Introduction
Selenium is a trace element that is fundamental and essential for several metabolic pathways such as thyroid hormone metabolism, antioxidant defence system and immune function1. Selenium and its compounds are present in food in form of selenoamino acids, selenoproteins, selenide, selenite, and thus, exhibited significant biological activities through iodothyronine deiodinase, glutathione peroxidases, phospholipid hydroperoxide, sperm capsule, selenoprotein and thioredoxin reductase.1,2,3 Selenium compounds inhibit signaling enzymes such as protein kinase C (PKC) that is responsible for tumor promotion and exert antioxidant effect through selenoproteins and thioredoxin reductases.4
The quantity of Se in food depends on their content, varies regionally and nationally with Se level in soil relatively low in some countries.5 Although, soil conditions have been associated with Se toxicity in animals (selenosis) in some areas with high Se content but Se deficiency syndromes occurred in animals and humans in areas of low soil Se.5 Selenium content in foods has been reported as insufficient to facilitate the optimal activity of protective selenoenzymes.6 The nutritional deficiency of Se in human blood lead to some threatening diseases such as cancer, cardiovascular disease, increasing of viral infections, male infertility, asthma, Kashan disease, thyroid dysfunction and osteoarthritis.7 Considering the importance of Se in human diet, it is expedient to increase the Se content in foods to rectify the implication of Se deficiency in the body. One of the best methods is to produce foods with adequate amount of Se such as cultivating edible fungi with Se fortification.
Although, wild and edible mushrooms contain some amount of Se but it concentration varied based on the Se content in soil or growth substrate. Hence, supplementation of Se into Pleurotus mushrooms will improve the antioxidant status, eliminate dietary aliments and suppress the syndromes associated with deficiency of Selenium. Pleurotus spp. have been appreciated as medicinal foods and selected for Se biofortification due to their rapid growth within short time, high volume of production, seasonal independence, safety and wide acceptance by consumers as medicinal foods.8 Therefore, this study is meant to assess the effect of Se on mycelial growth and biological efficiency as well as Se accumulated by edible mushrooms of Pleurotus spp. cultivated on signal grass with Se.
Materials and Methods
Microorganism
The studied fungi namely: Pleurotus ostreatus (P93), Pleurotus ostreatus (PLO6), Pleurotus pulmonarius (Pindo), Pleurotus cornucopiae (Plocor), Pleurotus djamor (PLO13) and Pleurotus djamor var. roseus (Psfarm) were from fungal collection of the Laboratório de Associações Micorrízicas, BIOAGRO, Departamento de Microbiologia, Universidade Federal de Viçosa (UFV), Minas Gerais, Brazil. The fungi were sub cultured on potato dextrose agar (PDA, Merck, Darmstadt, Germany) and incubated at 25±1 °C for seven days.
Effect of Se on mycelial growth rate and biomass of fungi
The fungi were exposed to Se in culture media at different concentrations (0, 50.0 or 100.0 mg L−1). Briefly, one agar disc (8.0 mm) of each actively grown fungi was cut and transferred to the center of culture medium (PDA, Merck, Darmstadt, Germany) containing of Se in form of sodium selenite (Na2SeO3) at pH 5.4 ± 0.1. The plates were incubated at 25 °C ± 1°C for seven days. The fungal growth rate was determined after seven days by measuring the colony’s diameter in two directions that were perpendicular to each other.9
The biomass (dry mass) of the fungi was determined by emptying the entire content of Petri dish (mycelium and culture media) into a bottle with distilled water and heated using microwave (5-10 min) to dissolve culture medium. Thereafter, the solution was filtered and the mycelium was dried in an oven at 60 °C until a constant weight was reached.10
Pleurotus mushroom cultivated on Urochloa decumbens with Selenium
The signal grass was obtained from a farmland in Unai, Minas Gerais State, Brazil. It was dried at room temperature and milled into particle of different sizes (4.0-6.0 mm). The milled signal grass was soaked in lime water solution of 2% Ca(OH)2 for 24 h and centrifuged at 1800 rpm for 5 min to remove excess water.11 Then, experimental units of each fungus were cultivated on signal grass (150 g) supplemented with 50 mg Kg-1 of Se. This concentration was chosen based on the fact that there was minimal inhibition of fungal mycelia, which was supported by the findings of Da Silva et al.,12 who revealed the Se concentration (50 mg Kg-1) for the optimum production of P. ostreatus. The experimental units were incubated at 25 ± 3 ºC for 30 d. After this time, the bags were transferred to a room and incubated for fructification with light, at temperature of 25 ± 3 ºC in 90% air humidity for about 7 days. The biological efficiency (BE) was calculated as follow:
BE = (fresh weight of mushroom / dry weight of substrate used) x 100
Determination of Se content in cultivated mushrooms
Mushrooms were freeze- dried until they reached a constant weight and were ground in a 2-mm sieve mill. Ground sample of each mushroom (500 mg) was placed into the sealed tube, 6 mL of nitric acid (HNO3) was added and left in a block of graphite (brand DigiPREP MS) for 24 h at 65 °C. Thereafter, 3 mL of hydrogen peroxide (H2O2) was added and the block temperature was raised to 120 °C. The resulting contents of the digested sample was transferred to new tubes and the final volume was made up to 10 mL using ultra-pure water (Milli -Q, Millipore 18.2 MΩ cm-1). The Se in the mushroom samples was quantified using atomic absorption spectrometer Varian (Spectra AA 220FS) and compared to Se standard.
Statistical Analysis
Statistical analysis of data obtained from completely randomized experimental design were analysed with the aid of Statistical Package for Social Sciences (SPSS Inc., version 17.0, Chicago, Illinois, USA) using one-way analysis of variance (ANOVA). Mean were compared by Duncan’s New Multiple Range Test and considered statistically significant when P≤0.05.
Results and Discussion
Edible fungi of higher Basidiomycetes, especially Pleurotus spp. are used as alternative source of mineral supplement when there are deficient of minerals in natural foods.13 This is because, they are easily grown within short time and capable of storing essential elements that maintain balanced nutrient in human.14 The growth rate of Pleurotus species on media containing Se, biological efficiency of Pleurotus mushroom growing on signal grass added with Se and Se concentration in the produced mushrooms is reported in this study.
The increase in Se concentration from 0 to 100 mg mL-1 reduced the fungal growth rate and their biomass production (Table 1). The value of fungal growth rate ranged from 1.5 mm d-1 to 3.6 mm d-1 and 1.0 to 3.30 mm d-1 at 50 and 100 mg mL-1 respectively. The growth rate of P. cornucopiae and P. djamor var. roseus were not different (P<0.05) at 50 and 100 mg mL-1 of Se. Mycelial biomass obtained from the edible fungi at 50 and 100 mg mL-1 of Se were within 11.4 g to 30.5 g and 5.63 g to 10.8 g respectively. The biomass produced by P. cornucopiae, P. djamor and P. djamor var. roseus at 50 and 100 mg mL-1 were similar (P<0.05). The reduction in growth rate and biomass production indicated that fortification with higher concentration of Se inhibited the growth and production of Pleurotus spp. The finding of Da Silva et al.,15 revealed decrease in Pleurotus spp. growth rate as the concentration of Se increased from 25.4 mg mL−1 to 101.8 mg mL−1. Although, Se is required for fungi metabolic activities but found to be toxic when greater than required amount is present in the substrate.16 Edible mushrooms are known to accumulate Se but the quantity depend on the fungal species, stage of maturity, the amount in soil and substrates used for the cultivation of edible fungi.4 The concentration of Se required for Pleurotus mushroom enrichment may differ based on substrates and fungal species. It is therefore necessary to determine fungal growth rate on media to quantify the amount of Se required in the substrates. Findings of Kalac4 had earlier revealed that the Se requirement for species or strains of edible mushroom depend on their developmental stage, substrate, Se form and concentration.
Table 1: Mycelial growth rate (mm d-1) and biomass (mg) of edible fungi of genus Pleurotus cultivated on Se in culture media at different concentrations (0, 50.0 or 100.0 mg L−1)
|
Se conc. |
Mycelial growth rate | |||||
| P93 | PLO 06 | Pindo | Pcor | PLO 13 | Psfarm | |
| 0 | 8.40 a ± 0.10 | 8.42 a ± 0.14 | 6.40 a ± 0.18 | 5.80 a ± 0.15 | 6.11 a ± 0.10 | 6.57 a ± 0.13 |
| 50 | 3.60 b ± 0.20 | 3.30 b ± 0.10 | 3.40 b ± 0.20 | 1.50 b ± 0.08 | 1.60 b ± 0.00 | 3.60 b ± 0.02 |
| 100 | 1.40 c ± 0.14 | 1.57 c ± 0.40 | 2.80 c ± 0.20 | 1.20 b ± 0.02 | 1.00 c ± 0.30 | 3.30 b ± 0.10 |
|
Biomass |
||||||
| 0 | 71.90 a± 1.21 | 89.96 a ± 6.90 | 149.43 a ± 16.8 | 34.03 a ± 1.27 | 62.20 a ± 1.90 | 67.30 a ± 3.30 |
| 50 | 21.3 b± 3.60 | 21.4 b ±0. 40 | 30.5 b ±1.20 | 12.10 b± 0.30 | 16.40 b±1.60 | 11.4 b± 0.70 |
| 100 | 5.63 c ± 0.80 | 9.70 c ± 0.20 | 10.8 c ± 0.20 | 10.6 b± 0.60 | 7.30 b ± 0.20 | 8.06 b±0.05 |
Values are mean±SD of replicates n=3. Mean with different superscript alphabet along column are significantly different at P≤0.05 when Duncan test was adopted for P93: P. ostreatus, PLO6: P. ostreatus, Pindo: P. pulmonarius, Plocor: P. cornucopiae, PLO13: P. djamor and Psfarm: P. djamor v. roseus
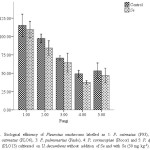 |
Fig. 1. Biological efficiency of Pleurotus mushrooms labelled as 1: P. ostreatus (P93), 2: P. ostreatus (PLO6), 3: P. pulmonarius (Pindo), 4: P. cornucopiae (Plocor) and 5: P. djamor (PLO13) cultivated on U. decumbens without addition of Se and with Se (50 mg kg-1). Click here to View figure |
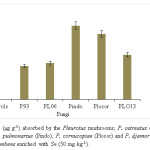 |
Figure 2: Se content (µg g-1) absorbed by the Pleurotus mushrooms; P. ostreatus (P93), P. ostreatus (PLO6), P. pulmonarius (Pindo), P. cornucopiae (Plocor) and P. djamor (PLO13) cultivated on U. decumbens enriched with Se (50 mg kg-1). Click here to View figure |
The biological efficiency of Pleurotus mushrooms produced from Se containing substrate was different (P<0.05) from control (without Se) as shown in Figure 1. P. ostreatus (P93) displayed higher (P<0.05) biological efficiency of 115.23% than other cultivated Pleurotus spp. The biological efficiency, 62.8% of P. pulmonarius (Pindo) from substrate containing Se was similar (P<0.05) to what obtained for its control without Se (70.6%). Higher content of Se was obtained for P. Pulmonarius (135.50 µg g-1) and was significantly different (P<0.05) from other Pleurotus spp. (Figure 2). This shows that the fungi may have mechanism for metabolizing Se. No Se detected in mushrooms cultivated on substrate without Se (control), as previously reported by Nunes et al.,17 in shiitake. This is in support of Baldrian18 who had earlier revealed that some heavy metals are essential for the fungal metabolism, whereas others have no known biological role even when present in excess.
The amount of Se (61.45-135.50 µg g-1) absorbed by the cultivated mushrooms are still within the range required for daily dose. The bioavailability of Se in agricultural residue utilized as substrate and supplementation of Se into substrates for the cultivation of Pleurtotus species has contributed to the content of amino acids and protein.6 Mushrooms possess effective mechanism that enables them to readily take up some metals from the ecosystem, which involved bioaccumulation; the active transportation of mineral into cell and intracellular components, biosorption methods like adsorption, ion exchange processes and covalent binding.19, 20
The ability of Pleurotus species to absorb Se and some metals has led to their applications in mycoremediation, often used in the bioconversion of industrial wastes and cultivated on agro-waste residues into fruit bodies of mushroom to control pollution.21 The uptake of organic Se compounds; selenocysteine, selenomethionine, selenite and selenate into various forms of Se metabolites by fungal species is an effective approach to acquire some secondary metabolites, which will promote their therapeutic properties such as antioxidant, antimicrobial, anticancer, cholesterol lowering and immunostimulatory effects.22,23,24 Hence, the production of edible mushrooms fortified with macro and micro elements can be used as medicinal foods to solve the problem of malnutrition and mineral deficiency in foods.
Conclusion
This research shows that signal grass can be used to produce edible mushrooms enriched with Se, which may serve as nutritional supplement in foods to control the Se deficiency symptoms and syndromes. Hence, biofortification of edible fungi, Pleurotus spp. with Se could be a healing option for the Se deficiency symptoms and associated ailments.
Acknowledgements
The authors express their gratitude to National Council for Scientific and Technological Development (CNPq), Brazil and The World Academy of Sciences (TWAS), Italy for the financial support. Olusola Clement Ogidi (OCO) is a recipient of CNPq/TWAS sandwich postgraduate fellowship award to Universidade Federal de Viçosa (UFV), Brazil. Prof. M. C. M. Kasuya is gratefully acknowledged for accepting OCO in the Laboratório de Associações Micorrízicas, Departmento de Microbiologia/BIOAGRO, UFV.
References
- Brown K. M., Arthur J. R. Selenium, selenoproteins and human health: a review. Public Health Nutrition. 2001;4(2B):593-599.
CrossRef - Burk R.F., Levander O.A. Selenium -in: Shils M. E., Olson J. A., Shike M., Ross, A.C. (Eds) Modern nutrition in health and disease. Lippincott Williams & Wilkins, Baltimore, MD. 1999;265-276.
- Sunde R. A. Regulation of glutathione peroxidase-1 expression. In: Hatfield D. L., Berry J., Gladyshev V. N. editors. Selenium: its molecular biology and role in human health. 2nd ed. New York: Springer Science Media. 2006;149–60.
CrossRef - Kalac P. Chemical composition and nutritional value of European species of wild growing mushrooms. A review. Food Chemistry. 2009;113;9-16.
CrossRef - Kabata-Pendias A. Geochemistry of Selenium. Journal of Environmental Pathology Toxicology Oncology. 1998;17(3-4):173-7.
- Bhatia P., Bansal C., Prakash R., Nagaraja T. P. Selenium uptake and associated anti-oxidant properties in Pleurotus fossulatus cultivated on wheat straw from seleniferous fields. Acta Alimentaria. 2014;43(2):280–287.
CrossRef - Papp L. V., Holmgren A., Khanna K. K. Selenium and selenoproteins in health and disease. Antioxidants Redox Signaling. 2010;12:793–795.
CrossRef - De Assuncao L. S., da Luz J. M. R., da Silva M. C. S., Vieira P. A. F., Bazzolli D. M. S., Vanetti M. C. D., Kasuya M. C. M. Enrichment of mushrooms: An interesting strategy for the acquisition of lithium. Food Chemistry. 2012;134:1123–1127.
CrossRef - Kim Y. K., Xiao C. L., Rogers J. D. Influence of culture media and environmental factors on mycelial growth and pycnidial production of Sphaeropsis pyriputrescens. Mycologia. 2005;97(1): 25–32.
CrossRef - Reeslev M., Kjoller A. Comparison of Biomass Dry Weights and Radial Growth Rates of Fungal Colonies on Media Solidified with Different Gelling Compounds. Applied and Environmental Microbiology. 1995;61(12):4236-4239.
- Kasuya M. C. M., da Luz, J. M. R., Nunes M. D., Da Silva M. C. S., Carvalho D. R., Assunção, L. S., Bento C. B. P. Production of Se-Enriched Mushrooms in Coffee Husks and Use of This Colonized Residue: In coffee in health and disease prevention. 2014;ISBN: 20149780124095175.
- Da Silva M. C. S., Naozuka J., da Luz, J. M. R., de Assunção L. S., Oliveira P. V., Vanetti M. C. D., Bazzolli D. M. S., Kasuya, M. C. M. Enrichment of ostreatus Mushrooms with Selenium in Coffee Husks. Food Chemistry. 2012;131(2):558-563.
CrossRef - Maftoun P., Johari H., Soltani M., Malik R., Othman N. Z., El Enshasy H. A. The Edible Mushroom Pleurotus: I. Biodiversity and Nutritional Values. International Journal of Biotechnology for Wellness Industries. 2015;4:67-83.
CrossRef - Correa R. C. G., Brugnari T., Bracht A., Peralta M. R., Ferreira I. C. F. R. Biotechnological, nutritional and therapeutic uses of Pleurotus (Oyster mushroom) related with its chemical composition: A review on the past decade findings. Trends in Food Science and Technology. 2016;50:103- 117.
CrossRef - Da Silva M. C. S., Nunes M. D., da Luz J. M. R., Kasuya M. C. M. Mycelial Growth of Pleurotus in Se-Enriched Culture Media, Advances in Microbiology. 2013;3:11-18.
CrossRef - Spallholz J. E. On the Nature of Se Toxicity and Carcinostatic Activity. Free Radical Biology and Medicine. 1994;17(1):45-64.
CrossRef - Nunes R. G. F. L., da Luz J. M. R., Freitas R. B., Higuchi A., Kasuya M. C. M., Vanetti, M. C. D. Selenium Bioaccumulation in Shiitake Mushrooms: A Nutritional Alternative Source of this Element. Journal of Food Science. 2012;77(9):983-986.
CrossRef - Baldrian P. Interactions of Heavy Metals with White- Rot Fungi. Enzyme and Microbial Technology. 2003;32(1):78-91.
CrossRef - Kulshreshtha S., Mathur N., Bhatnagar P. Mushroom as a product and their role in mycoremediation. AMB Express. 2014;4:29-35.
CrossRef - Kieliszek M., Błażejak S., Gientka I., Bzducha-Wróbel A. Accumulation and metabolism of Selenium by yeast cells. Applied Microbiology and Biotechnology. 2015;99:5373–5382.
CrossRef - Sanchez C. Cultivation of Pleurotus ostreatus and other edible mushrooms. Applied Microbiology and Biotechnology. 2010;85:1321–1337.
CrossRef - Tinggi U. Selenium: its role as antioxidant in human health. Environmental Health and Preventive Medicine. 2008;13:102–108.
CrossRef - Estrada A. E. R., Lee H-J., Beelman R. B., Jimenez-Gasco M. M., Royse D. J. Enhancement of the antioxidants ergothioneine and selenium in Pleurotus eryngii eryngii basidiomata through cultural practices. World Journal of Microbiology and Biotechnology. 2009;25:1597–1607.
CrossRef - Milovanovic I., Stajic M., Cilerdzic J., Stanojkovic T., Knezevic A., Vukojevic J. Antioxidant, antifungal and anticancer activities of se-enriched Pleurotus spp. mycelium extracts. Archives of Biological Sciences. 2014;66(4):1379-1388.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





