Introduction
There are more than 200 species of the Plantago genus of which Psyllium is a common species. Mucilage-rich Plantago seeds are primarily acquired from Plantago ovata (blond psyllium) and from Plantago psyllium (PP) (black psyllium).1 Mucilage in Psyllium consists of D-galactoronic acid, D-xylose, L-arabinose and other sugar traces.1 Owing to its powerful capacity for forming a gelatinous material with water, Psyllium is classified as a mucilaginous fiber. It is a valuable medicinal plant with compounds such as phenols and flavonoids. Plantago plants have potent antioxidant activity due to their high content of phenolic compounds. Psyllium contains nutritious polyunsaturated fatty acids (PUFAs), as the essential fatty acids (Omega-3 and Omega-6), metabolites with bioactivities and, sulfur containing amino acids, making it a good choice for the development of functional food. Psyllium has hypolipidemic effect as it decreases the level of serum cholesterol due to attaching bile acids in intestine and decreasing coronary artery disease risk.2
Because of the nutritional and physicochemical characteristics of PP, and stability at various pH levels and temperatures, adding Psyllium to baked products have been studied extensively e.g. in supplementing wheat flour3 in mixtures free of gluten4. Other studies’ findings revealed increased absorption of water and enhanced dough workability with the incorporation of PP5. Based upon the rate of hydration this inclusion also affected starch gelatinization and retro-gradation6. The forming characteristics of Psyllium fiber; when it is well hydrated can affect the batter volume texture and the bread’s crumb structure and thus help increase the dietary fiber concentration and give a lengthy shelf life to baked products.4,5
Plantago products are becoming more and more important due to their nutritional and technical functions. This research was therefore intended to examine the hypolipidemic impact of psyllium seeds and antioxidative activities in liver and blood when cake supplemented with Psyllium 10% and 30% -using whole commercial Psyllium- introduced to hyperlipidemic rats.
Materials and Methods
Plant Material and Preparation of Seeds
Plantago psyllium seeds were purchased (October 2018) from a local market in Alexandria. To remove impurities, seeds were cleaned and then ground to fine powder and stored in an air-tight container till analysis.
Ingredients
Plantago Psyllium seeds powder and other cake ingredients (Listed in Table 1) were purchased from a local market in Alexandria, Egypt. For all formulations, psyllium was ground and the particle fraction was between 0.5 and 1 mm.
Preparation of cake
Cake formulations using wheat flour in different ratios are given in Table 1. Cake making was prepared according to Sudha, et al.7. Wheat flour blends with of 0, 10 and 30 % of Psyllium seed powder were used for making cake. Sugar and oil were mixed for 10 minutes to prepare the cake batter. Eggs were then added and blended. Thereafter, dry ingredients were added (milk powder, whey protein, baking powder, vanilla and flour). As a functional food ingredient, psyllium powder was introduced to the formulation. At the end, water and batter were added and blended for 5 min. All cakes have been manufactured on equal terms. An amount of 1500 g of cake batter were prepared. Then, cakes of 40 g batter were baked at 180-190 ° C for 20-25 minutes. Cakes were kept at room temperature in polyethylene packs.7
Table 1: Recipe of cake supplemented with Psyllium seed powder (Grams based on the weight of cake flour).
| Ingredient | Blends | ||
| C∗ | 10% PP* | 30% PP* | |
| Flour | 1000 | 900 | 700 |
| Psyllium seed powder | 0 | 100 | 300 |
| Oil | 200gm | 200gm | 200gm |
| Refined sugar | 250gm | 250gm | 250gm |
| Eggs | 250gm | 250gm | 250gm |
| Baking powder | 25gm | 25gm | 25gm |
| Milk powder | 40gm | 40gm | 40gm |
| Vanilla | 2gm | 2gm | 2gm |
| Water | 150ml | 150ml | 150ml |
∗C= 100% wheat flour; ∗∗PP = Psyllium seed powder.
Proximate Analysis of Plantago Psyllium Seeds and Their Supplemented Cake
In accordance with the AOAC methods 942.05 and 930.15,8 the ash content and moisture were determined. A Kjeldahl system was used for calculating the total nitrogen content. After Soxhlet extraction of dried specimens with hexane the raw fat content was measured gravimetrically. Triplicate measurements have been made. Difference method was used to calculate the total carbohydrate.
Extraction of Plantago psyllium Seeds
Preparation of the Extracts
Twenty grams of powdered Plantago psyllium seeds were extracted in 200 ml water by conventional extraction method at room temperature for 6 hours. For the removal of peel particles, the mixture was filtered through a Whatman No. 2 filter paper. The residues have been re-extracted twice, to make sure particles are fully recovered. The extract yield was lyophilized in a jar at -4 °C before testing.
Determination of Antioxidant Activity of Plantago psyllium Seeds Extract and Their Cakes Products
The 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radicals scavenging assay (Brand-Williams, et al.9) was used. For antioxidant activity determination, 2, 2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical scavenging assay was adopted (Re et al.10)
Sensory Evaluation
Products were subjected to sensory evaluation test along with the control; by using a 9-point hedonic scale from 1(extremely dislike) to 9 (extremely like).
The in-Vivo Study
Experimental animals: A total of 28 Albino rats (180-200 g) were obtained from the Faculty of Pharmacy and Drug Manufacturing Animal House, Pharos University in Alexandria, Egypt. They were acclimated for 3 days before starting the experiment.
Induction of Dyslipidemia
High fat diet (HFD) (46% of calories from fat); 1 kg (standard laboratory diet), 1/2kg plant margarine rich in saturated fatty acids (SFA) from palm oil, and 1/4 kg lard.11
All animals had standard cages (7 / cage), a 21-23°C of air conditioning, 60-65% relative humidity and a 12-hour light/12-hour dark period. Consuming water ad libitum until the experiment was completed. The animals received proper care according to the Scientific Research Committee of Pharos University, Manual for Care and Use of Laboratory Animals.
Four Groups Comprising 7 rats in Each Group
Group a)
Rats fed on normal diet (Standard laboratory diet consists of: 60% Carbohydrates, 19% protein, 6% fats, 2.3% fibers and more than 5%vitamins and minerals, Energy: 3200 Kcal/kg). served as a negative control;
Group b)
Rats fed on HFD for 4 weeks, and (control cake) (without added Plantago psyllium powder); served as a positive control
Group c)
Rats fed on HFD for 4 weeks, and PP cake 10% for 4 weeks;
Group d)
Rats fed on HFD for 4 weeks, and PP cake 30% for 4 weeks.
Table 2: proximate composition of the standard diet and High fat diet:
| Ingredient | Negative control(C) | Positive control(HFD) |
| Carbohydrates | 60% | 60% |
| Protein | 19% | 19% |
| Fats | 6% | 46% |
| Fibers | 2.3% | 2.3% |
| Vitamins and minerals | 5% | 5% |
Measurements of Body Weight and Biochemical Blood Analysis
Body weights of all groups under investigation were assessed twice per week and at the end of experiment period, for 8 weeks the percentage of gain weight was expressed as (final weight-beginning weight)/ beginning weight) x 100.
After pentobarbital anesthesia, the blood samples were collected to obtain serum for assessment of aspartate aminotransferase (AST), and alanine aminotransferase (ALT) total protein, albumin content, total cholesterol and high-density lipoprotein (HDL) cholesterol, and triacylglycerol. Standard enzymatic spectrophotometery was performed for glucose and cholesterol using Bio-diagnostic kits (Giza – Egypt).
Antioxidant Enzyme Analysis
Reduced liver glutathione (GSH) was determined according to the method of Jollow.12 Catalse (CAT; EC 1.11.1.6) activity was determined13. Superoxide dismutase (SOD, EC 1.15.1.1) activity was measured in different tissues according to Misra and Fridovich.14 The activity of Glutathione peroxidase (GPX, EC. 1.1.1.9) was assayed according to Chiu.15 Liver thiobarbituric acid-reactive substances (TBARS) were measured at 532 nm by using 2-thiobarbituric (2,6-dihydroxy pyrimidine -2-thiol; TBA). An extinction coefficient of 156,000 M-1 cm -1 was used for calculation.16
Histopathology of Liver
Immediately after necropsy, the rats’ livers were dissected out carefully and weighed. The rats’ livers then were fixed in 10% formalin. Liver tissues were further embedded in paraffin blocks and sectioned into 5-6μm sections using Microtome (Leica, Italy) for serial specimens, mounted on glass slides and stained with hematoxylin eosin (HE) stain for histological light microscopic evaluation.17
Statistical Analysis
Mean and standard deviation were calculated for each studied variable. The interpretation of data obtained was done by analysis of variance (ANOVA) test. Level of significance was accepted at P<0.05.
Results and Discussion
Proximate Analysis of Plantago psyllium Seeds and Their Supplemented Cake
Plantago psyllium seeds are known to increase the final product’s nutritional value in wheatmeal flour18. Table 3 shows the chemical composition of the cakes. The fiber content of Plantago psyllium cake has been gradually increased with increasing Plantago psyllium ratio; from 7.77 ± 1.19% in PP cake 10% to 9.43 ± 1.29% in PP cake 30%. Improved cake fiber content corresponds to the high Plantago Psyllium fiber content (23.83 ± 1.94%). Carbohydrate, ash, and crude fiber contents of the PP cake mixes 30 percent, surpass PP cake 10% mixes and protein and fat content in Plantago Psyllium cupcake mixes 30% was lower than 10% (Table 3).
Table 3: Chemical composition of Plantago psyllium seeds and supplemented cake (g/100 g dry weight)
| Determination (%) | PP seeds | PP Cake 10% | PP Cake 30% |
| Protein | 9.34 ± 0.81 | 15.36 ± 0.70 | 11.70 ± 1.57 |
| Fat | 5.16 ± 0.85 | 5.48 ± 0.41 | 5.15 ± 0.80 |
| Carbohydrate | 59.26 ± 1.30 | 68.45 ± 1.55 | 70.33 ± 3.81 |
| Fiber | 23.83 ± 1.94 | 7.77 ± 1.19 | 9.43 ± 1.29 |
| Ash | 2.38 ± 0.70 | 2.92 ± 0.73 | 3.38 ± 0.74 |
Results are expressed as means ± standard deviation of three measurements; PP: Plantago psyllium
Compared with psyllium cake samples, the seed sample had lower ash levels. A sufficient consumption of dietary fiber is well documented to reduce the risk of a variety of chronic diseases. The significant mineral content in the psyllium sample (2.67 per cent w / w) was responsible for a rise in ash; similar results were found by Raymundo.19
Findings concerning Plantago psyllium’s cake were similar to the previous data stated by Beikzadeh18; that adding PP to wheat flour improves the nutritional value and gives cakes with higher carbohydrate, fiber and ash content than the control from wheat flour alone. On the other hand, Kaushal and Bhatia20 showed that protein, fat, carbohydrate and ash contents of PP seeds ranged from 23 – 30 %, 2.9 – 3.5%, 50 – 60% and 3.0 – 3.5%, respectively. Romero-Baranzini21 cited that, PP contained crude proteins (17.5%), crude fats (6.7%), ash (2.7%) and carbohydrates (49%). The amount of protein determines the effect of inclusion of psyllium seed on the consistency of the cake. An increase in the amount of protein has been recorded to improve cookie firmness by Fustier.22 The total fiber was observed to be affected positively by increased content of PP. Arabinose, galactose, galacturonic acid, and rhamnose are the most common types of soluble fiber in psyllium seed.23
Antioxidant Activity of Plantago psyllium Seeds Extract and Their Supplemented Cake
Antioxidants play a key role in health promotion in the nutraceutical sector and inhibit functional food ingredient oxidation procedures. In addition, their activity can be measured with various mechanisms.24 In the present study several antioxidant analyses, including radical scavenging of ABTS+ and DPPH, were conducted at several concentrations (15.62 to 1000 μgml−1) to determine the antioxidant and various scavenging actions of Plantago psyllium plants and their fortified cake (Fig. 1). The reducing capacity, antioxidant and scavenging activities of the extract of psyllium seeds increased concomitantly with the concentration as well as increase supplementation of seeds in cake product (Fig. 1).
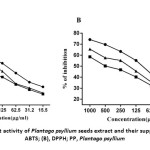 |
Figure 1: Antioxidant activity of Plantago psyllium seeds extract and their supplemented cake. |
The capacity of ABTS radicals to scavenge (expressed as the percent inhibition) was used as an indicator for estimating total antioxidant activity.25,26 The maximum percent inhibition of Plantago Psyllium seeds was found to be 83.4% for 1000 µg/ml concentration. In accordance, Pourmorad27 found that the antioxidant activity of PP is 89.3% for 1000 µg/ml concentration. Similarly, in the present study 65.5% and 75.4 % inhibition were observed for PP cake 10% and PP cake 30%, respectively at 1000 µg/ml concentration (Fig. 1B). The DPPH radicals ‘ stability has been commonly used as a tool to estimate plant samples by free radical scavenging activities.25 Similar to ABTS radicals, the highest DPPH scavenging activity for 1000 μg / ml was noted (Fig. 1). The maximum scavenging percentage 74.2% was found for Plantago seeds extract.
It was also observed that PP cake 30% extract showed significantly higher DPPH scavenging activity compared to PP cake 10% extract (Fig. 1A). The free-radical DPPH is more stable and can accept electron or hydrogen28. The findings showed that Plantago seeds extract could behave as a donor of hydrogen to scavenge DPPH free radicals.25, 26, 28 In this regard, Kobeasy29 reported that, Plantago contained the following biologically active compounds: (vanyllic acid), phenolics (caffee and ferulic acids) and flavonoids (luteolin). The findings of our study are consistent with Beara30who studied the antioxidant activities of Plantago species and stated that it could be viewed as a potential new natural antioxidant source.
Sensory Evaluation of Plantago Psyllium -Based Cake
The current trial explored the effect of Plantago psyllium seed powder and its utilization into cake products (Fig 2). The sensory characteristics of the product were analyzed by 9 point hedonic scale. The PP products were desirable in term of color, taste and texture (Fig 3). The result of sensory evaluation showed that the incorporation of PP powder up to 10% was found to be the most acceptable and achieved the highest score for cake products. Generally, PP incorporation makes the crumb more reddened, darker and less yellowish. The natural pigments in psyllium seed are among the main reasons for color change.18 Meanwhile, Sudha31 noted that inclusion of fiber caused darkening of the resultant products.
 |
Figure 2: Plantago psyllium seeds raw material (A), functional plantago psyllium cake 10% (B)and functional plantago psyllium cake 30 % (C). |
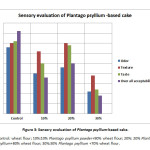 |
Figure 3: Sensory evaluation of Plantago psyllium-based cake. |
(1) Dislike extremely; (2) dislike very much; (3) dislike moderately; (4) dislike slightly; (5) neither like nor dislike; (6) like slightly; (7) like moderately; (8) like very much and like extremely (9).
Values are mean +SE of thirty of panelists
Data of the present study indicated that among samples with 10 and 30 % PP, no significant differences in textural toughness were found. Psyllium seeds at lower concentrations are favorably improving the gluten network, as found by Raymundo.19 An increase in cakes’ fiber content; from 10 to 30 percent results in an increase in textural hardness. When approximately ten percent of psyllium seeds were added to cake in different studies, form, shape, crust, pores and taste have improved. However, the texture softness and hardness or chewiness were not significantly affected.18
In addition to difficulties in handling the product a negative effect on sensory tests was observed at higher psyllium added concentrations in the Park32 report. Similar effect has been shown by Raymundo.19 Moreover, the inclusion of 7.5 percent of psyllium seeds can usually improve the overall sample quality.18
Table (4) reveals a significant increase in the body weight gain of the positive controls has also been found with the addition of HFD (cake free) in comparison to the negative control, a significant difference between body weight gain of the two PP cake 10% & 30% (24.16 and19.16). There has also been a positive link between the lower weight gain and the 30 percent PP cake consumption. This may be due to the presence of soluble gel-forming mucilage from the Plantago psyllium seeds.33 The development of viscous materials can help satiety and aid weight loss.
Table 4: Effects of normal, high-fat diet and Plantago psyllium cake diet supplementation on weight change, weight gain and feed intake of rats after 8weeks.
| Parameters | Negative Control | (PP- control cake) | PP Cake 10% | PP Cake 30% |
| Initial weight (g) | 181.5±5.3a | 180.5±3.2a | 177±3.2d | 178±4.2a |
| Final weight (g) | 240.3±5.3a | 268.8±8.9b | 233.4±2.1c | 220.2±2.6c |
| weight change(g) | 32.39 ±7.74a | 32.84 ±5.61b | 24.16±6.10d | 19.16±3.62d |
| Feed intake(g) | 22.5 ±0.7a | 28.4 ±0.04a | 24.7±0.7a | 22 ±1.5a |
Results are expressed as mean ± SD (n= 7). Means which are not significantly different have the same symbol; significance level at P ≤ 0.05.
Serum Lipids
The current findings showed significantly increased levels of TC, TG and LDL-C in rats fed a high-fat diet (cake free) compared with the control group (Table 5). However, there has been a reduction in HDL-C concentration in the HFD group (P < 0.05). Lard fat is abundant in saturated fatty acids, associated with boosting total and LDL-cholesterol, and is rich in monounsaturated fatty acids, that may increase serum triacylglycerol.34,35 Moreover, Plantago psyllium has shown a significant decrease in total cholesterol and LDL cholesterol when administered to humans with high cholesterol. It is also found effective in animals.36 Increased serum triglyceride concentration may be ascribed to reduced triglyceride clearance secondary to reduced lipoprotein lipase (LPL) activity.37 The elevated LDL-cholesterol concentration observed in hyperlipidemic rats can be ascribed to a reduction of saturated fatty acids and cholesterol included in the diet in LDL receptors.38
Table 5: Effects of normal, high-fat diet and Plantago psyllium cake diet supplementation on serum lipid profile.
| Treatments | TG (mg/dL) | TC (mg/dL) | LDL-C (mg/dL) | HDL-C (mg/dL) |
| Negative Control | 157.30±1.64c | 121.73±2.02c | 87.83±2.25c | 33.90±2.27c |
| (PP- control cake ) | 204.37±2.27a | 140.67±1.04a | 107.90±1.47a | 32.76±2.51d |
| PP Cake 10% | 166.57±3.45b | 132.10±1.32b | 93.13±0.55b | 38.96±1.30c |
| PP Cake 30% | 154.67±1.50d | 125.70±0.65d | 81.33±1.46d | 44.36±1.09a |
TG, Triglycerides; TC, Total cholesterol; LDL-C, low density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol
Data are expressed as mean ± SD (n= 7). Means which are not significantly different have the same symbol; significance level at P ≤ 0.05.
Plantago psyllium soluble fiber attaches to small intestine bile acids, forming a complex which prevents reabsorbing bile from the small intestine, thus increasing the production and secretion of the bile acids by drawing cholesterol to be used as a substitution for the missed acids39; thereby lowering blood cholesterol levels.
Feeding the rats on HFD with PP cake 30% induced significant decreases of triglycerides (24.31%), total cholesterol (10.64 %) and LDL-cholesterol (24.62%) concentrations in comparison with the high-fat diet group. A significant decrease of four percent in total serum cholesterol and a 7 percent decrease in LDL cholesterol compared to the control diets were found by Anderson.40 Cake 30 percent showed enhanced significant levels of HDL-cholesterol (26.14 %, compared with the PP-free cake group) due to PP seeds ‘ ability to accelerate the decline of radical species produced during the administration of cholesterol.41As a free radical scavenger, HDL-C prevents the peroxidation of beta-lipoproteins.42 Plantago psyllium seeds demonstrated enhanced anti-hyperlipidemic activity in rats but the extract regulation mechanism for TC, TG and LDL-C is not known. Meanwhile, PP can boost HDL cholesterol levels, which are still the main risk factors for coronary artery disease in the regular diet suggested by medical guides to fight against hypercholesterolemia.39
Data presented here are in agreement with Xing39 who recorded the PP extract and powder protection impact on hypercholesterolemic rats evidenced by a significant lower value of serum cholesterol, triglycerides, LDL level, cholesterol/ HDL, LDL / HDL. Furthermore, Anderson40 reported that PP extract administered orally for four weeks to hypercholesterolemic and diabetic rats resulted in a superior lipid profile and a reduction in blood sugar level.2 Similar observations were made by a number of workers, demonstrating a hypolipidemic effect of PP in experimental animals.
Liver Enzymes Activity, Total Protein and Albumin
The two main measures of liver damage and an evaluation toxicology test are the aspartate transaminase (AST) and the alanine transaminase (ALT).43 The activities of serum AST and ALT in the present study were significantly high in the HFD compared to normal rats; these findings are in agreement with Sudhahar.44 No significant differences were found in ALT & AST between the negative control group and PP cake 30%.
Consumption of HFD induced high levels of ALT and AST in hyperlipidemic rats compared to normal rats (Table 6). Thus, if any toxic interaction is produced by the Plantago psyllium cake diet, this could be noted by increasing liver damage, the findings show that no toxicity or damage to the liver could be recorded. this result is consistent with those of Obatolu.45 The total protein and albumin levels were not significantly different in all studied groups; they were within the normal range. As diets used in the present study were protein sufficient.
Table 6: Effects of normal, high-fat diet and Plantago psyllium cake diet supplementation on liver biomarkers.
| Treatments | Total protein (mg/dL) | Albumin(mg/dL) | AST (U/L) | ALT (U/L) |
| Negative Control | 8.50±0.26a | 6.60±0.10a | 50.66±1.41c | 39.20±1.31c |
| HFD + (PP-control cake) | 8.36±0.20a | 5.95±0.33ab | 66.33±1.04a | 54.166±1.60a |
| PP Cake 10% | 8.31±0.18a | 5.76±0.50b | 57.96±1.25b | 45.43±0.70b |
| PP Cake 30% | 8.44±0.15a | 5.80±0.42b | 52.13±1.46c | 40.21±1.23c |
Control: Normal diet; HFD: High Fat Diet; PP: Plantago psyllium
Activity of Antioxidant Enzymes
The unwanted effects of oxidative agents are affected by endogenous enzyme and non-enzyme antioxidants. Water-soluble antioxidants are SOD, GPx and CAT. Antioxidant enzymes like SOD, GPx, and CAT, may remove the reactive oxygen (ROS) and reactive nitrogen (RNS) species. The SOD is the first defensive line against oxidants, leading to oxygen disputes with H2O2. The GPx eliminates the need for glutathione (hydrogen donor and scavenger of H2O2, radical hydroxyl and chlorinated oxidants), to reduce organic peroxides H2O2 to H2O and O2. The H2O2 is reduced to water by CAT.46,47
Enzyme complexes for free radical scavenging as superoxide dismutase (SOD), glutathione peroxidase (GPx) and catalase (CAT) were tested to determine excessive oxidant stimulation (Table 7). The PP cake 30% was found to increase the activity of GSH, SOD, GPx, CAT enzymes by 64.80%, 32.07%, 49.96% and 36.37 %, respectively, in comparison to hyperlipidemic the control rats. Reduction in enzyme activity is linked to the aggregation of highly reactive free radicals which lead to adverse effects such as the loss of integrity and function of the cell membrane.48 When free radicals are present in elevated levels, they can associate with the enzymes and inactivate them.49 Moreover, Jung50 observed an improvement in antioxidant potential in Sprague-Dawley (SD) rats on HFD supplemented with chitosan and psyllium husk. Patel51 reported high antioxidant (in vitro) activity in psyllium-extracted polysaccharide (P. ovata).
Pigeolet49 revealed that PP has high concentration of phenolics which allow free radicals to break down, one of the key anti-oxidation mechanisms inhibiting lipid peroxidation chain reactions. The effect on polyphenol production was reported to be substantial, which makes possible that increased antioxidant activity to be related to increased production of polyphenols and flavonoids as well as improved hepatic antioxidant status, as measured by SOD and GPx activity when supplemented by PP.
In the present study hyperlipidemic rats showed elevation of hepatic TBARS concentration relative to negative control group (30.63±1.15 nmol/gm/ tissue) vs. treatment group (PP Cake 30%) (31.96±0.95 nmol/gm/ tissue) (Table7). A rise in TBARS levels has previously been recorded in HFD animals.52 A positive correlation between total cholesterol and levels of triacylglycerol and generation of free radicals was demonstrated.53 Furthermore, during the in vitro callus culture of this plant, the use of Plantago psyllium seed additives demonstrated an increasing polyphenol and flavonoid contents. Therefore, present study would is useful for the use of tissue cultivation in plant tissue production and their enhanced recovery from the tissue of the callus.31 This can be linked to enhanced polyphenols accumulation during callus aging. The same kind of comment was made by Ozyigit.54
The PP also contains high levels of polyphenolic antioxidants including flavonoids, ellagic, ferulic and gallagic acids, penoids, and catechins. The polyphenols have important biological activity, including the inhibition of oxidation, free radical eradication and risk reduction of cardiovascular diseases. The anti-mutagenic and promising antioxidants activity of PP may be a responsiblity of ellagitannins.55 A product which improves the overall antioxidant status and reduces the oxidative stress markers of healthy individuals may use psyllium as an additive.51
Table 7: Effects of normal, high-fat diet and Plantago Psyllium cake diet supplementation on tissue antioxidant enzymes.
| Treatments | TBARS(nmol/gm/tissue) | GSH(IU/gm tissue) | SOD(IU/gm tissue) | GPx(IU/gm tissue) | CAT(IU/gm tissue) |
| Negative Control | 30.63±1.15b | 3.30±0.43c | 66.23±1.70b | 35.26±1.53b | 34.46±1.45c |
| HFD + (PP-control cake) | 45.23±1.64a | 1.83±0.20d | 50.47±1.45c | 20.38±0.47c | 26.06±0.88d |
| PP Cake 10% | 32.86±1.34b | 4.54±0.21b | 67.73±0.68b | 37.53±1.67b | 37.36±0.66b |
| PP Cake 30% | 31.96±0.95b | 5.20±0.10a | 74.30±1.85a | 40.73±1.53a | 40.96±1.68a |
Data are expressed as mean ± SD (n= 7). Means which are not significantly different have the same symbol; significance level at P ≤ 0.05.
TBARS: Thiobarbituric acid reactive substances; GSH: Glutathione; SOD: Superoxide dismutase; GPx: Glutathione peroxidase; CAT: Catalase.
Liver Histology
The effects of Plantago psyllium on liver histopathology are shown in Fig. 4. Hematoxylin and eosin staining revealed variations in the composition of the liver tissue and the lipid aggregation in rats with various dietary treatments.
The negative control group’ livers had a structure that is well organized, but the HFD group revealed significant morphological changes. Their livers’ structures showed a high degree of damage and signs of necrosis and macro-vacuolization in hepatocytes. While the supplementation with PP visibly alleviated hepatocyte steatosis and lipid accumulation. In contrast with the negative control, the rats with HFD exhibited increased adipocyte size (Fig. 4). This was confirmed by the improvements in number of adipocytes per spot.
Higher levels of visceral fat in the liver often are accompanied by higher levels of serum lipid and are associated with insulin resistance, cardio-vascular risk, and dyslipidemia.56 Furthermore, excess fat accumulation in the liver can cause liver oxidative stress and liver damage.57 Histological findings showed a significant decrease in lipid deposition in rats on Plantago psyllium. Plantago psyllium permits substantial reductions in liver and adipose tissue volume, which indicates that the administration of Psyllium may minimize hepatic steatosis and obesity caused by HFD.
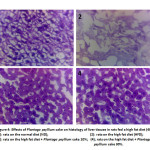 |
Figure 4: Effects of Plantago psyllium cake on histology of liver tissues in rats fed a high fat diet (40×). |
Conclusions
Regarding to its high nutritional value, Plantago psyllium seeds are promising sources of new biological molecules, and multi-functional polysaccharides and Plantago psyllium products. The Plantago psyllium seeds have a positive effect as a prebiotic that can decrease the risk of various chronic conditions. To date, however, limited data have proven these health benefits. Further studies may be required to investigate the oxidative stress reduction mechanisms of this remarkable crop and its safety for humans or animals.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors declare no conflict of interest with any person or Organisation in publishing this article.
References
- Saeedi, M., Morteza-Semnani, K., Ansoroudi, F., Fallah, S., Amin, G. Evaluation of binding properties of Plantago psyllium seed mucilage. Acta Pharmaceutica. 2010; 60: 339–348.
- Van Rosendaal, G.M., Shaffer, E.A., Alun, L.E, Brant, R. Effect of administration on cholesterol-lowering by psyllium: a randomized cross-over study in normocholesterolemic or slightly hypercholesterolemic subjects. J. Nutr. 2004; 28: 3–17.
- Sim, S. Y., Noor Aziah, A. A., & Cheng, L. H. Quality and functionality of Chinese steamed bread and dough added with selected non-starch polysaccharides. JFST. 2015; 52(1): 303–310.
- Mancebo, C. M., San Miguel, M. A., Martínez, M. M., & Gómez, M. Optimisation of rheological properties of gluten-free doughs with HPMC, psyllium and different levels of water. J Cereal Sci. 2015; 61: 8–15.
- Mariotti, M., Lucisano, M., Pagani, M. A., Ng, P. K. The role of corn starch, amaranth flour, pea isolate, and Psyllium flour on the rheological properties and the ultrastructure of gluten-free doughs. Food Res. Int. 2009; 42: 963–975.
- Aprodu, I., Banu, I. Influence of dietary fiber, water, and glucose oxidase on rheological and baking properties of maize based gluten-free bread. Food Technol. Biotechnol. 2015; 24(4): 1301–1307.
- Sudha, M., Rathinam, L.V. and KrishnaRau, L. Effect of maltodextrin and emulsifiers on the viscosity of cake batter and on the quality of cakes. 2005; 86: 706-712.
- AOAC International. Official methods of analysis. (G. W. Latimer, Ed.) (20th ed.). AOAC International, Rockville, Maryland. 2016; 20850–3250, USA.
- Brand-Williams, W., Cuvelier, M. E., Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol. 1995; 28: 25–30.
- Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med. 1999; 26(9–10):1231-1237.
- Buettner, R., Scholmerich, J., Bollheimer, L.C. High fat diets: modeling the metabolic disorders of human obesity in rodents. Obesity. 2007; 15:798-808.
- Jollow, D.J., Mitchell, J.R., Zampaglione, N., Gillette, J.R. Bromobenzene-induced liver necrosis. Protective role of glutathione and evidence for 3,4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacology. 1974; 11(3):151-69.
- Luck, H. Catalase. In, methods of enzymatic analysis. (Ed.Begmeyer HU) Academic press, Newyork, 1963, pp. 895-897.
- Misra, H.P., Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase.J Biol Chem. 1972; 25, 247(10):3170-5.
- Chiu D. T. Y., Stults F. H. and Tappel A. L. Purification and properties of rat lung soluble glutathione peroxidase. Biochim. Biophys. Acta, 1976; 445:558-566.
- Tappel, A.L., Zalkin, H. Inhibition of lipide peroxidation in mitochondria by vitamin E. Archives of Biochemistry and Biophysics. 1959; 80, 2: 333-336.
- Bancroft, J. D., Stevens, A. Theory and practice of histological techniques. 4thedn, Churchill living stone, New York, 1996.
- Beikzadeh, S., Peighambardoust, S., Beikzadeh, M., AsghariJavar-Abadi, M., Homayouni-Rad, A. Effect of Psyllium Husk on Physical, Nutritional, Sensory, and Staling Properties of Dietary Prebiotic Sponge Cake. Czech J. Food Sci. 2016; 34, (6): 534–540
- Raymundo A., Fradinho P., Nunes M.C. Effect of psyllium fibre content on the textural and rheological characteristics of biscuit and biscuit dough. Bioactive Carbohydrate and Dietary Fibre, 2014; 3: 96–105
- Kaushal, G.P., Bhatia, S. A study of polyphenols in the seeds and leaves of guar (Cyamopsis tetragonoloba L. Taub). J. Sci. Food Agric. 2006; 33: 461- 470.
- Romero-Baranzini, A.L., Rodriguez, O.G., Yanez-Farias, G.A., Barron-Hoyos, J.M., Rayas-Duarte, P. Chemical, Physicochemical, and Nutritional Evaluation of Plantago (Plantago ovata Forsk). Cereal Chem. 2006; 83(4): 358-362.
- Fustier, P., Castaigne, F., Turgeon, S.L., Biliaderis, C.G. Impact of commercial soft wheat flour streams on dough rheology and quality attributes of cookies. J Food Eng. 2009; 90: 228–237.
- Nelson A.L. High Fiber Ingredients. St. Paul, Egan Press, 2001.
- Shahidi, F., Zhong, Y. Measurement of antioxidant activity, J. Funct. Foods. 2015; 18: 757–781.
- Patel, M.K., Mishra, A., Jha, B. Non-targeted metabolite profiling and scavenging activity unveil the nutraceutical potential of psyllium (Plantago ovata Forsk), Front. Plant Sci. 2016; 7: 431.
- Pandey, S., Patel, M.K., Mishra, A., Jha, B. Physio-biochemical composition and untargeted metabolomics of cumin (Cuminum cyminum L.) make it promising functional food and help in mitigating salinity stress, PLoS One. 2015; e0144469.
- Pourmorad, F., Hosseinimehr, S.J., Shahabimajd, N. Antioxidant activity, phenol and flavonoid contents of some selected Iranian medicinal plants. Afr. J. Biotechnol. 2006; 5(11): 1142-1145.
- Fan, J., Wu, Z., Zhao, T., Sun, Y., Ye, H., Xu, R. Characterization, antioxidant and hepatoprotective activities of polysaccharides from Ilex latifolia Thunb, Carbohydr. Polym. 2014; 101: 990–997.
- Kobeasy, M. I., Abdel-Fatah, O. M., Abd El-Salam, S. M., Mohamed, Z. E. M. Biochemical studies on Plantago major and Cyamopsis tetragonoloba L. Inter J Biodivers Conserv.2011; 3(3): 83-91.
- Beara, N., Lesjak, M.M., Jovin, E.Đ., Balog, K.J., Mimica-Duki, N.M. Plantain (Plantago L.). Species as Novel Sources of Flavonoid Antioxidants. J. Agric. Food Chem., 2009, 57(19): 9268-9273.
- Sudha M.L., Vetrimani R., Leelavathi K. Influence of fiber from different cereals on the rheological characteristics of wheat flour dough and on biscuit quality. Food Chem. 2007; 100: 1365–1370
- Park H., Seib P.A., Chung O.K. Fortifying bread with a mixture of wheat fiber and psyllium husk fiber plus three antioxidants. Cereal Chem. 1997; 74: 207–211.
- Talukder, R., Talapatra, S., Ghoshal, N. and Raychaudhuri, S. S. Antioxidant activity and high-performance liquid chromatographic analysis of phenolic compounds during in vitro callus culture of Plantago ovata Forsk and effect of exogenous additives on accumulation of phenolic compounds J Sci. Food Agric., 2016; 96: 232–244.
- Higashi, K., Ishikawa, T., Shige, H., Tomiyasu, K., Yoshida, H., Ito, T. Olive oil increases the magnitude of postprandial chylomicron remnants compared to milk fat and safflower oil. J. Am. Coll. Nutr. 1997; 16 (5): 429–434.
- Lichtenstein, A.H., Kennedy, E., Barrier, P., Danford, D., Ernst, N.D., Grundy, S.M., Leveille, G.A., Van Horn, L., Williams, C.L., Booth, S.L., 1998. Dietary fat consumption and health. Nutr. Rev. 1998; 56 :3–19.
- Terpstra, A. H., Lapre, J. A., de Vries, H. T. & Beynen, A.C. Hypocholesterolemic Effect of Dietary Psyllium in Female Rats. Ann. Nutr. Metab. 2000; 44: 223-228.
- Nofer, J.R., Kehrel, B., Fobker, M., Levkau, B., Assmann, G., von Eckardstein, A. HDL and arteriosclerosis: beyond reverse cholesterol transport. Atherosclerosis, 2002; 161 (1):1–16.
- Mustad, V.A., Etherton, T.D., Cooper, A.D., Mastro, A.M., Pearson, T.A., Jonnalagadda, S.S., Kris-Etherton, P.M. Reducing saturated fat intake is associated with increased levels of LDL receptors on mononuclear cells in healthy men and women. J. Lipid Res., 1997; 38 (3):459–468.
- Xing, L.C., Santhi, D., Shar, A. G., Saeed, M., Arain, M.A., Shar, A.H., Bhutto, Z.A., Kakar, M.U., Manzoor, R., Abd El-Hack, M.E., Alagawany, M., Dhama, K. and ling, M.C. Psyllium Husk (Plantago ovata) as a Potent Hypocholesterolemic Agent in Animal, Human and Poultry. Int J Pharmacol. 2017; 13: 690-697.
- Anderson, J.W., Allgood, L.D., Lawrence, A., Altringer, L.A, Jerdack, G.R., Hengehold, D.A, Morel, J.G. cholesterol-lowering effects of psyllium intake adjunctive to diet therapy in men and women with hypercholesterolemia: Meta –analysis of 8 controlled trials Am. J. Clin. Nutr. 2000; 71:472-479.
- Raghavendran, H.R., Sathivel, A., Devaki, T. Effect of Sargassum polycystum (Phaeophyceae)-sulfated polysaccharide extract against acetaminophen-induced hyperlipidemia during toxic hepatitis in experimental rats. Mol. Cell. Biochem. 2005; 276 (1-2): 89–96.
- Chander, R., Kapoor, N.K. High-density lipoprotein is a scavenger of superoxide anions. Biochem. Pharmacol. 1990; 40:1663–1665.
- Giannini, E. G., Testa, R., & Savarino, V. Liver enzyme alteration: A guide for clinicians. CMAJ. 2005; 172: 367–379.
- Sudhahar, V., Kumar, S.A., Sudharsan, P.T., Varalakshmi, P. Protective effect of lupeol and its ester on cardiac abnormalities in experimental hypercholesterolemia. Vascul. Pharmacol. 2007; 46 (6): 412–418.
- Obatolu, V., Ketiku, A. & Adebowale, E. Effect of feeding maize / legume mixtures on biochemical indices in rats. Ann Nutr Metab. 2003; 47:170–175
- Modaresi, A., Nafar, M., Sahraei, Z. Oxidative stress in chronic kidney disease. Iran J Kidney Dis. 2015; 9(3):165–79.
- Andrade, K.Q., Moura, F.A., dos Santos, J.M., de Araujo, O.R., de Farias Santos, J.C., Goulart, M.O. Oxidative stress and inflammation in hepatic diseases: Therapeutic possibilities of n-acetylcysteine. Int J Mol Sci. 2015; 16(12):30269–308.
- Sheela, C.G., Augusti, K.T. Antiperoxide effects of S-allyl cysteine sulphoxide isolated from Allium sativum Linn and gugulipid in cholesterol diet fed rats. Indian J. Exp. Biol. 1995, 33 (5): 337– 341.
- Pigeolet, E., Corbisier, P., Houbion, A., Lambert, D., Michiels, C., Raes, M. Glutathione peroxidase, superoxide dismutase, and catalase inactivation by peroxides and oxygen derived free radicals. Mech. Ageing Dev. 1990; 51 (3): 283–297.
- Jung, E. Y., Hong, Y. H., Chang, U. J., Suh, H. J. Anti-obese effects of chitosan and psyllium husk containing vitamin C in Sprague-Dawley (SD) rats fed a high fat diet. Prog. Nutr. 2016; 18 (2): 152-160.
- Patel, M. K., Tanna, B., Mishra, A., Jha, B. Physicochemical characterization, antioxidant and anti-proliferative activities of a polysaccharide extracted from psyllium ( ovata) leaves. Int. J. Biol. Macromol. 2018; 118: 976–987
- Visavadiya, N.P., Narasimhacharya, A.V. Hypocholesteremic and antioxidant effects of Withania somnifera (Dunal) in hypercholesteremic rats. Phytomedicine. 2007; 14 (2-3): 136–142.
- Chen, J., Li, X. Hypolipidemic effect of flavonoids from mulberry leaves in triton WR-1339 induced hyperlipidemic mice. Asia Pac. J. Clin. Nutr. 2007; 16: 290–294.
- Ozyigit, I.I., Kahraman, M.V., Ercan, O. Relation between explant age,total phenols and regeneration response in tissue cultured cotton (Gossypium hirsutum L.). Afr J Biotechnol. 2007; 6:3–8.
- Czerska, M., Mikolajewska, K., Zielinski, M., Gromadzinska, J., Wasowicz, W. Today’s oxidative stress markers. Med Pr. 2015; 66(3): 393–405.
- Satapathy, S. K., Ochani, M., Dancho, M., Hudson, L. K., RosasBallina, M., Valdes-Ferrer, S. I., Olofsson, P. S., Harris, Y. T., Roth, J., & Chavan, S. Galantamine alleviates inflammation and other obesity-associated complications in high-fat diet-fed mice. Mol. Med. 2011; 17: 599–606.
- Bao, Y., Wang, Z., Zhang, Y., Zhang, J., Wang, L., Dong, X., Su, F., Yao, G., Wang, S., & Zhang, H. Effect of Lactobacillus plantarum P-8 on lipid metabolism in hyperlipidemic rat model. Eur J Lipid Sci Tech. 2012; 114: 1230–1236.

This work is licensed under a Creative Commons Attribution 4.0 International License.







