Recent Advances in Non-Sugar Components of Sugarcane Juice: A Review
1Department of Tea and Food Science, Xinyang Normal University, Xinyang, China
2Dabie Mountain Laboratory, Xinyang, China
3Huaihe Campus Administrative Committee, Xinyang Normal University, Xinyang, China
Corresponding Author Email: fangkun@xynu.edu.cn
DOI : http://dx.doi.org/10.12944/CRNFSJ.14.1.2
ABSTRACT:As the global economy booms, the quality of refined sugar and its derivatives is attracting increasing attention. Mixed juice extracted from sugarcane contains many impurities other than sucrose, such as pigments, nitrogenous substances, organic acids, colloidal substances, lipids, and inorganic substances. Although some of these impurities (e.g., amino acids, vitamins, flavonoids, and minerals) are beneficial to human health, they degrade the quality of sugar and its derivatives, affecting their production and utilization. Understanding the non-sugar components and colored substances in sugarcane juice mixtures is essential to ensure product quality, prolong the storage period, and improve the organoleptic properties. This review summarizes the non-sugar components of sugarcane juice, including their composition, chemical structures, and contents. Additionally, it highlights four colored substances (natural pigments, melanoidins, caramels, and hexose alkaline degradation products) in sugarcane juice and their formation mechanisms.
KEYWORDS:Chemical Structures; Colored Substances; Formation Principle; Non-Sugar Components; Sugarcane Juice.
Introduction
Sugarcane (Saccharum officinarum L.) originated in the subtropical region near the Tropic of Cancer, is one of the most economically valuable graminaceous plants in the world, and is a key player in the agricultural economy of many countries around the globe, such as Brazil, India, China, and Thailand.1 Sugarcane rosettes are erect, thick, and juicy, often covered with white powder; sugarcane leaves are alternate, with small, sharp teeth on the edges; and the flower spikes are in compound racemes. The widespread cultivation of sugarcane emphasizes its status as an important cash crop.2 The main use of sugarcane is in the production of sugar, which is a key ingredient in the food and beverages industry, meeting approximately 80% of global sugar demand.3 The cultivation of sugarcane can promote the development of related industries, such as sugar production,4 chemical industries,5 paper industry,6 etc. A small portion of sugarcane is also used to produce fuel ethanol. The use of sugarcane for fuel ethanol production offers significant advantages.7 In addition, a small portion of it is used to produce refined sugar, white sugar, and brown sugar, etc., and its high-value utilization is less and not thoroughly researched.8
Sugarcane is a complex material whose composition and characteristics are influenced by factors such as the climate, phytosanitary conditions, soil, harvesting process, age, and variety. Generally speaking, sugarcane juice contains not only 10-15% of saccharides, but also many substances needed by the human body, such as proteins, amino acids, vitamins, organic acids (malic, citric, succinic, fenugreek, nicotinic, aconitic, methyl fenugreek, and glycolic acid), polyphenols, flavonoids, and trace elements needed by the human body, such as calcium, iron, and zinc and other nutritive and active substances.9 Drinking sugarcane juice not only replenishes energy and water needed by the human body, but also clears heat and quenches thirst, nourishes lungs and stomach, and has the effect of preventing dental caries, lowering blood pressure, anticarcinogenic, antioxidant, cellular protection, protection against skin damage, enhancement of immunity, and diuretic, and nourishment.10 The appearance of sugarcane juice is a critical factor affecting its quality. Its color problem is mainly caused by the phenolic substances, where small-molecule phenolic acids are easily oxidized by air and certain oxidative enzymes and become colored, undergoing the so-called browning, which makes the juice look turbid and affects the quality, clarity, and color of sugarcane juice.11 Considering the numerous other uses of sugarcane constituents, such as sugarcane wax, leaves, and especially phytocompounds derived from sugarcane juice, compositional studies could be carried out to obtain their nutritional properties.12
Despite the many benefits of sugarcane juice, unfortunately, studies on the relationship between the nutrient content of sugarcane juice and its byproducts have not yet appeared in the literature, and an understanding of this relationship could be useful in the development of quality standards for foods based on sugarcane and its processing. This review provides an overview of the non-sugar components of sugarcane juice, focusing on the color-forming substances and their formation mechanisms, intending to provide information on the range of variability of the different sugarcane components, which can contribute to the understanding of their nutritional attributes and characteristics. This review can support databases such as the International Sugar Society, FAOSTAT, the Ministry of Agriculture, and the Food Composition List, and provide a basis for new research on the sugarcane industry.
Non-sugar components in sugarcane
Sugarcane’s physical composition is primarily segmented into four key constituents: fibers, insoluble solids, and water.13 The relative abundance of these components is subject to variation based on the specific agricultural and industrial procedures used in sugar production, as depicted in Fig. 1. The fiber component, originating from the stems, comprises organic solid fractions and exhibits notable heterogeneity. Insoluble solids, which resist dissolution in water, are predominantly composed of inorganic matter, soil particles, and various extraneous elements. The prevalence of these substances is dictated mainly by the methodologies employed in processing the sugarcane, including cutting techniques and harvesting approaches.14 Conversely, the soluble solid portion primarily comprises sucrose, but it may also encompass a minor presence of other chemical constituents such as waxes.15 In terms of its overall makeup, sugarcane consists of 53.6% juice (based on wet weight) and 26.7% fiber (based on dry weight). Its components are enriched with carbohydrates (including glucose, fructose, and sucrose), amino acids, and organic acids.16 The chemical composition of sugarcane stalks varies, typically including 12% to 16% of soluble sugars, 2% to 3% of non-sugar carbohydrate compounds, and 63% to 73% water content.17 Significantly, the juice extracted from sugarcane is rich in starch, pectin, proteins, and polyphenols. As sugar products undergo processing, non-sugar colloids accumulate and coalesce into stable and unstable aggregates. These aggregates detrimentally affect the clarity, viscosity, and color of sugarcane juice, thereby impacting the quality of the derived sugar products.18 Furthermore, the non-sugar content in sugarcane juice includes a variety of colloids, nitrogenous substances, organic acids, and ash.19 Although these components are essential nutrients for sugarcane growth, they pose significant challenges in sugar milling due to their impact on critical stages such as clarification, filtration, crystallization, and purification. The process of producing white sugar predominantly involves the separation and purification of cane sugar from these non-sugar elements.20 A thorough understanding of the nature and properties of these non-sugar fractions is imperative for their effective separation, removal, or transformation into byproducts beneficial to the sugar industry. This section aims to delve deeper into the impurities present in sugarcane, with a particular focus on analyzing their chemical composition.
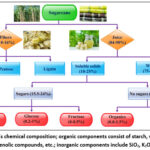 |
Figure 1: Sugarcane’s chemical composition; organic components consist of starch, waxes, amino acids, organic acids, phenolic compounds, etc.; inorganic components include SiO2, K2O, P2O5, Fe2O3, etc |
Colloidal substances
Polysaccharides, abundant and essential macromolecules in living organisms, naturally occur in sugarcane. Sugarcane juice typically contains colloidal polysaccharides, such as cell wall polysaccharides, pectin, starch, and dextran, at concentrations ranging from 0.1% to 0.2%.21 The characteristics of these polysaccharides are distinct:
Cell wall polysaccharides, intrinsic to sugarcane, comprise xylose, arabinose, galactose, rhamnose, glucose, and glucuronic acid. During the pressing of sugarcane, these polysaccharides partially disperse in the juice and undergo gradual hydration and dissolution upon heating.22
Pectin, a high-molecular-weight polymer of D-galacturonic acid connected through α-1,4 glycosidic bonds, is abundant in immature sugarcane but less so in mature stalks. Soluble in hot water, pectin is a typical hydrophilic colloid that forms pectic acid under acidic or basic conditions, and it can precipitate as calcium salt complexes, especially in higher pH environments.23
Starch, a glucose polymer linked by α-1,4 (Amylose) and α-1,6 (Amylopectin) glycosidic bonds, does not dissolve in cold water but forms a paste and hydrophilic colloids in hot water. This characteristic is a primary factor in reducing the filtration rate of back-soluble syrup in sugar refineries.24
Dextran, a glucose polymer with molecular masses ranging from tens of thousands to millions, mainly results from the action of the microorganism Leu. mesenteroides on sucrose.25 Fresh sugarcane contains minimal dextran, but its content increases significantly in sugarcane that is not fresh or is infected with bacteria.26 Dextran contributes to the cloudiness of sugarcane juice, increases viscosity, and impacts filtration and crystallization processes in sugar production.27
Due to their high molecular weight, sugarcane polysaccharides form colloidal solutions upon dissolution in water. This transformation is primarily due to the abundance of hydrophilic hydroxyl groups in their molecular structure, which are responsible for the turbidity observed in sugarcane juice.28 Furthermore, the presence of these polysaccharides notably escalates the viscosity of the solution, thereby decelerating the filtration process. This increase in viscosity also impedes the growth of sucrose crystals and often leads to substantial incorporation of polysaccharides into the crystals. Such integration adversely affects the quality of the sugar product. Additionally, polysaccharides contribute to an increased retention of sugar in the molasses, a factor detrimental to the efficiency of the sugar production process.29
Nitrogenous organic substances
In sugarcane, nitrogenous organic compounds are predominantly proteins, amino acids, and amides. The concentration of these substances varies with the sugarcane variety, geographical region, and the amount of nitrogen fertilizer used, generally constituting approximately 0.3% to 0.5% of the sugar juice brix.30 In freshly matured sugarcane, proteins comprise about 65% to 70% of the nitrogenous content. However, this proportion can decrease to 40% or even below 20% in sugarcane that is not fresh, immature, or affected by natural adversities such as wind damage, frost, or insect infestations. This decrease is due to the hydrolysis of proteins, altering the composition of nitrogenous substances.31 Proteins are high molecular weight, nitrogen-containing organic compounds, typically comprising about 16% nitrogen and having molecular masses generally ranging from 10,000 to 100,000, with some extending to several hundred thousand. They are formed by the dehydration condensation of 200-1000 different amino acids and amino amides. Research indicates that the principal amino acids in sugarcane include aspartic acid, threonine, serine, alanine, valine, isoleucine, leucine, tyrosine, phenylalanine, histidine, and proline, present in concentrations around 10-100 mg/mL. Additionally, trace amounts of glutamic acid, glycine, cystine, methionine, lysine, and arginine are present (Fig. 2).32 During the clarification of sugar juice, uncoagulated proteins remaining in the clear juice are not eliminated and pose challenges to subsequent sugar production. As colloidal substances, they increase the viscosity of the sucrose juice, impede filtration, and react with reducing sugars during evaporation and sugar processing, thereby deepening the color of sugar products.33 Although proteins composed of amino acids are vital nutrients, these uncoagulated proteins become a primary source of “harmful nitrogen” in the sugar-making process.34
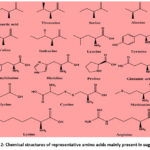 |
Figure 2: Chemical structures of representative amino acids mainly present in sugarcane |
Colored substances
The coloration of sugarcane juice results from a wide array of substances, some inherent to the sugarcane while others develop through chemical transformations. Several of these colored compounds originate naturally in the juice. In contrast, others evolve from initially colorless or pale precursors in sugarcane. These precursors undergo various chemical reactions, ultimately leading to the formation of dark-colored substances.35
Natural pigments in sugarcane
Sugarcane’s natural pigments are categorized into fat-soluble and water-soluble pigments.36 Fat-soluble pigments, which do not dissolve in water but are soluble in organic solvents such as acetone and benzene, primarily include chlorophyll a (C55H72O5N4Mg) and chlorophyll b (C55H70O6N4Mg), lutein (C40H56O2), and carotene (C40H56). Their chemical structures are detailed in Fig. 3. In sugarcane juice, these pigments are typically found mixed with various fats or encapsulated in protein coverings, existing as suspended particles. During the heating of sugarcane juice, proteins coagulate, causing these particles to solidify along with them.37 If the clarification step is efficiently executed in the sugar production process, most of these pigments are removed and are in the filtrate. Conversely, ineffective clarification resulting in cloudy clear juice allows some of these pigments to remain dispersed within the juice and other lipids, leading to undesirable effects.
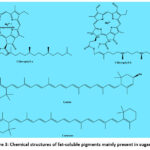 |
Figure 3: Chemical structures of fat-soluble pigments mainly present in sugarcane |
Phenolic compounds
Phenolic compounds, a class of secondary metabolites found abundantly in plants, encompass a variety of substances such as flavonoids, stilbenes, phenolic acids, tannins, and lignans.38 These compounds are renowned for their health-promoting properties,39 including anti-tumor,40 anti-inflammatory,41 antioxidant,42 antibacterial,43 and cardioprotective activities.44 Owing to these benefits, they find extensive applications in foods,45 pharmaceuticals,46 and cosmetics.47 Like other plants, such as fruits, vegetables, nuts, and seeds, sugarcane is rich in phenolic acids and flavonoids, among other phenolics.48 In a notable study, Nakasone et al.49 identified five antioxidant compounds in kokuto (a traditional Japanese sugarcane confectionery), including syringaresinol, medioresinol, and others. Takara et al.50 later expanded this list to 13 phenolic compounds in Kokuto non-centrifuged cane sugar. Recently, more phenolics have been isolated from sugarcane and its derivatives, such as chlorogenic acid51 and ferulic acid.52 These compounds vary depending on the sugarcane variety, plant tissues, environmental conditions, and processing methods. The total phenolic content in commercial sugarcane juice is reported to be between 418 and 567 mg GAE/L.53 The primary phenolic compounds in sugarcane are flavonoids, derivatives of trichothecene, apigenin, or lignan, and members of the hydroxycinnamic acid group like caffeic acids and ferulic acids.54 Studies by Almeida et al.55 showed that sugarcane culms mainly contain hydroxycinnamic acid, but this profile changes to predominantly flavonoids post-juicing. Research by Zhao et al.56 identified eight phenolic acids in sugarcane bagasse extract, including vanillic acid and sinapic acid. Furthermore, several flavone glycosides have been found in sugarcane leaves and juice.57 Wang et al.58 quantified the sugarcane polyphenol extract (SPE) using high-performance liquid chromatography (HPLC), revealing (-)-epicatechin (EC) as its major component, as indicated in Fig. 4, which depicts the chemical structures of significant phenolic compounds found in sugarcane.
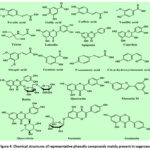 |
Figure 4: Chemical structures of representative phenolic compounds mainly present in sugarcane |
Policosanols and long-chain aldehydes
Policosanol, commonly derived from the waxes of sugarcane epidermis and leaves, is a term for long-chain (ranging from C20 to C36) aliphatic primary alcohols. In sugarcane, the predominant constituents of policosanols are tetracosanol (C28H58O, representing 60%-70% of the composition), triacontanol (C30H62O, comprising 10%-15%), and hexacosanol (C26H54O, making up 4%-10%), along with lesser amounts of docosanol (C22H46O) and octacosanol (C24H50O).59 The policosanols have been known for their health benefits, contributing to lipid-lowering, anti-aging, and tissue regeneration, and providing cellular, hepatic, cardiovascular, and cerebral vascular protection. They can also help prevent diseases such as diabetes, hypercholesterolemia, Alzheimer’s disease, heart disease, stroke, inflammation, ulcers, and cancer.60 In addition to policosanols, sugarcane contains long-chain aldehydes, vital components of natural plant waxes.61 These aldehydes, typically found in plant epidermal waxes, are crucial for leaf protection and are recognized as lipid biomarkers.62 Research by Asikin et al.63 showed that octamer (C28H56O) accounted for 73-80% of the total aldehydes in sugarcane, with lower levels of the trimer (C30H60O) and hexamer (C26H52O). Qi et al.64 analyzed waxes from sugarcane stems and found them to contain long-chain aldehydes, policosanols, fatty acids, hydrocarbons, and esters. Further studies identified n-hexaformaldehyde, octaformaldehyde, and n-trialdehyde in different sugarcane varieties.65 Fig. 5 presents the chemical structures of prominent polyphenols and long-chain aldehydes in sugarcane.
 |
Figure 5: Chemical structures of representative policosanols and long-chain aldehydes mainly present in sugarcane |
Miscellaneous (Other organics)
In studies on sugarcane, it has been found that this plant contains a variety of organic compounds, including triterpenoids, phytosterols, and fatty acids.66 Phytosterols, classified under the triterpene category, are ubiquitous in the plant kingdom, akin to cholesterol in animal cells. These compounds are primarily found in the unsaponifiable segment of plant lipids.67 Owing to their structural resemblance to cholesterol and their capacity to impede the absorption of cholesterol in the intestines, phytosterols have garnered considerable interest. This inhibition is beneficial as it reduces overall plasma cholesterol and low-density lipoprotein (LDL) levels.68 Investigations into sugarcane and its industrial derivatives have revealed that this crop is a rich source of phytosterols, including β-sitosterol, soya sterols, and other less prevalent steroids.69 Additionally, glycosylated sterols found in sugarcane are recognized for their efficacy in diminishing cholesterol absorption. Therefore, a detailed comprehension of sugarcane sterol types and concentrations is pivotal for developing and researching related products.
Fatty acids, both energy sources and integral membrane components, exhibit a range of biological activities influencing metabolism, function, and hormonal responsiveness in cells and tissues.70 These activities encompass the modulation of membrane architecture and function, the orchestration of intracellular signal transduction pathways, the regulation of transcription factors and gene expression, and the control of bioactive lipid mediator production.71 Considering that sugarcane juice is predominantly water (70-75%), the likelihood of it containing lipophilic substances is minimal. However, sugarcane wax is known to contain long-chain aliphatic fatty acids, albeit in lower amounts. Research by Ledon et al.72 identified various fatty acids in sugarcane wax oil, such as palmitic, oleic, linoleic, and linolenic acids. Attard et al.63 also explored different sugarcane waste types (peel, leaf, bagasse), uncovering distinct phytoconstituents leading to waxes with varied compositions. Notably, peel waxes were rich in n-cholesterol and long-chain fatty aldehydes, accounting for 83% of the total wax content. In contrast, waxes from residual leaves were a significant source of triterpenoids (169 mg/g wax), alongside fatty acids and phytosterols. Predominantly, sugarcane leaf wax contains unsaturated fatty acids such as linolenic, linoleic, and oleic acids. Reflecting on the literature, Fig. 6 illustrates the sterols and fatty acids found in parts of the sugarcane.
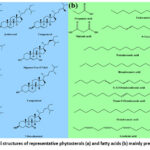 |
Figure 6: Chemical structures of representative phytosterols (a) and fatty acids (b) mainly present in sugarcane |
Nitrogen-free organic acids
The organic acid profile of sugarcane juice varies with cane variety, cultivation region, and growth conditions. Non-nitrogenous organic acids predominantly found in sugarcane juice include aconitic, oxalic, lactic, malic, and acetic acids, along with lesser quantities of fumaric, citric, hydroxyacetic, and succinic acids.73 Fig. 7 depicts the chemical structures of these organic acids in sugarcane juice. Notably, immature sugarcane tends to have higher concentrations of organic acids, particularly ornithine and lactic acid, while stale sugarcane shows increased levels of acetic and lactic acids. Literature indicates that the ratio of aconitic acid to Bx in the juice from the middle section of mature sugarcane stems is approximately 0.4% Bx.74 However, juice from the stem ends exhibits a higher aconitic acid ratio, around 1.1% Bx. This ratio can rise to 1.5% Bx and 2% Bx in immature sugarcane, contributing to its higher acidity. The presence of more organic acids in sugarcane juice necessitates the addition of more lime, thereby increasing the formation of soluble calcium salts. This process can adversely affect the reduction of evaporation scale and complicate the sugar cooking process.
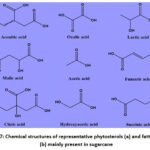 |
Figure 7: Chemical structures of representative phytosterols (a) and fatty acids (b) mainly present in sugarcane |
Inorganic substances
The composition of various inorganic substances found in sugarcane juice is influenced mainly by the soil conditions, fertilization practices, and irrigation methods in which the sugarcane is cultivated. These inorganic components in sugarcane juice comprise both anions and cations.75 Predominantly, the anions in sugarcane include phosphate, sulfate, silicate, and chloride ions. Regarding cations, the most abundant in cane juice are potassium and calcium, followed by magnesium, iron, sodium, and aluminum.76 Most of these inorganic elements, except phosphoric acid, which aids the clarification process, are considered detrimental. Calcium, sulfate, and silicic acid primarily contribute to scale accumulation in equipment maintenance. Conversely, potassium and chlorine are vital contributors to the formation of treacle molasses.77
Principle of colored substance formation in the sugar production process
During sugarcane processing, compounds that cause white sugar to turn yellow or brown are easily introduced during the juice and syrup stages. These coloring substances vary in molecular weight, structure, and characteristics, and may get trapped within the sugar crystals, negatively impacting the yield and quality of the white sugar.78 A lower color intensity in crystal sugar typically correlates with a higher market value. Hence, controlling color formation is critical in sugar manufacturing due to the difficulty in removing these substances and the consequent increase in production costs. The process of color development in sugarcane juice is multifaceted, involving not only the natural pigments in the cane but also the degradation of sugars, changes in pH, thermal influences, and interactions between amino compounds and carbohydrates during sugar processing. There are four primary chromogenic factors in sugar processing: natural plant pigments (such as chlorophylls, lutein, and anthocyanins), the Maillard reaction, caramelization, and the degradation products of alkaline fructose.79 The ensuing section will explore the mechanisms behind the formation of these colored substances in more detail.
Plant pigments in nature
Section 2.3 previously detailed the composition of colored substances in sugarcane juice, which includes natural pigments and phenolic compounds. The coloration in sugarcane predominantly arises from enzymatic reactions involving phenolic compounds inherently present in the plant.80 Phenolic compounds, characterized by a hydroxyl group attached to an aromatic ring, are categorized based on the number of phenol units they possess, ranging from monophenols to polyphenols. Sugarcane juice contains phenolic acids like hydroxycinamic acids, exemplified by caffeic acid and sinapinic acid, and flavonoids such as vitamin E and diosmetin-8-C-glycoside.81 However, pigmentation is more closely associated with polymerized phenolic compounds than simple, low molecular weight phenolics.82 Melanins, which are oxidized products of phenolics like tyrosine and also phenylalanine reaction products, are responsible for darker pigmentation. These melanins are formed through enzymatic oxidation (e.g., by polyphenol oxidase), yielding colors ranging from yellow to brown. Additionally, pigments such as anthocyanins, chlorophylls, and carotenoids contribute to the appealing color of sugarcane juice.83 Tarafdar et al.84 noted that changes in the color of sugarcane during ripening, such as a reduction in green color or an increase in yellow color, may correspond to a decline in chlorophyll content.
Maillard reaction
In sugarcane juice, amino acids and intrinsic amino compounds do not inherently cause coloration. However, their presence alongside monosaccharides and carbonyl derivatives of these sugars initiates the Maillard reaction, culminating in the development of a brownish product. Melanoidins, the dark brown to black products of this reaction, result from the interaction of reducing sugars with amino-group-containing compounds. The Maillard reaction is intricate, typically unfolding in three distinct phases:85 The initial phase, known as the Marschall reaction, begins with the dehydration reaction between compounds containing reactive amines and the α-hydroxycarbonyl group of reducing sugars. Aldose sugars, like glucose, react to form N-substituted aldehyde-based amines. These condensation products rapidly dehydrate to Schiff bases, which are then converted to allylamines. Following this, an Amadori rearrangement occurs, leading to the formation of ketoamines. In the case of ketoses, such as fructose, they undergo the Haynes reaction with amines, forming aminoaldoses, precursors to Amadori compounds. The second phase involves the degradation of Amadori compounds, heavily influenced by the pH level. Above pH 7, this degradation primarily involves 2,3 enolization, during which reductones and various other breakdown products are formed.86 These reactive compounds use additional condensation reactions, incorporating hydrogen into the structure of melanoidins. Dicarbonyl compounds and amino acids react to yield aldehydes and α-amino ketones, a process identified as Strecker degradation, marked by the release of carbon dioxide. In the final stage, the highly reactive interim product generated during the Maillard reaction forms a polycondensation reaction, resulting in melanin. This process encompasses a series of chemical reactions, including cyclization, dehydration, transaldolization, rearrangement, isomerization, and condensation. The culmination of these reactions is the creation of brown nitrogen-containing polymers and co-polymers, collectively referred to as melanin. As the browning process advances, the atomic weights of these colored compounds consequently increase. Recent studies87 have revealed that the color of sugarcane juice often changes immediately after extraction, serving as an indicator of various chemical and biochemical reactions. These color changes are attributed to precursors such as reducing sugars and amino acids in sugarcane, which, during the juicing process, form high-molecular-weight colored polymers.88 The diverse precursors present in different sugarcane varieties lead to variable pathways and rates of color degradation. This variability in the degradation process significantly influences the initial color of fresh sugarcane juice, leading to differences across varieties.
Caramels
Caramel compounds, which emerge as byproducts in the breakdown of sucrose into fructose and glucose, typically require heat to form.89 Intriguingly, even low concentrations of certain impurities, particularly iron, can significantly reduce the temperature necessary for caramelization, bringing it down to around 40 °C. These compounds are gelatinous and tend to adhere to the surfaces of sugar crystals, potentially impacting the quality of white sugar.90 Caramelization is a process that involves heating concentrated sucrose syrup above 210 °C. The initial step in forming color during this process involves the sugars, predominantly monosaccharides, undergoing an intramolecular rearrangement. Depending on the specific time and temperature conditions, the solution can turn yellow or brown. This reaction process leads to an increase in H+ ions, thereby gradually decreasing the pH of the solution to values between 4 and 5. Osuloses, α-dicarbonyl compounds like 3-deoxyhexulose, are formed during this sugar degradation. They are crucial for developing the characteristic color and taste of caramel. The formation of three typical O-heterocyclic compounds in d-glucose is facilitated by osuloses: hydroxymethylfurfural (HMF), hydroxydimethylfuranone (HDF), and hydroxyacetylfuran (HAF). During heat-induced caramelization, a significant role is played by the formation of antiglycation agents and oligomers through polymerization.
The process includes the generation of anhydrous sugars through intramolecular dehydration. The structures of the polymerisation products formed during caramelisation are not fully understood, despite the fact that there are fewer reactive intermediates involved in this process compared to the Maillard reaction. Compounds like HMF, HDF and HAF are regarded as precursors to conventional polymers, and Kroh has detailed their structures.91 HMF, being relatively stable, and its precursors (e.g., deoxy HMF) are less likely to polymerize regularly compared to more reactive hexose precursors suitable for such polymer formations. Yaylayan and his colleagues92 identified two nitrogen-free intermediates in the Maillard reaction, classified as ceramides, originating from glucosone and/or 3-or 1-deoxyglucosone. The pyrolysis of these isolated polymers produced furans that were similar to those derived from sucrose, indicating the presence of glycosidically linked sugar derivatives. Defaye93 suggested that the primary component of persistent Caramel is a combination of five fruit-based sugars (accounting for approximately 20% of the whole). Among the components, α, β-1,2′:2,1′-furan dicarboxylic acid dianhydride is the predominant one. A large number of these dicarboxylic acid dianhydrides fill important hydroxyl positions and have 1–6 glucose residues for glycosylation.
Alkaline degradation products of hexoses
Alkaline Hexose Degradation Products (HADPs) form another category of colorants in sugarcane processing.94 Before their formation, an alkaline solution catalyzes the hydrolysis of sucrose, subsequently causing glucose and fructose to break down under high temperatures in alkaline conditions. These monosaccharides experience a series of reversible and irreversible transformations in alkaline aqueous environments.95 The reversible transformations encompass ionization, mutarotation, and a combination of enolization and isomerization, forming enediol anions. These anions are often considered common intermediates in monosaccharide isomerization reactions, undergoing the Lobry de Bruyn-Alberda van Ekenstein rearrangement.96 Following the primary generation of alkenediol anionic species, a series of irreversible key degradation reactions ensues, ultimately yielding organic acid products. The isomerisation of monosaccharides and alkaline degradation reactions are initiated by the intermediary function of the enediol species. The process of β-elimination can be initiated by the enediol anion, resulting in the formation of α-dicarbonyl compounds. These compounds can either experience a benzyl rearrangement or α-dicarbonyl cleavage.97 The benzyl rearrangement process produces α-hydroxy carboxylic acids, including lactic and saccharinic acids, whereas dicarbonyl cleavage results in the formation of carboxylic acids like formic and acetic acid, and aldehydes, such as formaldehyde and acetaldehyde. Aldolization and retro-aldolization of carbonyl compounds are responsible for the elongation and fragmentation of carbon chains, respectively. Essential degradation reaction products can be categorized into two groups based on their carbon atom count: 1) Carboxylic acids with six or fewer carbon atoms (≤ C6). Most monosaccharides are transformed into colorless carboxylic acids, including lactic acid, saccharin, formic acid, acetic acid, and oxalic acid. 2) Carboxylic acids with more than six carbon atoms (> C6). The variety of these carboxylic acid products is wide and depends on the degradation reaction conditions. Compounds with a molecular weight greater than C6 can polymerise to form high-molecular-weight substances that are associated with color changes during alkali degradation reactions. The way C6 compounds are structured and the nature of what they are is something that is still not fully understood. Polymer formation is quite likely due to extensive aldol condensation reactions of the intermediate (II) carbonyl compounds under alkaline degradation conditions. These > C6 carboxylic acids are known to contain carboxylates, CH3, CH2, CH2OH, CHOH, and (alkenylated) β-dicarbonyl molecules.98
Future perspectives
Through this review, specific knowledge gaps have been identified. In what follows, practical and exciting new insights are presented for further exploration.
Different sugarcane varieties, growing environment, harvesting time, industrial means, and many other factors affect the fractions of non-sugar substances in sugarcane juice, especially color-forming substances. Future research should compare the differential effects of these factors.
In the development and research of new technologies, it should be committed to reducing the sulfur content in white sugar and harmlessly treating the sewage produced in the production process, the loss of sucrose components, and the removal of byproducts should be taken into account, and environmentally and sustainably clarified technologies should be committed to.
In the process of developing sugar solution, more attention should be paid to combining the characteristics of colored substances (phenolic substances) and other functional components (proteins and amino acids) in the sugar solution, to ensure the quality of the production and at the same time to achieve selective adsorption and recycling of colored substances and other active ingredients (phenolic active ingredients, etc.), and increase the benefits of the enterprise.
The non-sugar nutrients in sugarcane juice are more comprehensive, rich in amino acids, minerals, flavonoids, polyphenols, and other substances, and have certain antioxidant, free radical scavenging, and immune function-maintenance properties. Sugarcane juice concentrate deserves further development and promotion as a health product, especially as a functional beverage, rather than being used solely for sugar production.
Conclusion
In summary, the non-sucrose constituents comprise pigments, amino acids, organic acids, and inorganic salts in sugarcane juice. The pigment composition is notably complex, originating partly from chlorophyll, flavonoids, and polyphenolic compounds present in sugarcane. Additionally, dark substances such as humic and melanin form through various chemical reactions from colorless or pale compounds within sugarcane. Although natural pigments in sugarcane juice, such as phenols, are beneficial to health and have low toxicity, pigments from white sugar production and processing affect the sensory properties of sugar, such as color and flavor. Therefore, the content of these pigments in sugar production should be limited. A good understanding of the types, sources, and formation mechanisms of non-sugar substances in sugarcane juice, especially pigments, can help in better applications in the sugar industry.
Acknowledgement
We extend our gratitude to Editor Chen of Yifan Company for providing language editing services for this article.
Funding Sources
This work was financially supported by the Natural Science Foundation of Henan (252300423643), the Nanhu Scholars Program for Young Scholars of XYNU, and the Open Fund of Dabie Mountain Laboratory (DMLOF2024012).
Conflicts of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trail Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable.
Author Contributions
- Kun Fang: Writing–original draft, Validation, Resources, Methodology, Funding acquisition, Project administration.
- Pei Li: Writing-review and editing, Validation, Funding acquisition.
- Xiangrui Huang: Validation, Data curation, Formal analysis.
- Yihan Li: Visualization, Resources, Investigation.
- Hanbing Wang: Software, Methodology, Conceptualization.
References
- Koetle M. J., Snyman S. J., Rutherford R. S. Ex vitro morpho-physiological screening of drought tolerant sugarcane epimutants generated Via 5-azacytidine and imidacloprid treatments. Trop Plant Biol. 2022;15(4): 288-300.
CrossRef - Wang Q., Xie C. F., Shi C. R., et al. Sugar product diversification and its Opportunities in China. Sugar Tech. 2022;24(4): 1093-1106.
CrossRef - Ungureanu N., Vlăduț V., Biriș S. Ş. Sustainable valorization of waste and byproducts from sugarcane processing. Sustainability. 2022; 14(17): 11089.
CrossRef - Li Y. R., Yang L. T. Sugarcane agriculture and sugar industry in China. Sugar Tech. 2015; 17(1): 1-8.
CrossRef - Sahu O. Assessment of sugarcane industry: Suitability for production, consumption, and utilization. Ann Agrar Sci. 2018; 16(4): 389-395.
CrossRef - Agarwal, N. K., Kumar, M., Pattnaik, F., et al. Exploring the valorization potential of sugarcane bagasse pith: a review. BioEnerg Res.2023;16(3)L: 1280-1295.
CrossRef - Quintero J A, Montoya M I, Sánchez O J, et al. Fuel ethanol production from sugarcane and corn: comparative analysis for a Colombian case. 2008; 33(3): 385-399.
CrossRef - Edwards C. H., Rossi M., Corpe C. P., et al. The role of sugars and sweeteners in food, diet and health: Alternatives for the future. Trends Food Sci Technol. 2016; 56: 158-166.
CrossRef - Fito J., Tefera N., Van Hulle S. W. Sugarcane biorefineries wastewater: bioremediation technologies for environmental sustainability. Biol. Technol. Agric. 2019; 6: 1-13.
CrossRef - Sperança M. A., Nascimento P. A. M., Pereira F. M. V. Impurity in sugarcane juice as mineral content: a prospect for analysis using energy-dispersive X-ray fluorescence (EDXRF) and chemometrics. Microchem J. 2021; 164: 105951.
CrossRef - Wang L., Wu J., Huang H., et al. Coloration mechanisms of fresh sugarcane juice: Investigating the critical components and enzyme activity. J Food Sci. 2022; 87(4): 1552-1562.
CrossRef - Ali S. E., Yuan Q., Wang S., et al. More than sweet: A phytochemical and pharmacological review of sugarcane (Saccharum officinarum L.). Food Biosci. 2021; 44: 101431.
CrossRef - Costa M. V. A., Fontes C. H., Carvalho G., et al. Ultrabrix: A device for measuring the soluble solids content in sugarcane. Sustainability. 2021; 13(3): 1227.
CrossRef - Pryor S. W., Smithers J., Lyne P., et al. Impact of agricultural practices on energy use and greenhouse gas emissions for South African sugarcane production. J Clean Prod. 2017; 141: 137-145.
CrossRef - Canilha L, Chandel A K, Suzane dos Santos Milessi T, et al. Bioconversion of sugarcane biomass into ethanol: an overview about composition, pretreatment methods, detoxification of hydrolysates, enzymatic saccharification, and ethanol fermentation. BioMed Res Int. 2012, 2012(1): 989572.
CrossRef - Kim M., Day D. F. Composition of sugar cane, energy cane, and sweet sorghum suitable for ethanol production at Louisiana sugar mills. Ind. Microbiol. Biotechnol. 2011; 38(7): 803-807.
CrossRef - Kumar S., Prakasha G., Hanumanthapa D., et al. Diversified use of byproducts of sugarcane and cotton-a review. Int J Curr Microbiol Appl Sci. 2018; 7: 1616-1634.
CrossRef - Uchimiya M. Roles of reversible and irreversible aggregation in sugar processing. Crit Rev Food Sci Nutr. 2017; 57(6): 1206-1214.
CrossRef - Meng L., Li K., Li J., et al. Understanding the pathways for irreversible aggregate clusters formation in concentrated sugarcane juice derived from the membrane clarification process. LWT. 2021; 151: 112204.
CrossRef - Zidan D., Azlan A. Non-centrifugal sugar (NCS) and health: A review on functional components and health benefits. Appl Sci. 2022; 12(1): 460.
CrossRef - Meng L., Shi C., Wen T., et al. Acidity rectified aggregation behaviour of colloids in sugarcane juice. Int J Food Sci Technol. 2022; 57(12): 7929-7941.
CrossRef - De Souza A. P., Leite D. C., Pattathil S., et al. Composition and structure of sugarcane cell wall polysaccharides: implications for second-generation bioethanol production. BioEnergy Res. 2013; 6: 564-579.
CrossRef - Ling G. Z., Xiao J. L., Yang S., et al. The alleviation of manganese toxicity by ammonium in sugarcane is related to pectin content, pectin methyl esterification, and nitric oxide. GCB Bioenergy. 2022; 14(5): 585-596.
CrossRef - Fang K., Li K., Yang T. H., et al. Starch-based magnetic nanocomposite as an efficient absorbent for anticancer drug removal from aqueous solution. J. Biol. Macromol. 2021; 184: 509-521.
CrossRef - Alves F. V., Polesi L. F., Aguiar C. L., et al. Structural and physicochemical characteristics of starch from sugar cane and sweet sorghum stalks. Carbohydr Polym. 2014; 111: 592-597.
CrossRef - Panigrahi C., Shaikh A. E. Y., Bag B. B., et al.technological review on processing of sugarcane juice: Spoilage, preservation, storage, and packaging aspects. J Food Process Eng. 2021; 44(6): e13706.
CrossRef - Moodley P., Kana E. G. Optimization of xylose and glucose production from sugarcane leaves (Saccharum officinarum) using hybrid pretreatment techniques and assessment for hydrogen generation at semi-pilot scale. Int J Hydrogen Energy. 2015; 40(10): 3859-3867.
CrossRef - Prati P., Moretti R. H. Study of clarification process of sugar cane juice for consumption. Food Sci. Technol. 2010; 30: 776-783.
CrossRef - Cuddihy J. A., Porro M. E., Rauh J. S. The presence of total polysaccharides in sugar production and methods for reducing their negative effects. J Am Soc Sugar Cane Technol. 2001; 21: 73-91.
- Thorburn P. J., Meier E. A., Probert M. E. Modelling nitrogen dynamics in sugarcane systems: Recent advances and applications. Field Crops Res. 2005; 92(2-3): 337-351.
CrossRef - Reis R. S., de Moura Vale E., Heringer A. S., et al. Putrescine induces somatic embryo development and proteomic changes in embryogenic callus of sugarcane. J Proteomics. 2016; 130: 170-179.
CrossRef - Loiret F., Grimm B., Hajirezaei M., et al. Inoculation of sugarcane with Pantoea sp. increases amino acid contents in shoot tissues; serine, alanine, glutamine and asparagine permit concomitantly ammonium excretion and nitrogenase activity of the bacterium. Plant Physiol. 2009; 166(11): 1152-1161.
CrossRef - Fang K, Li P, Huang X, et al. Recent Advances in the Adsorption and Mechanisms of Colored Substances in Sugarcane Juice: A Review. Sugar Tech. 2025; 27(3): 654-666.
CrossRef - Vinall K., Schmidt S., Brackin R., et al. Amino acids are a nitrogen source for sugarcane. Funct Plant Biol. 2012; 39(6): 503-511.
CrossRef - Kaavya R., Pandiselvam R., Kothakota A., et al. Sugarcane juice preservation: A critical review of the state of the art and way forward. Sugar Tech. 2019; 21: 9-19.
CrossRef - Li J., Liang L., Cheng J., et al. Extraction of pigment from sugarcane juice alcohol wastewater and evaluation of its antioxidant and free radical scavenging activities. Food Sci Biotechnol. 2012; 21(5): 1489-1496.
CrossRef - Velásquez F., Espitia J., Mendieta O., et al. Non-centrifugal cane sugar processing: A review on recent advances and the influence of process variables on qualities attributes of final products. J Food Eng. 2019; 255: 32-40.
CrossRef - Costa D C, Costa H S, Albuquerque T G, et al. Advances in phenolic compounds analysis of aromatic plants and their potential applications. Trends Food Sci Technol. 2015; 45(2): 336-354.
CrossRef - Quideau S., Deffieux D., Douat-Casassus C., et al. Plant polyphenols: chemical properties, biological activities, and synthesis. Angew Chem Int Ed. 2011; 50(3): 586-621.
CrossRef - Nan Y., Su H., Zhou B., et al. The function of natural compounds in important anticancer mechanisms. Front Oncol. 2023; 12: 1049888.
CrossRef - Sun W., Shahrajabian M. H. Therapeutic potential of phenolic compounds in medicinal plants—Natural health products for human health. Molecules. 2023; 28(4): 1845.
CrossRef - Kautzmann C., Castanha E., Dammann C. A. J., et al. Roasted yerba mate (Ilex paraguariensis) infusions in bovine milk model before and after in vitro digestion: Bioaccessibility of phenolic compounds, antioxidant activity, protein–polyphenol interactions and bioactive peptides. Food Res Int. 2024; 183: 114206.
CrossRef - Sun Z., Li Q., Zhang J., et al. Phenolic constituents with antibacterial activity from Eleutherine bulbosa. Fitoterapia. 2024; 172: 105787.
CrossRef - Gayibov U. G., Gayibova S. N., Karimjonov H. M., et al. Antioxidant and cardioprotective properties of polyphenolic plant extract of Rhus glabra L. Plant Sci Today. 2024; 11(3): 1-11.
CrossRef - Nemli E., Ozkan G., Subasi B. G., et al. Interactions between proteins and phenolics: effects of food processing on the content and digestibility of phenolic compounds. J Sci Food Agric. 2024; 104(5): 2535-2550.
CrossRef - Sadeghi A., Rajabiyan A., Nabizade N., et al. Seaweed-derived phenolic compounds as diverse bioactive molecules: A review on identification, application, extraction and purification strategies. J. Biol. Macromol. 2024;266: 131147.
CrossRef - Rispo F., De Negri Atanasio G., Demori I., et al. An extensive review on phenolic compounds and their potential estrogenic properties on skin physiology. Front Cell Dev Biol. 2024; 11: 1305835.
CrossRef - Hewawansa U. H., Houghton M. J., Barber E., et al. Flavonoids and phenolic acids from sugarcane: Distribution in the plant, changes during processing, and potential benefits to industry and health. Compr Rev Food Sci Food Saf. 2024; 23(2): e13307.
CrossRef - Nakasone Y., Takara K., Wada K., et al. Antioxidative compounds isolated from Kokuto, non-centrifugal cane sugar. Biosci Biotechnol Biochem. 1996; 60(10): 1714-1716.
CrossRef - Takara K., Matsui D., Wada K., et al. New antioxidative phenolic glycosides isolated from Kokuto non-centrifuged cane sugar. Biosci Biotechnol. 2002; 66(1): 29-35.
CrossRef - Sabino A. R., Tavares S. S., Riffel A., et al. 1H NMR metabolomic approach reveals chlorogenic acid as a response of sugarcane induced by exposure to Diatraea saccharalis. Ind Crops Prod. 2019; 140: 111651.
CrossRef - Gottschalk L. M. F., Oliveira R. A., da Silva Bon E. P. Cellulases, xylanases, β-glucosidase and ferulic acid esterase produced by Trichoderma and Aspergillus act synergistically in the hydrolysis of sugarcane bagasse. Biochem Eng J. 2010; 51(1-2): 72-78.
CrossRef - Eggleston G. Positive aspects of cane sugar and sugar cane derived products in food and nutrition. J Agric Food Chem. 2018; 66(16): 4007-4012.
CrossRef - Rodrigues N. P., Brochier B., de Medeiros J. K., et al. Phenolic profile of sugarcane juice: Effects of harvest season and processing by ohmic heating and ultrasound. Food Chem. 2021; 347: 129058.
CrossRef - Duarte-Almeida J. M., Salatino A., Genovese M. I., et al. Phenolic composition and antioxidant activity of culms and sugarcane (Saccharum officinarum L.) products. Food Chem. 2011; 125(2): 660-664.
CrossRef - Zhao Y., Chen M., Zhao Z., et al. The antibiotic activity and mechanisms of sugarcane (Saccharum officinarum L.) bagasse extract against food-borne pathogens. Food Chem. 2015; 185: 112-118.
CrossRef - Ali S. E., El Gedaily R. A., Mocan A., et al. Profiling metabolites and biological activities of sugarcane (Saccharum officinarum Linn.) juice and its product molasses via a multiplex metabolomics approach. Molecules. 2019; 24(5): 934.
CrossRef - Wang L, Yue Z, Guo M, et al. Dietary flavonoid hyperoside induces apoptosis of activated human LX‐2 hepatic stellate cell by suppressing canonical NF‐κB signaling. BioMed Res Int. 2016, 2016(1): 1068528.
CrossRef - Irmak S., Dunford N. T., Milligan J. Policosanol contents of beeswax, sugar cane and wheat extracts. Food Chem. 2006; 95(2): 312-318.
CrossRef - Shen J., Luo F., Lin Q. Policosanol: Extraction and biological functions. J Funct Foods. 2019; 57: 351-360.
CrossRef - Asikin Y., Chinen T., Takara K., et al. Determination of long-chain alcohol and aldehyde contents in the non-centrifuged cane sugar Kokuto. Food Sci Technol Res. 2008; 14(6): 583-588.
CrossRef - Samuels L., Kunst L., Jetter R. Sealing plant surfaces: cuticular wax formation by epidermal cells. Annu Rev. Plant Biol. 2008; 59: 683-707.
CrossRef - Asikin Y., Takahashi M., Hirose N., et al. Wax, policosanol, and long‐chain aldehydes of different sugarcane (Saccharum officinarum L.) cultivars. Eur J Lipid Sci Technol. 2012; 114(5): 583-591.
CrossRef - Qi G., Peng F., Xiong L., et al. Extraction and characterization of wax from sugarcane bagasse and the enzymatic hydrolysis of dewaxed sugarcane bagasse. Prep Biochem Biotechnol. 2017; 47(3): 276-281.
CrossRef - del Río J. C., Marques G., Lino A. G., et al. Lipophilic phytochemicals from sugarcane bagasse and straw. Ind Crops Prod. 2015; 77: 992-1000.
CrossRef - Singh A., Lal U. R., Mukhtar H. M., et al. Phytochemical profile of sugarcane and its potential health aspects. Rev. 2015; 9(17): 45.
CrossRef - Choudhary S. P., Tran L. S. Phytosterols: perspectives in human nutrition and clinical therapy. Curr. Med. Chem. 2011; 18(29): 4557-4567.
CrossRef - Ostlund Jr R. E. Phytosterols in human nutrition. Annu Rev Nutr. 2002; 22: 533-549.
CrossRef - Georges P., Sylvestre M., Ruegger H., et al. Ketosteroids and hydroxyketosteroids, minor metabolites of sugarcane wax. Steroids. 2006; 71(8): 647-652.
CrossRef - Das U. N. Essential fatty acids: biochemistry, physiology and pathology. Biotechnol J Healthc Nutr Technol. 2006; 1(4): 420-439.
CrossRef - Calder P. C. Functional roles of fatty acids and their effects on human health. JPEN J Parenter Enteral Nutr. 2015; 39(1_suppl): 18S-32S.
CrossRef - Ledon N., Casaco A., Rodriguez V., et al. Anti-inflammatory and analgesic effects of a mixture of fatty acids isolated and purified from sugar cane wax oil. Planta Med. 2003; 69(04): 367-369.
CrossRef - Dowd M. K., Johansen S. L., Cantarella L., et al. Low molecular weight organic composition of ethanol stillage from sugarcane molasses, citrus waste, and sweet whey. J Agric Food Chem. 1994; 42(2): 283-288.
CrossRef - Bruni G O, Klasson K T. Aconitic acid recovery from renewable feedstock and review of chemical and biological applications. 2022, 11(4): 573.
CrossRef - Varga I., Jović J., Rastija M., et al. Efficiency and management of nitrogen fertilization in sugar beet as spring crop: A review. Nitrogen. 2022; 3(2): 170-185.
CrossRef - De Godoi L. A. G., Camiloti P. R., Bernardes A. N., et al. Seasonal variation of the organic and inorganic composition of sugarcane vinasse: main implications for its environmental uses. Sci. Pollut Res. 2019; 26: 29267-29282.
CrossRef - Walford S. N. Applications of ion chromatography in cane sugar research and process problems. Chromatogr. A. 2002; 956(1-2): 187-199.
CrossRef - Coca M., García M. T., González G., et al. Study of coloured components formed in sugar beet processing. Food Chem. 2004; 86(3): 421-433.
CrossRef - Rao M. J., Duan M., Yang M., et al. Sugarcane rind secondary metabolites and their antioxidant activities in eleven cultivated sugarcane varieties. Sugar Tech. 2022; 24(5): 1570-1582.
CrossRef - Vickers J., Grof C., Bonnett G., et al. Overexpression of polyphenol oxidase in transgenic sugarcane results in darker juice and raw sugar. Crop Sci. 2005; 45(1): 354-362.
CrossRef - Duarte-Almeida J. M., Novoa A. V., Linares A. F., et al. Antioxidant activity of phenolics compounds from sugar cane (Saccharum officinarum L.) juice. Plant Foods Hum Nutr. 2006; 61: 187-192.
CrossRef - Manohar M., Nayaka M. H., Mahadevaiah. Studies on phenolic content and polyphenol oxidase activity of sugarcane varieties with reference to sugar processing. Sugar Tech. 2014; 16: 385-391.
CrossRef - Tarafdar A., Nair S. G., Kaur B. P. Identification of microfluidization processing conditions for quality retention of sugarcane juice using genetic algorithm. Food Bioprocess Technol. 2019; 12: 1874-1886.
CrossRef - Tarafdar A., Kaur B. P., Pareek S. Effect of microfluidization on deteriorative enzymes, sugars, chlorophyll, and color of sugarcane juice. Food Bioprocess Technol. 2021; 14: 1375-1385.
CrossRef - Ke C., Li L. Influence mechanism of polysaccharides induced Maillard reaction on plant proteins structure and functional properties: A review. Carbohydr Polym. 2023; 302: 120430.
CrossRef - Martins S. I., Jongen W. M., Van Boekel M. A. A review of Maillard reaction in food and implications to kinetic modelling. Trends Food Sci Technol. 2000; 11(9-10): 364-373.
CrossRef - Adulvitayakorn S., Azhari S. H., Hasan H. The effects of conventional thermal, microwave heating, and thermosonication treatments on the quality of sugarcane juice. J Food Process Preserv. 2020; 44(2): e14322.
CrossRef - Mersad A., Lewandowski R., Heyd B., et al. Colorants in the sugar industry. Int Sugar J. 2003; 105(1254): 269-281.
- Samborska K., Bonikowski R., Kalemba D., et al. Volatile aroma compounds of sugarcane molasses as affected by spray drying at low and high temperature. LWT. 2021; 145: 111288.
CrossRef - Liu J, Zhang H, Jiao R, et al. An approach to regulate Maillard reaction and caramelization reaction in brown sugar: the addition of epicatechin naturally occurring in sugarcane. Food Control. 2025: 111901.
CrossRef - Kroh L. W. Caramelisation in food and beverages. Food Chem.1994; 51(4): 373-379.
CrossRef - Yaylayan V., Kaminsky E. Isolation and structural analysis of Maillard polymers: caramel and melanoidin formation in glycine/glucose model system. Food Chem. 1998; 63(1): 25-31.
CrossRef - Golon A., Kuhnert N. Unraveling the chemical composition of caramel. J Agric Food Chem. 2012; 60(12): 3266-3274.
CrossRef - Rupa T., Asokan S. Effect of rind pigments and juice colorants on juice claribility, settling time and mud volume of sugarcane. Sugar Tech. 2008; 10: 109-113.
CrossRef - Zhao X., Zhou Y., Liu D. Kinetic model for glycan hydrolysis and formation of monosaccharides during dilute acid hydrolysis of sugarcane bagasse. Technol. 2012; 105: 160-168.
CrossRef - Yamatsugu K., Kanai M. Catalytic approaches to chemo-and site-selective transformation of carbohydrates. Rev. 2023; 123(10): 6793-6838.
CrossRef - Li H., Yang S., Saravanamurugan S., et al. Glucose isomerization by enzymes and chemo-catalysts: status and current advances. ACS Catal. 2017; 7(4): 3010-3029.
CrossRef - Hinkova A., Bubník Z., Kadlec P., et al. Potentials of separation membranes in the sugar industry. Sep Purif Technol. 2002; 26(1): 101-110.
CrossRef
Abbreviations List
HADPs – Alkaline Hexose Degradation Products
HADPs – High-performance liquid chromatography
HAF – Hydroxyacetylfuran
HDF – Hydroxydimethylfuranone
HMF – Hydroxymethylfurfural
SPE – Sugarcane polyphenol extract













