Stress-Driven Changes in Ascorbic Acid Levels in Raphanus Sativus: A Comparative Study
Department of Life Sciences, Christ University, Bangalore, Karnataka, India
Corresponding Author Email: mridul.umesh@christuniversity.in
DOI : http://dx.doi.org/10.12944/CRNFSJ.14.1.26
ABSTRACT:Abiotic stresses such as extreme temperatures and salinity are known to significantly influence the nutritional quality of vegetables during cultivation and postharvest handling. However comparative study on how different abiotic stresses alter ascorbic acid stability in root vegetables like red radish is understudied. Thus, the present study was conducted to evaluate the effect of various abiotic stresses (heat, cold and salinity) on ascorbic acid (AA) levels and antioxidant activity in red radish (Raphanus sativus L.). DCPIP titration and DNPH UV-Vis spectrophotometric techniques were used to analyse the ascorbic acid content, and the DPPH radical scavenging assay was used to measure antioxidant capacity. The findings showed that as stress duration and intensity increased, the total AA content decreased significantly (p < 0.05). At high temperature (90 °C), heat treatment resulted in a progressive decrease from 27.7 mg/100g to 20.8 mg/100g. Under cold stress, AA content increased slightly initially and then dropped by 22% on day 8. A two-phase response to salt stress was seen mild salt concentration leading to a moderately increased AA content, while severe salt concentration caused a 55% reduction after 72 hours. With an IC₅₀ value of 8.27mg/mL, the antioxidant activity increased as extract concentration increased from 3.8% to 62.1% inhibition. According to these findings, red radish exhibits a short-term adaptive defence in mild stress situations but a significant decrease in AA in severe stress situations. These findings are consistent with previous reports in other related vegetables such as tomato, broccoli and spinach, where moderate abiotic stress increases antioxidant defences before degradation occurs. These results suggest the need for efficient postharvest management and stress-aware storage techniques to preserve the functional and health-promoting qualities of red radish.
KEYWORDS:Abiotic Stress; Antioxidant Activity; Ascorbic Acid (AA); Reactive Oxygen Species (ROS); Red Radish (Raphanus Sativus)
Introduction
With the increasing focus on food security and nutritional value at the global scale, it is now a priority in food and nutrition science to know how the health-benefiting compounds in crops are influenced by environmental stress. Modern agriculture is increasingly facing threats of abiotic stress factors such as salinity, heat, and cold that constrain plant production and affect the postharvest nutritional quality of vegetables being consumed globally.
Ascorbic acid (AA) or vitamin C is a vital water-soluble vitamin that is a key nutrient in both plant physiology and human nutrition, first isolated and purified by Szent-Györgyi in 1928. It exists in two forms: L-ascorbic acid, which is a reduced form and L-dehydroascorbic acid (DHAA) oxidised form. AA can neutralise reactive oxygen species (ROS) and acts as a coenzyme in various biosynthetic processes, such as forming cell wall structures, producing plant hormones like gibberellins and ethylene, and synthesising secondary metabolites like pigments and alkaloids.1 Ascorbic acid is an essential micronutrient for humans, which cannot be synthesized in the body due to the lack of the gene encoding for L-gulonolactone oxidase. The body uses sodium-dependent vitamin C transporters (SVCT1 and SVCT2) to distribute vitamin C effectively and maintain redox balance encoded by the SLC23 family.2 A deficiency in vitamin C can cause scurvy, a disease that weakens connective tissues, causes anaemia, and weakens immunity, underlining the importance of overall health and well-being.3
Plants do produce AA through the Smirnoff-Wheeler pathway, with the help of major mitochondrial enzymes L-galactono-1,4-lactone dehydrogenase (GLDH) that converts L-galactono-1,4-lactone into ascorbate. Several environmental stressors, including salinity, temperature, light, and growth cues, might influence this process. AA influences gene expression of stress related proteins such as superoxide dismutase (SOD) and catalase.1 Studies indicate exogenous AA has been reported to increase stress tolerance of crops like wheat, maize, and rice by improving osmotic adjustment, antioxidant activity and membrane integrity under drought, salinity and heat stress.4
Red radish is a root crop of the Brassicaceae family known for its rich phytochemical and nutritional content, often used as traditional medicine and cuisine. Studies show that AA content in red radish varies between 20-40mg/100g in roots, even more in leaves, which makes them a very effective food source of vitamin C.5 However, its physiological and nutritional responses to environmental stress are less understood when it comes to AA modulation by abiotic stress.
It is essential to understand how AA’s biochemistry reacts to environmental stressors to develop functional foods and stress-resistant vegetables. Several studies have shown that moderate abiotic stress may increase antioxidant defences, even though prolonged or extreme stress frequently results in severe reductions in AA through oxidative destruction or repression of biosynthetic processes. Additionally, post-harvest storage has been shown to significantly lower levels of ascorbic acid through oxidative breakdown in cold conditions. Galani et al. reported significant drops in vitamin C content and antioxidant activity in fruits and vegetables stored at 4 °C, underlining the need for an immediate extraction after harvest for precise determination.6
Recent climate fluctuations have increased the frequency of temperature, drought and salinity worldwide which directly threatens the nutritional quality and shelf life of vegetables. Many global studies have reported vitamin C reduction under abiotic stress in crops like tomato, broccoli, spinach and pepper where high temperature, extended cold storage and salinity were observed to drastically reduce the ascorbic acid content.1,7 Despite this global research the biochemical response of Ascorbic acid levels of Raphanus sativus to different stress levels is still not well understood. Filling this knowledge gap is important for creating storage and processing methods that can maintain nutritional value.
With increased focus on climate-resistant crops and functional food production, it is important to know the mechanism of AA content under abiotic stress conditions to ensure the nutritional quality of the vegetables. AA not only enhances plant stress tolerance but also acts as a primary nutritional marker for the consumer.8 Thus, the determination of the effect of abiotic stress on AA content and antioxidant activity in red radish is of agricultural, food science and nutrition practical significance. This research presents a new, integrated method by treating red radish to heat, cold, and salt stress, each mimicking actual postharvest or climatic stresses, and contrasting AA degradation with two complementary estimation methods: DCPIP titration (AOAC-standardised) and DNPH UV-Vis spectrophotometry. In addition, antioxidant activity was determined by the DPPH assay. Through assessment of red radish functional integrity under short- and long-duration stress, this research offers a novel biochemical insight into stress-induced spoilage of nutrients in vegetables and provides insights for climate-resilient crop improvement as well as functional food development.
Materials and Methods
The contents of ascorbic acid were estimated by applying both the titration method using DCPIP and DNPH spectrophotometry to ensure both accuracy and sensitivity. The DCPIP method, which is based on the redox reaction between ascorbate and 2,6-dichlorophenol-indophenol, is quick and accurate for estimating total ascorbate, while in the DNPH method, both oxidised and reduced forms are measurable which gives a full picture of redox shifts due to stress.9,10
Among different methods that exist for quantifying antioxidant capacity, the DPPH assay was chosen due to its widespread use in stress studies and its ability to provide an indication of free radical scavenging efficiency.11,12 These methods together provide a robust view of how abiotic stress alters ascorbic acid concentration and functional antioxidant potential in red radish.
Materials
Fresh red radishes (Raphanus sativus) were collected from a local market in Bengaluru, India. All reagents used were of analytical grade. 2,6-dichlorophenol-indophenol (DCPIP), 2,4-dinitrophenylhydrazine (DNPH), thiourea, DPPH(2,2-diphenyl-1-picrylhydrazyl), oxalic acid, sulfuric acid, methanol and NaCl.
Stress Treatments
All experiments were performed under controlled laboratory conditions, and each treatment consisted of three independent biological samples and was performed in triplicate (n=3). Untreated fresh R. sativus samples served as controls for each type of stress. The edible root portion of red radish was used for all analyses. Leaves were removed, and roots were washed thoroughly, peeled and sliced before stress treatments.
Heat stress: samples were exposed to 45°C, 65°C, and 90°C for 1 hour.
Cold stress: samples were stored at 4°C for 1, 4, 6, and 8 days in a refrigerated chamber.
Salt stress: slices were treated with 100,150, 200mM NaCl solutions for 24 and 72 hours at room temperature to replicate osmotic stress conditions.
Ascorbic Acid Extraction
Two separate extractions were followed to ensure accuracy and comparability. For titrimetric analysis, 5g of red radish samples were homogenised in 4% oxalic acid and made up to a volume of 25 mL, and from that, 5ml of an aliquot was diluted with 15ml of oxalic acid before titration. For spectrophotometric analysis, 1g of red radish was extracted with 25ml of 5% HPO3, further centrifuged at 7000rpm for 10 minutes and processed immediately.8
2,6-dichlorophenol-indophenol (DCPIP) Titrimetric Estimation of Ascorbic Acid
Ascorbic acid was measured by the DCPIP titration procedure, following the official AOAC (2005) method with adaptations made to fit red radish tissue.9 The sample was titrated against 0.02% DCPIP solution until a pale pink endpoint. The volume of dye used was noted, and the ascorbic acid level was determined using a calibration curve of standard ascorbic acid. Results were reported as mg of ascorbic acid/100 g fresh weight (mg/100g FW).
2,4-Dinitrophenylhydrazine UV-Vis spectrophotometric estimation
The DNPH procedure measures both oxidised and reduced AA. 2ml of the extract was combined with 1ml of 0.02% DCPIP, 2ml of 2% thiourea and 1ml of 2% DNPH (made in 4.5M H₂SO₄). And incubated at 50 °C for 70 minutes, and 4ml of 85% sulfuric acid was added to it after cooling. Absorbance was read at 520nm in a UV-Vis spectrophotometer. The level of ascorbic acid was determined by a standard curve with R² ≥ 0.99. This approach, according to Niroula et al. (2021), was adapted to fit the red radish matrix and sensitivity needs.10
DPPH Radical Scavenging Assay
The method of Akinmoladun et al. (2010) was used for the determination of the scavenging activity of DPPH free radical in the extract solution.11 Extracts were tested at different concentrations in methanol. Absorbance was read at 517 nm, and the percentage inhibition was calculated relative to the blank using the formula:
DPPH Scavenging activity (%) = [(Abscontrol − Abssample)] / (Abscontrol)] × 100
Where Abscontrol is the absorbance of DPPH + methanol; Abssample is the absorbance of DPPH radical + sample extract or standard.
Statistical analysis
All the experiments were performed in triplicate (n=3). Data were analysed using SPSS IBM (version 27.0). Results were expressed as mean ± standard deviation (SD). Data analysis was carried out by one-way ANOVA. All evaluations were done at 5% significance level (p-value < 0.05), and Confidence intervals (95%) were calculated where applicable.13
Results
Effect of heat stress on ascorbic acid content:
The AA content in fresh red radish was found to be 27.7 ± 1.2 mg/100 g. When exposed to heat stress gradual decline in the levels of ascorbic acid was seen as the temperature increased. At 45°C, there was a slight decrease to 26.0 ± 1.7 mg/100 g. As the temperature increased to 65°C and 90°C, it showed a noticeable decline, 23.5 ± 2.6 and 20.8 ± 3.7 mg/100 g, respectively (Fig. 1A). The reduction was statistically significant (p < 0.05).
Effect of cold stress on ascorbic acid content:
Under cold storage at 4°C, AA content initially increased from 24.7 ± 2.7 mg/100 g to 25.0 ± 2.5 mg/100 g on day 2 (Fig. 1B). However, a progressive decline was observed thereafter, with values decreasing to 21.0 ± 2.0 mg/100 g on day 4 and 19.2 ± 3.2 mg/100 g on day 8. The reduction observed after prolonged storage was statistically significant (p < 0.05).
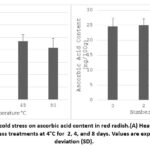 |
Figure 1: Effect of heat and cold stress on ascorbic acid content in red radish.(A) Heat stress treatments at 45°C, 65°C, and 90°C, (B) Cold stress treatments at 4°C for 2, 4, and 8 days. Values are expressed as mean ± standard deviation (SD). |
Effect of salt stress on ascorbic acid content:
Salt stress for 24hours showed results AA based upon the salt concentration, at mild stress (100mM) showed increased levels 25.9 ± 3.8 to 26.4 ± 4.6 mg/100 g. This is possible due to stress-induced upregulation of biosynthesis as an adaptive response. However, when exposed to 150mM and 200mM, the ascorbic acid levels declined to 24.6 ± 5.4 and 20.1 ± 7.6 mg/100 g. Extended salt stress for 72hours affected the degradation of AA more significantly. Whereas the fresh sample had AA content of 34.3 mg/100g, levels decreased significantly at 100mM, 150mM and 200mM. This loss was time and concentration-dependent (p < 0.05) (Fig. 2). It showed a 55% loss at the highest stress level.
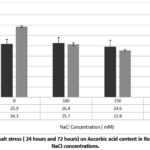 |
Figure 2: Effect of salt stress ( 24 hours and 72 hours) on Ascorbic acid content in Red Radish at varying NaCl concentrations. |
Antioxidant activity
The antioxidant activity of red radish extract increased in a concentration-dependent manner as determined by the DPPH radical scavenging assay (Fig. 3). Percentage inhibition increased from 3.8% to 62.1% with increasing extract concentration. The IC₅₀ value was calculated to be 8.27 mg/mL.
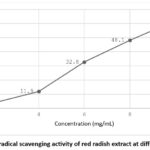 |
Figure 3: DPPH radical scavenging activity of red radish extract at different concentrations |
Discussion
Heat stress
The present findings suggest that heat exposure reduces ascorbic acid content in a temperature-dependent manner in red radish. This trend is in line with other research on vegetables and fruits. In radish leaves, leafy greens and spinach, similar temperatures led to declines in AA were observed, with losses of 16% to 51% depending upon varying cooking conditions.14 Similarly, cabbage and lettuce exhibited fast AA degradation when treated at 70°C, with first-order kinetics.15 In tropical fruits pulps like guava, papaya, ohmic heating resulted in a continuous loss of ascorbic acid levels, depending on the temperature of the heat.16 Thus thermal denaturation of enzymes involved in the AA recycling system including dehydroascorbate reductase may be attributed to further cause of the losses observed.17 The results confirm that vitamin C retention compromised by high temperature processing and emphasise the need for mild temperature treatments during cooking or drying.
Cold stress
The biphasic pattern most likely represents postharvest metabolic adjustment in which antioxidant enzymes are momentarily activated by low temperatures before oxidative degradation.18 This trend aligns with previous findings, where storage induced a drastic decrease in AA content in tomato with a loss of 71.8%.6 A similar type of kinetics was seen in capsicum showed a decrease in vitamin C after 13 days of storage.19 Some studies have reported that AA content increased in the immediate postharvest period but decreased gradually later. Mangaba fruits, for example, increased slightly during the first few days of storage but decreased by 50% after being exposed for a longer period of time.20 Similarly, fresh peas and broccoli maintained AA levels during short-term cold storage and then showed a 20% loss over 14 days.21 This prolonged storage has been associated with enhanced oxidative degradation of ascorbic acid leading to reduced vitamin C levels.22 Thus, these findings indicate that short-term exposure to cold may slightly increase AA, but prolonged exposure accelerates its oxidative degradation.
Salt stress
The findings are consistent with the biphasic salt stress response seen in various plant species Chen et al., and Neumann et al. Mild salt exposure increases ascorbate biosynthesis through increased expression of L-galactono-1,4-lactone dehydrogenase in tomato, whereas a slight increase is due to the oxidative damage and AA depletion.4,23 The same trend was seen in brassica crops, moderate salinity first promoted AA accumulation, but then caused a loss with prolonged and severe exposure.24 Therefore, red radish results show a widespread physiological response, where low salinity initiates antioxidant upregulation, but high salt stress leads to ascorbic acid degradation.
Antioxidant activity
Antioxidant activity of extracts of red radish was determined with the DPPH radical scavenging assay. This trend reflects a common dose-dependent antioxidant pattern such that higher concentrations of extracts were associated with greater radical scavenging capacity (Fig. 3). The derived IC₅₀ value was about 8.27mg/mL, reflecting moderate antioxidant activity. These observations were similar to earlier studies in Raphanus sativus, Broccoli and other cruciferous vegetables, where DPPH activity is highly correlated with phytochemical and ascorbic acid contents.25 The research of Goyeneche et al (2015), found antioxidant potential in roots and leaves of Raphanus sativus antioxidant capacity of 1.36 mmol TE/100g in roots and greater in leaves ratio of 1:3.5 Although their research employed a Trolox standard for TE unit expression, the pattern of lower antioxidant capacity in roots than in leaves is in agreement with present study.
Conclusion
In this study, it was determined that red radish ascorbic acid content is under the direct impact of abiotic stress conditions. Extreme heat, cold or salt stress resulted in declines in ascorbic acid levels, whereas mild stress, short-term cold or low salt temporarily increased AA biosynthesis as a sort of adaptive response. The antioxidant activity, determined by DPPH radical scavenging, agreed with AA levels and proved that red radish maintains functional antioxidant activity under a variety of stress levels. These results show the control of ascorbic acid under stress and its central role in plant defence. Practical uses of these outcomes in post-harvest management, food processing and improvement of crops bring into focus the need for regulating temperature and salinity to prevent degradation of vitamin C levels during storage and to retain nutritional quality.
In addition, they provide useful guidance for stress tolerance screening in crops. Further studies on molecular pathways of AA regulation including the role of key enzymes such as L-galactono-1,4-lactone dehydrogenase and ascorbate peroxidase under combined or cyclic stress assess possible ways to promote AA stability and antioxidant defences in red radish and other vegetables related to it.
Acknowledgement
The authors sincerely thank Department of Life Sciences, Christ University, for providing laboratory facilities and institutional support necessary for the successful completion of this research work.
Funding Sources
This work was supported by the Centre for Research Projects, Christ University, under the Internal Seed Money Grant (Minor Project Scheme- Ref No: CU-ORS-SM-24/57). The funding agency had no role in the design of the study; collection, analysis, or interpretation of data; writing of the manuscript; or in the decision to submit the article for publication.
Conflict of Interest
The authors do not have any conflicts of interest.
Data Availability Statement
The data that support the findings of the present study are available from the corresponding authors upon reasonable request.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Materials from Other Sources
Not Applicable
Author Contributions
- Asvika Sridhar: Experimental and writing the initial draft
- Mridul Umesh: Supervision, editing and final revision of the article.
Reference
- Mellidou I, Koukounaras A, Kostas S, Patelou E, Kanellis AK. Regulation of vitamin C accumulation for improved tomato fruit quality and alleviation of abiotic stress. Genes (Basel). 2021;12(5):694.
CrossRef - Bürzle M, Suzuki Y, Ackermann D, et al. The sodium-dependent ascorbic acid transporter family SLC23. Mol Aspects Med. 2013;34(2-3):436-454. doi:10.1016/j.mam.2012.12.002
CrossRef - Abdullah M, Jamil RT, Attia FN. Vitamin C (Ascorbic Acid). StatPearls [Internet] Treasure Island. Published online 2023.
- Shalata A, Neumann PM. Exogenous ascorbic acid (vitamin C) increases resistance to salt stress and reduces lipid peroxidation. J Exp Bot. 2001;52(364):2207-2211. doi:10.1093/jexbot/52.364.2207
CrossRef - Goyeneche R, Roura S, Ponce A, et al. Chemical characterization and antioxidant capacity of red radish (Raphanus sativus L.) leaves and roots. J Funct Foods. 2015;16:256-264. doi:10.1016/j.jff.2015.04.049
CrossRef - Galani JHY, Patel JS, Patel NJ, Talati JG. Storage of fruits and vegetables in refrigerator increases their phenolic acids but decreases the total phenolics, anthocyanins and vitamin C with subsequent loss of their antioxidant capacity. Antioxidants (Basel). 2017;6(3):59. doi:10.3390/antiox6030059
CrossRef - Feszterová M, Kowalska M, Mišiaková M. Stability of vitamin C content in plant and vegetable juices under different storing conditions. Appl Sci (Basel). 2023;13(19):10640. doi:10.3390/app131910640
CrossRef - Cho KS, Jeong HJ, Cho JH, et al. Vitamin C content of potato clones from Korean breeding lines and compositional changes during growth and after storage. Hortic Environ Biotechnol. 2013;54(1):70-75. doi:10.1007/s13580-013-0089-8
CrossRef - Nielsen SS. Vitamin C Determination by Indophenol Method. In: Food Analysis Laboratory Manual. Springer International Publishing; 2017:143-146. doi:10.1007/978-3-319-44127-6_15
CrossRef - Niroula A, Amgain N, Kc R, Adhikari S, Acharya J. Pigments, ascorbic acid, total polyphenols and antioxidant capacities in deetiolated barley (Hordeum vulgare) and wheat (Triticum aestivum) microgreens. Food Chem. 2021;354(129491):129491. doi:10.1016/j.foodchem.2021.129491
CrossRef - Igbinosa EO, Uzunuigbe EO, Igbinosa IH, Odjadjare EE, Igiehon NO, Emuedo OA. In vitro assessment of antioxidant, phytochemical and nutritional properties of extracts from the leaves of Ocimum gratissimum (Linn). Afr J Tradit Complement Altern Med. 2013;10(5):292-298. https://www.ncbi.nlm.nih.gov/pubmed/24311838
CrossRef - Baliyan S, Mukherjee R, Priyadarshini A, et al. Determination of antioxidants by DPPH radical scavenging activity and quantitative phytochemical analysis of Ficus religiosa. Molecules. 2022;27(4):1326. doi:10.3390/molecules27041326
CrossRef - Yin X, Chen K, Cheng H, et al. Chemical stability of ascorbic acid integrated into commercial products: A review on bioactivity and delivery technology. Antioxidants (Basel). 2022;11(1):153. doi:10.3390/antiox11010153
CrossRef - Mehrotra N, Head, Department of Biochemistry, SVKM’s Mithibai College of Arts, Chauhan Institute of Science & Amrutben Jivanlal College of Commerce and Economics (Autonomous), Vile-Parle (West), Mumbai-400056, Maharashtra, India. Effects of thermal treatments on ascorbic acid content of leafy vegetables: An in vitro analysis. jpps. 2021;1(1):10-15. doi:10.21276/jpps.2021.1.1.3
CrossRef - f A, E.o E, A.m K, M.m A. Kinetic modeling of vitamin C (ascorbic acid) degradation in blanched commonly consumed salad vegetables using computer simulation analysis. IOSR J Appl Chem. 2017;10(04):59-66. doi:10.9790/5736-1004015966
CrossRef - Athmaselvi KA, Kumar C, Poojitha P. Influence of temperature, voltage gradient and electrode on ascorbic acid degradation kinetics during ohmic heating of tropical fruit pulp. J Food Meas Charact. 2017;11(1):144-155. doi:10.1007/s11694-016-9381-5
CrossRef - Hasanuzzaman M, Bhuyan MHMB, Anee TI, et al. Regulation of ascorbate-glutathione pathway in mitigating oxidative damage in plants under abiotic stress. Antioxidants (Basel). 2019;8(9):384. doi:10.3390/antiox8090384
CrossRef - Brizzolara S, Manganaris GA, Fotopoulos V, Watkins CB, Tonutti P. Primary metabolism in fresh fruits during storage. Front Plant Sci. 2020;11:80. doi:10.3389/fpls.2020.00080
CrossRef - Rahman MS, Al-Rizeiqi MH, Guizani N, Al-Ruzaiqi MS, Al-Aamri AH, Zainab S. Stability of vitamin C in fresh and freeze-dried capsicum stored at different temperatures. J Food Sci Technol. 2015;52(3):1691-1697. doi:10.1007/s13197-013-1173-x
CrossRef - Carnelossi MAG, Sena HC de, Narain N, Yaguiu P, Silva GF da. Physico-chemical quality changes in mangaba (hancornia speciosa gomes) fruit stored at different temperatures. Braz Arch Biol Technol. 2009;52(4):985-990. doi:10.1590/s1516-89132009000400023
CrossRef - Favell DJ. A comparison of the vitamin C content of fresh and frozen vegetables. Food Chem. 1998;62(1):59-64. doi:10.1016/s0308-8146(97)00165-9
CrossRef - Zhao K, Xiao Z, Zeng J, Xie H. Effects of different storage conditions on the browning degree, PPO activity, and content of chemical components in fresh Lilium bulbs (liliumbrownii F.e.brown var. Viridulum baker.). Agriculture. 2021;11(2):184. doi:10.3390/agriculture11020184
CrossRef - Chen X, Zhou Y, Cong Y, et al. Ascorbic acid-induced photosynthetic adaptability of processing tomatoes to salt stress probed by fast OJIP fluorescence rise. Front Plant Sci. 2021;12:594400. doi:10.3389/fpls.2021.594400
CrossRef - Hernandez M, Fernandez-Garcia N, Diaz-Vivancos P, Olmos E. A different role for hydrogen peroxide and the antioxidative system under short and long salt stress in Brassica oleracea roots. J Exp Bot. 2010;61(2):521-535. doi:10.1093/jxb/erp321.
CrossRef - Li Z, Lee HW, Liang X, et al. Profiling of phenolic compounds and antioxidant activity of 12 cruciferous vegetables. Molecules. 2018;23(5). doi:10.3390/molecules23051139
CrossRef













