Drying Kinetics and Activation Energy of Asparagus Root (Asparagus racemosus Wild.) for Different Methods of Drying
1Department of Post-Harvest Process and Food Engineering, G. B. Pant University of Agriculture and Technology, Pantnagar, India, 263145
2Department of Horticulture, G. B. Pant University of Agriculture and Technology, Pantnagar, India, 263145.
Corresponding Author Email: deepikaprp27@gmail.com
DOI : http://dx.doi.org/10.12944/CRNFSJ.6.1.22
ABSTRACT:Drying reduces the water activity of food product and hence increases the shelf life of the food. In the present study, fresh asparagus roots were pretreated in hot water at 80oC for 5 min. The methods of drying used for the study were tray drying, solar drying, vacuum drying and fluidized bed drying at four temperature levels 40, 50, 60 and 70oC. The complete drying of asparagus follow falling rate period only. The total time for drying decreases with increase in temperature of drying air from 40oC – 70oC. Fluidized bed dryer has a highest average drying rate as compare to tray dryer, solar dryer and vacuum dryer. Four empirical models, namely Page’s, Exponential, Generalized exponential and Logarithmic model were fitted in the drying data to describe the phenomena of drying process using a linearized regression technique. It was found that the Page’s model was best as it describe most precisely about the drying behavior of asparagus roots. The effective moisture diffusivity of asparagus roots varies from 7.14 × 10-9 to 3.70 × 10-8 m2/s and it was also found that diffusivity increases with increase in temperature. The activation energy was found to be from 11.797 to 30.318 kJ/mol.
KEYWORDS:Asparagus roots; Activation energy; Drying; Diffusivity; Empirical models
Introduction
Asparagus (Asparagus racemosus L.) growing in the Mediterranean region is a rare wild species of Asparagus. It is commercially cultivated in Madhya Pradesh, Uttar Pradesh and Uttrakhand. It is a slender climber and belonging to the family Liliaceae. It is gaining popularity due to its texture and flavor.1 It includes about 300 species in the world and out of which the 22 species are recorded in India. It is most commonly used in traditional medicine.2 It is used for treatment and prevention of dyspepsia, gastric ulcers and as a galactogogue, nervous disorders, liver diseases, inflammation and infectious diseases. It is also used for the treatment of epilepsy, excessive heat, chronic fevers, kidney disorders, stomach ulcers and liver cancer, it also increases milk secretion in nursing mothers and also regulates sexual behaviors. It contains vitamins (A, B1, B2, C, E), minerals (Mg, P, Ca, Fe), and folic acid with few primary chemical constituents like asparagine, essential oils, arginine, tyrosine, flavonoids (kaempferol, quercetin, and rutin), resin, and tannin.3 Crude fiber, crude protein, ash content, ether extract and nitrogen free extract analysis found that it is very rich in nitrogen free extract and minerals like Ca, Mg, Fe, Cu, Zinc etc.3
Fresh Asparagus roots are highly perishable which leads to wastage and losses of the fresh product. It degrades rapidly, which results in a short shelf life of roots of 3–5 days under normal postharvest handling at room temperature.4 The short shelf life of fresh root is mainly due to its high respiration rate which continues even after harvesting.5 It leads to degradation in quality, loss in quantity and reduction in economic values of the product. The reduction of these losses is important, especially to balance the supply and demand at the time of off-season. Therefore, several methods including refrigeration, freezing and drying can be used which increases the shelf life of product.6
Blanching is the pre-treatment method used to arrest few physiological processes. It helps for inactivation of the enzymes, acceleration of drying rate and for reduction of the quality loss. It expels intercellular air from the tissues and also softens the product.7 Generally, blanching of fruits and vegetables is done by heating in steam or hot water.8
Drying is one of the most generally used methods for preservation. It preserves food by reducing water activity which hinders the growth and reproduction of microorganisms which are responsible for deterioration and also minimizes the moisture induced problems.9 Thin layer drying equations are used to estimate the drying time for several food products and also for the generalization of the drying curves. Drying kinetics is greatly affected by the temperature of air, velocity of air, thickness of material, and others.10 Physical and thermal properties of food products, such as the heat and mass transfer, and moisture diffusion data are required for the ideal design of dryer.11 The knowledge of effective moisture diffusivity is also important for designing and modeling of the mass transfer processes such as drying or moisture adsorption during storage. Drying characteristics & kinetics of fruits and vegetables have been studied by many investigators, cauliflower,12 carrot,13 yam,7 asparagus,14 kiwifruit and apricot.15 However, the drying characteristic of Asparagus has not been investigated and a very few information are available in the literature.
The objective of this work was to compare the effect of different dryers (solar, tray fluidized bed and vacuum drying) and temperature of drying air (40, 50, 60 and 70°C) on drying time and also to find a best suitable empirical mathematical model for drying, and to estimate effective moisture diffusivity for Asparagus roots.
Material and Method
Sample: Fresh wild Asparagus (Asparagus racemosus L.) roots were procured from the Medicinal Plant Research and Development Centre, Pantnagar, Uttrakhand. Sample was selected carefully for uniform shape, size and samples were also checked for any defect on visual inspection. Selected roots were cleaned and washed by removing damaged fruits and all foreign matters such as dust and dirt. The selected samples were cut into 2-4 mm long slices. The sliced sample was used for initial moisture content determination by a vacuum oven drying method.16 Finally, pre-treatments were applied and drying experiments were performed.
Experimental procedure: For drying, pre-treatment was carried out by blanching with hot water at 80ºC for 5 min.17 Pretreated sample was subjected to the thin layer drying in the “Tray, Solar, Fluidized bed, and Vacuum” dryers at the temperature ranges of 40 – 70oC with an increment of 10oC.18,19,20 The dryer was started before 60 min for steady-state conditions to dry the samples and the drying process was started when the required drying conditions for drying were achieved. Then, the pre-treated samples of Asparagus root were put in a single layer in dryer for drying. The weight of the samples was continuously taken with an interval of 1 hour to check the change of sample moisture or to determine the moisture content at different drying time. The drying was carried until two to three consecutive weights which didn’t vary more than 3 – 5 mg and that final weight was recorded. The dried samples were packed in polyethylene bags of 200 gauge in air tight condition and stored at room temperature in desiccators. The initial moisture content of the sample was calculated on dry basis by using formula Mc = W1 – W2/W0 Where, W0 is dry weight of the sample (g), W1 is sample weight before drying (g) and W2 is sample weight after drying (g). Moisture Ratio: The moisture content of Asparagus roots is expressed in moisture ratios (MR) with the following equation;21,22

The Me values were neglected because as compared to Mo and M the values of equilibrium moisture content were very small for long drying time and therefore the moisture ratio was calculated as the following relationship23,24:

Where, M is the mean moisture content; Mo is the initial moisture content; and Me is the equilibrium moisture content (EMC).
Determination of effective moisture diffusivity: For slab geometry food particles the fick’s diffusion equation was used for effective moisture diffusivity calculation of biological products.25 The fresh cut Asparagus roots were assumed to be similar as slab geometry and for calculation of moisture ratio the following equation was used26:
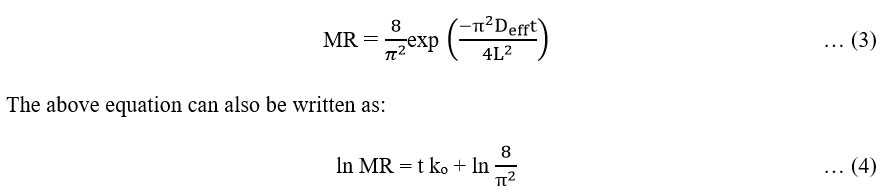
Where, Deff is the effective moisture diffusivity (m2/s); L is the half of thickness of slab (m). According to Equation (4) by plotting graph of ln (Moisture Ratio) versus time, the slope (ko) was calculated. On comparing the above equation with linear equation Y = mX + C, where m is the slope of the straight line and the slope was calculated as

Determination of activation energy: The effective moisture diffusivity is related with temperature by Arrhenius equation27:
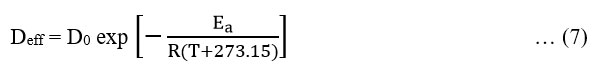
where, D0 is the constant of Arrhenius equation in m2s-1, Ea is the activation energy in kJ.mol-1, T is the temperature in °C and R is universal gas constant in kJ.mol-1.K-1.
The rearranged form of the equation (7):
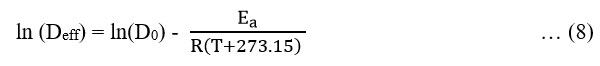
The activation energy was calculated using the curve between ln(Deff) versus 1/(T+273.15).
Modeling of the thin-layer drying curves: The curve of moisture ratio and time explains better about the drying behavior as compare to the moisture content and time curve because the initial value of moisture ratio was one for each of the experiments. These moisture ratio values of drying data were also used for prediction of the best drying model for pre-treated samples of Asparagus roots. MR data were fitted into four models listed in Table 1 to select the best model for drying of Asparagus. The thin-layer equations were used to fit the drying experimental data of Asparagus roots in their linearised form using regression technique in a spread sheet (EXCEL) and CURVE EXPERT 1.3 software package on personal computer. The comparison of the all models was done by comparing coefficient of determination (R2) and standard error of estimation (SEE).
Table 1: Empirical Models
| S. No. | Model expression | Name of model | References |
| 1 | MR = | Exponential model [28] | Henderson, 1974 |
| 2 | MR = A | Generalized exponential model [29] | Zhang and Litchfield,1991 |
| 3 | MR = | Page’s model [30] | Guarte, 1996 |
| 4 | MR = a e-kt + c | Logarithmic model [31] | Karathanos, 1999 |
Where, MR is moisture ratio, t is time in h, k is drying constant in h-1 and A, n, a, c are the drying parameters.
Result and Discussion
Characteristics of the Asparagus roots
The value of moisture content of the root samples was found to be 86.44% (w.b.) and 637.46% (d.b.) which shows that the Asparagus roots can be grouped under a highly perishable medicinal plant. It was also be found that the final moisture content of sample decreases with the increase in drying temperature.
Moisture ratio
The variation of moisture ratio verses time for an experimental range of temperature (40-70oC) is shown in Figures 1 to 4. The relationship of moisture ratio and time shows that there was a rapid decrease in moisture ratio with a faster rate at initial stage of 60 to 120 min of drying and in later stages the moisture ratio decrease in slower rate as moisture content approached to equilibrium moisture content. Moisture ratio curves for all the four levels of temperatures showed that the drying of 70oC was faster than other drying temperatures. It was also found that the drying rate was higher for higher air temperature and the drying was fast in case of fluidized bed dryer as compare to other dryers. The constant drying rate period was not observed and the complete drying process took place in the falling drying rate period which shows that diffusion is a dominant physical mechanism which governs moisture movement in the samples. Similar results were also obtained by other authors working with other foods.13,32
 |
Figure 1: Variation in moisture ratio with drying time in solar dryer Click here to View figure |
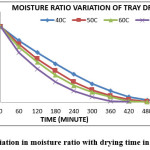 |
Figure 2: Variation in moisture ratio with drying time in tray dryer Click here to View figure |
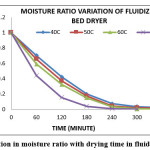 |
Figure 3: Variation in moisture ratio with drying time in fluidized bed dryer Click here to View figure |
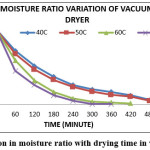 |
Figure 4: Variation in moisture ratio with drying time in vacuum dryer Click here to View figure |
Effective Moisture Diffusivity
The result indicate that internal mass transfer resistance controls the drying time therefore the falling rate period dominated the drying process. For estimation of diffusion coefficients (Deff), the slope of ln(MR) versus time (Figures 5 to 8) was used. The value of effective moisture diffusivity is shown in Table 2. The effective diffusivity was determined to be 7.14 × 10-9 to 1.75 × 10-8 m2/s for the solar drying, 8.44 × 10-9 to 2.19 × 10-8 m2/s for vacuum drying, 1.31 × 10-8 to 1.95 × 10-8 m2/s for tray drying and 1.85 × 10-8 to 3.70 × 10-8 m2/s for fluidized bed drying in the temperature range of 40–70ºC. It was found that the effective diffusivity of samples increased with increase in drying air temperature, which may be because of the increase in diffusion with the increase of temperature of samples.33 The values of effective moisture diffusivity obtained from this study are in the range of 10-12 to 10-8 for biological materials.34
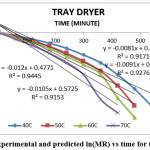 |
Figure 5: Experimental and predicted ln(MR) vs time for tray dryer Click here to View figure |
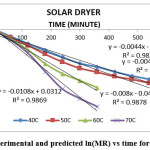 |
Figure 6: Experimental and predicted ln(MR) vs time for solar dryer Click here to View figure |
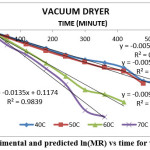 |
Figure 7: Experimental and predicted ln(MR) vs time for vacuum dryer Click here to View figure |
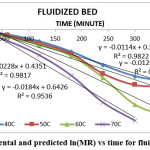 |
Figure 8: Experimental and predicted ln(MR) vs time for fluidized bed dryer Click here to View figure |
Table 2: Effective moisture diffusivity at different conditions
| Temp. (oC) | Dryers | Equation | ko | Deff (m2/s) |
| 40 | Vacuum | y = -0.0052x – 0.0242 | -0.0052 | 8.44 × 10-9 |
| Tray | y = -0.0081x + 0.5532 | -0.0081 | 1.31 × 10-8 | |
| Solar | y = -0.0044x – 0.1463 | -0.0044 | 7.14 × 10-9 | |
| Fluid | y = -0.0114x + 0.2527 | -0.0114 | 1.85 × 10-8 | |
| 50 | Vacuum | y = -0.0056x – 0.0534 | -0.0056 | 9.09 × 10-9 |
| Tray | y = -0.0091x + 0.478 | -0.0091 | 1.48 × 10-8 | |
| Solar | y = -0.0047x – 0.2156 | -0.0047 | 7.63 × 10-9 | |
| Fluid | y = -0.0129x + 0.2558 | -0.0129 | 2.09 × 10-8 | |
| 60 | Vacuum | y = -0.0097x – 0.0313 | -0.0097 | 1.57 × 10-8 |
| Tray | y = -0.0105x + 0.5725 | -0.0105 | 1.70 × 10-8 | |
| Solar | y = -0.008x – 0.0466 | -0.0080 | 1.30 × 10-8 | |
| Fluid | y = -0.0184x + 0.6426 | -0.0184 | 2.99 × 10-8 | |
| 70 | Vacuum | y = -0.0135x + 0.1174 | -0.0135 | 2.19 × 10-8 |
| Tray | y = -0.012x + 0.4771 | -0.0120 | 1.95 × 10-8 | |
| Solar | y = -0.0108x + 0.0312 | -0.0108 | 1.75 × 10-8 | |
| Fluid | y = -0.0228x + 0.4351 | -0.0228 | 3.70 × 10-8 |
Activation Energy
The activation energy for the drying methods was calculated by using the curves between ln(Deff) and 1/(T+273.15) (Figures 9 to 12). By using slope of the straight lines, the activation energy was found to 11.797 kJ/mol for tray dryer, 21.687 kJ/mol for fluidized bed dryer, 28.667 kJ/mol for solar dryer 30.318 kJ/mol.
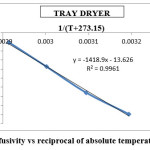 |
Figure 9: Effective diffusivity vs reciprocal of absolute temperature for tray dryer Click here to View figure |
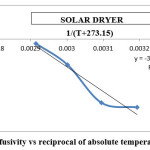 |
Figure 10: Effective diffusivity vs reciprocal of absolute temperature for solar dryer Click here to View figure |
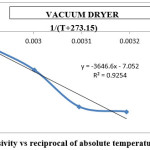 |
Figure 11: Effective diffusivity vs reciprocal of absolute temperature for vacuum dryer Click here to View figure |
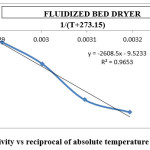 |
Figure 12: Effective diffusivity vs reciprocal of absolute temperature for fluidized bed dryer Click here to View figure |
Validity of drying models
As there was absence in constant rate period in complete drying of Asparagus roots, so the models which describe the falling rate period phenomenon of drying were only attempted. The standard error of estimation was less in Page’s model for all conditions of experiments as well as the coefficient of determination was high but very closely near to logarithmic model in 16 experiments. Therefore, on the basis of R2 and SEE values, both the logarithmic model and the page’s model were observed to be more satisfactory compare to other two models for experimental data. To analyze further about the validity of one these two models namely the logarithmic and the Page’s models, the variation of moisture ratio with time for observed and predicted values were plotted. This is because of the fact that for best fit drying model, the latter part of the moisture ratio curve is more important that initial part of the curve. The residual of observed and predicted MR (MR in the logarithmic and the Page’s models) are compared. Standard error of estimation of moisture ratio for the sample set was minimum for Page’s model followed by logarithmic model. Therefore the Page’s model was selected as the best fit curve. The value of statistical parameters for drying models is shown in Table 3 and the value of the regression coefficient for drying models are shown in Table 4.
Table 3: Statistical parameters for drying models
| Temp. (oC) | Dryers | Exponential model | Page’s model | Generalized Exponential model | Logarithmic model | ||||
| R2 | SEE | R2 | SEE | R2 | SEE | R2 | SEE | ||
| 40 | Vacuum | 0.9984 | 0.0172 | 0.9989 | 0.0149 | 0.9985 | 0.0177 | 0.9991 | 0.0147 |
| Tray | 0.9828 | 0.0645 | 0.9976 | 0.0254 | 0.9855 | 0.0628 | 0.9983 | 0.0233 | |
| Solar | 0.9969 | 0.0235 | 0.9989 | 0.0143 | 0.9972 | 0.0236 | 0.9999 | 0.0047 | |
| Fluid | 0.9889 | 0.0560 | 0.9995 | 0.0131 | 0.9901 | 0.0579 | 0.9957 | 0.0427 | |
| 50 | Vacuum | 0.9985 | 0.0169 | 0.9996 | 0.0086 | 0.9987 | 0.0168 | 0.9995 | 0.0116 |
| Tray | 0.9898 | 0.0499 | 0.9985 | 0.0207 | 0.9911 | 0.0495 | 0.9992 | 0.0164 | |
| Solar | 0.9961 | 0.0268 | 0.9981 | 0.0199 | 0.9963 | 0.0276 | 0.9997 | 0.0078 | |
| Fluid | 0.9911 | 0.0502 | 0.9992 | 0.0168 | 0.9919 | 0.0527 | 0.9964 | 0.0392 | |
| 60 | Vacuum | 0.9993 | 0.0133 | 0.9998 | 0.0058 | 0.9993 | 0.0138 | 0.9995 | 0.0134 |
| Tray | 0.9929 | 0.0412 | 0.9986 | 0.0198 | 0.9937 | 0.0415 | 0.9994 | 0.0142 | |
| Solar | 0.9982 | 0.0206 | 0.9989 | 0.0179 | 0.9984 | 0.0218 | 0.9984 | 0.0244 | |
| Fluid | 0.9946 | 0.0388 | 0.9988 | 0.0202 | 0.9949 | 0.0413 | 0.9985 | 0.0250 | |
| 70 | Vacuum | 0.9983 | 0.0204 | 0.9988 | 0.0185 | 0.9984 | 0.0220 | 0.9984 | 0.0246 |
| Tray | 0.9957 | 0.0326 | 0.9985 | 0.0194 | 0.9960 | 0.0341 | 0.9996 | 0.0120 | |
| Solar | 0.9970 | 0.0283 | 0.9996 | 0.0116 | 0.9972 | 0.0299 | 0.9981 | 0.0275 | |
| Fluid | 0.9982 | 0.0236 | 0.9999 | 0.0061 | 0.9982 | 0.0260 | 0.9993 | 0.0194 | |
Table 4: Regression coefficient for drying models
| Temp. (oC) | Dryers | Exponential model | Page’s model | Generalized Exponential model | Logarithmic model | ||||
| K | K | n | A | k | a | k | C | ||
| 40 | Vacuum | 0.00539 | 0.00739 | 0.9412 | 0.9894 | 0.00533 | 0.9750 | 0.0057 | 0.0221 |
| Tray | 0.00454 | 0.00047 | 1.4093 | 1.0632 | 0.00481 | 1.3526 | 0.0027 | -0.3350 | |
| Solar | 0.00532 | 0.00983 | 0.8864 | 0.9808 | 0.00523 | 0.9369 | 0.0063 | 0.0652 | |
| Fluid | 0.00815 | 0.00104 | 1.4135 | 1.0413 | 0.00844 | 1.1562 | 0.0064 | -0.1337 | |
| 50 | Vacuum | 0.00608 | 0.00976 | 0.9104 | 0.9843 | 0.00598 | 0.9580 | 0.0066 | 0.0371 |
| Tray | 0.00549 | 0.00103 | 1.3102 | 1.0447 | 0.00572 | 1.2194 | 0.0038 | -0.2079 | |
| Solar | 0.00618 | 0.01165 | 0.8796 | 0.9828 | 0.00607 | 0.9437 | 0.0073 | 0.0606 | |
| Fluid | 0.00908 | 0.00148 | 1.3685 | 1.0331 | 0.00936 | 1.1189 | 0.0074 | -0.1009 | |
| 60 | Vacuum | 0.00963 | 0.00608 | 1.0943 | 1.0094 | 0.00972 | 1.0188 | 0.0094 | -0.0121 |
| Tray | 0.00611 | 0.00164 | 1.2548 | 1.0341 | 0.00630 | 1.1448 | 0.0047 | -0.1370 | |
| Solar | 0.00828 | 0.00538 | 1.0866 | 1.0122 | 0.00838 | 1.0150 | 0.0083 | -0.0034 | |
| Fluid | 0.01009 | 0.00285 | 1.2609 | 1.0207 | 0.01026 | 1.0861 | 0.0085 | -0.0780 | |
| 70 | Vacuum | 0.01227 | 0.01848 | 0.9133 | 0.9918 | 0.01218 | 0.9913 | 0.0122 | 0.0006 |
| Tray | 0.00754 | 0.00304 | 1.1774 | 1.0194 | 0.00767 | 1.0954 | 0.0061 | -0.0943 | |
| Solar | 0.00989 | 0.00365 | 1.2060 | 1.0175 | 0.01003 | 1.0479 | 0.0091 | -0.0368 | |
| Fluid | 0.01496 | 0.00529 | 1.2304 | 1.0081 | 0.01505 | 1.0370 | 0.0137 | -0.0327 | |
Discussion
The present study concludes that the drying of Asparagus was carried out only in the falling rate stage which shows that the moisture removed from the product was governed by diffusion phenomenon. The drying was fast in case of the fluidized bed dryer. Page’s model was best for prediction of the data. The drying time of Asparagus decreases and the effective diffusivity increases as the air temperature was increased. The highest effective diffusion was found to be 3.70 × 10-8 m2/s for a fluidized bed dryer at temperature 70°C. The lowest effective diffusion was 7.14 × 10-9 m2/s for a solar dryer at temperature 40°C. Activation energy was found to 11.797 kJ/mol for tray dryer, 21.687 kJ/mol for fluidized bed dryer, 28.667 kJ/mol for solar dryer 30.318 kJ/mol. This activation energy is the main basic consideration for the design of any drying system and calculation of required drying energy.
Acknowledgments
This research work was technically supported by the G.B. Pant University of Agriculture and Technology, Pantnagar, India.
References
- Lau M. H., Tang J., Swanson B. G. Kinetics of Textural and Colour Changes in Green Asparagus During Thermal Treatments. Journal of Food Engineering: 45: 231–236: (2000).
CrossRef - Bopana N., Saxena S. Asparagus Racemosus – Ethnopharmacological Evaluation and Conservation Needs. Journal of Ethnopharmacology: 110: 1–15: (2007).
CrossRef - Sharma, A., Sharma, V. A Brief Review of Medicinal Properties of Asparagus Racemosus (Shatawari). International Journal of Pure & Applied Bioscience: 1(2): 48-50: (2013).
- An J., Zhang M., Wang S., Tang J. Physical, Chemical and Microbiological Changes in Stored Green Asparagus Spears as Affected by Coating of Silver Nano Particles-PVP. Lebensmittel-Wissenschaft and –Technology: 41: 1100–1107: (2008).
- Albanese, D., Russo, L., Cinquanta, L., Brasiello, A., Di Matteo, M. Physical and Chemical Changes in Minimally Processed Green Asparagus During Cold Storage. Food Chemistry: 101: 274–280: (2007).
CrossRef - Mrkic V., Cocci E., Rosa M. D., Sacchetti G. Effect of Drying Conditions on Bioactive Compounds and Antioxidant Activity of Broccoli (Brassica oleracea). Journal of the Science of Food and Agriculture: 86: 1559–1561: (2006).
CrossRef - Xiao H. W., Yao X. D., Lin H., Yang W. X., Meng J. S., Gao Z. J. Effect of SSB (Superheated Steam Blanching) Time and Drying Temperature on Hot Air Impingement Drying Kinetics and Quality Attributes of Yam Slices. Journal of Food Process Engineering: 35: 370–390: (2012).
CrossRef - Mate J. I., Zwietering M., van’t Riet K. The Effect of Blanching on the Mechanical and Rehydration Properties of Dried Potato Slices. European Food Research and Technology: 209: 343–347: (1999).
CrossRef - Mujumdar, A. S. Advances in Drying, Volume II. Washington, New York. (1995).
- Erenturk S., Erenturk K. Comparison of Genetic Algorithm and Neural Network Approaches for the Drying Process of Carrot. Journal of Food Engineering: 78: 905–912: (2007).
CrossRef - Aghbashlo M., Kianmehr M. H., Samimi A. H. Influence of Drying Conditions on the Effective Moisture Diffusivity, Energy of Activation and Energy Consumption During the Thin-Layer Drying of Beriberi Fruit (Berberidaceae). Energy Conversion and Management: 49: 2865–2871: (2008).
CrossRef - Gupta M. K, Sehgal V. K., Arora S. Optimization of Drying Process Parameters for Cauliflower Drying. Journal of Food Science and Technology: 50: 62–69: (2013).
CrossRef - Zielinska M., Markowski M. Air Drying Characteristics and Moisture Diffusivity of Carrots. Chemical Engineering and Processing: 49: 212–218: (2010).
CrossRef - Kohli D., Shahi N.C., Pandey J.P., Singh A. Drying of Asparagus Roots in Solar and Fluidised Bed Dryer. International Journal of Agriculture Sciences: 9(13): 4072-4076: (2017).
- Diamante L.M. Drying Characteristics of Sweet Potato Slices. In: Proceedings of International Conference of Preservation and Security, National Academy of Science and Technology, Philippines: 187–199: (1994).
- Ranganna, S. Handbook of Analysis and Quality Control for Fruit and Vegetable Product, 2nd Tata McGraw hill publication Co. Ltd., New Delhi: 112: (1986).
- Janjai, S., Tang, P. Performance of a Solar Dryer using Hot Air from Roof Integrated Solar Collectors for Drying Herbs and Spices. Renewable Energy: 30(14): 2085-2095: (2005).
CrossRef - Jokic S., Mujic I., Martinov M., Velic D., Bilic M., Lukinac J. Influence of Drying Procedure on Colour and Rehydration Characteristic of Wild Asparagus. Czech J. Food Sci: 27: 171-177: (2009).
- Bala, B. K., Hoque, M. A., Hossain, M. A., Uddin, M. B. Drying Characteristics of Asparagus Roots (Asparagus racemosus Wild.). Drying Technology: An International Journal: 28(4): 533-541: (2010).
CrossRef - Nindo C. I., Sun T., Wang S. W., Tanga J., Powers J.R. Evaluation of Drying Technologies for Retention of Physical Quality and Antioxidants in Asparagus (Asparagus officinalis L.). Lebensmitel-Wissenschaft und –Technologie: 36: 507–516: (2003).
- Erenturk S., Gulaboglu M. S., Gultekin S. The Thin Layer Drying Characteristics of Rosehip. Eng: 89(2): 159-166: (2004).
CrossRef - Midilli A. Determination of Pistachio Drying Behaviour and Conditions in Solar Drying Systems. Int. J. Energy Res: 25: 715-725: (2001).
CrossRef - Doymaz I. Effect of Pre-Treatments Using Potassium Metabisulphite and Alkaline Ethyloleate on the Drying Kinetics of Apricots. Eng: 89(3): 281-287: (2004).
- Goyal R. K., Kingsly A. R. P., Manikanthan M. R., Ilyas S. M. Mathematical Modeling of Thin Layer Drying Kinetics of Plum in a Tunnel Dryer. Food Eng: 79: 176-180: (2007).
CrossRef - Doymaz Thin Layer Drying Behaviour of Mint Leaves. J. Food Eng: 74: 370-375: (2006).
CrossRef - Crank J. Mathematics of Diffusion. UK: Clarendon Press, oxford. (1975).
CrossRef - Akpinar E., Midilli A., Bicer Y. Single Layer Drying Behavior of Potato Slices in a Convective Cyclone and Mathematical Modeling. Energy Conversion and Management: 44: 1689–1705: (2003).
- Henderson S. M. Progress in Developing the Thin Layer Drying Equation. Transactions of the ASAE. 17: 1167–1172: (1974).
CrossRef - Zhang Q, Litchfleld J. B. An Optimization of Intermittent Corn Drying in a Laboratory Scale Thin Layer Dryer. Dry Technol: 9: 383–395: (1991).
- Guarte R. C. Modelling the drying behaviour of copra and development of a natural convection dryer for production of high quality copra in the Philippines. Ph.D. dissertation, 287. Hohenheim University, Stuttgart, Germany. (1996).
- Karathanos V.T. Determination of Water Content of Dried Fruits by Drying Kinetics. J Food Eng: 39: 337–344: (1999).
CrossRef - Madhiyanon T., Phila A., Soponronnarit A. Models of Fluidized Bed Drying for Thin-Layer Chopped Coconut. Applied Thermal Engineering: 29: 2849–2854: (2009).
CrossRef - Diamante L. M., Ihns R., Savage G. P., Vanhanen L. A New Mathematical Model for Thin Layer Drying Fruits. International Journal of Food Science and Technology: 45: 1956–1962: (2010).
CrossRef - Zogzas N.P., Maroulis Z.B., Marinos-Kouris D. Moisture Diffusivity Data Compilation in Foodstuffs. Drying Technology: 14: 2225–2253: (1996).
CrossRef













