Introduction
Lactic acid bacteria are a wide microorganism group which is known for formation of lactic acid as a dominant metabolite with their sugar metabolism (Zhong et al. 2014). Lactic acid fermentation is the most common and the oldest way of food preservation. Besides preservation aspects of lactic acid bacteria are seen from mouth to the end of the intestinal system of human and other mammals (Molin 2001, de Vries et al. 2006).
Lactic acid bacteria commonly named as probiotics have a broad range of utilization area in human diet and food industry. Probiotics are defined as “living microorganisms when consumed adequate amounts provide health benefit to the host” by FAO/WHO working group (FAO/WHO, 2002). The use of probiotics has an increasing demand for their prevention and treatment of diseases mainly intestinal diseases (Zhong et al. 2014). On the other hand, in recent years many researchers have focused on their anti-genotoxic called DNA-bioprotective effects.
Human gets exposure with genotoxins such as mycotoxins, polycyclic aromatic hydrocarbons, and n-nitroso-compounds through diet and environmental contaminations. Mycotoxins are the secondary metabolite of fungus. Production requirements change with ambient temperature, water activity, pH, utilizable nutrients and competition with the other micro-organisms (El-Nezami et al. 1998). Aflatoxin B1 is mycotoxin which was reported one of the most potent hepato-carcinogen and its exposure stems from human diet. Once the food is contaminated with AFB1, it can be removed physical and chemical treatments. However, these treatments may lead to undesirable properties of food such as loss of nutritional quality, flavor etc. and need expensive investments.
Recent years, there is a growing interest of decontaminating DNA-reacting compounds with microbial processes. The first reported study by Ciegler et al. (1966), indicate many kind of microorganisms, bacteria, yeasts, moulds, actinomycetes and algae are able to reduce aflatoxin in food and feed. DNA-bioprotective effect depends on probiotic strain (Cenci et al. 2005, Caldini et al. 2008). One strain may show the antigenotxic activity efficiently, one strain may be the genotoxic itself. Mainly, antigenotoxic effect of these probiotic strains is a new functional property (Cenci et al. 2005).
In this study we aimed to investigate DNA-bioprotective effect of Lactobacillus plantarum on human colon adenocarcinoma (Caco-2) enterocytes against Aflatoxin B1 (AFB1) with comet assay without metabolic activation.
Materials and Methods
Bacterial culture
Lactobacillus plantarum isolated from fermented food in previously study was maintained in deMan Rogosa Sharpe (MRS) Broth at 37°C for 18 hours in 5% CO2 supplemented incubator. After 18 h incubation total viable lactic acid bacteria was determined with plate count method on MRS Agar incubated at 37°C for 24-48 h in CO2 incubator.
Caco-2 cell culture
Human intestinal colon adenocarcinoma, Caco-2 cell line, was used in the study. Caco-2 cells were grown in Dulbecco’s Modified Eagle Medium F-12 (DMEM) supplemented with 10% (v/v) fetal bovine serum (FBS), 1% (v/v) L-glutamine, %1 (v/v) penicillin-streptomycin solution. The cells were maintained at 37°C in a 5% CO2 atmosphere and sub-cultivated at 80% confluences with Trypsin-EDTA.
Cell viability test (Trypan blue dye exclusion test)
Cell viability was determined with trypan blue dye exclusion test which is based on the exclusion of the dye by the dead cells. Viable and non-viable cells were counted with Neubauer hemocytometer on the light microscope using x10 objectives.
Genotoxin preparation
Genotoxin, AFB1, was prepared by following method by Topçu et al.(2010).
L. plantarum and AFB1 co-incubation
L. plantarum was centrifuged 10000 rpm for 5 minutes and supernatant discarded. Pellet was washed twice with Ca++ and Mg++ free phosphate buffered saline (PBS). Pellet was solved in AFB1 containing PBS and 5 mL of suspension placed in 6-well-plates and agitated 37°C for 6 hours on orbital microplate shaker. Negative control (PBS) and AFB1 without L. plantarum placed the 6-well-plate either. L. plantarum viability and total number was assigned at both 0. and 6th hours.
Comet assay (Single cell gel electrophoresis)
Comet assay was performed as the Comet Assay datasheet manual with minor modifications (Enzo Life Sciences, New York, ABD). Cells were seeded on the 6-well-plate two days before the test. After 18 hours of seeding, cells were exposed to genotoxins for 16 hours. Test was conducted with positive and negative controls. For this purpose, 50 μM H2O2 was used as positive; PBS was used as negative control. Positive control was prepared just before the assay on the ice.
After genotoxin exposure, medium in the wells was discarded using sterile single-use Pasteur pipette, and washed with ice cold PBS to remove medium and metabolite residues and trypsinized the cells at 37°C for 5-7 minutes. Then cells were dissolved in PBS and cell viability was checked. PBS+cell suspension was used for the Comet assay.
Cells were counted on the Neubauer hemocytometer; cell suspension was added in the low melting agarose (LMA), mixed well and transferred onto the comet slide. Slides were placed on a flat surface in the refrigerator. Agarose was hardened and slides were placed into the lysis solution. Slides were immersed cold lysis solution at least 2 hours. In the end of lysis, slides were washed with deionized water and put in the freshly prepared alkali solution .This step let cleave the double bonds in the alkali labile sites of DNA molecule. Slides were placed onto the electrophoresis tank and power supply was set 1 V/cm and current 300 mA for 40 minutes. Electrophoresis was conducted in alkali (pH>13) conditions. CyGreen nucleic acid dye use for imaging comets. x10000 stock dye solution diluted x10 concentration and slides were dyed. Comets were screened using x10 objectives with FITC filter equipped Nikon C2 confocal laser scanning microscope.
Results
L. plantarum-AFB1 Co-incubation
Lactic acid bacteria+genotoxin co-incubation did not reduce the viable lactic acid bacteria cell count.
Caco-2 cell viability
Comet test was started with same cell count (105 cells). AFB1 and negative control cells showed higher viability (77.5% and 81%, respectively) than AFB1+LAB1 and positive control cells. This means current doses of AFB1 did not lead any cytotoxic effect. However, when AFB1 was together with L. plantarum cell viability was reduced (64%). This means lactic acid bacteria have cytotoxic effect on colon adenocarcinoma cells.
L. plantarum+AFB1 application to the Caco-2 cells caused to diminish Caco-2 cell size and viability (Data not shown). This result indicates cytotoxic effect of L. plantarum on Caco-2 cells. Similar results were taken by Er et al. (2015). They found that L. plantarum’s cell-free filtrate, after 24 hour incubation with Caco-2 cells, showed the highest (50% inhibition) cytotoxic effect.
Comet Assay
Comet assay results were scored visually. Images was categorized as; No-migration, low-migration, high-migration (Kadioglu et al. 2009). Samples were divided into 4 categories; negative and positive controls, AFB1 and AFB1+LAB applied groups. While no tail was observed in the negative controls and AFB1+LAB group; positive and AFB1 group was showed high migration in the tails.
Tail appearance in the alkaline comet assay indicates single stranded DNA breaks, double-stranded DNA breaks and majority of apurinic, apyrimidinic sites as well as alkali labile sites in the DNA molecule. Tail length is related with the damage quantity. Lactobacillus plantarum is able to reduce/bind AFB1 13-54% according to our HPLC study. Reducing amount by Lactobacillus plantarum is based on incubation period/time. The most efficient binding rate was found at 6th hour. LAB+AFB1 complex is a reversible formation. After 6 hours, LAB+AFB1 complex started to break down, and binding rate reduced. In spite of having uncertainty in the literature about by-products of AFB1 that was eliminated with lactic acid bacteria, our results showed that Lactobacillus plantarum is able to reduce to AFB1 without any toxic by-products.
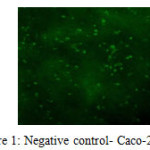 |
Figure 1: Negative control- Caco-2 cells Click here to View figure |
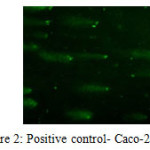 |
Figure 2: Positive control- Caco-2 cells Click here to View figure |
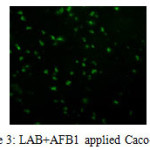 |
Figure 3: LAB+AFB1 applied Caco-2 cells Click here to View figure |
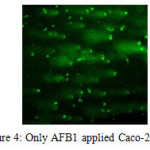 |
Figure 4: Only AFB1 applied Caco-2 cells Click here to View figure |
Discussion
While Lactobacillus rhamnosus and L. paracasei are abundant in dairy products, L. plantarum generally is responsible for plant originated fermentation. L. plantarum play a key role especially olive and sauerkraut fermentations (de Vries et. al. 2006). Thus, human consume fermented plant products also consume substantial amount of L. plantarum (Molin 2001). To enhancement of diet with fermented food is easy and healthy way for protecting genotoxins.
Acknowledgements
This project was funded by Abant Izzet Baysal University, Scientific Research Projects Coordination Unit (Project number: 2015.09.04.843). We would like to thank Abant Izzet Baysal University, Novel Food Technologies Development, Application and Research Center (YENIGIDAM)
References
- Caldini, G., Trotta, F., Villarini, M, Moretti, M., Pasquini, R., Scassellati Sforzolini, G. Cenci,G. Screening of Potential Lactobacilli Antigenotoxicity by Microbial and Mammalian Cell-Based Tests. International Journal of Food Microbiology; 102: 37-47: 2005.
CrossRef - Caldini, G., Trotta, F., Corsetti, A., Cenci, G. Evidence for In Vitro Anti-genotoxicity of Cheese Non-starter Lactobacilli. Antonie van Leuwenhoek; 93: 51-59: 2008
CrossRef - Cenci, G., Caldini, G. and Trotta, F. Inhibition of DNA Reactive Agents by Probiotic Bacteria. Recent Research Developments in Applied Microbiology & Biotechnology; 2: 103-121: 2005.
- Ciegler, A., Lillehoj, E.B., Peterson, R.E., Hall, H.H. Microbial Detoxification of Aflatoxin. Applied Microbiology; 14 (6): 934-939: 1966.
- de Vries, M.C., Vaughan, E.E., Kleerebezem, M., de Vos, W.M. Lactobacillus plantarum– Survival, Functional and Potential Probiotic Properties in the Human Intestinal Tract. International Dairy Journal; 16: 1018-1028: 2006.
CrossRef - El-Nezami, H, Kankaanpaa, P, Salminen, S, Ahokas, J. Ability of Dairy Strains of Lactic Acid Bacteria to Bind a Common Food Carcinogen, Afatoxin B1. Food and Chemical Toxicology; 36: 321-326: 1998.
CrossRef - Er, S., Koparal, A.T. and Kıvanç, M. Cytotoxic Effects of Various Lactic Acid Bacteria on Caco-2 Cells. Turkish Journal of Biology; 39: 23-30: 2015.
CrossRef - Fuchs, S., Sontag, G., Stidl, R., Ehrlich, V., Kundi, M., Knasmüller, S. Detoxification of Patulin and Ochratoxin A, Two Abundant Mycotoxins, by Lactic Acid Bacteria. Food and Chemical Toxicology; 46: 1398-1407: 2008.
CrossRef - Guidelines for the evaluation of probiotics in food, London Ontario, Canada. April 30 and May 1, 2002.
- Kadıoğlu, E., Sardas, S., Ertürk, S., Ozatamer, O., Karakaya, A.E. Determination of DNA Damage by Alkaline Halo and Comet Assay in Patients Under Sevoflurane Anesthesia. Toxicology and Industrial Health; 25: 205-212: 2009.
CrossRef - Molin, G. Probiotics in Foods not Containing Milk or Milk Constituents, with Special Reference to Lactobacillus plantarum. The American Journal of Clinical Nutrition ; 73 (suppl):380S–5S, 2001.
- Peltonen, K., El-Nezami, H., Haskard, C., Ahokas, J., Salminen, S. AflatoxinB1 Binding by Dairy Strains of Lactic Acid Bacteria and Bifidobacteria. Journalof Dairy Science.; 84: 1256–2152: 2001.
CrossRef - Topçu, A., Bulat, T., Wishah, R., Boyacı, IH. Detoxification of Aflatoxin B1 and Patulin by Enterococcus faecium Strains. International Journal of Food Microbiology; 139: 202-205: 2010.
CrossRef - Zhong, L., Zhang, X., and Covasa, M. Emerging Roles of Lactic Acid Bacteria in Protection Against Colorectal Cancer. World Journal of. Gastroenterology; 20 (204): 7878-7886: 2014.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





