Alteration of Bioactive Compounds and Antioxidative Properties in Thermal, Ultra-High Pressure and Ultrasound Treated Maoberry (Antidesma Bunius L.) Juice during Refrigerated Storage
1Institute of Nutrition, Mahidol University, Nakorn Pathom Campus, Nakorn Pathom, Thailand.
2Faculty of Science and Technology, Phranakhon Si Ayutthaya Rajabhat University, Phranakhon Si Ayutthay, Thailand.
Corresponding Author Email: pittaya.chaikham@gmail.com
DOI : http://dx.doi.org/10.12944/CRNFSJ.9.3.17
ABSTRACT:Maoberry (Antidesma bunius L.) is a tropical fruit locally referred to as “Mao-Luang” in Thailand. The fruit contains high amounts of ascorbic acid and phenolic compounds with antioxidative potential, which has demonstrated medicinal value in terms of anti-cancer and anti-diabetic effects. In this term, this research purposed to determine the changes of predominant bioactive phytochemicals, antioxidant capacity and microbiological quality of pasteurized (85ºC/1 min), pressurized (500 and 600 MPa/30ºC/30 min), and ultra-sonicated (20 kHz/60% and 80% amplitude/30 min) maoberry juices during storage at 4°C for 30 days. The results displayed that ascorbic acid, phenolic acids (gallic and vanillic acids), anthocyanins (cyanidin 3-o-glucoside and cyanidin 3-rutinoside), flavonoids [(+)-catechin and (˗)-epicatechin), 2,2-diphenyl-1-picryl hydrazyl hydrate (DPPH) radical inhibition and ferric reducing antioxidant power (FRAP) value in pressurized and ultra-sonicated juices displayed higher reduction rate during storage than those in pasteurized juice. Nevertheless, at the final stage of storage, both juices still contained higher levels of antioxidant compounds and properties than in thermally treated juice. All the treated samples were shown to reduce initial microbial load of fresh maoberry juice to a non-detectable amount, while maintaining their quality during prolonged refrigerated storage.
KEYWORDS:Antioxidants; Maoberry; Pasteurization; Pressurization; Ultra-Sonication
Introduction
As the presence and variety of natural nutraceuticals and novel functional food products continue to grow, maoberry juice is widely recognized as a nutrient-rich and healthy beverage option in today’s market. Antioxidant compounds in fruits, for example, polyphenolic compounds and ascorbic acid (vitamin C), are believed to contribute to the prevention of oxidative stress related diseases, viz. coronary artery disease, cancers, diabetes, and aging-related disorders.1-2 Maoberry fruits (Antidesma bunius L.) are vastly grown in the northern and northeastern regions of Thailand, and are commonly used to make jam, jelly, red wine, ready-to-drink juice, and juice concentrates.3-5 They comprise of significant amounts of bioactive phytochemicals, viz. ascorbic acid, phenolic acids, anthocyanins, and flavonoids, thus contain pharmacological properties.6-7
There is strong research evidence regarding the health benefits of maoberry fruit extracts, such as pathogen inhibition and prevention of human breast epithelial cell apoptosis, contributed by their antioxidant constituents.8-9 While maoberry juice is widely consumed for health-related purposes, traditional methods used in fruit juice thermal processing, such as pasteurization and sterilization, were shown to induce detrimental effects characterized by high levels of reduction in such beneficial compounds.5, 10-11 In terms of microbiological stability, previous studies have indicated significant reductions in microbial counts following ultra-sonication of mulberry and blueberry juices, with correspondingly greater effects observed as sonication intensity increased.12-13 Similarly, ultra-high hydrostatic pressure (HHP) and ultra-sonic (US) techniques were shown to maintain various bioactive compounds and to inactivate microbial growth in grapefruit, pomegranate, jabuticaba, and aronia juices.14-17 Only few studies to date have observed the effectiveness of applying these advanced technologies to maoberry juice preservation. Therefore, the objective of this research was to investigate the influence of pasteurization (85ºC/1 min), pressurization (500 and 600 MPa/30ºC/30 min) and ultra-sonication (20 kHz/60% and 80% amplitude/30 min) on antioxidative properties and microbiological quality of maoberry juice. The storage stability of each treated juices at 4ºC for 30 days was also monitored.
Materials and Methods
Chemicals
Folin–Ciocalteu phenol reagent, 2,2-diphenyl-1-picryl hydrazyl hydrate (DPPH), 2,4,6-tripyridyls-triazine (TPTZ), ferric chloride hexahydrate, (+)-catechin, (-)-epicatechin, gallic acid, vanillic acid, cyanidin-3-o-glucoside and cyanidin 3-rutinoside were obtained from Sigma–Aldrich (St. Louis, MO, USA). High performance liquid chromatography (HPLC) grade methanol, absolute ethanol, acetonitrile, acetic acid, sulphuric acid and phosphoric acid were purchased from Merck (Darmstadt, Germany). Plate Count Agar (PCA), Potato Dextrose Agar (PDA) and Violet Red Bile Lactose Agar (VRBL) were supplied from Hi-Media (Mumbai, India). Deionized water and castor oil were provided from Chemical & Lab Supplies (Bangkok, Thailand).
Preparation of Fresh and Processed Maoberry Juices
Organic maoberry fruits were purchased from a local market in Pupan district, Sakon Nakhon province, Thailand. The fresh fruits were washed twice prior to extraction with a juice extractor before packing and processes. The extracted juice (200 mL) was filled in a retort pouch bag (P.P. Pack, Mahasarakham, Thailand) before pasteurization at 85ºC for 1 minute using a thermostatic water bath (LAUDA, Lauda-Königshofen, Germany). Pressurized maoberry juice was prepared using a ‘Food Lab’ high-pressure rig (Stansted Fluid Power, Stansted, Essex, UK). The pressure transmitting medium was a mixture of castor oil and absolute ethanol at a ratio of 1:4 (v/v). The juice (200 mL) was filled in a nylon-polyethylene bag (P.P. Pack, Mahasarakham, Thailand), followed by pressure levels of 500 and 600 MPa at 30ºC for 30 minutes. Besides, maoberry juice (200 mL) was filled into a 300 mL sterile beaker, then ultra-sonicated at a frequency of 20 kHz and 60-80% amplitudes (VCX 130 PB 130 W, Sonics & Materials Inc., Newtown, CT) in an aseptic area. After processing for 30 minutes, the core temperature of ultra-sonicated juices increased from the baseline of 18.63 ± 0.45ºC to 55.02 ± 2.14ºC and 70.36 ± 1.83ºC at wave amplitudes of 60% and 80%, respectively. Subsequently, those pasteurized and ultra-sonicated juices were immersed in an ice-water bath for cooling down to ambient temperature. All the processed samples were then refrigerated at 4ºC for 30 days. During storage, sampling was carried out at 5 day-intervals to determination of bioactive compounds, antioxidative properties and microbiological quality.
Determination of Bioactive Phytochemicals
Ascorbic Acid
To extract ascorbic acid, fresh and processed maoberry juices (10 mL) were mixed with 40 mL of diluted sulphuric acid (pH 2.2) for 10 min using a sonicator bath before filtering through a 0.20-μm nylon membrane (Millex Syringe Filter; Sigma–Aldrich, St. Louis, MO, USA). A 20-μL filtrate was injected into the HPLC system, which consisted of a YMC-Pack ODS-AM C18 column (5 μm, 4.6 mm ID × 250 mm).4 The detector was adjusted to wavelength of λmax 250 nm. Acetic acid solution (0.1 M) was used as a mobile phase with a flow rate of 1.5 mL/min and column temperature was adjusted to 30°C.7 Ascorbic acid identification was carried out by comparing its retention time and spectra with the standard using a calibration curve. Peak area was identified and converted to the concentration of ascorbic acid.
Predominant Phenolic Compounds
All predominant phenolic compounds, viz. gallic acid, vanillic acid, cyanidin 3-o-glucoside, cyanidin 3-rutinoside, (+)-catechin and (˗)-epicatechin, in fresh, processed and stored samples were determined according to the modified protocol of Jorjong et al.6 To extract those phenolic compounds, the juice (20 mL) was well-mixed with 30 mL of methanol using a sonicator bath for 10 min. After that, each mixture was filtered through a 0.20-μm nylon membrane and 20 μL filtrate were injected to the HPLC system.4 Acetonitrile (eluent A) and diluted phosphoric acid (pH 2.58, eluent B) was used as a mobile phase with a flow rate of 0.8 mL/min for separation both gallic and vanillic acids. The gradient system of the mobile phase commenced from 0 min (100% A) to 15 min (91% A), 22 min (89% A), 38 min (82% A), 43 min (77% A), 45 min (70% A), 55 min (20% A), and 60 min (95% A). The detector was adjusted to wavelength of λmax 280 nm. For determination of cyanidin 3-o-glucoside and cyanidin 3-rutinoside concentrations, 4% (v/v) phosphoric acid (eluent A) and acetonitrile (eluent B) was used as a mobile phase with a flow rate of 1 mL/min and column temperature of 40°C. The UV–Vis detection was at λmax 520 nm. The linear gradient started with 94% B at 0 min, 75% B at 55 min, 75% B at 65 min at isocratic elution for 70 min.6 Besides, the mobile phase for (+)-catechin and (˗)-epicatechin was acetonitrile/deionized water (2/97.8, v/v) plus 0.2% phosphoric acid (eluent A) and acetonitrile/deionized water (97.8/2, v/v) plus 0.2% phosphoric acid (eluent B). The flow rate of mobile phase and column temperature were set at 0.6 mL/min and at 40°C, respectively. The UV–Vis detection was at λmax 520 nm. Gradient elution started with 20% B, 50% B at 30 min, 60% B at 35 min, 20% B at 40 min at isocratic elution for 55 min.3 Quantification was carried out by comparing the retention times and absorbance along the spectra, as well as by the use of reference standards. The concentrations of phenolic compounds were calculated using a corresponding external standard.
Determination of Antioxidant Capacity
DPPH Assay
Briefly, the sample (2 mL) was well-mixed with 8 mL of 100% methanol using an orbital shaker for 10 min before centrifugation at 4,500 rpm for 10 min. Subsequently, the supernatant (1.6 mL) was poured into 0.4 mL of 1.5 μM DPPH radical in methanol, mixed, and left to stand for 30 min in the dark. The absorbance of the mixture was then measured at wavelength of λmax 517 nm using a UV–Visible spectrophotometer (UV1800; Shimadzu, Tokyo, Japan). DPPH radical inhibition was calculated, where A0 is absorbance of the control (1.6 mL of methanol), and A1 is absorbance of the sample.18
DPPH radical inhibition (%) = [1 − (A1/A0)] × 100
FRAP assay
FRAP value was determined according to the modified method as described by Benzie and Stain.19 One mL juice was poured into 9 mL deionized water and well-mixed for 20 min before filtering through a 0.20-μm nylon membrane. Afterwards, 3 mL FRAP reagent (10:1:1 of 300 mM sodium acetate buffer at pH 3.6, 10 mM TPTZ and 20 mM ferric chloride hexahydrate reagent) was added to the filtrate, mixed and incubated at 37°C for 30 min in the dark room. The absorbance of the mixture was then measured at wavelength of λmax 593 nm. The results were expressed as mM of ferrous sulfate per 100 mL sample (mM FeSO4/100 mL).
Assessments of Microbiological Quality
The indicator microbes, viz. total bacteria, yeast-mold and coliform counts, in the sample were carried out using the PCA, PDA and VRBL media, respectively. The media were poured into the Petri dish and agitated gently to homogenize with the diluted sample. After formed a gel, all plates were incubated at 37°C for 24 h before counting the colonies. Those microbes were calculated as colony forming units per milliliter sample (CFU/mL).20
Statistical Analysis
All data were existed as mean ± standard deviation (S.D.) of six replications (n = 6). One-way analysis of variance (ANOVA) was carried out using SPSS software (SPSS Inc., Chicago, IL) at a significance level of 95%. Duncan’s New Multiple Range Test (DMRT) was used to differentiate the means.
Results and Discussion
Bioactive Compounds and Antioxidative Properties of Maoberry Juices
Ascorbic acid is a strong reducing agent with high antioxidant potential. However, it is also susceptible to oxidation or degradation by light, oxygen, heat, peroxide and enzymes.18, 21 In this study, the change in ascorbic acid concentrations in maoberry juice affected by thermal, ultra-high pressure, and ultra-sonic treatments were determined, as shown in Table 1. The amount of ascorbic acid in all processed samples was significantly lower (P≤0.05) than that of fresh juice, with the lowest contents in pasteurized juice. In pressurized maoberry juices, there was no significant difference on ascorbic acid concentrations (P>0.05), regardless of different levels of pressure treatment. On the other hand, the increased amplitude of ultra-sonication was found to significantly correlate with the reduced levels of the compound (P≤0.05). The internal temperature of the sample increased from 22.19 ± 2.31ºC to 74.12±1.45ºC after ultra-sonication at 80% amplitude. Pressurization and ultra-sonication treatments tended to degrade ascorbic acid in maoberry juice, which is similar in effect of heat treatment (i.e. pasteurization).22 Landl et al. also revealed that ascorbic acid can be degraded to dehydroascorbic acid, and further irreversibly converted into 2,3-diketogulonic acid.23 Overall, our results showed that ultra-high pressure and ultra-sonic processing could retain ascorbic acid concentration in maoberry juice by 85.49–86.66%, and 64.15–69.78%, respectively, and are shown to have greater conservative effects compared to pasteurization. Similarly, the findings of Vega-Galvez et al.24 and Khandpur and Gogate25, reported that pasteurization processing had led to significantly increased degradation of ascorbic acid in fruit and vegetable juices when compared to pressurization and ultra-sonication.
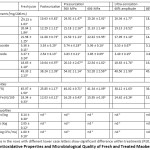 |
Table 1: Antioxidative Properties and Microbiological Quality of Fresh and Treated Maoberry Juices. |
Gallic and vanillic acids are predominant phenolic acids found in maoberry fruits. According to Jorjong et al., there are 15 cultivars of maoberry fruits grown in Northeastern Thailand, containing, on average, around 159.47 and 179.75 mg/100 g (dry basis) of gallic and vanillic acids, respectively.6 Gallic acid is commonly regarded as a bioactive phenol compound with antioxidative, anti-carcinogenic, antibacterial, antifungal and antiviral properties.26 Vanillic acid was also found to be associated with a variety of pharmacologic activities, viz. inhibiting carcinogenesis, apoptosis and inflammation, and anti-hypertensive, and anti-colitis effects.27-29 From the results presented in Table 1, both phenolic acids have portrayed greater sensitivity to heat treatments. Phenolic acid concentrations in thermally and ultra-sonic treated juices were notably lower (P≤0.05) than those in other juices. While there was no significant effect of varied pressure levels on these components as compared to those in fresh juice, both phenolic acid contents showed a declining trend when the amplitude level increased. Keenan et al. stated that pressure could affect the structure of large polymers by keeping small molecules, such as phenolic acids, intact.30 Similarly, in the study of Chaikham and Apichartsrangkoon, phenolic acids (i.e. gallic and ellagic acids) in longan juice were not affected by pressurization conditions.31 This might suggest that gallic and vanillic acids tend to remain stable during pressure treatment. According to the study of Chaikham et al. about ultra-sonification effects on maoberry juice, an increase in total phenolic content was observed following treatment at 80% amplitude.18 Similar trends were observed in ultra-sonication of grapefruit juice10 and purple cactus pear juice.32 Higher wave amplitudes during ultra-sonication could facilitate the release of phenolic compounds from plant-cell walls, which may contribute to the overall effect.22 Contrarily, the study of Kwaw et al., a significant increase in phenolic concentrations, including gallic and vanillic acids, was observed on ultra-sonication of fermented mulberry (Morus nigra) juice.5 Although little is known about the effect of ultra-sonification on polyphenolic content of foods, it was suggested that combining ultra-sonication with pulsed-light treatment may facilitate greater release of antioxidants via phytonutrient degradation into phenolic compounds.5
Besides, major anthocyanins (cyanidin 3-o-glucoside and cyanidin 3-rutinoside) and flavonoids [(+)-catechin and (˗)-epicatechin] in fresh and processed maoberry juices were also investigated (Table 1). It was found that the levels of cyanidin 3-o-glucoside, cyanidin 3-rutinoside, (+)-catechin and (˗)-epicatechin in maoberry juice were noticeably enhanced after pressurization, increasing by roughly 6.69-9.66%, 13.65-14.24%, 7.77-20.86%, and 6.04-9.57%, respectively, while such components in ultra-sonicated maoberry juice at 60% amplitude were similar in concentration to those of unprocessed juice. The increase in levels of anthocyanins and flavonoids may be related to the increased extractability as a result of processing. Contrarily, it was observed that ultrasonication at 80% amplitude resulted in significant reduction of all anthocyanins and flavonoids in maoberry juice, as compared to fresh juice. Overall, the pasteurized samples contained the lowest contents of compounds. According to the results, it could be derived that harsh thermal processing, including pasteurization and ultra-sonication with high frequency amplitude, may have caused the higher levels of predominant anthocyanin and flavonoid degradation in maoberry juice.
DPPH radical inhibition has been used to determine the antioxidant activity of maoberry juice, as indicated by the ability of antioxidants to donate a hydrogen atom or electron in radical stabilization. FRAP assay is generally used to measure the capacity of a substance in reducing TPTZ–Fe(III) complex to TPTZ–Fe(II) complex.1, 31, 33 From the results in Table 1, DPPH radical inhibition and FRAP values of all processed maoberry juice samples were lower than that of fresh juice. Significantly lower levels (P≤0.05) were observed in pasteurized juice and juice ultra-sonicated at 80% amplitude for 30 minutes. Lowest values were found in pasteurized juice, similar to that observed for ascorbic acid, phenolic acids, anthocyanins and flavonoids. However, in this study, only high correlation coefficients of ascorbic acid versus antioxidant activities were derived (R2 = 8.85-9.05). In the study of Chaikham and Baipong, it was elucidated that high hydrostatic pressure processing demonstrated significantly greater effectiveness in preserving bioactive components, i.e. ascorbic acid, total anthocyanins, total phenols, and antioxidant activities (DPPH and FRAP assays) of maoberry juice compared to thermal processing.7
Storage Stability of Bioactive Compounds and Antioxidant Activities of Processed Maoberry Juices
Figure 1 illustrates significant decrease in levels of ascorbic acid in all processed maoberry juices (P≤0.05) with increasing storage time. At the final stage of storage, ascorbic acid concentrations in all samples had reduced by around 50% from their initials. The highest rate of reduction was observed in pressurized juices, following by ultra-sonicated and pasteurized juices, respectively. These findings were confirmed by the slopes of linear trend-lines (linear equations) from the correlation of ascorbic acid content versus storage time (Table 2). However, as observed in Figure 1, the pressurized juices still retained higher amounts of ascorbic acid than that of ultra-sonicated juices, followed by thermally treated juice throughout the entire storage duration.
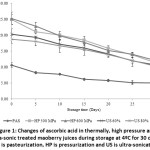 |
Figure 1: Changes of ascorbic acid in thermally, high pressure and ultra-sonic treated maoberry juices during storage at 4ºC for 30 days. PAS is pasteurization, HP is pressurization and US is ultra-sonication. |
Table 2: Linear Equations and R-square Values of Linear Trend-Lines from the Correlation of Antioxidant Properties (y) Versus Storage Period (x).
| Antioxidant properties | Treatments | Linear equations | R-square values (R2) |
| Ascorbic acid | Pasteurization | y = -0.178x + 9.650 | 0.899 |
| Pressurization at 500 MPa | y = -0.425x + 24.31 | 0.985 | |
| Pressurization at 600 MPa | y = -0.443x + 24.80 | 0.988 | |
| Ultra-sonication at 60% amplitude | y = -0.334x + 20.67 | 0.993 | |
| Ultra-sonication at 80% amplitude | y = -0.287x + 19.32 | 0.981 | |
| Gallic acid | Pasteurization | y = -1.142x + 14.04 | 0.944 |
| Pressurization at 500 MPa | y = -2.285x + 22.81 | 0.961 | |
| Pressurization at 600 MPa | y = -2.437x + 24.07 | 0.959 | |
| Ultra-sonication at 60% amplitude | y = -1.645x + 19.66 | 0.944 | |
| Ultra-sonication at 80% amplitude | y = -1.460x + 16.59 | 0.987 | |
| Vanillic acid | Pasteurization | y = -0.798x + 12.09 | 0.947 |
| Pressurization at 500 MPa | y = -1.876x + 23.02 | 0.990 | |
| Pressurization at 600 MPa | y = -1.871x + 23.87 | 0.993 | |
| Ultra-sonication at 60% amplitude | y = -1.521x + 18.61 | 0.990 | |
| Ultra-sonication at 80% amplitude | y = -1.019x + 15.52 | 0.984 | |
| Cyanidin 3-o-glucoside | Pasteurization | y = -0.195x + 3.377 | 0.952 |
| Pressurization at 500 MPa | y = -0.292x + 5.887 | 0.939 | |
| Pressurization at 600 MPa | y = -0.386x + 6.384 | 0.907 | |
| Ultra-sonication at 60% amplitude | y = -0.316x + 5.950 | 0.958 | |
| Ultra-sonication at 80% amplitude | y = -0.267x + 5.594 | 0.934 | |
| Cyanidin 3-rutinoside | Pasteurization | y = -0.209x + 2.894 | 0.924 |
| Pressurization at 500 MPa | y = -0.249x + 4.282 | 0.863 | |
| Pressurization at 600 MPa | y = -0.246x + 4.241 | 0.921 | |
| Ultra-sonication at 60% amplitude | y = -0.204x + 3.675 | 0.982 | |
| Ultra-sonication at 80% amplitude | y = -0.143x + 3.238 | 0.969 | |
| (+)-Catechin | Pasteurization | y = -0.600x + 10.48 | 0.989 |
| Pressurization at 500 MPa | y = -1.658x + 22.30 | 0.979 | |
| Pressurization at 600 MPa | y = -2.224x + 25.42 | 0.981 | |
| Ultra-sonication at 60% amplitude | y = -1.718x + 21.29 | 0.951 | |
| Ultra-sonication at 80% amplitude | y = -1.026x + 18.03 | 0.942 | |
| (˗)-Epicatechin | Pasteurization | y = -1.523x + 39.72 | 0.987 |
| Pressurization at 500 MPa | y = -2.166x + 54.81 | 0.958 | |
| Pressurization at 600 MPa | y = -2.049x + 53.69 | 0.969 | |
| Ultra-sonication at 60% amplitude | y = -2.037x + 50.18 | 0.976 | |
| Ultra-sonication at 80% amplitude | y = -1.737x + 47.72 | 0.976 | |
| DPPH inhibition | Pasteurization | y = -2.516x + 28.08 | 0.996 |
| Pressurization at 500 MPa | y = -2.548x + 42.31 | 0.971 | |
| Pressurization at 600 MPa | y = -2.651x + 43.39 | 0.977 | |
| Ultra-sonication at 60% amplitude | y = -2.880x + 41.26 | 0.982 | |
| Ultra-sonication at 80% amplitude | y = -2.147x + 31.87 | 0.997 | |
| FRAP value | Pasteurization | y = -0.937x + 12.44 | 0.961 |
| Pressurization at 500 MPa | y = -1.349x + 16.47 | 0.928 | |
| Pressurization at 600 MPa | y = -1.373x + 16.77 | 0.978 | |
| Ultra-sonication at 60% amplitude | y = -1.192x + 15.30 | 0.977 | |
| Ultra-sonication at 80% amplitude | y = -1.018x + 14.18 | 0.994 |
In this study, concentrations of predominant phenolic acids, anthocyanins and flavonoids in treated maoberry juices were directly affected by prolonged storage time (Figures 2-4). The amounts of gallic acid, vanillic acid, cyanidin 3-o-glucoside, cyanidin 3-rutinoside, (+)-catechin, and (˗)-epicatechin had notably declined (P≤0.05) during refrigerated storage for 30 days. The degradation rates of these phytochemical compounds, as indicated by the slopes from the linear equations (Table 2), were greater in pressurized juices, followed by ultrasonicated and pasteurized juices, respectively, with exception to the slope of linear trend-line of cyanidin 3-rutinoside in pasteurized juice being higher than that observed for ultra-sonicated juices. However, on the 30th day of storage, the concentrations of these components were significantly higher in pressurized juices compared to ultra-sonicated juices, followed by pasteurized juice, with exceptions to cyanidin 3-o-glucoside, cyanidin 3-rutinoside, and (+)-catechin concentrations in ultra-sonicated juices. Results from Figures 3 and 4 have observed no significant difference (P>0.05) between pressurized and ultra-sonicated juices in terms of levels of those phenolic acids. According to the report of da Silveira et al., it was found that high-pressure effect was more pronounced at preserving the thermal sensitivity of bioactive compounds (i.e. anthocyanins, non-anthocyanin, phenolic compounds, and tocopherols) and antioxidant capacity (peroxyl radical scavenging, hypochlorous acid scavenging, and hydrogen peroxide scavenging assays) in açaí juice than the thermal processing.34
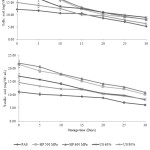 |
Figure 2: Changes of predominant phenolic acids in thermally, high pressure and ultra-sonic treated maoberry juices during storage at 4ºC for 30 days. AS is pasteurization, HP is pressurization and US is ultra-sonication. |
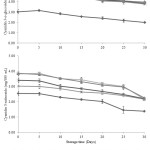 |
Figure 3: Changes of predominant anthocyanins in thermally, high pressure and ultra-sonic treated maoberry juices during storage at 4ºC for 30 days. PAS is pasteurization, HP is pressurization and US is ultra-sonication. |
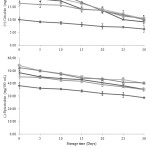 |
Figure 4: Changes of predominant flavonoids in thermally, high pressure and ultra-sonic treated maoberry juices during storage at 4ºC for 30 days. PAS is pasteurization, HP is pressurization and US is ultra-sonication. |
Furthermore, the antioxidant activities (DPPH and FRAP assays) of all treated maoberry juice samples were also determined. In this case, DPPH radical inhibition and FRAP values of maoberry juice substantially decreased with prolonged storage duration (Figure 5). Moreover, similar reduction rates of both values in the processed juices were observed with increased storage time, as indicated by the slopes of their linear trend-lines from the equations in Table 2. Similarly, Morales-de la Peña et al. reported a depletion of ascorbic acid content and antioxidant capacity in fruit juice-soy milk beverages with prolonged chilled storage.35 A study on cold storage of blueberry juice demonstrated stability in antioxidant capacity for up to 10 days of refrigeration at 4°C, yet a significant loss of 83% anthocyanins was observed.36 This could be possibly be due to reactions with other phenolic compounds in the juice.
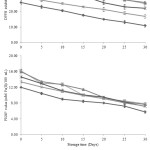 |
Figure 5: Changes of antioxidant activities in thermally, high pressure and ultra-sonic treated maoberry juices during storage at 4ºC for 30 days. PAS is pasteurization, HP is pressurization and US is ultra-sonication. |
Microbiological Changes in Maoberry Juice after Processing and During Storage
From Table 1, the initial amounts of total plate counts, yeasts-molds and fecal coliforms in fresh maoberry juice were 6.14, 2.05, and 1.60 log CFU/mL, respectively. In this term, it was found that all indicator microorganisms were effectively eliminated via applied processing methods. The microbial quality of the treated samples complied with the standard for ready-to-drink maoberry juice.37 Many scientific findings have demonstrated the inhibitory effects of high pressure treatment on the survival of microorganisms, resulting from changes in cell morphology and inhibition of metabolic reactions essential for cell maintenance and genetic mechanisms. The ultra-sonication process enhances formation of biocides, including free radicals and hydrogen peroxide, through cavitation, thus preventing the growth of microbial vegetative cells.22
Following storage conditions, the microbiological quality of processed juice samples remained within the general standard of ready-to-drink maoberry juice.37 During microbiological testing, none of the indicator microbial species were detected throughout the entire storage period (data not shown). Similarly, Wu et al. reported that thermal and high hydrostatic pressure processing was able to effectively reduce microbial count of pineapple fruit juice to a satisfactory level.38 According to a study by Donsì et al., pulsed high pressure processing was shown to reduce initial microbial count of Annurca apple and orange juices, while maintaining low levels of microbial proliferation following 21 days of chilled storage, which complied with the (EC) Regulation no. 2073/2005 on the microbiological criteria for foodstuffs.39 Furthermore, significantly lower levels of microbial load and increased shelf-stability was observed following pasteurization, high pressure treatment and refrigerated storage of acidic apple and sugarcane juices.40-41
Conclusion
In summary, different processing methods were evaluated based on their effectiveness in preserving maoberry juice. Pressurization and ultra-sonication of maoberry juice were shown to retain higher ascorbic acid compared to conventional method. Phenolic content, on the other hand, noticeably reduced following thermal processing methods of pasteurization and high-amplitude ultra-sonication. With prolonged refrigerated storage, the antioxidant capacity of all maoberry juices declined. All processing methods were shown to reduce initial microbial load of fresh maoberry juice to a non-detectable amount, while maintaining their microbial quality during prolonged refrigerated storage. Although pressurization has demonstrated the ability to preserve bioactive compounds and enhance storage stability, few studies have observed the effects of novel processing adaptations on maoberry juice. Further research would be necessary to fully determine the capacity and effectiveness of alternative preservation methods.
Acknowledgments
We would like to express our gratitude towards Phranakhon Si Ayutthaya Rajabhat University and Mahidol University for their facilities in conducting this research study.
Funding Source
The authors received no financial support for the research, authorship, and publication of this article.
Conflict of Interest
The authors declare no conflict of interest.
References
- Bendary, E., Francis, R.R., Ali, H.M.G., Sarwat, M.I., El Hady, S. Antioxidant and structure-activity relationships (SARs) of some phenolic and anilines compounds. Annals of Agricultural Science. 2013; 58(2): 173-181.
CrossRef - El-Tantawy, W.H., Soliman, N.D., El-naggar, D., Shafei, A. Investigation of antidiabetic action of Antidesma bunius extract in type 1 diabetes. Archives of Physiology and Biochemistry. 2015; 121(30): 116-122.
CrossRef - Butkhup, L., Samappito, S. An analysis on flavonoids contents in Mao Luang fruits of fifteen cultivars (Antidesma bunius), grown in Northeast Thailand. Pakistan Journal of Biological Sciences. 2018; 11(7): 996-1002.
CrossRef - Chaikham, P., Prangthip, P., Seesuriyachan, P. Ultra-sonication effects on quality attributes of maoberry (Antidesma bunius ) juice. Food Science and Technology Research. 2016; 22(5): 647-654.
CrossRef - Kwaw, E., Ma, Y., Tchabo, W., Apaliya, M.T., Sackey, A.S., Wu, M., Xiao, L. Impact of ultrasonication and pulsed light treatments on phenolics concentration and antioxidant activities of lactic-acid-fermented mulberry juice. LWT – Food Science and Technology. 2018; 92: 61-66.
CrossRef - Jorjong, S., Butkhup, L., Samappito, S. Phytochemicals and antioxidant capacities of Mao-Luang (Antidesma bunius) cultivars from Northeastern Thailand. Food Chemistry. 2015; 181: 248-255.
CrossRef - Chaikham P., Baipong, S. Comparative effects of high hydrostatic pressure and thermal processing on physicochemical properties and bioactive components of Mao Luang (Antidesma bunius ) juice. Chiang Mai Journal of Science. 2016; 43(4): 851-862.
- Puangpronpitag, D., Yongvanit, P., Boonsiri, P., Suttajit, M., Areejitranusorn, P., Na, H-K., Surh, Y-J. The molecular mechanism underlying anti-apoptotic and anti-inflammatory effects of Mamao (Antidesma thwaitesianum Müell. Arg.) Polyphenolics in human breast epithelial cells. Food Chemistry. 2011; 127: 1450-1458.
CrossRef - Butkhup, L., Samappito, S. Changes in physicochemical properties, polyphenol compounds and antiradical activity during development and ripening of Maoluang (Antidesma bunius Spreng) fruits. Journal of Fruit and Ornamental Plant Research. 2011; 19(1): 85-99.
- Aadil, R.M., Zeng, X-A., Han, Z., Sun, D-W. Effects of ultrasound treatments on quality of grapefruit juice. Food Chemistry. 2013; 141: 3201-3206.
CrossRef - Chen, D., Xi, H., Guo, X., Qin, Z., Pang, X., Hu, X., Wu, J. Comparative study of quality of cloudy pomegranate juice treated by high hydrostatic pressure and high-temperature short time. Innovative Food Science & Emerging Technologies. 2013; 19: 85–94.
CrossRef - Engmann, F.N., Ma, Y., Tchabo, W., Ma, H. Ultrasonication treatment effect on anthocyanins, color, microorganisms and enzyme inactivation of mulberry (Moraceae nigra) Juice. Journal of Food Processing and Preservation. 2015; 39(6): 854-862.
CrossRef - Mohideen, F.W., Solval, K.M., Li, J., Zhang, J., Chouljenko, A., Chotiko, A., Prudente, A.D., Bankston, D., Sathivel, S. Effect of continuous ultra-sonication on microbial counts and physico-chemical properties of blueberry (Vaccinium corymbosum) juice. LWT – Food Science and Technology. 2015; 60(1): 563-570.
CrossRef - Varela-Santos, E., Ochoa-Martinez, A., Tabilo-Munizaga, G., Reyes, J.E., Pérez-Won, M., Briones-Labarca, V., Morales-Castro, J. Effect of high hydrostatic pressure (HHP) processing on physicochemical properties, bioactive compounds and shelf-life of pomegranate juice. Innovative Food Science & Emerging Technologies. 2012; 13: 13-22.
CrossRef - Uckoo, R.M., Jayaprakasha, G.K., Somerville, J.A., Balasubramaniam, V.M., Pinarte, M., Patil, B.S. High pressure processing controls microbial growth and minimally alters the levels of health promoting compounds in grapefruit (Citrus paradise Macfad) juice. Innovative Food Science & Emerging Technologies. 2013; 18: 7-14.
CrossRef - Błaszczak, W., Amarowicz, R., Górecki, A.R. Antioxidant capacity, phenolic composition and microbial stability of aronia juice subjected to high hydrostatic pressure processing. Innovative Food Science & Emerging Technologies. 2017; 39: 141-147.
CrossRef - Inada, K.O.P., Torres, A.G., Perrone, D., Monteiro, M. High hydrostatic pressure processing affects the phenolic profile, preserves sensory attributes and ensures microbial quality of jabuticaba (Myrciaria jaboticaba) juice. Journal of the Science of Food and Agriculture. 2017; 98(1): 231-239.
CrossRef - Chaikham, P., Apichartsrangkoon, A., Seesuriyachan, P. Physical and biochemical qualities of pressurized and pasteurized longan juices upon storage. Emirates Journal of Food and Agriculture. 2014; 26(3): 218-228.
CrossRef - Benzie, I.F.F., Stain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power; the FRAP assay. Analytical Biochemistry. 1996; 239: 70–76.
CrossRef - US Food and Drug Administration. Bacteriological analytical manual (BAM). Washington DC: New Hampshire Avenue, Department of Health and Human Services. 2001.
- Igual, M., García-Martínez, E., Camacho, M.M., Martínez-Navarrete, N. Effect of thermal treatment and storage on the stability of organic acids and the functional value of grapefruit juice. Food Chemistry. 2010; 118: 291–299.
CrossRef - Chaikham, P., Prangthip, P. Alteration of antioxidative properties of longan flower-honey after high pressure, ultra-sonic and thermal processing. Food Bioscience. 2015; 10: 1-7.
CrossRef - Landl, A., Abadias, M., Sárraga, C., Viñas, I., Picouet, P.A. Effect of high pressure processing on the quality of acidified Granny Samith apple purée product. Innovative Food Science and Emerging Technologies. 2010; 11: 557–564.
CrossRef - Vega-Galvez, A., Miranda, M., Aranda, M., Henriquez, K., Vergara, J., Tabilo-Munizaga, G., Perez-Won., M. Effect of high hydrostatic pressure on functional properties and quality characteristics of Aloe vera gel (Aloe barbadensis Miller). Food Chemistry. 2011; 129(3): 1060–1065.
CrossRef - Khandpur, P., Gogate, P.R. Effect of novel ultrasound based processing on the nutrition quality of different fruit and vegetable juices. Ultrasonics Sonochemistry. 2015; 27: 125–136.
CrossRef - Rangkadilok, N., Worasuttayangkurn, L., Bennett, R.N., Satayavivad, J. Identification and quantification of polyphenolic compounds in longan (Euphoria longana ) fruit. Journal of Agricultural and Food Chemistry. 2005; 53: 1387–1392.
CrossRef - Kim, S., Kim., M., Um, J., Hong S. The beneficial effect of vanillic acid on ulcerative colitis. Molecules. 2010; 15: 7208-7217.
CrossRef - Kumar, S., Prahalathan, P., Raja, B. Antihypertensive and antioxidant potential of vanillic acid, a phenolic compound in L-NAME-induced hypertensive rats: a dose-dependence study. Redox Report. 2011; 16(5): 208-215.
CrossRef - Gitzinger, M., Kemmer, C., Fluri, D.A., El-Baba, M.D., Weber, W., Fussenegger, M. The food additive vanillic acid controls transgene expression in mammalian cells and mice. Nucleic Acids Research. 2012; 40(5): E37.
CrossRef - Keenan, D.F., Rößle, C., Gormley, R., Butler, F., Brunton, N.P. Effect of high hydrostatic pressure and thermal processing on the nutritional quality and enzyme activity of fruit smoothies. LWT – Food Science and Technology. 2012; 45: 50–57.
CrossRef - Chaikham, P., Apichartsrangkoon, A. Comparison of dynamic viscoelastic and physicochemical properties of pressurised and pasteurised longan juices with xanthan addition. Food Chemistry. 2012; 134: 2194–2200.
CrossRef - Zafra-Rojas, Q.Y., Cruz-Cansino, N., Ramírez-Moreno, E., Delgado-Olivares, L., Villanueva-Sánchez, J., Alanís-García, E. Effects of ultrasound treatment in purple cactus pear (Opuntia ficusindica) juice. Sonochemistry. 2013; 20: 1283-1288.
CrossRef - Chuaychan, S., Benjakul, S. Effect of maltodextrin on characteristics and antioxidative activity of spray-dried powder of gelatin and gelatin hydrolysate from scales of spotted golden goatfish. Journal of Food Science and Technology. 2016; 53(9): 3583-3592.
CrossRef - da Silveira, T.F.F., Cristianini, M., Kuhnle, G.G., Ribeiro, A.B., Filho, J.T., Godoy, H.T. Anthocyanins, non-anthocyanin phenolics, tocopherols and antioxidant capacity of açaí juice (Euterpe oleracea) as affected by high pressure processing and thermal pasteurization. Innovative Food Science & Emerging Technologies. 2019; 55: 88–96.
CrossRef - Morales-de la Peña, M., Salvia-Trujillo, L., Rojas-Graü, M.A., Martín-Belloso, O. Impact of high intensity pulsed electric field on antioxidant properties and quality parameters of a fruit juice–soymilk beverage in chilled storage. LWT – Food Science and Technology. 2010; 43(6): 872-881.
CrossRef - Reque, P.M., Steffens, R.S., Jablonski, A., Flôres, S.H., Rios, A.O., Jong, E.V. Cold storage of blueberry (Vaccinium) fruits and juice: Anthocyanin stability and antioxidant activity. Journal of Food Composition and Analysis. 2014; 33: 111-116.
CrossRef - Thai Industrial Standard Institute. Thai Community Product Standard (TCPS) No. 486/2004: Mao juice. Bangkok, Thailand: Ministry of Industry. 2001.
- Wu, W., Xiao, G., Yu, Y., Xu, Y., Wu, J., Peng, J., Li, L. Effects of high pressure and thermal processing on quality properties and volatile compounds of pineapple fruit juice. Food Control. 2021; 130: 108293.
CrossRef - Donsì, G., Ferrari, G., Maresca, P. Pasteurization of fruit juices by means of a pulsed high pressure process. Journal of Food Science. 2010; 75(3): 167-177.
CrossRef - Luong, T.S.V., Moir, C., Chandry, P.S., Pinfold, T., Olivier, S., Broussolle, V., Bowman, J.P. Combined high pressure and heat treatment effectively disintegrates spore membranes and inactivates Alicyclobacillus acidoterrestris spores in acidic fruit juice beverage. Innovative Food Science & Emerging Technologies. 2020; 66: 102523.
CrossRef - Sreedevi, P., Jayachandran, L.E., Srinivasa Rao, P. (2020). Application of highpressure processing for extending the shelf life of sugarcane juice under refrigerated conditions. Journal of Food Processing and Preservation. 2020; 44(12): e14957.
CrossRef













