Introduction
Protein is an essential nutrient needed by the human body on a daily basis. In Indonesia, legumes, particularly soybeans, are common source of vegetable protein. However, domestic soybeans production only meet approximately 25-30% of the demand, with the remaining relying on imports.1 Therefore, it is necessary to explore other alternatives. Among the many legumes, white jack bean stand out as a potential commodity, with a total protein content of 31.4%, comparable to 37.58% of soybeans. 2 According to Agustia et al. 3, white jack bean has high bioavailability and excellent amino acid profile.
A product derivable from white jack bean is tempe which enhances the value. The transformation process includes several stages, namely boiling, soaking, yeast inoculation, and fermentation. These stages positively impact the food, enhancing taste, digestibility, likability, safety from microorganism spoilage, and inactivating antinutritional factors. 4 However, due to the high moisture content in tempe leading to a limited shelf life, the product needs to be processed into flour. This process also aid in reducing antinutritional compounds such as HCN. 5
The conversion of white jack bean tempe into protein concentrate further amplified its potential in the food industry, making it a versatile ingredient for various food products. 6 However, protein concentrate needs to be encapsulated to protect it from damage and maintain its stability. Encapsulation is an effective method that can improve the chemical and biological stability of food ingredient, protecting it from possible reactions. 7 Its common approaches are spray drying and freeze drying. According to Wang et al. 8, to maintain the quality of soybean protein hydrolysate, it is more effective to conduct spray drying. Encapsulation is covering or wrapping a material using various types of encapsulant or coating material, namely maltodextrin (MD), Gum Arabic (GA), WPC, and WPI.
Maltodextrin and Gum Arabic are the most commonly used encapsulants, valued for their excellent protective qualities, high water solubility, as well as neutral taste, smell, and colour. 9 This ensures that protein concentrate of white jack bean tempe remains unaffected in terms of the physical properties. When employing spray drying method, the encapsulant should exhibit good solubility, emulsification properties, film-forming capability, and low viscosity at high concentration. 10 While maltodextrin possesses most of these properties, the emulsifying capacity is limited. Therefore, combining it with other high-emulsifying biopolymers, such as Gum Arabic, is recommended. 11 According to Arifan et al 12 and Rao 13, the combination of maltodextrin and Gum Arabic yield better stability and encapsulation efficiency.
Various ratios of maltodextrin and Gum Arabic result in diverse physical and chemical properties of encapsulated materials. Several studies stated that a higher percentage of Gum Arabic increased protein content and encapsulation efficiency. 13–16 However, a higher maltodextrin percentage leads to lower moisture content and higher yields. 16–19 This study investigated the encapsulation of white jack bean tempe protein concentrate using different ratios of maltodextrin and Gum Arabic as the encapsulant. Experiments were conducted with varying ratios of maltodextrin: Gum Arabic, namely 0, 80:20, 70:30, 50:50, 30:70, and 20:80, to evaluate the effects on protein content, moisture content, encapsulation efficiency, and yield.
This study aimed to determine the impact of varying ratios of maltodextrin and Gum Arabic on the encapsulation of white jack bean tempe protein concentrate through spray drying method. Furthermore, the objective is to identify the optimal encapsulant ratio to preserve the physical and chemical characteristics of white jack bean tempe protein concentrate. The results were intended to increase the use of the commodity in the food industry and promote the application as a superfood.
Materials and Methods
Materials
The materials used in this study included white jack bean (Canavalia ensiformis) and yeast (Raprima, Indonesia) for tempe production. The protein concentrate was prepared using white jack bean tempe flour, distilled water, HCl, NaOH, and 70% alcohol. Furthermore, the materials used in the encapsulation process were protein concentrate of white jack bean tempe, distilled water, maltodextrin, and Gum Arabic. Additional supporting materials were employed for sample analysis.
White Jack Bean Tempe
The process began by sorting and cleaning white jack bean under running water. The bean was weighed as needed and boiled for 30 minutes, followed by a 24 hours soaking period with periodic water changes. Subsequently, the skin was peeled and the bean was cut into six pieces. After another round of washing and cleaning. They were steamed for 45 minutes. Inoculation was conducted by adding yeast (1.5% w/w) to cooled, cooked bean. Following this, the bean was packed with pp ziplock plastic with a hole and left to ferment at 27 °C for 36 hours. Finally, the fermented product was sliced into 0.5 cm thick pieces, dried in an oven at 50 °C for 24 hours, and ground using a grinder. The resulting milled tempe was sifted through an 80-mesh sieve.
White Jack Bean Tempe Protein Concentrate
White jack bean tempe flour was mixed with distilled water at a ratio of 1:10 (w/v) and stirred continuously on a hot plate stirrer until fully dissolved. The initial pH of the solution was measured. To initiate extraction, 2 N NaOH was added to the mixture until a pH of 10 was reached, and this state was maintained for 30 minutes at 30 °C. The solution was then subjected to centrifugation at 4000 rpm for a duration of 15 minutes, yielding a clear liquid (supernatant). Protein separation was achieved by introducing 2 N HCl until the pH decreased to 4.2. This condition was sustained for an additional 10 minutes, followed by another round of centrifugation at 4000 rpm for 15 min. The residue obtained represented the white jack bean tempe protein concentrate. It was then weighed and mixed with 70% alcohol at a ratio of 1:3 (w/v). The mixture was homogenized for 1 hour and subjected to centrifugation at 4000 rpm and 4 °C for 10 minutes, resulting in the final protein concentrate.
Encapsulation
The encapsulation process used spray drying method. To prepare the encapsulant solution, maltodextrin and Gum Arabic were blended in a specified proportion. This mixture was then dissolved in distilled water and agitated for 24 hours using a stirrer, in order for it to get hydrated. Simultaneously, the protein concentrate solution was homogenized in distilled water. The two solutions were combined and subjected to spray drying at an inlet temperature of 160 °C to produce the encapsulated powder.
Protein Content
The protein content was determined using the Kjeldahl method. A 0.5 g sample was weighed and placed into the Kjeldahl flask. Subsequently, a Kjeldahl tablet and 10 ml concentrated H2SO4 were introduced. Digestion was conducted for 2 hours, followed by cooling for 1 hour. The digested sample was diluted with 25 ml distilled water. Separately, in an Erlenmeyer flask, a solution was prepared with 30 ml of 3% H3BO3 and 2-3 drops of the methyl red indicator. The sample in the Kjeldahl flask was then subjected to distillation. Subsequently, the distillate was titrated using 0.02 N HCl until a pink hue appeared. The volume of HCl consumed during the titration was recorded. 20
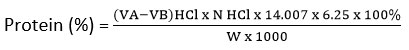
Given
VA = Volume of HCl used for titrating the sample (ml)
VB = Volume of HCl used for titrating the blank (ml)
N = Normality of the standardized HCl solution
14.007 = Atomic weight of nitrogen
6.25 = Conversion factor for protein content in legumes
W = Weight of the sample (g)
Moisture Content
The moisture content was determined using the gravimetric technique. A cup was placed in an oven set to 105 °C for 1 hour. It was then allowed to cool in a desiccator for 15 minutes, before being weighed using a precision balance. The drying process was repeated until a constant weight was achieved, after which 1 g sample was added to the cup. This assembly was heated in the oven at 105 °C for 5-6 hours. Subsequently, the cup was placed in a desiccator for 20 minutes and reweighed using an analytical balance. The drying process was repeated until the combined weight of the cup and the sample remained unchanged. 20
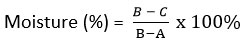
Given:
A = Weight of the constant cup (g)
B = Weight of the wet sample combined with the constant cup (g)
C = Weight of the dry sample combined with the constant cup (g)
Yield
The yield is determined based on the ratio of ingredients used to the mass of encapsulant produced. 20
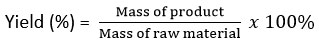
FTIR (Fourier Transform-Infrared Spectroscopy) Analysis
A 5 mg dried sample was blended with potassium bromide (KBr) until a uniform mixture was achieved. Subsequently, this powdered mixture was compressed into pellets using a hydraulic press. Finally, the resulting pellets were subjected to analysis using an FTIR Spectrophotometer (Thermo Scientific Nicolet iS5, US) in the 4000-500 cm-1 range, with a resolution of 4 cm-1. 21
Statistical Analysis
The data were six treatments and three replicates examined using Analysis of Variance Test (ANOVA) at the 95% confidence level (α=0.05). When the significance probability value (p value) is >0.05, it indicated no significant difference. Value (p value) <0.05 implied a significant difference, hence, the analysis with Duncan’s Multiple Range Test (DNMRT).
Results and Discussion
Encapsulation comprises the application of an encapsulating substance to envelop a material. This method serves as a means to safeguard the physical, chemical, and biological attributes. Furthermore, the process has the potential to alter the state of the material, transforming it from a liquid to a solid. 22 In the food industry, the use of encapsulation is frequently advantageous, as it proves valuable in enhancing and preserving the stability of bioactive constituents. These include fats, vitamins, peptides, fatty acids, antioxidants, minerals, and live microorganisms such as probiotics in various stages of food processing and storage. Additionally, the process prevent undesirable interactions between these bioactive components and the food matrix. 23,24 In this study, spray drying was employed to encapsulate a protein concentrate extracted from white jack bean tempe. Various ratios of maltodextrin and Gum Arabic were used in the encapsulation process. Figure 1 shows a visual comparison of the white jack bean tempe protein concentrate before and after encapsulation.
 |
Figure 1: White jack bean tempe (a) before encapsulation, (b) after encapsulation |
Protein Content
Protein is a macronutrient that contains carbon, hydrogen, oxygen, and nitrogen. 25 When processing white jack bean into encapsulated protein concentrate, changes in protein content were observed. As a result, this variable was quantified at each stage using the Kjeldahl method, a method based on nitrogen content measurement. The analysis comprised white jack bean tempe, the flour, protein concentrates in wet form, and encapsulated protein concentrate for each treatment. The protein content analysis for these materials is presented in Table 1.
Table 1: Protein contents of the materials
|
Material |
Protein Content (%db) |
|
White Jack Bean Tempe |
33.34 ± 0.76 |
|
White Jack Bean Tempe Flour |
30.99 ± 0.04 |
|
Wet Protein Concentrate |
56.76 ± 0.28 |
Notes: Protein contents are presented as mean ± standard deviation.
Based on the results, the protein content of white jack bean tempe was 33.34 ± 0.76%, meeting the quality requirements outlined in SNI 3144:2015 concerning soybean tempe, where the minimum value was set at 15%. The decrease in protein from tempe to flour was attributed to the 24 hours heating process during flour production.
When transforming tempe flour into protein concentrate, there was a significant increase in protein content from 30.99 ± 0.04% to 56.76 ± 0.28%. The rise indicated that the acid-based approach with an isoelectric point and the defatting process used in concentrate production yield satisfactory result, since the threshold was set at a minimum of 50%-70%. However, the protein content of the wet concentrate changed after encapsulation. Table 2 presents the protein content of encapsulated protein concentrate under different treatments.
Table 2: Protein content of the white jack bean tempe protein concentrate with each treatment.
|
Treatment (MD: GA) |
Protein Content (%db) |
|
P1 (0:0/control) |
56.36 ± 0.00d |
|
P2 (80:20) |
24.02 ± 0.04a |
|
P3 (70:30) |
25.55 ± 0.09b |
|
P4 (50:50) |
25.94 ± 0.11c |
|
P5 (30:70) |
25.78 ± 0.07bc |
|
P6 (20:80) |
26.10 ± 0.45c |
Notes: Protein content was presented as mean ± standard deviation, and different subscript notations indicated significant differences between treatments in the 5% Duncan test.
According to Duncan further test in Table 2, some treatments showed significant differences in protein content at 5% significance level. Specifically, P1, without an encapsulant, produces the highest protein content due to the absence of encapsulant, which causes an increase in total solids in the concentrate, preventing the content from decreasing. In comparison, P2 to P6 had a decrease in protein content due to the addition of encapsulant.
The highest protein content at 26.10 ± 0.45%, in the encapsulation results, was obtained from the treatment using an expansion of encapsulant MD:GA 20:80, labelled as P6. This treatment showed a significant difference with P1, P2, and P3. However, it did not show any significant difference between P4 and P5. P6 also had the highest addition of Gum Arabic. This was in line with adding more Gum Arabic will produce higher protein content. 16 The phenomenon was attributed to the presence of arabinogalactan protein (AGP) and glycoprotein (GP) in Gum Arabic, which exhibit a propensity to associate with another protein. As the concentration of encapsulant increases, the influence of AGP and GP in introducing nitrogen into the product becomes more pronounced.26,27
The lowest result of 24.02 ± 0.04% was obtained from P2, the treatment with the slightest addition of Gum Arabic, MD:GA (80:20). P2 showed results that were significantly different from the other treatments. This is because there was no protein binding.28
Moisture Content
Moisture content is the total water contained in an ingredient or food product. It is one of the main factors in microbial growth which is being inhibited with a concentration of below 10%. 29 Similar to protein content, there are variations in water at each stage of making white jack bean tempe protein concentrate. Therefore, moisture content was analyzed using the gravimetric method or weighing. The results of the material moisture content are presented in Table 3.
Table 3: Moisture contents of the materials.
|
Material |
Moisture Content (%db) |
|
White Jack Bean Tempe |
56.03 ± 1.00 |
|
White Jack Bean Tempe Flour |
9.93 ± 0.06 |
|
Wet Protein Concentrate |
86.02 ± 0.85 |
Notes: Moisture content is presented as mean ± standard deviation.
Table 3 shows that the moisture content of white jack bean tempe is in accordance with SNI 3144:2015 concerning soybean tempe. In the regulation, the maximum moisture content was set at 65%. Specifically, white jack bean tempe had moisture content of 56.03 ± 1.00%. This high moisture content is a result of several steps in the tempe-making process, such as soaking, boiling, fermentation, and others.
Table 3, the moisture content of white jack bean tempe flour stands at 9.93 ± 0.06%, in accordance with the standard quality specifications outlined in SNI 01-3751-2006. According to this standard, the maximum permissible moisture content for flour was set at 14.5%. The reduction in moisture content was attributed to the drying process applied, which included the use of a forced convection oven for a duration of 24 hours. In the wet protein concentrate, the concentration of water was 86.02 ± 0.85%. This is because in the making process, flour was dissolved using distilled water in a ratio of 1:10 (flour: distilled water), with an addition of NaOH and HCl. Therefore, encapsulation was conducted using spray drying with different treatments, including adding various encapsulants. Table 4 present the results on the moisture content of each treatment.
Table 4: Moisture content of white jack bean tempe protein concentrate with each treatment.
|
Treatment (MD: GA) |
Moisture Content (%db) |
|
P1 (0:0/control) |
7.00 ± 0.13d |
|
P2 (80:20) |
4.42 ± 0.37a |
|
P3 (70:30) |
5.63 ± 0.07b |
|
P4 (50:50) |
6.40 ± 0.20c |
|
P5 (30:70) |
5.86 ± 0.18b |
|
P6 (20:80) |
7.93 ± 0.42e |
Notes: Moisture content is presented as mean ± standard deviation, and different subscript notations indicated significant differences between treatments in the 5% Duncan test.
According to the Duncan further test in Table 4, some treatments showed significantly different results in moisture content at 5% significance level. In P1, which was the control, the concentration of water was 7.00 ± 0.13% and significantly differs from other treatments. The results fall above the range of moisture content in spray drying, ranging from 2-6% according to Yolanda. 30 Meanwhile, P2 to P6, treatments with added encapsulant, showed that P2 was significantly different from others and had the lowest concentration of water at 4.42 ± 0.37%. It also had the most maltodextrin ratio of 80:20 (MD: GA). This was in line with the report of Premi & Sharma 31, where it was reported that adding more this encapsulant results in lower moisture content. Maltodextrin used in this study was DE 10-12, with a molecular weight of 15,000-15,400 g/mol, smaller than Gum Arabic 32,33, implying a lower moisture content.
P6, which was the addition of encapsulant with the highest Gum Arabic ratio of 20:80 (MD:GA), produced the highest concentration of water at 7.93 ± 0.42% and showed significantly different results with other treatments. This follows several studies, where it was reported that adding Gum Arabic can increase moisture content due its molecular weight of 250,000 to 1,000,000. 34 Additionally, the encapsulant has hydrophilic properties, meaning it retains bound water for longer. 26
Yield
Yield is a value obtained from calculating the powder mass from spray drying divided by the initial mass of material entering spray dryer multiplied by 100. The viscosity of the material before drying is a factor that determines the amount of yield which was also affected by the type and added ratio of encapsulant. Table 5 shows the yield results from the treatment of maltodextrin and Gum Arabic variations in white jack bean tempe concentrate.
Table 5: Yield of white jack bean tempe protein concentrate with each treatment.
|
Treatment (MD:GA) |
Yield (%db) |
|
P1 (0:0/control) |
5.33 |
|
P2 (80:20) |
18.67 |
|
P3 (70:30) |
17.17 |
|
P4 (50:50) |
21.60 |
|
P5 (30:70) |
20.93 |
|
P6 (20:80) |
14.27 |
Based on Table 5, the yield results from each treatment showed different results. The most negligible value was obtained from the control treatment (P1), which did not include the addition of encapsulant. This is because, without the expansion of an encapsulant, the total solids will not increase, as they are directly proportional. Meanwhile, the highest yield was produced by P4 or the addition of MD:GA (50:50) encapsulant. The amount was influenced by emulsion stability. A stable emulsion and appropriate encapsulant ratio will facilitate the powder formation, resulting in high yields. 18 Gum Arabic in P4 significantly contributed to the high yield due to its good film-forming ability and emulsion-stabilizing properties. 35 Maltodextrin, producing low viscosity with high total solids, also contributed to the yield. 28 However, from the results obtained, MD:GA (20:80) or P6 had the most negligible yield for encapsulation. This is because the encapsulant concentration is directly proportional to the viscosity of the mixture of ingredient, resulting in a stickier mix and lower yield during spray drying. 16
FTIR (Fourier transform infrared Spectroscopy)
FTIR is a scientific instrument used to identify functional groups within both organic and inorganic compounds. This identification is achieved through the measurement of the absorption of infrared radiation at different wavelengths. 36 The operating principle of FTIR includes the exposure of a sample to infrared light and the subsequent observation of which specific wavelengths in the spectrum were absorbed by the sample. Each compound possesses unique absorption regions, allowing for the differentiation and characterization based distinctive spectral signatures. 37
The absorption regions in the spectrum of proteins and polypeptides were categorized into several types, each denoted by an “amide”. These include amide A, B, I, II, III, IV, V, VI, and VII. These divisions were instrumental in characterizing and analyzing the infrared spectra of these biomolecules, providing valuable information about the structural features and chemical composition.38
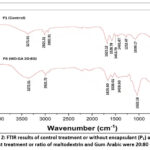 |
Figure 2: FTIR results of control treatment or without encapsulant (P1) and the best treatment or ratio of maltodextrin and Gum Arabic were 20:80 (P6). |
Figure 2 shows the spectra of P1 (control) and P6 (best treatment). However, there is still a shift and difference in peaks due to the addition of encapsulant in the best treatment. A broad peak at wave number 3271.91 cm-1 in P1 and shifts to 3272.51 cm-1 in P6, indicated the presence of OH or hydroxyl groups. Furthermore, sharp peaks at 2921.31 cm-1 and 2851.51 cm-1 in P1 and 2921.72 cm-1 in P6, demonstrated the presence of CH stretching groups. Additionally, a sharp peak at 1623.12 cm-1 in P1 and shifts to 1625.60 cm-1 in P6 showed the presence of NH bending or amine groups. It was mentioned that these numbers include amide I groups C = O stretching, as they fall in the range of 1600 cm-1 – 1690 cm-1. Other peaks, such as those at wave numbers 1528.79 cm-1 and 1453 cm-1 in P1 and 1538.01 cm-1 in P6, corresponded to CN stretching and NH bending groups, which include amide II. Peaks at 1401.67 cm-1 in P1 and 1409.60 cm-1 in P6 indicated OH bending groups.
In P1, a peak at 1232.47 cm-1 corresponds to a CN stretching and NH bending group or amide III, while P6 lacks a peak in that region. Furthermore, there is a peak at wave number 1039.17 cm-1 in P1 and shifts to 1022.19 cm-1 in P6, indicating a CN stretching or amine group. A peak at 631.62 cm-1 in P1, associated with a bending OCN group or amide IV, was absent in P6. Meanwhile, in P6, there is a peak at 599.65 cm-1, which belongs to the amide VI or C=O bending functional group, a feature not observed in P1.38,39
Conclusion
In conclusion, the protein concentrate derived from jack bean tempe, with varying ratios of maltodextrin and Gum Arabic, exhibited significant differences in protein content, moisture content, and yield. The highest and lowest protein content was observed in ratios of maltodextrin to Gum Arabic at 20:80 and 80:20, respectively. Additionally, the highest yield was achieved at 50:50. Among the tested ratios, the combination of maltodextrin and Gum Arabic in a 20:80 ratio was the most favorable treatment. This formulation exhibited enhanced functional groups responsible for protein absorption, including amide A, B, I, II, III, IV, V, VI, and VI.
Acknowledgment
The authors are grateful to the Universitas Padjadjaran and The Ministry of Education and Culture of Indonesia for supporting this study.
Conflict of Interest
There was no conflict of interest in this study.
Funding Sources
This study received funding from Universitas Padjadjaran through the Riset Disertasi Doktor (RDDU) scheme research with Number Grant 1932/UN6.N/LT/2019.
References
- Ramadhani D. A., Sumanjaya R. Analysis of factors affecting the availability of soybeans in Indonesia. Economica. 2015; 4(1):30-41. DOI:10.22202/economica.2015.v4.i1.261
CrossRef - Ekafitri R., Isworo R. Use of peanuts as a raw material for protein sources for emergency food. Pangan. 2014; 23(2): 134-144.
- Agustia F. C., Subardjo Y. P., Ramadhan G. R., Betaditya D. Formula of mohiro flakes made of mocaf-black rice supplementated with jack bean as alternative breakfast high protein and dietary fiber.Indonesian Food Technologists. 2019; 8(4): 130-136. DOI:10.17728/jatp.3009
CrossRef - Purwandari, F. A., Annisa, E. D. N., Rachmawati, A. T., Puspitasari, D., Wikandari, R., Setyaningsih, W., Ningrum, A., Sardjono. Effect of different cooking methods on chemical composition, nutritional values, and sensory properties of Jack bean (Canavalia ensiformis) tempe. Food Res. 2021; 5(3): 327-333.
CrossRef - Putro N. T. L., Arisasmita J. H., Srianta I. Study of HCN Content and Starch Characteristics in Jack Bean Flour (Canavalia ensiformis) based on Seeds Soaking Time in Hot Water and Sodium Bicarbonatesolution (NaHCO3). J Teknol Pangan dan Gizi. 2015; 14(2): 72-82.
- Jarpa-Parra M. Lentil protein: a review of functional properties and food application. An overview of lentil protein functionality. Int J Food Sci Technol. 2018; 53(4): 892-903. DOI:10.1111/ijfs.13685
CrossRef - Rezvankhah A., Emam-Djomeh Z., Askari G. Encapsulation and delivery of bioactive compounds using spray and freeze-drying techniques: A review. Dry Technol. 2020; 38(1-2): 235-258. DOI:10.1080/07373937.2019.1653906
CrossRef - Wang H., Tong X., Yuan Y., Peng X., Zhang Q., Zhang S., Xie C., Zhang X., Yan S., Xu J., Jiang L., Qi B., Li Y. Effect of Spray-Drying and Freeze-Drying on the Properties of Soybean Hydrolysates. J Chem. 2020; 1-8. DOI:10.1155/2020/9201457
CrossRef - Todorović A., Šturm L., Salević-Jelić A. Encapsulation of Bilberry Extract with Maltodextrin and Gum Arabic by Freeze-Drying: Formulation, Characterisation, and Storage Stability. Processes. 2022; 10(1991): 1-16. DOI:10.3390/pr10101991
CrossRef - Landoni L., Alarcon R., Vilca L., Chasquibol N., Pérez-Camino M.C., Gallardo G. Physicochemical characterization and oxidative stability of microencapsulated edible sacha inchi seed oil by spray drying. Grasas Aceites. 2020; 71(4): 1-10. DOI:10.3989/gya.1028192
CrossRef - Fernandes L. P., Turatti I. C. C., Lopes N. P., Ferreira J. C., Candido R. C., Oliveira W. P. Volatile Retention and Antifungal Properties of Spray- Dried Microparticles of Lippia sidoides Essential Oil. Dry Technol. 2008; 26(12): 1534-1542. DOI:10.1080/07373930802464034
CrossRef - Arifan F., Wikanta D. K. Microcapsules formulation of cinnamon oleoresin (Cinnamon burmanni) and cloves (Caryophillus aromaticus Linn). J Ilmu Pertan. 2020; 4(1): 30-35.
- Rao P. S., Bajaj R. K., Mann B., Arora S., Tomar S. K. Encapsulation of antioxidant peptide enriched casein hydrolysate using maltodextrin–gum arabic blend. J Food Sci Technol. 2016; 53(10): 3834-3843. DOI:10.1007/s13197-016-2376-8
CrossRef - Cian R. E., Campos-Soldini A., Chel-Guerrero L., Drago S. R., Betancur-Ancona D. Bioactive Phaseolus lunatus peptides release from maltodextrin/gum arabic microcapsules obtained by spray drying after simulated gastrointestinal digestion. Int J Food Sci Technol. 2019; 54(6): 2002-2009. DOI:10.1111/ijfs.14031
CrossRef - Giroldi M., Grambusch I. M., Schlabitz C., Kuhn D., Lehn D. N., Volken de Souza C. F. Encapsulation of protein hydrolysates by spray drying: feasibility of using buffalo whey proteins. Int J Food Sci Technol. 2022; 57(6): 3419-3427. DOI:10.1111/ijfs.15665
CrossRef - Rasyid R.P. Influence of addition of gum arab and maltodextrin on physicochemical features of albumin fish powers (Channa striata) by vacuum drying methods. Thesis. Universitas Brawijaya.2019. http://repository.ub.ac.id/id/eprint/170764
- Kang Y. R., Lee Y. K., Kim Y. J., Chang Y. H. Characterization and storage stability of chlorophylls microencapsulated in different combination of gum Arabic and maltodextrin. Food Chem. 2019; 272: 337-346. DOI:10.1016/j.foodchem.2018.08.063
CrossRef - Kunarto B, Sani E.Y. Characteristics of durian skin antioxidant extract (Durio zibethinus murr.) encapsulated using durian seed maltodextrin and gum arabic. Pros SNST Univ Wahid Hasyim. 2018: 84-90.
- Sonone E. V. S, Unde P. A, Kad V. P. Effect of process parameters on physical properties of spray dried banana pseudostem juice powder. Int J Adv Eng Manag Sci. 2016; 2(8): 1301-1312.
- AOAC International. Official Methods of Analysis. AOAC International. 18th ed. 2005; Method 922.09.
- Kumari A., Kaushik N., Slizyte R., Khushboo. Production and microencapsulation of protein hydrolysate of pink perch (Nemipterus japonicus) by-products obtained from Surimi Industry for its sustainable utilization. Waste Biomass Valorization. 2022; 14(1): 209-226. DOI:10.1007/s12649-022-01853-3
CrossRef - Agustin D. A., Wibowo A. A. Encapsulation Technology: Techniques And Applications. Distilat J Teknol Separasi. 2021; 7(2): 202-209. DOI:10.33795/distilat.v7i2.210
CrossRef - Nedovic V., Kalusevic A., Manojlovic V., Levic S., Bugarski B. An overview of encapsulation technologies for food applications. Procedia Food Sci. 2011; 1:1806-1815. DOI:10.1016/j.profoo.2011.09.265
CrossRef - de Vos P., Faas M. M., Spasojevic M., Sikkema J.. Encapsulation for preservation of functionality and targeted delivery of bioactive food components. Int Dairy J. 2010; 20(4): 292-302. DOI:10.1016/j.idairyj.2009.11.008
CrossRef - Callahan A, Leonard H, Powell T. Nutrition: Science and Everyday Application. Open Oregon Educational Resources. 2020. https://openoregon.pressbooks.pub/nutritionscience2e/
- Sugianto, Suprayitno E., Budi S. B. The effect of addition of dextrin and arabic gum to the quality of crude albumin Fish Cork (Ophiocephalus striatus). Int J Sci Res Publ. 2019; 9(7): 396-400. DOI:10.29322/ijsrp.9.07.2019.p9153
CrossRef - Merillon J. M, Ramawat K. G. Bioactive Molecules in Food. Spinger Nature. 2019. DOI:10.1007/978-3-319-78030-6
CrossRef - Kania W., Andriani M. M., Siswanti. The effect of a binder ratio variation to physical and chemical characteristics in hyacinth bean sprouts granul functional instant drink (Lablab purpureus (L.) sweet). Teknosains Pangan. 2015; 4(3): 16-29.
- Zambrano V. M, Dutta B, Mercer D. G, MacLean H. L, Touchie M. F. Assessment of moisture content measurement methods of dried food products in small-scale operations in developing countries: A review. Trends Food Sci Technol. 2019;88:484-496. DOI:10.1016/j.tifs.2019.04.006
CrossRef - Yolanda, C. E. Y. Effect of using wall materials on spray drying method to physic and chemical properties of quail Egg Powder. Thesis. University of Katolik Soegijapranata. 2019. http://repository.unika.ac.id/id/eprint/21079
CrossRef - Premi M., Sharma H. K. Effect of different combinations of maltodextrin, gum arabic and whey protein concentrate on the encapsulation behavior and oxidative stability of spray dried drumstick (Moringa oleifera) oil. Int J Biol Macromol. 2017; 105: 1232-1240. doi:10.1016/j.ijbiomac.2017.07.160
CrossRef - Avaltroni F., Bouquerand P. E, Normand V. Maltodextrin molecular weight distribution influence on the glass transition temperature and viscosity in aqueous solutions. Carbohydr Polym. 2004; 58(3): 323-334. DOI:10.1016/j.carbpol.2004.08.001
CrossRef - Castro N., Durrieu V., Raynaud C., Rouilly A. Influence of DE-value on the physicochemical properties of maltodextrin for melt extrusion processes. Carbohydr Polym. 2016; 144: 464-473. DOI:10.1016/j.carbpol.2016.03.004
CrossRef - Prasad N., Thombare N., Sharma S. C., Kumar S. Gum arabic – A versatile natural gum: A review on production, processing, properties and applications. Ind Crops Prod. 2022; 187: 2-6. DOI:10.1016/j.indcrop.2022.115304
CrossRef - Karrar E., Mahdi A. A., Sheth S., Ahmed I. A. M., Manzoor M. F., Wei W., Wang X. Effect of maltodextrin combination with gum arabic and whey protein isolate on the microencapsulation of gurum seed oil using a spray-drying method. Int J Biol Macromol. 2021; 171: 208-216. DOI:10.1016/j.ijbiomac.2020.12.045
CrossRef - Berna F. Fourier Transform Infrared Spectroscopy (FTIR)- Encyclopedia of Geoarchaeology. In: Gilbert AS, ed. Dordrecht: Springer Netherlands. 2017; 285-286. DOI:10.1007/978-1-4020-4409-0_15
CrossRef - Gallagher W. FTIR Analysis of Protein Structure. eLS. 2009; 662-666. DOI:10.1002/9780470015902.a0021885
CrossRef - Bunaciu A. A., Fleschin Ş., Aboul-Enein H.Y. Evaluation of the protein secondary structures using fourier transform infrared spectroscopy. Gazi Univ J Sci. 2014; 27(1): 637-644.
- Nandiyanto A. B. D, Oktiani R., Ragadhita R. How to read and interpret ftir spectroscope of organic material. Indones J Sci Technol. 2019; 4(1): 97-118. DOI:10.17509/ijost.v4i1.15806
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.






