Introduction
Breast cancer is the most common cancer and cause of cancer-related deaths among women, accounting for 1.67 million (25.2%) new cases and 521,907 (14.7%) deaths worldwide.1 Breast cancer is a cancer that develops from the breast tissue. This is an invasive cancer that often appears in women. Some signs of breast cancer are a change of breast shape, dimpled skin, nipple discharge or red scaly patches of skin, and a lump in the breast.2 Based on data of the International Agency for Research on Cancer (IARC) in 2012, the incidence of cancer in women has reached 134 per 100,000 of the population. The highest incidence is breast cancer with 40 cases per 100,000 women followed by cervical cancer reaching 17 cases per 100,000. The estimate of the death toll in Indonesia Globocan for breast cancer is 16.6 deaths per 100,000 population.3
Cancer cases are linked to diet and lifestyle.4 Although surgery and chemotherapy can lower the risks of breast cancer in some women who suffer the disease, the side effects cause them to limit the use. Currently, preventive efforts have become more important. Some natural products have an important role in the discovery and development of drugs for the treatment of various types of deadly diseases including cancer.5 Several studies have investigated the anti-proliferation effects of some phytochemicals derived from fruits and vegetables. Research examines cancer prevention by utilizing foods that contain bioactive compounds such as antioxidants.6,7 More than 1,000 different phytochemicals have been identified as having potential effects against various cancers. These phytochemicals offer considerable advantages because they are safe and may target multiple cell-signaling pathways.8 Related to the high occurrence of breast cancer in Indonesia, T47D cancer cells are often used as research models to identify the ability of anti-proliferation of bioactive compounds contained in fruits, vegetables, cereals, legume or tubers.
Coleus tuberosus is a source of carbohydrate. Some research suggests that it has potential antioxidant and anti-proliferation effects. Bioactive compounds identified in the Coleus tuberosus which include phenols, flavonoids, oleanolic acid, ursolic acid, maslinic acid, and phytosterol have antioxidant, and antiproliferation, apoptosis effects.9-11 Further research on the antioxidant activity and cancer prevention activity is still needed. Thus, this study examines the potential of Coleus tuberosus extract as natural antioxidant and breast cancer prevention agents using T47D cancer cells
Materials and methods
Chemicals
This study investigated the effects of ethanol extracts of Coleus tuberosus flesh and peel on T47D cancer cell line. MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) (Sigma Aldrich), RPMI (Sigma Aldrich), phosphate buffered saline (PBS) (Gibco), DMSO (Sigma Aldrich), and acridine orange – ethidium bromide (Bio Rad) were used.
Preparation of ethanol extracts of Coleus tuberosus flesh and peel
The peels of Coleus tuberosus were separated with peeler from the flesh with a thickness of 1 mm. The flesh and peel were dried in a cabinet dryer for twenty four hours and then crushed and extracted using maceration with 95% ethanol for 7 days. The extracts were stored in a frozen condition.
Evaluation of cellular antioxidant activity of ethanol extracts of Coleus tuberosus flesh and peel in T47D cancer cells
The cellular antioxidant activity was evaluated by referring to Wolfe and Liu12 and Liu and Finley.13 T47D cells grown on a 96-well microplate containing RPMI were supplemented with fetal bovine serum 10% (v/v), penicillin 100 U, and streptomycin 100 mg/ml at 37oC under 5% CO2. After 24 hours of growth, growth medium was removed, and the wells were washed with sterile PBS. The cells were pretreated with ethanol extracts of Coleus tuberosus flesh or peel in concentrations of 100, 200, 400, and 800 µg/ml for 20 mins in triplicate. After 20 mins of pretreatment, growth media containing 25 µM DCFH-DA and PMA (100 ng/ml in DMSO) were added and incubated for 30 minutes. The fluorescence of the cells was measured using a flow cytometer (FACS Calibur BD) at a wavelength of 535 nm. The cellular antioxidant activity was determined by calculating the percentage decrease in the intensity of the reactive oxygen species (ROS) fluorescence:
![]()
where Fito is the oxidative stress control, Fit1 is the cells with bioactive compound treatment, and Fit2 is that of the control without oxidative stress.14
Evaluation of anti-proliferation of T47D cancer cell lines
The anti-proliferation effects were evaluated by referring to Hogan et al.,15 Cells were placed on a 96-well plate at 1.5×104 cells/ml with RPMI plus 10% (v/v) fetal bovine serum, penicillin 100 U and streptomycin (100 mg/ml, 37°C, 5% CO2). These conditions were kept for a one-hour period. Next, cancer cells were treated for about one hour in the experiment media with ethanol extract of Coleus tuberosus peel or flesh extracts at concentrations of 62.5-2,000 µg/ml with the final volume of 100 µl. Cell viability was determined by MTT assay. After one-hour incubation, the media were treated and eliminated at the end of the incubation period and then washed with HBSS. The cells were incubated with 10 µL of MTT reagent solution (5 mg/ml in RPMI) added to each of the wells for 24 hours.
The mixtures were left for one night. The absorbance was recorded at 570 nm with a multipliable plate reader. The absorbance data required for cell viability are expressed as a percentage of the control (the number of living cells in control cells) during the experiment. After each treatment, the MTT assay was expressed as: Cell viability (%) =
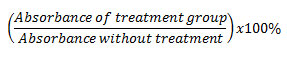
Cell Cycle Arrest
A total of 106 cell/well were distributed in 6-well plates and incubated at 37oC for cell adaptation. T47D cells incubated with ethanol extract of Coleus tuberosus peel or flesh extracts with concentrations of 7.8125, 15.625, 31.25, 62.5 and 125 µ g/ml for 24 hours. After 24 hours, cells incubated were taken and washed twice using ice-cold PBS. The cells were fixed and permeabilized with 70% ice-cold ethanol at 4oC for one hour. Then, T47D cells were washed with PBS and resuspended in a solution containing propidium iodide stain (50 ml/ml) and RNase A (250 mg/ml). Cell suspensions were incubated for 30 min at room temperature, followed by fluorescence-activated cell sorting (FACS; cater-plus flow cytometry; Becton Dickinson co., Germany) using 10,000 cells per group.16 The percentages of cells in the G0-G1, S, and G2/M phases were analyzed using Modfit LT Cell Cycle 3.0 analysis software (Becton Dickinson).
Induction of apoptosis
The morphological changes due to apoptosis induction after treatment with the extracts were evaluated using acridine orange staining and ethidium bromide. T47D cells were cultured on the cover slip on 105 cells/well. The medium was replaced with medium containing ethanol extract samples of Coleus tuberosus flesh or peel extracts (62.5 µg/ml). The cells were then incubated for 24 hours at 37oC in 5% CO2. The medium was removed and solution of acridine orange and ethidium bromide (100 µg/ml of acridine orange (Bio-Rad) in PBS and 100 µg/ml of ethidium bromide (Bio-Rad) in PBS for 5 minutes) was added. The cover slips were mounted on a glass object and then observed under a fluorescence microscope (Zeiss MC 80).17
Statistical analysis
Each experiment was performed three times and data were expressed as mean ± SD. The testing was performed by two way anova of the antioxidant activity, anti-proliferation effects and cell cycle arrest. If the obtained results are different significantly, then it was followed with Duncan’s Multiple Range Test (DMRT)
Results and discussion
Celullar antioxidant activity of ethanol extracts of Coleus tuberosus flesh and peel on T47D
Treatment of T47D cancer cells with ethanol extracts of Coleus tuberosus flesh (EECF) or ethanol extract of Coleus tuberosus peel (EECP) inhibited PMA-induced ROS generation in dose-dependent manner. The percentage decrease of ROS in cancer cells incubated with peel extract T47D was higher than that of cells incubated with ethanol extract of Coleus tuberosus flesh or peel (Fig. 1).
The percentage decreases of ROS in the T47D cancer cells incubated with ethanol extract of Coleus tuberosus peel extract at concentration of 100, 200, 400, and 800 µg/ml were 31.30 ± 3.27, 46.93 ± 9.78, 64.96 ± 7.23, and 77.09 ± 1.78, respectively (Fig.1). The percentage decreases of ROS in the T47D cells incubated with ethanol extract of Coleus tuberosus flesh at concentrations of 100, 200, 400, and 800 µg/ml were 22 ± 7.93, 38.70 ± 5.49, 55.96 ± 2.04, and 62.08 ± 1.54, respectively.
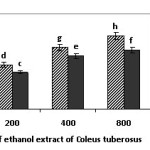 |
Figure 1: The percentage decrease in reactive oxygen species (ROS) with the treatment of ethanol extract of Coleus tuberosus flesh and peel on T47D cells induced with phorbol myristate acetate,Note: Different notations suggest a significant difference (p < 0.05). Click here to View figure |
Table 1: IC50 cellular antioxidant activities of ethanol extracts of Coleus tuberosus flesh and peel on T47D cancer cells
| Bioactive compounds | Celullar antioxidant activity IC50 (µg/ml) |
| Ethanol extract of Coleus tuberosus flesh | 287.13±10.35 |
| Ethanol extract of Coleus tuberosus peel | 217.86±12.96 |
The current study investigated the antioxidant activity and anti-proliferation activity of ethanol extracts of Coleus tuberosus flesh and peel on T47D cancer cell lines. The results show that the extracts have the ability to decrease ROS in T47D cells induced by PMA. The ability is associated with content of bioactive compounds in Coleus tuberosus, such as phenols, flavonoids, oleanolic acid, ursolic acid, maslinic acid, and phytosterol.9,18,19 Some research suggests that incubation with extracts containing bioactive compounds may lower the ROS in cancer cells induced by free radicals and improve the antioxidant defense system.20-24 Therefore, treatment with bioactive compounds could prevent DCFH oxidation and decrease the formation of fluorescent DCF.14,25,26
The antioxidative ability of the flesh and peel extracts comes from the interaction between bioactive compounds in the extract, which have a synergistic effect. This supports the research of Náthia-Neves et al.,27 and Li et al.,28 indicating that the different antioxidant ability is related to the different content of bioactive compounds in extracts. A high amount of bioactive compounds tends to result in high ability to neutralize free radicals.
The mechanism of bioactive compounds could have the ability to maintain the fluidity of cell membranes by capturing ROS, so that cellular level communication can occur normally, including the entire signals associated with activation of antioxidant enzymes (Nrf-2-ACRE).29 Increased expression of Nrf-2-ARE can increase the antioxidant defense system (GPx, SOD, glutathione, CAT, vitamin C, vitamin E, and beta-carotene). Further, an increase in the antioxidant defense system of cells can increase the ability to neutralize singlet oxygen (1O2), hydrogen peroxide (H2O2), superoxide anion radicals (O2•-), and hydroxyl radicals (OH•) due to the induction of PMA. An increase in glutathione, vitamin C, vitamin E, and beta-carotene in the cell can increase the capture of free radicals that are present in the cell and reduce the amount of free radicals in T47D cells.
The effects of ethanol extract of Coleus tuberosus flesh and peel extracts on the proliferation of T47D cancer cells
The anti-proliferative activity of flesh and peel extract on T47D cancer cells was characterized by MTT assay. Cells were treated by applying different extract concentrations of 62.5, 125, 250, 500, 1000, and 2000 µg/ml and incubating the cells for 24 hours (Fig. 2).
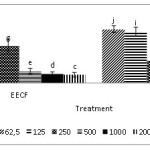 |
Figure 2:. The percentage of viability cells produced by treatments with an ethanol extract of Coleus tuberosus flesh (EECF) and peel (EECP) at different concentrations after a 24-hour incubation. Data are presented as the mean of three independent experiments. Means followed by different letters is significant difference (p < 0.05). Click here to View figure |
Fig. 2 indicates that the incubation of T47D cancer cells with flesh extract and peel extract can decrease the percentage of viable cells compared to the control in a dose-dependent manner. The anti-proliferative activity of peel extract and flesh extract is expressed as the inhibition concentration (IC50). A lower IC50 value indicates a higher anti-proliferative effect of a sample. Table 2 shows that the IC50 levels after a 24-hour incubation period are 887.05 ± 5.03 µg/ml (peel extract) and 548.18 ± 4.52 µg/ml (flesh extract). The peel extract seems to have a higher anti-proliferative effect than the flesh extract at all concentrations, which corresponds to its lower IC50.
Table 2: IC50 anti-proliferation activities of ethanol extracts of Coleus tuberosus flesh and peel on T47D cancer cells
| Bioactive compounds | IC50 (µg/ml) |
| Ethanol extract of Coleus tuberosus flesh | 887.05±5.03 |
| Ethanol extract of Coleus tuberosus peel | 548.18±4.52 |
The peel extract and flesh extract may cause a significant growth inhibition of T47D in a dose-dependent manner. In particular, the peel extract showed higher anti-proliferative activities than the flesh extract towards T47D cancer cells. These results are in line with previous research by Nugraheni et al.,10 who proved that peel extract has a greater anti-proliferation activity than flesh extract. The inhibition ability for the proliferation of cancer cells is supported by the presence of various bioactive compounds in the flesh and peel, such as carotenoids, vitamins, and other polyphenol phytochemicals. The peels also contain more bioactive compounds than the flesh.30
The anti-proliferation activities of the flesh extract and peel extract on T47D cancer cells may have been caused by the interactions between bioactive compounds in the extracts. These bioactive compounds include ursolic acid, oleanolic acid,9 phenol and flavonoids compounds,18 maslinic acid, and phytosterols, such as beta-sitosterol, stigmasterol, and campesterol.11 These provide a synergy effect in determining the anti-proliferation ability. Prior research also suggests that maslinic acid, phytosterols, and phenolic compounds may have anti-proliferative activity in some cancer cells.31-35 In particular, ursolic acid and oleanolic acid are known as being capable of inhibiting the proliferation of cancer cells.10,36-37
Cell cycle arrest
Evaluation using flow cytometer used to identify the effect of ethanol extract of Coleus tuberosus flesh and peel on cell cycle arrest (125, 62.5, 31.25, 15.625, and 7.8125 µg/ml). Cell cycle arrest occurs in the S and G2/M phases (Fig. 3 and Fig. 4). This indicates that treatment with flesh extract or peel extract causes changes in the cancer cell cycles
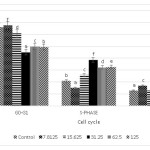 |
Figure 3: Effect of ethanol extract of Coleus tuberosus flesh on cell cycle arrest. T47D cells were treated with 0 (control), 7.8125, 15.625, 31.25, 62.5, and 125 µg/ml of peel extract for 24 h, after which the cells were stained with PI and analyzed for DNA content by flow cytometer. Means followed by different letters differ statistically (p < 0.05). Click here to View figure |
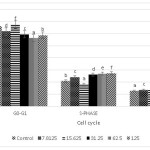 |
Figure 4: Effect of ethanol extract of Coleus tuberosus peel extract on cell cycle arrest. T47D cells were treated with 0 (control), 7.8125, 15,625, 31.25, 62.5, and 125 µg/ml of peel extract for 24 h, after which the cells were stained with PI and analyzed for DNA content by flow cytometer. Means followed by different letters differ statistically (p < 0.05). Click here to View figure |
T47D cells incubated with ethanol extract of Coleus tuberosus flesh or peel showed cell cycle arrest in the S and G2/M phases, in a dose dependent manner. Some research suggests that bioactive compounds such as flavonoids, polyphenols, ursolic acid and oleanolic acid are potent regulators which can cause the S-phase and G2-M cell cycle arrest.38-42 The ability of ethanol extract of flesh and peel of Coleus tuberosus induces the cell cycle arrest could cause an inhibition of cell proliferation.
Acridine orange staining-ethidium bromide on T47D cells
The phenotypic characteristics of cells, which are separately treated with flesh extract and peel extract, were evaluated by conducting a microscopic inspection of the overall morphology. The results show that either the flesh extract or peel extract performs a significant evidence of cells death even after the 24-hour incubation period. Staining with acridine orange and ethidium bromide on all treatment was used to find out the capabilities of extracts in inducing apoptosis on T47D cells. The treated cells indicated a nuclear condensation after the 24-hour treatment. The positive control cells showed bright green nuclei (Fig. 5A), while the negative control cells produced an orange-red color with uniform intensity (Fig. 5B). The apoptotic cells had an orange color (Fig. 5C).
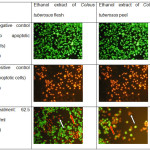 |
Figure 5: (A) Untreated cells, (B) treated cells with doxorubicin, (C) treatment with concentration of 62.5 µg/ml. Click here to View figure |
Treatment with T47D cancer cells with ethanol extract of Coleus tuberosus flesh and peel induced apoptosis, which was characterized by changing shape, condensation, and DNA degradation of the cells (Figure 5). The combination of acridine orange and ethidium bromide can be used on both living and dead cells undergoing apoptosis. Green fluorescence occurs when bound to double-stranded DNA in living cells, and red fluorescence occurs when bound to single-stranded DNA, predominantly in dead cells. Cells that experience apoptosis early will experience fragmentation of DNA, which results in the green color of the nucleus. When apoptotic processes end, DNA fragmentation results in a red or orange,43 because cells lose the integrity of the membrane,44 so that the ethidium bromide can enter into the cell and intercalate with the DNA that has undergone fragmentation. Shikha Srivastava et al.,45 indicated that the treatment of cancer cells by flavonoids led to the fragmentation and degradation of cellular DNA, resulting in apoptosis.Fig. 5. A. Untreated cells, B. treated cells with doxorubicin, C. treatment with concentration of 62.5 µg/ml. Cellular and nuclear morphological changes of T47D cancer cells due to exposure to various concentrations of Coleus tuberosus flesh or peel extract after a 24-hour incubation. Viable cells have a uniform colour, bright green with an organized structure. Early apoptotic cells have green nuclei, but the perinuclear chromatin condensation is visible as bright green fragment or patches (red arrow). Late apoptotic cells indicated have orange to red-colored nuclei with fragmented chromatin (white arrow).
This research shows that ethanol extracts of Coleus tuberosus flesh and peel have the ability as antioxidant for T47D cells, inhibiting the proliferation, inducing cell cycle arrest and inducing apoptosis. This results show the potential of Coleus tuberosus as a source of natural antioxidants which the ability to inhibit proliferation of T47D cancer cells.
Conclusion
Ethanol extracts of Coleus tuberosus flesh and peel can reduce reactive oxygen species in T47D cells induced with the radical generator PMA. The ability of the peel extract to reduce oxidative stress is better than that of the flesh extract. Incubation of cells with the extracts can reduce proliferation, causing cell cycle arrest in the S and G2-M phases, and induced apoptosis.
Conflict of interest
The authors declare no conflict of interest in this work
Acknowledgements
The author would like to thank the Directorate General of Higher Education of the Republic of Indonesia for funding the research under contract number 447a/HB-Multitahun/UN34.21/2013.
References
- Mardela A. P, Maneewat K, Sangchan H. Breast cancer awareness among Indonesian women at moderate-to-high risk. Nursing and Health Sciences. Nursing and Health Sciences.2017;19:301–306
CrossRef - Kabel A. M, Baali F. H. Breast Cancer: Insights into Risk Factors, Pathogenesis, Diagnosis and Management. Journal of Cancer Research and Treatment.2015;3:28-33
- The Ministry of Health of Republic Indonesia. Infodatin, Data and information centre of the Ministry of health of Indonesia, the month of Breast Cancer Care. ISSN. 2016; 2442-7659
- Salem A. A, Mackenzie G. G. Pancreatic Cancer: A Critical Review of Dietary Risk. Nutrition Research: https://doi.org/10.1016/j.nutres.2017.12.001 (2017)
CrossRef - Muhammad N, Steele R, Isbell T. S, Philips N, Ray R. B. Bitter melon extract inhibits breast cancer growth in preclinical model by inducing autophagic cell death. Oncotarget.2017;8:66226-66236
CrossRef - Katiyara, S. K, Pala, H. C, Prasa R. Review Dietary proanthocyanidins prevent ultraviolet radiation-induced nonmelanoma peel cancer through enhanced repair of damaged DNA-dependent activation of immune sensitivity. Seminars in Cancer Biology.2017;46:138–145
CrossRef - Sinhaa D, Biswasb J, Nabavic S. M, Bishayee A. Review Tea phytochemicals for breast cancer prevention and intervention: From bench to bedside and beyond, Seminars in Cancer Biology.2017;46:33–54
CrossRef - Russo G.L, Tedesco I, Spagnuolo C, Russo M. Review Antioxidant polyphenols in cancer treatment: Friend, foe or foil?. Seminars in Cancer Biology.2017;46: 1–1
CrossRef - Nugraheni M, Santoso U, Hastari W. Potential of Coleus tuberosus as an antioxidant and cancer chemoprevention agent. International Food Research Journal.2011;18: 1471-1480
- Nugraheni M, Santoso U, Hastari W. In vitro antioxidant, antiproliferative and apoptosis effect of Coleus tuberosus L. African Journal of Food Science. 2011;5:232 – 241
- Hsum Y. W, Yew W. T, Hong P. L. V, Soo K. K, Hoon L. S, Chieng Y. C, Mooi L. Y. Identification and evaluation of potential anti-tumor promoting compounds from tubers of Coleus tuberosus. International PSE Symposium on Natural Products in cancer Therapy, Naples, Italy 23-26 September 2008
- Wolfe K, Wu X, Liu R. H. Antioxidant activity of Apple peels. Journal of Agricultural and Food Chemistry.2003;51: 609-14
CrossRef - Liu H, Finley J. Potential cell culture models for antioxidant research. Journal of Agricultural and Food Chemistry. 2005;53:4311-4314
CrossRef - Muanda F. N, Bouayed J, Djilani A, Yao C, Soulimani R, Dicko A. Chemical composition and, cellular evaluation of the antioxidant activity of Desmodium adscendens leave. Evidence-Based Complementary and Alternative medicine : Article ID 620862 (2011).
CrossRef - Hogan S, Chung H, Zhang L, Li J, Lee Y. W, Yumin, Zhou K. Antiproliferative and Antioxidant Properties of Antocyanin-rich extract from Acai. Food Chemistry: 118: 208-214 (2010)
CrossRef - Cho J. H, Lee, J. G, Yang Y. I, Kim, J. H, Ahn J. H, Nam I, Baek, Lee K. T, . Choi J. H. Eupatilin, a dietary flavonoid, induces G2/M cell cycle arrest in human endometrial cancer cells. Food and Chemical Toxicology: 49: 1737-1744 (2011).
CrossRef - Meiyanto E, Agustina D, Suparjan A. M, Da’I M. PVG-O induces apoptosis on T47D Breast cancer cells line through caspase-3 activation. Jurnal Kedokteran Yarsi: 15: 075-079 (2007).
- Nugraheni M, Lastariwati B, Windarwati. Antioxidant Activity of The Ethanolic Extracs of Peel and Flesh of Coleus tuberosus, 1st International Conference Biodiversity Proceedings.2016
- Mooi L. Y, Wahab N. A, Lajis N. H, Ali, A. M. Chemopreventive properties of phytosterols and maslinic acid extracted from Coleus tuberosus in inhibiting the expression of EBV early-antigen in Raji cells. Journal Chemistry and Biodiversity.2010; 7: 1267-1275
CrossRef - Ighodaro O. M, Akinloye O. A. First line defence antioxidant-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid, Alexandria Journal of Medicine article in press https://doi.org/10.1016/j.ajme.2017.09.001 .2017
CrossRef - Martinez-Martos J. M, Mayas M. D, Carrera P, de Saavedra J. M. A, Agesta R. S, Arrazola M, Jesus M. Phenolic compounds oleuropein and hydroxytyrosol exert differential effects on glioma development via antioxidant defense systems. Journal of Functional Foods.2014;11:221-234
CrossRef - Li D, Ren D, Luo Y, Yang X. Protective effects of ursolic acid against hepatotoxicity and endothelial dysfunction in mice with chronic high choline diet consumption. Chemico- Biological Interaction.2016;258:102-107
- Kondeva-Burdina M, Shkondrov A, Simeonova R, Vitcheva V, Krasteva I, . Ionkova I. In vitro/in vivo antioxidant and hepatoprotective potential of defatted extract and flavonoids isolated from Astragalus spruneri Boiss. (Fabaceae). Food and Chemical Toxicology.2017;111: 631-640
CrossRef - Chen J, Song M, Li Y, Zhang Y, Taya K, Li C. M. The effect of phytosterol protects rats against 4-nitrophenol-induced liver damage. Environmental Toxicology and Pharmacology.2016;41: 266-271
CrossRef - Liu S, Huang H. Assessments of antioxidant effect of black tea extract and its rationals by erythrocyte haemolysis assay, plasma oxidation assay and cellular antioxidant activity (CAA) assay, Journal of Functional Foods.2015;18:1095-1105
CrossRef - Pichette K. G. A, Legault J. Sensitive cell-based assay using DCFH oxidation for the determination of pro- and antioxidant properties of compounds and mixtures: Analysis of fruit and vegetable juices. Food Chemistry.2009;115:720-726
CrossRef - Nathia-Neves G, Tarone A. G, Tosi M. M, Junior M. R. M, Meireles M. A. A., Extraction of bioactive compounds from genipap (Genipa americana L.) by pressurized ethanol: Iridoids, phenolic content and antioxidant activity. Food Research International.2017;102:595-604
CrossRef - Li X, Wang T, Zhou B, Gao W, Cao J, Huang L. Chemical composition and antioxidant and anti-inflammatory potential of peels and flesh from 10 different pear varieties (Pyrus spp.). Food Chemistry.2014;152:531-538
CrossRef - Zhao H. D, Zhang F, Shen G, Li Y. B, Jing H. R, Ma L. F, Yao J. H, Tian X. F., Sulforaphane protects liver injury induced by intestinal ischemia reperfusion through Nrf2-ARE pathway. World Journal of Gastroenterology.2010;16:3002-3010
CrossRef - Kim H, Kim H, Mosaddik A, Gyawali R, Ahn K.S, Cho S. K, Induction of apoptosis by ethanolic extract of mango peel and comparative analysis of the chemical constitutes of mango peel and flesh. Food Chemistry. 2012;133: 416-422
CrossRef - Parra A, Rivas F, Martin-Fonseca S, Garcia-Granados A, Martinez A. Maslinic acid derivatives induce significant apoptosis in b16f10 murine melanoma cells, European Journal of Medicinal Chemistry.2011;46:5991-6001
CrossRef - Shahzad N, Khan W, Shadab M. D, Ali A, Saluja S. S, Sharma S, Al-Allaf F. A,. Abduljalee Z, Ibrahim I. A. A, Abdel Wahab A. F, Afifiy M. A, Al-Ghamdi S. S., Phytosterols as a natural anticancer agent: Current status and future perspective. Biomedicine & Pharmacotherapy.2017;88:786-794
CrossRef - Cilla A, Attanzio A, Barbera R, Tesoriere L, Livrea M.A. Anti-proliferative effect of main dietary phytosterols and β-cryptoxanthin alone or combined in human colon cancer Caco-2 cells through cytosolic Ca+2 – and oxidative stress-induced apoptosis. Journal of Functional Foods.2015;12:282-293
CrossRef - Yaglioglu A. S, Eser F. Screening of some Juniperus extracts for the phenolic compounds and their antiproliferative activities. South African Journal of Botany.2017;113:29-33
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.







