Introduction
Hippophae rhamnoides, known as sea buckthorn (SB), belongs to the Elaeagnaceae family. It appears as a large shrub in parts of Eurasia. It is one of the oldest land plants, dating from the Ice Age with beneficial effects appearing since the 4th century BC. According to historical sources, SB was part of Alexander the Great’s army diet. The legend revealed that leaves and fruits of this plant treated both patients and injured horses. References to its usage are also found in both Tibetan tradition and Chinese medicine. Other sources report that during the 13th century, Jenkins Khan had used it in his campaigns. Finally, in 1929 the first biochemical analysis of SB fruits took place.1 Since then, our knowledge of its health properties has increased.
Sea buckthorn contains vitamins A and C, alpha-tocopherol, large amounts of carotenoids, minerals (K, Na, Mg, Ca, Fe, Zn, Se), monosaccharides, amino acids, flavonoids, fatty acids, glycerol phospholipids, phytosterols, zeaxanthin esters, polyphenolic compounds (see Table 1), etc. Its composition varies according to origin, climate, and the extraction procedure used. The fruit in SB provides about 400 mg vitamin C/100 gr.1
Table 1: Hippophae rhamnoides L. constituents 1, 36, 37
| Category | Constituent examples |
| Triterpenoids | Oleanolic acid, 3-o-trans-p-coumaryl-oleanolic acid, 3-o-cafeenyl-oleanolic acid, 2-o-trans-p-coumaryl-maslinic acid, 2-o-caffenyl-maslinic acid, ursolic acid, 19-hydroxy-methyl-ursolic acid |
| Vitamins | C, A, E, zeaxanthin esters, b-carotene, a-tocopherol, folate |
| Minerals | Ca, Mg, K, Se, Na, Fe, Zn, P, Co, Cr, Cu, Mn, Ni, Sr, Va, Mb, Al, Li, Cd, As |
| Heavy metals | Cd, Pb, Hg |
| EFA | Palmitoleic acid, oleic acid, palmitic acid, linoleic acid, a-linolenic acid |
| Flavonoids | Querqetin, kaempferol, isohamnetin, cathechin, rutin, myricetin |
| Tannins | Hippophaenin A, hippophaenin B |
| Lipopolysaccharide | Octacosananoic acid |
| Phenols | Ellagic acid, ferulic acid |
| Monosaccharides | Xylose |
| Volatile esters | Ethyl-dodecanoate, ethyl-octanoate, ethyldecanoate |
| Glucosides | 1-0-hexadecanolenic acid |
| Sterols | b-sitosterol, stigmastonol, campesterol, stigmastadievol |
| Aldehydes | 1-decanol, circiumaldehyde, 5-hydroxy-methyl-2-furancarboxaldehyde |
| Polyalcohols | Mannitol, sorbitol, xylitol |
| Glycosphingolipids | Hippophae cerebroside |
| Free aminoacids | Aspartic acid, praline, threonine, serine, lysine, valine, alanine, phenylalanine, glutamine, isoleucine, glycine, histidine, tyrosine, arginine, cysteine, methionine |
Various studies have linked the functionality of diet lipids with the prevention and treatment of many diseases. These functional lipids are the omega-3 and omega-6 fatty acids, conjugated linoleic acids, medium chain triglycerides, and phytosterols have many beneficial effects on human chronic diseases such as in obesity, bone health, and in treating and managing depression, blood pressure, cardiovascular health, etc. Sea buckthorn seeds contain most of the nutrients and phytochemicals, including high amounts of unsaturated fatty acids. The oil from the pulp/peel fraction is combined, due to difficulty in separation. Both seeds and berry pulp have high total lipid content, including tocopherols, tocotrienols, carotenoids, sterols, as well as omega-3 and omega-6 fatty acids.2
The seeds are source of highly unsaturated oil, containing saponifiable and unsaponifiable matter. Phytosterols are the major constituents of the unsaponifiable fraction of sea buckthorn, which are capable of lowering plasma cholesterol upon consumption by humans. Of them, β-sitosterol is present in both seed and seed oil, and constitutes 57-76% and 61-83%, of the seed and pulp/peel sterols, respectively.3,4 Phytosterols are reported to improve microcirculation in the skin, to regulate the inflammatory process, whereas exhibit anti-atherogenic, anti-ulcerative and anti-cancer actions.5
The seed oil contains over 95% of the recoverable tocopherols which are present at a high concentration (140 mg/100 ml), 1% phytosterols and small amounts of tocotrienols.6 On the other hand, the major fatty acids in the seed oil are linoleic (34%), alpha-linolenic (25%), and oleic (19%) acids, whereas the major fatty acids in the pulp oil are palmitic (33%), oleic (26%), and palmitoleic (25%) acids.7,8 Palmitoleic acid (16:1n-7) is also a component of skin fat and is known to support cell tissue and wound healing. Sea buckthorn oil provides naturally a 1:1 ratio of n-3:n-6 fatty acids (linolenic and linoleic acid respectively).9 Stereochemical types of the major fatty acids of SB oil are depicted in Figure 1.
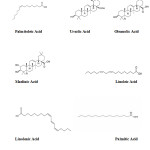 |
Figure 1: Stereochemical types of major Sea buckthorn oil fatty acidsClick here to View figure |
Furthermore, due to its constituents, the oil from fruits and seeds has been tested in various pathological conditions6 such as liver diseases, inflammation, disorders of the gastrointestinal system, eczema, wounds, inflammation, burns, lupus erythematosus, and chronic dermatoses. Oil from seed or pulp absorbs ultraviolet light and promote healthy skin.9 Thus we reviewed the studies, either on humans or animals, concerning the effects of sea buckthorn oil on various pathological conditions.
Cardiovascular health
Several animal studies have shown beneficial effects on cardiovascular health produced by sea buckthorn oil. Basu et al.,10 fed 20 male albino rabbits and divided them into four groups: group I – control, group II – SB seed oil, group III – cholesterol 1% for 60 days, group IV – cholesterol plus SB seed oil. Group IV received 1 ml of SB seed oil/day for 30 days, following 30 days of high cholesterol diet. They also studied in vitro the vasorelaxant effect of SB seed oil with aortic ring model technique, and the accumulation of cholesterol in the aorta with Sudan-IV staining technique. Results showed that SB oil caused significant decrement in plasma cholesterol, LDL-C, atherogenic index and LDL/HDL ratio; whereas HDL-C, HDL-C/total cholesterol ratio, and the vasorelaxant activity of the aorta were significantly increased. Oil consumption following a high fat diet resulted in decline in total cholesterol, triacylglycerides, LDL, and atherogenic index.
In the same context, Malik et al.,11 reported that SB oil could curtail experimental induced cardiac dysfunction in rats. The animals received different doses of SB oil (5, 10, and 20 ml/kg) daily for 30 days. On 29th and 30th day, both control and intervention group were given additionally 85 mg/kg isoproterenol, which produces cardiac dysfunction, lipid peroxidation, and reduction in antioxidant activities. It was shown that SB oil at the dose of 20 ml/kg/day attenuates histological damage caused by isoproterenol; its protective effect was mediated through free radical scavenging and antioxidant activities.
In a small scale cross-over study, Johansson et al.,7 recruited 12 healthy normolipidemic men to consume 5 g/day SB oil and coconut oil (placebo) for 4 weeks, with a wash-out period of 4-8 weeks. SB oil did not affect phospholipid fatty acids, plasma lipids and glucose, but it did significantly reduce adenosine-5-diphosphate induced platelet aggregation, and maximum aggregation. In other words, SB oil affects blood clotting chain reactions and thus could also have practical implications.
Moreover, Lehtonen et al.,12 compared the effects of SB and bilberries (BBs) on metabolic disease variables. Eighty female, overweight or obese, subjects (that concluded the study) received randomly 4 different berry diets (BB, SB, SB phenolic extract, SB oil) for 33-35 days; in between diets a 30-39 wash-out period was involved. After BB (Δ, -1.2 cm; P = 0.041) and SB (Δ, -1.1 cm; P = 0.008) consumption, a statistically significant decline in waist circumference was measured. Vascular cell adhesion molecule decreased after BB (Δ, -49.8 ng/ml; P = 0.002) and SB oil (Δ, -66.1 ng/ml; P = 0.001) periods, whereas following SB phenolic extract diet intercellular adhesion molecule (ICAM) declined (Δ, -6.1 ng/ml; P = 0.028). These results reveal that SB fractions are beneficial on various variables of cardiovascular and metabolic disease.
As studying the pathophysiology of stroke, with presence of cerebral edema caused by vascular leakage, Purushothaman et al.,13 investigated the role of SB oil supplementation in preventing tissue hypoxia. The latter is thought to be a major induction factor of vascular leakage in the brain. Rats exposed to hypobaric hypoxia (9144 m, 5h) suffered a subsequent increase in the transvascular leakage, measured by water content and leakage of sodium fluoresce in dye in the brain. Hypoxic stress in turn, promoted free radicals and malondialdehyde production, whereas decreased levels of antioxidants (glutathione, glutathione peroxidase, and superoxide dismutase) are observed. When animals were pretreated with SB seed oil, tissue hypoxia was significantly attenuated, as well as free radicals and malondialdehyde production. SB seed oil increased animals’ hypoxic tolerance, as measured by increased hypoxic gasping time and survival time, and curtailed catecholamine plasma levels, compared to control. Thus, it was evident that SB oil can lessen hypoxia-induced vascular leakage in the brain, and attenuate hypoxic stress injury.
Insulin and glucose response
Given that berries, and similar antioxidant containing fruits and vegetables, are thought to implicate positively to insulin sensitivity and glucose response,14 sea buckthorn’s role in postprandial glucose metabolism was also studied. Lehtonen et al15 recruited 10 healthy normal-weight male subjects to consume four different breakfasts on four distinct study days: meal A (control), meal B1 (dried and crushed whole berries), meal B2 (supercritical fluid-carbon dioxide SF-CO2 extracted oil free berries), and meal B3 (ethanol extracted SF-CO2 residue). All meals contained yoghurt and 50g glucose. Results showed that meal B1 attenuated postprandial peak insulin response compared to control (p=0.039), and established postprandial hyperglycemia and subsequent hypoglycemia (p=0.036). Meal B2 resulted in more stable insulin response compared to control (p=0.037). Thus, we conclude that the oily part of berries is not effective in glucose metabolism modulation. On the contrary, the EtOH soluble components containing the majority of antioxidants are beneficial in both insulin and glucose responses.
Moreover, in Table 2 a comparison between SB components, dosage, duration and effect is outlined.
 |
Table 2: Comparison between SB components, dosage, duration and effect Click here to View table |
Renal health
Rodhe et al.,16 conducted a randomized, double blinded, placebo-controlled crossover trial to evaluate effects of SB oil extract – rich in vitamins, phytochemicals, polyunsaturated fatty acids – on oxidative stress, saliva production, and inflammation in hemodialysis patients. The 45 patients participated received either 500mg SB oil or placebo, for 8 weeks. Before crossing over to the other treatment, a 4-weeks wash-out period was involved. Following SB oil supplementation, no significant changes in DNA breaks, oxidative DNA lesions, salivary flow rates, of inflammation were measured. Only plasma levels of sodium and phosphate increased, whilst plasma iron levels decreased. In all, SB oil did not protect hemodialysis patients from oxidative stress of inflammation.
Hepatic health
Several studies in humans or animals imply that sea buckthorn oil exerts hepatoprotective activities. Hsu et al.,17 studied the protective effects of SB seed oil on carbon tetrachloride (CCl(4))-induced hepatic damage in male ICR mice. Oral administration of SB seed oil at doses of 0.26, 1.30, and 2.60 mg/kg for 8 weeks, significantly reduced the CCl(4)-induced elevated levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), triglyceride (TG), and cholesterol at least 13% in serum, and the level of malondialdehyde (MDA) in liver at least 22%. In addition, SB seed oil significantly increased the activities of hepatic enzymes, such as superoxide dismutase (SOD), catalase, glutathione peroxidase (GSH-Px), glutathione reductase (GSH-Rd), as well as GSH hepatic content, up to 134%. These findings have been also confirmed by liver histopathology evaluation. Likewise, Liu et al.,18 compared SB seed oil and sarcocarp oil in rats with CCl4-induced hepatocirrhosis. For 45 days, rats were fed with different concentration of seed oil and sarcocarp oil. As it was shown, both oils modulate serum ALT increase and liver SOD decline, whilst seed oil was found to be more effective than sarcocarp one.
In a clinical trial, Gao et al.,19 recruited 45 cirrhotic patients to receive either 15 g SB extract (group A, n=30) or vitamin-B complex (group B, n=15) 3 times a day for 6 months. Following SB extract treatment serum levels of laminin, hyaluronic acid, collagen types III and IV, and total bile acid (TBA) decreased significantly compared to control group, both before and after treatment. In addition serum levels of TNFa, IL-6, laminin and type IV collagen in treatment group were significantly higher than those in the control group, whereas SB reduced time needed for aminotransferases normalization. This hepatoprotective activity was attributed to retinol contained in SB extract.
Gastric health
There is also an animal study that suggests SB oil possesses gastroprotective properties. Xing J et al.,20 administrated SB seed and pulp oil to rats with reserpine-induced gastric injury. Oral administration of 7 ml/kg/day SB seed or pulp oil resulted in decline of ulcer formation in water-immersion (P < 0.05) and reserpine-induced (P < 0.01) injury. Combination of the two oils (at a dose of 3,5ml/kg/day each) significantly reduced the index of pylorus ligation-induced gastric ulcer (P < 0.05) and accelerated the healing process of acetic acid-induced gastric ulcer (P < 0.01); indicating important gastrointestinal prophylactic properties of SB oils.
Epidermis health
Sea buckthorn oil is thought to be beneficial when applied on skin burns or traumas. In an animal study21, SB oil was administrated both per os, at a dose of 2.5ml/kg, and topical (200 microl) for 7 days on burn wounds in rats. SB oil promoted wound healing, as hydroxyproline, hesosamine, DNA and total protein contents were significantly increased compared to control treated with sulfadiazine ointment. This effect was also confirmed by histopathological findings. In addition, gene expression of collagen type-III and matrix metalloproteinases (MMP-2 and 9) was up-regulated, in granulation tissue. It was also observed that SB oil exhibits antioxidant activities, as it increased glutathione levels and reduced reactive oxygen species (ROS) in the wound granulation tissue. No adverse effects or toxicity signs were observed in any group receiving SB oil treatment.
In 1994, Zhao Y22 reported that topical application of SB seed oil on 32 burn cases resulted in tissue regeneration. This effect was attributed to palmitoleic acid of SB oil, which is also a component of the skin. Its emollient properties are thought to promote tissue rebuilt. These observations are in accordance with the outcome of a 2006 study conducted by Wang ZY et al.,23 SB oil dressing was applied on burn wounds of 151 patients, and attenuated the swelling and effusion of wounds, as well as relieving the pain. Patients receiving the oil dressing exhibited significantly faster epithelial cell growth and wound healing, more obvious exudation decline and pain relief, compared to control group treated with Vaseline gauze.
Nevertheless, studies concerning skin diseases, such as atopic dermatitis or eczema, are inconclusive. In a systematic review24 of randomized controlled trials, 11 studies with a total of 596 participants were included, concerning supplements such as oral zinc, selenium, vitamins D, B6, and E, SB seed or pulp oil, hempseed oil, sunflower oil, fish oil or DHA, for atopic eczema treatment. None of them had such statistically significant results so as to be recommended for public of clinical practice.
In a placebo-controlled, double blind trial Yang et al.,8 studied the effects of SB oil from both seed and pulp, on atopic dermatitis. Seed oil is rich in linoleic (34%), alpha-linolenic (25%), and oleic (19%) acids, whereas pulp oil contains great amounts of palmitic (33%), oleic (26%), and palmitoleic (25%) acids. 49 atopic dermatitis patients received 5g of seed oil, pulp oil, or paraffin oil (placebo) daily for 4 months. Results revealed that dermatitis improved significantly with pulp oil (p<0.01) and paraffin oil (p<0.001) treatment, whereas seed oil treatment was not effective (p=0.11). It seems that pulp oil increased the proportion of palmitoleic acid (p<0.05) and decreased the one of pentadecanoic acid (p<0.01) in both plasma phospholipids and neutral lipids. After receiving seed oil for 1 month, patients experienced symptoms improvement, correlated as proved with the increase in alpha-linolenic percentage in plasma phospholipids (Rs=0,84; p=0.001) and neutral lipids (Rs=0.68; p=0.02). A year later, the same research group25 reported that seed oil produced both a small increase in docosapentanoic acid and a decrement in palmitic acids proportions in skin glycerophospholids (0,05<p<0,1), whereas the other fatty acids remained invariant. Something that implies skin glycerophospholipids are not sensitive to dietary modification.
Likewise, in a prospective, randomized, double blind, placebo controlled trial26 53 atopic dermatitis patients were divided into 3 groups: a 20% SB-containing cream (group 1), a 10% SB-containing cream (group 2), or a miglyol cream (placebo, group 3). All patients applied their treatment on skin for 4 weeks. All groups experienced skin condition improvement, but none of the treatments had statically significant results compared to control group.
Cancer
Data concerning the role of sea buckthorn in prevention and control of cancer are inadequate, though there are several animal trials reporting beneficial effects. Cancer cell growth was inhibited by SB oil, but not as effectively as medicine (such as phosphamide).27 In addition, in mice fed with SB oil, a faster recovery rate of their hemopoietic system was observed, following high chemotherapy dose. SB oil was thought to promote non-specific immunity and to exert anti-tumor effects.28 In a similar model of myelosuppression built by 5-Fu intra-peritoneal infusion,29 mice fed with SB oil had elevated counts of erythrocytes compared to control group. Treatment group had also decreased mortality. Thus, SB oil is considered to stimulate the recovery of hematopoiesis after chemotherapy.Moreover, Mingyu et al.,30 report that both intraperitoneal injection and oral administration of SB oil, suspended tumor development in mice.
Ophthalmologic health
Sea buckthorn oil content in (n-3) and (n-6) fatty acids, and antioxidants, is thought to be beneficial in conditions, such as dry eye. In a double-blind, randomized, parallel trial,31 86 women and men with dry eye symptoms received 2 g/day of SB oil or placebo for 3 mo from fall to winter. Participants in SB oil group had significantly less increase in tear film osmolarity, compared to control group. Noted that tear film hyperosmolarity is a focal factor in dry eye. SB oil also attenuated maximum values of burning and redness symptoms, during the cold season. Due to the fact that dry eye is known to be affected positively by linoleic, γ-linolenic, and n-3 fatty acids intake, Järvinen et al.,32 studied whether SB oil supplementation affects the composition of tear film fatty acids in dry eye patients. Evaporative dry eye is associated with meibomian gland dysfunction and abnormalities of the tear film lipids. Patients received either 2g SB oil/day or placebo for 3 months. Results revealed that no changes in fatty proportions occurred, between the two groups. This implies that SB oil effect is not mediated through tear film fatty acids changes. However, carotenoids, tocopherols in SB oil, or eicosanoids produced from oil fatty acids may affect positively inflammation and differentiation of meibomian gland cells.
Oxidative stress and toxicity
Sea buckthorn seed oil was investigated for its antioxidant properties with several in vitro and in vivo trials, in terms of an animal model.33 Mice were fed with various concentrations of SB seed oil, and suffered carbon tetrachloride (CCl4)-induced oxidative stress. As a result, DPPH radical scavenging activity, ferrous ion chelating activity, reducing power and inhibition of lipid peroxidation activity all increased with increasing concentrations of SB seed oil. In vivo, SB seed oil significantly suppressed CCl4-toxicity, as evidenced from aggregated antioxidant enzymes activity (superoxide dismutase, catalase, glutathione peroxidase and glutathione reductase). Liver glutathione levels were increased, whereas hepatic malondialdehyde was reduced. These outcomes are in line with those of hypoxic stress animal model by Purushothaman et al.,13 concerning the possible antioxidant role of SB oil.
Ruan et al.,34 studied in vivo the injury caused by inhalation of the common air pollutant sulfur dioxide on organs and germ plasm of mouse, and the role of SB seed oil supplementation in it. Micronuclei in the polychromatic erythrocytes (PCE) of mouse bone marrow and the ratio between organ and body weight of treatment mouse were determined. It was evident that SB seed oil attenuated the increase of micronuclei in polychromatic erythrocytes and the change organ/body of mouse organs ratio.
Conclusions
Only a few clinical trials testing applications and efficiency of SB oil are found in scientific literature. Amongst them, it is evident that SB oil supplementation can inhibit platelet aggregation7 and decrease vascular adhesion molecules (VCAMs),12 exhibiting cardioprotective properties. It also appears to enhance the activity of antioxidant hepatic enzymes under cirrhotic conditions, as well as attenuate disease markers as laminin, hyaluronic acid, total bile acid, and collagen III and IV levels19; suggesting a possible beneficial role.
Moreover, studies on patients with atopic dermatitis25,26 or burn traumas22,23 revealed that SB oil has advantageous properties in reducing symptoms intensity, and promoting tissue regeneration and wound healing. Possibly to due to its palmitoleic content, that is also naturally present in the skin. Because of its ability to absorb UV radiation,9,35 SB oil could be used in sun blocking lotions.
Interesting though might be the role of SB oil as relief agent in dry eye condition31,32; it was shown that it attenuated inflammation and tear film osmolarity. Retinol in SB oil might be responsible for such actions.
In all, SB oil could have several applications in everyday life, given that further clinical studies are conducted to prove, beyond any doubt, its beneficial role in several health conditions.
References
- Bilaloglu Guliyeva V, Gulb M, Yildirima A. Hippophae rhamnoides L: chromatographic methods to determine chemical composition, use in traditional medicine and pharmacological effects. Journal of Chromatography B. 2004;812:291-397.
CrossRef - Yang B, Kallio H. Composition and physiological effects of sea buckthorn (Hippophaė) lipids. Trends in Food Science and Technology. 2002;13(5):160-167.
CrossRef - Dulf F. Fatty acids in berry lipids of six buckthorn (Hippophae rhamnoeides L, subspecies carpatica) cultivars grown in Romania. Chem Central J. 2012;6:106-117.
CrossRef - Yang B, Karlsson R, Oksman P, Kallio H. Phytosterols in sea buckthorn (Hippophaë rhamnoides L.) berries: identification and effects of different origins and harvesting times. J Agric Food Chem. 2001;49(11):5620-5629.
CrossRef - Yang B, Kallio H. Fatty acid composition of lipids in sea buckthorn (Hippophaë rhamnoides L.) berries of different origins. J Agric Food Chem. 2001;49(4):1939-1947.
CrossRef - Bal L, Meda V, Naik S, Satya S. Sea buckthorn berries: A potential source of valuable nutrients for nutraceuticals and cosmoceuticals. Food Research Int. 2011;44:1718-1727.
CrossRef - Johansson A, Korte H, Yang B, Stanley J, Kallio H. Sea buckthorn berry oil inhibits platelet aggregation. J Nutr Biochem. 2000;11(10):491-495.
CrossRef - Yang B, Kalimo K, Mattila L, et al. Effect of dietary supplementation with sea buckthorn (Hippophae rhamnoides) seed and pulp oils on atomic dermatitis. J Nutr Biochem. 1999;10(622-30).
CrossRef - Geetha S, Asheesh G. Medicinal and therapeutic potential of Sea buckthorn (Hippophae rhamnoides L). J Ethnopharmacol. 2011;138:268-278.
CrossRef - Basu M, Prasad R, Jayamurthy P, Pal K, Arumughan C, Sawhney R. Anti-atherogenic effects of seabuckthorn (Hippophaea rhamnoides) seed oil. Phytomedicine. 2007;14(11):770-777.
CrossRef - Malik S, Goyal S, Ojha S, et al. Seabuckthorn attenuates cardiac dysfunction and oxidative stress in isoproterenol-induced cardiotoxicity in rats. Int J Toxicol. 2011;30(6):671-680.
CrossRef - Lehtonen H, Suomela J, Tahvonen R, et al. Different berries and berry fractions have various but slightly positive effects on the associated variables of metabolic diseases on overweight and obese women. Eur J Clin Nutr. 2011;65(3):394-401.
CrossRef - Purushothaman J, Suryakumar G, Shukla D, et al. Modulatory effects of seabuckthorn (Hippophae rhamnoides L.) in hypobaric hypoxia induced cerebral vascular injury. Brain Res Bull. 2008;77(5):246-252.
CrossRef - Avignon A, Hokayem M, Bisbal C, Lambert K. Dietary antioxidants: Do they have a role to play in the ongoing fight against abnormal glucose metabolism? Nutrition. 2012;28(7-8):715-721.
CrossRef - Lehtonen H, Järvinen R, Linderborg K, et al. Postprandial hyperglycemia and insulin response are affected by sea buckthorn (Hippophaë rhamnoides ssp. turkestanica) berry and its ethanol-soluble metabolites. Eur J Clin Nutr. 2010;64(12):1465-1471.
CrossRef - Rodhe Y, Woodhill T, Thorman R, Möller L, Hylander B. The Effect of Sea Buckthorn Supplement on Oral Health, Inflammation, and DNA Damage in Hemodialysis Patients: A Double-Blinded, Randomized Crossover Study. J Ren Nutr. 2012;173-2(12):S1051-2276.
- Hsu Y, Tsai C, Chen W, Lu F. Protective effects of seabuckthorn (Hippophae rhamnoides L.) seed oil against carbon tetrachloride-induced hepatotoxicity in mice. Food Chem Toxicol. 2009;47(9):2281-2288.
CrossRef - Liu C, Xu J, Ye C, Huang C. [Effects and comparison of seed oil and sarcocarp oil of Hippophae rhamnoides on rats with experimental hepatocirrhosis]. Zhongguo Zhong Yao Za Zhi. 2006;31(13):1100-1102.
- Gao Z, Gu X, Cheng F, Jiang F. Effect of Sea buckthorn on liver fibrosis: A clinical study. World J. Gastroenterol. 2003;9(7):1615-1617.
CrossRef - Xing J, Yang B, Dong Y, Wang B, Wang J, Kallio H. Effects of sea buckthorn (Hippophae rhamnoides L.) seed and pulp oils on experimental models of gastric ulcer in rats. Fitoterapia. 2002;73:644-650.
CrossRef - Upadhyay N, Kumar R, Mandotra S, et al. Safety and healing efficacy of Sea buckthorn (Hippophae rhamnoides L.) seed oil on burn wounds in rats. Food Chem Toxicol. 2009;47(6):1146-1153.
CrossRef - Zhao Y. Clinical effects of Hippophae seed oil in the treatment of 32 burn cases. Hippophae. 1994;7:36-37.
- Wang Z, Luo X, He C. [Management of burn wounds with Hippophae rhamnoides oil]. Nan Fang Yi Ke Da Xue Xue Bao. 2006;26(1):124-125.
- Bath-Hextall F, Jenkinson C, Humphreys R, Williams H. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012;15(2):CD0052005.
CrossRef - Yang B, Kalimo K, Tahvonen R, Mattila L, Katajisto J, Kallio H. Effect of dietary supplementation with sea buckthorn (Hippophaë rhamnoides) seed and pulp oils on the fatty acid composition of skin glycerophospholipids of patients with atopic dermatitis. J Nutr Biochem. 2000;11(6):338-340.
CrossRef - Thumm E, Stoss M, Bayerl C, Schurholz T. Randomized trial to study efficacy of a 20% and 10% Hippophae rhamnoides containing creme used by patients with mild to intermediate atopic dermatitis. Aktuelle Dermatologie. 2000;26(8-9):285-290.
- Nersesian A, Zilfian V, Kumkumadzian V, Proshian N. Antimutagenic properties of sea buckthorn oil. Genetika. 1990;26:378-380.
- Yu Lea. Effects of Hippophae rhamnoides juice on immunologic and antitumor functions. Acta Nutrimenta Sinica. 1993;15(3):280-283.
- Chen Yea. Study on the effects of the oil from Hippophae rhamnoides in hematopoiesis. Chinese Herbal Drugs. 2003;26(8):572-575.
- Mingyu X. Anticancer effects of and direction of research on Hippophae. Hippophae. 1994;7(4):41-43.
- Larmo P, Järvinen R, Setälä N, et al. Oral sea buckthorn oil attenuates tear film osmolarity and symptoms in individuals with dry eye. J Nutr. 2010;140(8):1462-1468.
CrossRef - Järvinen R, Larmo P, Setälä N, et al. Effects of oral sea buckthorn oil on tear film Fatty acids in individuals with dry eye. Cornea. 2011;30(9):1013-1019.
CrossRef - Tinga H, Hsub Y, Tsaic C, Lua F, Choua M, Chend W. The in vitro and in vivo antioxidant properties of seabuckthorn (Hippophae rhamnoides L.) seed oil. Food Chemistry. 2011;125(2):652-659.
CrossRef - Ruan A, Min H, Meng Z, Lü Z. Protective effects of seabuckthorn seed oil on mouse injury induced by sulfur dioxide inhalation. Inhal Toxicol. 2003;15(10):1053-1058.
CrossRef - Beveridge T, Li T, Oomah B, Smith A. Sea buckthorn products: manufacture and composition. J Agric Food Chem. 1999;47(9):3480-3488.
CrossRef - Cakir A. Essential oil and fatty acid composition of the fruits of Hippophae rhamnoides L. (Sea Buckthorn) and Myrtus communis L. from Turkey. Biochemical Systematics and Ecology. 2004;32(9):809-816.
CrossRef - Zheng RX, Xu XD, Tian Z, Yang JS. Chemical constituents from the fruits of Hippophae rhamnoides. Natural Product Research. 2009;23(15):1451-1456.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





