketones, and heterocycles decreased in whole wheat sourdough. A similar trend was observed for esters and sulfur compounds in all-purpose sourdough. Peak area percent increased for hydrocarbons in whole wheat sourdough, and for aldehydes together with acids in all-purpose sourdough. There were clear differences in the aroma volatile composition of whole wheat and all-purpose sourdough. The former had a more complex aroma volatile compound composition than the latter. Evaluation of the aroma volatile compounds emanating from sourdough fermentation can aid consumers as well as manufacturers with regards to the quality, shelf-life, and what characteristic aromas the final bread product will possess.
Acknowledgements
This work did not receive any specific research project grant.
References
- FNRI-DOST (2015). Nutritional Guidelines for Filipinos: a prescription to good nutrition. Retrieved April 05, 2017, from http://region4a.dost.gov.ph/news/766-nutritional-guidelines-for-filipinos-a-prescription-to-good-nutrition.
- USDA (2015). Dietary Guidelines for Americans 2015-2020; U.S. Department of Agriculture: Washington, DC, 2016.
- Tighe, P., Duthie, G., Vaughan, N., Brittenden, J., Simpson, W. G., Duthie, S., Thies, F. (2010). Effect of increased consumption of whole-grain foods on blood pressure and other cardiovascular risk markers in healthy middle-aged persons: A randomized controlled trial. American Journal of Clinical Nutrition, 92(4), 733–740.
CrossRef - Ye, E. Q., Chacko, S. A., Chou, E. L., Kugizaki, M., & Liu, S. (2012). Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. Journal of Nutrition, 142(7), 1304–1313.
CrossRef - Chatenoud, L., Tavani, A., Vecchia, C. L., Jacobs, D. R., Negri, E., Levi, F., & Franceschi, S. (1998). Whole grain food intake and cancer risk. International Journal of Cancer Int. J. Cancer, 77(1), 24-28.
CrossRef - Buehler, E. (2006). Bread science: the chemistry and craft of making bread. Carrboro: Two Blue Books.
- Poutanen, K., Flander, L., & Katina, K. (2009). Sourdough and cereal fermentation in a nutritional perspective. Food Microbiology, 26(7), 693-699.
CrossRef - Schnürer, J., & Magnusson, J. (2005). Antifungal lactic acid bacteria as biopreservatives. Trends in Food Science & Technology, 16(1-3), 70–78.
CrossRef - Cizeikiene, D., Juodeikiene, G., Paskevicius, A., & Bartkiene, E. (2013). Antimicrobial activity of lactic acid bacteria against pathogenic and spoilage microorganism isolated from food and their control in wheat bread. Food Control, 31(2), 539-545.
CrossRef - Izzreen, M. N., Petersen, M. A., & Hansen, Å S. (2016). Volatile Compounds in Crumb of Whole-Meal Wheat Bread Fermented with Different Yeast Levels and Fermentation Temperatures. Cereal Chemistry Journal, 93(2), 209-216.
CrossRef - Plessas, S., Alexopoulos, A., Bekatorou, A., Mantzourani, I., Koutinas, A. A., & Bezirtzoglou, E. (2011). Examination of freshness degradation of sourdough bread made with kefir through monitoring the aroma volatile composition during storage. Food Chemistry, 124(2), 627-633.
CrossRef - Aponte, M., Boscaino, F., Sorrentino, A., Coppola, R., Masi, P., & Romano, A. (2013). Volatile compounds and bacterial community dynamics of chestnut-flour-based sourdoughs. Food Chemistry, 141(3), 2394-2404.
CrossRef - Hansen, A., & Schieberle, P. (2005). Generation of aroma compounds during sourdough fermentation: Applied and fundamental aspects. Trends in Food Science & Technology, 16(1-3), 85-94.
CrossRef - Birch, A. N., Petersen, M. A., & Hansen, Å S. (2014). REVIEW: Aroma of Wheat Bread Crumb. Cereal Chemistry, 91(2), 105-114.
CrossRef - Seitz, L. M., Chung, O. K., & Rengarajan, R. (1998). Volatiles in Selected Commercial Breads 1. Cereal Chemistry, 75(6), 847-853.
CrossRef - Ruiz, J. A., Quilez, J., Mestres, M., & Guasch, J. (2003). Solid-Phase Microextraction Method for Headspace Analysis of Volatile Compounds in Bread Crumb. Cereal Chemistry, 80(3), 255-259.
CrossRef - Bianchi, F., Careri, M., Chiavaro, E., Musci, M., & Vittadini, E. (2008). Gas chromatographic–mass spectrometric characterisation of the Italian Protected Designation of Origin “Altamura” bread volatile profile. Food Chemistry, 110(3), 787-793.
CrossRef - Maeda, T., Kikuma, S., Araki, T., Ikeda, G., Takeya, K., & Sagara, Y. (2009). The Effects of Mixing Stage and Fermentation Time on the Quantity of Flavor Compounds and Sensory Intensity of Flavor in White Bread. Food Science and Technology Research FSTR, 15(2), 117-126.
CrossRef - Moskowitz, M. R., Bin, Q., Elias, R. J., & Peterson, D. G. (2012). Influence of Endogenous Ferulic Acid in Whole Wheat Flour on Bread Crust Aroma. J. Agric. Food Chem. Journal of Agricultural and Food Chemistry, 60(45), 11245-11252.
CrossRef - Onishi, M., Inoue, M., Araki, T., Iwabuchi, H., & Sagara, Y. (2011). Odorant Transfer Characteristics of White Bread during Baking. Bioscience, Biotechnology, and Biochemistry, 75(2), 261-267.
CrossRef - Czerny, M., & Schieberle, P. (2002). Important Aroma Compounds in Freshly Ground Wholemeal and White Wheat FlourIdentification and Quantitative Changes during Sourdough Fermentation. J. Agric. Food Chem. Journal of Agricultural and Food Chemistry, 50(23), 6835-6840.
CrossRef - Kirchhoff, E., & Schieberle, P. (2001). Determination of Key Aroma Compounds in the Crumb of a Three-Stage Sourdough Rye Bread by Stable Isotope Dilution Assays and Sensory Studies. J. Agric. Food Chem. Journal of Agricultural and Food Chemistry, 49(9), 4304-4311.
CrossRef - Pico, J., Bernal, J., & Gómez, M. (2015). Wheat bread aroma compounds in crumb and crust: A review. Food Research International, 75, 200-215.
CrossRef - Lin, L., Peng, C., Wu, T., Yu, T., Chen, C., Wang, H., & Peng, R. Y. (2010). Flour Blending Based On Crude Fat Is More Crucial Than Ash Content In View Of Prolonged Quality Assurance. Journal of Food Processing and Preservation, 34(1), 104-126.
CrossRef - Jensen, S., Oestdal, H., Skibsted, L. H., Larsen, E., & Thybo, A. K. (2011a). Chemical changes in wheat pan bread during storage and how it affects the sensory perception of aroma, flavour, and taste. Journal of Cereal Science, 53(2), 259-268.
CrossRef - Paraskevopoulou, A., Chrysanthou, A., & Koutidou, M. (2012). Characterisation of volatile compounds of lupin protein isolate‐enriched wheat flour bread. Food Research International, 48(2), 568-577.
CrossRef - Obretenov, T., & Hadjieva, P. (1977). Gaschromatographische Massenspektralanalyse der Brot-Aromastoffe. Z Lebensm Unters Forch Zeitschrift Fr Lebensmittel-Untersuchung Und -Forschung, 165(4), 195-199.
CrossRef - Matsakidou, A., Blekas, G., & Paraskevopoulou, A. (2010). Aroma and physical characteristics of cakes prepared by replacing margarine with extra virgin olive oil. LWT – Food Science and Technology, 43(6), 949-957.
CrossRef - Castle, L., & Crews, C. (2006). FURAN: Methods applied by CSL and formation and mitigation in food. Joint DG Sanco/EFSA/DG JRC workshop furan in food: Analytical methods and brainstorming on the elements. To be included in database, 19. May, Brussels.
- Kim, J., & Lee, Y. (2009). Study of Maillard reaction products derived from aqueous model systems with different peptide chain lengths. Food Chemistry, 116(4), 846-853.
CrossRef - Martins, S. I., & Boekel, M. A. (2005). A kinetic model for the glucose/glycine Maillard reaction pathways. Food Chemistry, 90(1-2), 257-269.
CrossRef - Kolb, B., & Ettre, L. S. (2006). Static headspace-gas chromatography: Theory and practice. Hoboken, NJ: Wiley.
CrossRef - Ubeda, C., Callejón, R., Hidalgo, C., Torija, M., Mas, A., Troncoso, A., & Morales, M. (2011). Determination of major volatile compounds during the production of fruit vinegars by static headspace gas chromatography–mass spectrometry method. Food Research International, 44(1), 259-268.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.






