Phyto-Flavonoids in Cancer Therapy: Emerging Insights and Future Perspectives
1Department of Pharmacology, Centurion University of Technology and Management, Odisha, India
2Department of Pharmacology, Siksha O Anusandhan University, Bhubaneswar, India
Corresponding Author Email: deepankar.rath@gmail.com
Download this article as:
ABSTRACT:Cancer remains a major global health burden, with rising incidence and mortality rates largely attributed to the limitations of current therapies in effectively targeting cancer cell proliferation, genetic mutations, and drug resistance. The successful clinical use of phytochemical-based anticancer agents such as paclitaxel, vincristine, camptothecin, etoposide, and teniposide has highlighted the therapeutic value of phytochemicals in cancer therapy. Among all classes, flavonoids or phyto-flavonoids such as apigenin, epigallocatechin gallate, genistein, luteolin, naringenin, and quercetin have gained considerable attention as complementary and alternative agents for cancer prevention and treatment due to their multimodal activity and non-toxic profiles. Extensive in vitro and in vivo studies have demonstrated that phyto-flavonoids can modulate key hallmarks of cancer, such as apoptosis, angiogenesis, metastasis, and cell cycle progression, and their anticancer effects are mediated through the regulation of crucial molecular signalling pathways, including NF-κB, PI3K/Akt/mTOR, MAPK, and p53, with minimal toxicity to normal cells compared to conventional chemotherapeutics. Despite encouraging preclinical evidence, the clinical translation of phyto-flavonoids remains limited due to challenges related to pharmacokinetics, bioavailability, standardization, and regulatory approval. Nonetheless, recent advances in nanotechnology and drug delivery systems are improving their therapeutic efficacy and mitigating their drug-ability limitations. A more profound understanding of their molecular mechanisms and pharmacological profiles could pave the way for developing novel, safe, and effective anticancer agents from natural sources. Overall, this review uniquely integrates recent molecular, pharmacological, and nanotechnology platform uses enhance clinical and translational success as anticancer therapeutics, thereby providing invaluable suggestions or expert opinions for future drug development and precision oncology research.
KEYWORDS:Anticancer Agents; Cancer Therapy; Molecular Targets; Pathways; Phyto-flavonoids; Phytochemical-Based Therapy
Introduction
Cancer continues to be a leading cause of morbidity and mortality worldwide, representing a major public health concern.1,2 According to the World Health Organization (WHO) and Global Cancer Observatory (GLOBOCAN) report, around 20 million new cancer cases and more than 9.7 million cancer-related deaths have been recorded globally.1,2 The burden is rising significantly due to aging populations, lifestyle factors, and environmental exposures.3,4 Among all cancer types, lung cancer remains the most commonly diagnosed and lethal in men, while breast cancer leads in incidence and mortality rates among women.1,5 In a regional perspective, the Asian community contributed nearly 50% of global cancer cases and 58% of deaths, primarily due to limited access to early detection and the unavailability or unaffordability of treatment in low- and middle-income countries.1,5 Europe contributed around 23% of global cases with comparatively better survival outcomes through the implementation of advanced healthcare systems.6 Although Africa has a relatively lower incidence but an unreasonably high mortality, reflecting poor diagnosis-treatment availability. India, especially the South Asian region, ranks among the top contributors to global cancer cases (around 1.4 million new cases with high mortality rates).1,2,7 India faces a rising cancer trend due to changing lifestyles, increasing tobacco use, pollution, and limited awareness, particularly in rural areas.7-9 Despite having a lower per capita incidence than western continents, India’s cancer mortality-to-incidence ratio remains high, representing gaps in early detection, screening, and affordable treatment.8,9 Therefore, there is a need for the urgent implementation of region-specific cancer prevention strategies and the integration of alternative, affordable therapies in national cancer control programs.
Despite advances in oncology research with high-end instrumentation, the current therapeutic approaches, including surgery, radiotherapy, chemotherapy, immunotherapy, and targeted therapy, have several limitations.10-12 Chemotherapy and radiation often damage normal cells, leading to severe side effects, drug resistance, and recurrence in many patients.13,14 Moreover, emerging and aggressive cancer subtypes (triple-negative breast cancer, pancreatic ductal adenocarcinoma, and drug-resistant leukaemia), frequently show poor responses to available treatments.15-17 In low-resource settings, targeted therapies are less accessible and less effective due to their limitations, which include tumor heterogeneity, genetic mutations, and high treatment costs, despite their promising nature.11,18 Because of this, cancer is still a complicated illness that frequently eludes long-term therapeutic management, requiring the development of novel therapeutic approaches. Therefore, it is imperative to investigate complementary and alternative therapeutic approaches that are safer, more accessible, and more effective.11,12
In these prospective, natural products especially Phyto flavonoids derived from medicinal plants have shown promise as anticancer agents with a long history of therapeutic application.19-21 For example, taxol, topotecan, vincristine, irinotecan, vinblastine, etc., are some well-known examples of phytochemicals that got Food and Drug Administration (FDA) approval as clinical anticancer drugs.22,23 The current research supports the incorporation of phyto-flavonoids as adjuvant or standalone therapies in contemporary oncological strategies, which calls for further investigation into their potential for treatment. Accordingly, the present review is updating and highlights these potential flavonoids’ anticancer potency, mechanisms, and future opportunity to be used as alternative/ complementary anticancer regimens.
Phytochemicals: An alternative source for anticancer drug discovery
From the ancient eras, Ayurveda, Traditional Chinese Medicine (TCM), and Sidda have long utilized herbal regimens and formulations to treat tumor and related disorders.24,25 In modern anticancer therapy, several FDA approved anticancer drugs, including paclitaxel (from Taxus brevifolia), vincristine and vinblastine (from Catharanthus roseus), etoposide and teniposide (from Podophyllum species), and camptothecin derivatives (from Camptotheca acuminata), emphasize the enormous value of phytochemicals for contemporary cancer therapy.22,23 Compared with conventional chemotherapeutics, many phytochemicals show selective toxicity and are non-toxic to normal cells, offering an advantage in reducing systemic toxicity and side effects.26-28 Phytochemicals generally stand with multipotential (antioxidant, anti-inflammatory, and anticancer) properties and explored early-phase clinical trials suggest that phytochemicals can be used as complements to conventional chemotherapeutics for enhancing therapeutic potency against drug resistance and reducing the side effects.21,22,27-30
Phyto-flavonoids: A multipotential anticancer regimens
Flavonoids are a large family of polyphenolic class ubiquitously present in fruits, vegetables, tea, and medicinal plants.31,32 Based on their core chemical structure, flavonoids are primarily classified into six major subgroups: flavones (apigenin, luteolin), flavonols (quercetin, kaempferol), flavanones (naringenin), flavanols (catechins, epigallocatechin-3-gallate/ EGCG), anthocyanins (cyanidin), and isoflavones (genistein, daidzein) (Figure 1). Each subclass exhibits unique patterns of hydroxylation, methylation, glycosylation, and prenylation, contributing to their wide range of biological activities.28,33,34 The antioxidant, anti-inflammatory, and antiproliferative properties of flavonoids, particularly their ability to scavenge reactive oxygen species and modulate cell signaling pathways, make them promising candidates for cancer therapy. Their natural abundance and low toxicity profile further support their potential as complementary agents in chemotherapeutic regimens.
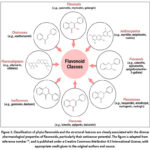 |
Figure 1. Classification of phyto-flavonoids and the structural features are closely associated with the diverse pharmacological properties of flavonoids, particularly their anticancer potential. |
Although many flavonoids demonstrated promise as anticancer agents and were non-toxic to healthy cells in various in vitro and in vivo models, only a small number of them have been approved or registered for clinical trial studies.28,33,34 Quercetin was investigated in a Phase I trial (NCT00003399). Doses as high as 1700 mg/m² were found to be well tolerated and to lower tumor markers in a range of solid tumor types. Prostate-specific antigen (PSA) levels in patients with prostate cancer are decreased by genistein, per Phase II trials (NCT00244933). EGCG in the form of green tea extract (NCT00917735) has been assessed in clinical trials for the prevention of breast and prostate cancer, and the results indicate positive effects on tumor suppression and oxidative stress markers (https://clinicaltrials.gov/). Despite these benefits, barriers such as limited systemic absorption, rapid metabolism, and low bioavailability prevent them from being used more widely. Thus, integrating flavonoids into modern cancer care frameworks through innovative delivery methods and combination therapy holds great promise for safer and more successful oncological outcomes. 28,33,34 As per the main focus of the review article, we have used two keywords, ‘flavonoids and cancer’ and ‘phyto-flavonoids and cytotoxicity,’ from four widely used literature databases, namely, PubMed, ScienceDirect, Wiley, and Springer online library (Table 1). Since we gathered numerous publications, we selected the most recent and comprehensive studies to avoid repetition and ensure uniqueness as well as minimize the research gaps in this area. In addition, those have been published in different languages, but we have selected those that have English translations available to minimize language-related bias. Both positive and negative findings from different literature databases related to the anticancer potential of phyto-flavonoids were critically analysed, mostly using indexed peer-reviewed journal articles published to present an unbiased overview.
Cancer-fighting flavonoids (individual with synergetic form) with mode of actions
Flavonoids are naturally occurring polyphenolic compounds that are abundant in fruits, vegetables, and medicinal plants. Because of their multi-targeted mechanisms of action, flavonoids are showing promise as anticancer agents clearly depicted in the Figure 2.28,33,34 By altering important cellular pathways involved in cell proliferation, apoptosis, angiogenesis, inflammation, and metastasis, apigenin, quercetin, kaempferol, luteolin, and genistein have strong anti-cancer effects.28,35-38 For example, apigenin inhibits pro-oncogenic transcription factors like NF-κB and STAT3, induces G2/M cell cycle arrest, and activates intrinsic apoptotic pathways through caspase-3 and caspage-9.28,36 Similarly, quercetin increases oxidative stress in cancer cells, disrupts the PI3K/Akt/mTOR signalling axis, and downregulates anti-apoptotic proteins like Bcl-2.39,40 Genistein also modifies epigenetic targets, reactivating dormant tumor suppressor genes.41 Luteolin, which inhibits the Wnt/β-catenin pathway and lowers the expression of thymidylate synthase, making colorectal cancer cells more sensitive to 5-fluorouracil (5-FU).36,42
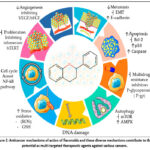 |
Figure 2: Anticancer mechanisms of action of flavonoids and these diverse mechanisms contribute to their potential as multi-targeted therapeutic agents against various cancers. |
The figure is adapted from reference number34 published under a Creative Commons Attribution 4.0 International License. Proper credit is given to the original authors and source.
The epithelial–mesenchymal transition (EMT) and tumour angiogenesis, both of which are essential for tumor growth and metastasis, are also known to be hampered by flavonoids (Table 1). It has been demonstrated that luteolin and kaempferol inhibit matrix metalloproteinases (MMPs) and downregulate VEGF expression, which lowers the potential for tumor vascularization and invasion. By inhibiting histone deacetylase (HDAC) and DNA methyltransferase (DNMT), genistein, an isoflavone derived from soy, reactivates tumour suppressor genes and performs epigenetic modulation (Table 1). Additionally, by boosting antitumor immune responses and suppressing immunosuppressive cytokines in the tumor microenvironment, a variety of flavonoids demonstrate immunomodulatory effects. When taken as a whole, these diverse bioactivities highlight the value of flavonoids as lead compounds or adjuvants in the creation of secure and efficient anticancer treatments (Table 1).
In addition to sensitizing resistant tumors and selectively targeting cancer cells with low toxicity to healthy tissues, flavonoids frequently work in concert with chemotherapeutic medications to enhance their pro-apoptotic effects.43 For instance, by encouraging G2/M phase arrest and mitochondrial-mediated apoptosis, apigenin dramatically raises the cytotoxicity of cisplatin in ovarian and cervical cancer cells.44-46 It increases intrinsic apoptotic signalling by downregulating Bcl-2 and activating caspase-3 and caspase-9.45,46 Similarly, by raising intracellular ROS levels and inhibiting the PI3K/Akt/mTOR pathway, which is frequently overactivated in resistant tumours, quercetin amplifies the effects of doxorubicin in breast cancer.47-49 In addition to encouraging cancer cell death, these synergistic effects also make it possible to use chemotherapeutics at lower dosages, which lessens systemic toxicity.48,49 When used in combination therapy, flavonoids can reverse the mechanisms underlying drug resistance. Inhibiting NF-κB and downregulating P-glycoprotein (P-gp), for instance, makes paclitaxel-resistant breast cancer cells more sensitive to drugs like 5-FU and oxaliplatin.50 Flavonoids are useful adjuvants for overcoming resistance in aggressive and refractory tumors because of these synergistic interactions.46-49
Beyond their direct cytotoxic effects, flavonoids work in concert with chemotherapeutics to target survival signaling and the tumor microenvironment.51 When combined with doxorubicin or cisplatin, kaempferol dramatically disrupts angiogenesis in liver and ovarian cancers.52 Additionally, it blocks signals that cancer cells need to survive by inhibiting the Akt and ERK1/2 pathways, which increases apoptosis.52 By blocking NF-κB-mediated inflammation and activating the p53 pathway to make cells more susceptible to apoptosis, the citrus flavonoid naringenin increases the effectiveness of docetaxel in prostate cancer.53 Additionally, flavonoids assist in altering the tumor milieu, lowering oxidative stress and inflammation, and creating an environment that is less conducive to cancer cell survival during chemotherapy.53
From a quantitative perspective, apigenin reduced tumor volume up to 45% at 50 mg/kg in a mouse xenograft model (p < 0.01) and increased median survival from 28 to 40 days.54 Similarly, quercetin exhibited an IC₅₀ of 12.5 μM in MCF-7 (ER-positive human breast cancer) cells and decreased tumor weight by 38% in vivo, and when it was combined with doxorubicin, it produced a combination index (CI) of 0.7 as a 20% improvement in survival versus doxorubicin alone.55 The well-known EGCG lowered intratumoral VEGF expression by 2.3-fold and reduced microvessel density by 35%, consistent with its antiangiogenic activity.56 Overall, most flavonoids reduced tumors 30 to 70% in individual and synergetic treatments against different cancers in vivo, but clinical data remain limited to early-phase trials with primarily safety endpoints. Larger and controlled studies need to be conducted for mainstream uses. In summary, this review is unique, as it comprehensively analyses twenty-three flavonoids with detailed cancer-specific mechanisms, integrating the latest in vitro, in vivo, and clinical findings. Mainly, the existing reviews highlight the multi-biological activity, and some of them are only on specific flavonoids and some of them on specific cancers with a lack of details. The present review fills these gaps by critically integrating updated experimental and clinical evidence, summarizing quantitative outcomes, and evaluating the clinical readiness of key flavonoids. By addressing these deficiencies, this review offers a more comprehensive and translational perspective on the role of phyto-flavonoids in modern cancer therapy. It also highlights novel molecular insights, bioavailability strategies, and future research directions using the nanoparticle platform to enhance translational success with examples, distinguishing it from existing reviews.
Discussion
Significant anticancer potential has been demonstrated by phyto-flavonoids in various preclinical models with demonstrated consistent tumor growth inhibition through modulation of pivotal signalling cascades, including PI3K/Akt, NF-κB, MAPK, and STAT3 pathways (Table 1). Recorded preclinical reports suggested that flavonoids significantly reduced tumor size around 20–70% and enhanced apoptosis rates even higher than controls in some cases (Table 1). In addition, quercetin, genistein, and apigenin-like compounds exhibited synergistic effects with standard chemotherapeutics, improving survival and reducing drug resistance. From a clinical and closer translation standpoint, quercetin, genistein, apigenin, and EGCG have advanced to early-phase clinical evaluation in both animal models and human trials. Similarly, baicalein, luteolin, kaempferol, and fisetin, at the late preclinical stage, demonstrating potent anticancer activity individually and synergism with conventional chemotherapeutics, need more investigation on their pharmacokinetic improvement for clinical applicability. Equally, morin, tangeretin, and eupatilin are primarily supported by in vitro data, representing promising but exploratory candidates needing in vivo validation. Overall, the current data suggests that further mechanistic, pharmacological, and clinical investigations are necessary to fully understand their therapeutic potential in oncology (Table 1). The majority of available clinical studies are small, non-randomized, or employ various dosages and formulations, making it challenging to draw firm conclusions. Furthermore, there is frequently inconsistency in the standardization of flavonoid content in botanical formulations, which results in a range of therapeutic outcomes.32,57,58 Overall, clinical validation of phyto-flavonoids is still scarce despite a large number of in vitro and in vivo studies.
Clinical challenges and limitations of Phyto flavonoids in cancer therapy
Despite the significant in vitro and in vivo reports, several obstacles still hinder their clinical use, including a poor pharmacokinetic profile, particularly low oral bioavailability, rapid metabolism, and short half-life.59 Reduced systemic availability results from the extensive glucuronidation and sulfation of flavonoids like quercetin, genistein, and EGCG in the liver and gastrointestinal tract.57,58 Because of these factors, they are less effective as a treatment and require higher dosages to produce pharmacological activity, which raises the possibility of off-target effects.58,59 Additionally, the low aqueous solubility of flavonoids limits their absorption, and their vulnerability to intestinal degradation diminishes their stability prior to systemic circulation.57,60 Another drawback is the possibility of drug-flavonoid interactions, which can alter the metabolism of concurrently administered anticancer medications, especially by inhibiting or inducing cytochrome P450 enzymes.60,61 These unresolved pharmacodynamic issues, which also present regulatory challenges, restrict the incorporation of phyto-flavonoids into mainstream cancer therapy.59 Last but not least, flavonoids are generally safe in terms of toxicity; however, their toxicity profiles differ based on dosage, metabolism, and bioavailability, potentially restricting their therapeutic use.62,63 For example, quercetin and genistein display mild hepatotoxicity or reproductive effects at elevated doses, whereas others demonstrate negligible adverse effects.62,63 Understanding these profiles is crucial for the development of drugs suitable for real-world and clinical settings with safe dosing and formulation strategies, which leads to high translational success.
Researchers are developing new and sustainable drug delivery methods to improve the solubility, stability, and tumor-targeting potential of phyto-flavonoids.64,65 Recent studies have shown enhanced bioavailability and targeted cancer cell delivery using nanotechnology-based strategies like nanoparticles, liposomes, dendrimers, and phytosomes.66 For instance, by extending their plasma half-life and tumor accumulation, quercetin-loaded nanoparticles showed markedly enhanced bioavailability and anticancer activity in colon cancer models.67,68 Likewise, genistein-encapsulated polymeric nanoparticles and EGCG-loaded liposomes have demonstrated improved therapeutic outcomes, decreased systemic degradation, and increased cellular uptake in prostate and breast cancers, respectively.69,70 As demonstrated by methylated derivatives of apigenin and luteolin, another strategy is to methylate, glycosylate, or acylate flavonoid structures to enhance their lipophilicity and metabolic stability.71,72 These tactics minimize off-target toxicity by enhancing systemic exposure and lowering the requirement for high dosage.71,72 Furthermore, flavonoid-based combination treatments with immunotherapies or chemotherapeutics may minimize side effects while producing synergistic effects.50 It has been demonstrated that flavonoids like wogonin, naringenin, and baicalein alter apoptosis, angiogenesis, and drug resistance pathways, making cancer cells more susceptible to traditional chemotherapeutics like doxorubicin, paclitaxel, and cisplatin.73 Tools from systems biology and network pharmacology can pinpoint flavonoid multi-target mechanisms and maximize therapeutic benefit by coordinating their co-administration with chemotherapeutic agents.21,74 Future perspectives suggest that biotechnological production using modified microbes (using E. coli and Saccharomyces cerevisiae) will ensure reliable, large-scale synthesis of flavonoid molecules with pharmaceutical-grade purity.75,76 Lastly, the application of genomics and metabolomics-guided personalized medicine can assist in determining which patient subgroups respond best to particular flavonoid regimens. Together, these developments provide a strong foundation for turning phyto-flavonoids into anticancer medications with clinically feasible and efficient.
Table 1: List of some leading anticancer flavonoids with their common natural sources and explored molecular mechanisms.
|
Flavonoid (natural sources) |
Explored mode of actions or molecular mechanisms |
|
Apigenin (Parsley, celery, chamomile) |
Inhibits STAT3, NF-κB, ERK1/2; G2/M arrest; pro-apoptotic (caspase-3/9); anti-invasive. 35,77 |
| Baicalein (Scutellaria baicalensis) |
Inhibits COX-2; apoptosis; downregulates MMP-2/9; anti-migration/invasion. 78,79 |
|
Biochanin A (Red clover, soy) |
Phytoestrogenic action, suppresses tumor cell invasion. 80,81 |
| Chrysin
(Honey, propolis, passion flower) |
Induces apoptosis; inhibits PI3K/Akt and NF-κB; anti-metastatic; aromatase inhibition82,83 |
|
Cyanidin-3-O-glucoside (Berries, red cabbage) |
Inhibits EGFR, MAPK pathway, promotes apoptosis84 |
| Delphinidin (Blue berries, egg plants) |
Anti-angiogenic, inhibits VEGFR and ERK1/2 signaling.85,86 |
|
Diosmetin (Citrus fruits) |
Activates caspase cascade, suppresses PI3K/Akt, inhibits tumor cell proliferation.87,88 |
|
Catechin/ Epigallocatechin-3-gallate (Green tea) |
Inhibits EGFR and receptor TKs; anti-angiogenic (downregulates VEGF; pro-apoptotic; PI3K/Akt, MAPK modulation).56,89 |
|
Eupatilin (Artemisia asiatica) |
Inhibits NF-κB and COX-2, anti-inflammatory and anti-proliferative)90,91 |
|
Fisetin (Strawberries, apples, cucumbers) |
Caspase-mediated apoptosis; G1 arrest; inhibits mTOR and Wnt/β-catenin; reduced Bcl-2.90,92 |
|
Genistein (Soybeans, legumes) |
ERα/β modulation; protein tyrosine kinase inhibition; suppresses NF-κB, Akt; epigenetic effects41,93 |
|
Hesperetin (Citrus) |
Antioxidant/anti-inflammatory; apoptosis; cell-cycle arrest; suppresses MAPK/NF-κB.55,94 |
|
Isorhamnetin (Sea buckthorn, onions) |
Inhibits metastasis, upregulates Nrf2, downregulates MMP-2 and -9).95,96 |
|
Kaempferol (Tea, spinach, broccoli) |
Inhibits Akt/mTOR and NF-κB; mitochondrial apoptosis; anti-angiogenesis (VEGF).97,98 |
|
Luteolin (Celery, thyme, broccoli) |
Anti-angiogenic (downregulates VEGF); anti-metastatic (downregulates MMP-2/9); activates p53; ROS modulation; Wnt/β-catenin inhibition).36,42 |
|
Morin (Citrus, oregano) |
Mitochondrial apoptosis; downregulates Bcl-2; inhibits VEGF, PI3K/Akt; G2/M arrest.99,100 |
|
Myricetin (Berries, grapes, tea) |
Modulates p53; apoptosis; inhibits proliferation and metastasis; kinase modulation.101,102 |
|
Naringenin / Pectolinarigenin (Citrus fruits, grapefruit, orange) |
G0/G1 arrest; inhibits PI3K/Akt/mTOR, TGF-β/Smad; anti-inflammatory (downregulates NF-κB).103,104 |
|
Quercetin (Onions, apples, berries, tea) |
Induces apoptosis; cell-cycle arrest (G1/S, G2/M); inhibits PI3K/Akt, MAPK, NF-κB; modulates p53, Bax/Bcl-2).40,105 |
|
Rutin (Buckwheat, citrus, apples) |
Induces apoptosis, inhibits angiogenesis and proliferation).106,107 |
| Tangeretin (Tangerine peel) |
Inhibits metastasis and angiogenesis; induces cell cycle arrest).108,109 |
|
Wogonin (Scutellaria baicalensis) |
Anti-inflammatory cytokine suppression; cell-cycle arrest; apoptosis; immune modulation.105,110 |
Note: COX2, cyclooxygenase-2; EGFR, epidermal growth factor receptor; HCC, hepatocellular carcinoma; NF-κB, Nuclear factor kappa B; MAPK, mitogen-activated protein kinase; MMP-2/9, matrix metalloproteinase-2/9; PI3K/Akt/mTOR, phosphoinositide 3-kinase/ protein kinase B/ mammalian target of rapamycin; STAT3, signal transducer and activator of transcription 3; TGF-β, transforming growth factor-beta; VEGF, vascular endothelial growth factor.
Conclusion
The pharmacological and multimodal effects of phyto-flavonoids, such as their anti-inflammatory, anti-proliferative, pro-apoptotic, antioxidant, and anti-angiogenic properties, have made them promising natural anticancer agents. Higher molecular interactions with the majority of the targets implicated in the development, progression, and metastasis of cancer are made possible by their medicinal chemistry profiles. Preclinical investigations have demonstrated the modulation of several key cancer signalling pathways by quercetin, apigenin, genistein, and luteolin-like flavonoids. They may also be used as supplements to traditional chemotherapy and radiation therapy, improving therapeutic efficacy and lowering toxicity, according to mounting data from clinical trials. Notwithstanding these benefits, flavonoids have several drawbacks, including low bioavailability, quick metabolism, and restricted tumor specificity. Platforms for combinatorial therapy, advanced nanotechnology, and structural modification are actively addressing these issues. With the aid of public-private partnerships and policy support, future directions should concentrate on carefully planned randomized clinical trials, standardization of flavonoid formulations, and clarification of pharmacokinetic profiles to establish dosing schedules and safety standards. In conclusion, phyto-flavonoids serve as a promising and non-toxic alternative to next-generation anticancer drugs, whether used individually or in combination with other treatments.
Acknowledgement
The author is thankful to the Honourable Vice-Chancellor of Centurion University of Technology and Management, Odisha, for academic support.
Funding Sources
The author(s) received no financial support for this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
All the information presented was obtained from previously published studies and publicly available databases with proper citations. The corresponding author can provide any datasets used for this study upon request.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable.
Author Contributions
- Deepankar Rath: Conceptualization, Data collection, Manuscript drafting, and Review and editing.
- Biswakanth Kar: Conceptualization, Manuscript drafting, Review and editing.
- Gurudutta Pattnaik: Conceptualization, Supervision, Critical Analysis, Review and editing.
- Pallishree Bhukta: Data collection and management, Manuscript drafting, Review and editing.
References
- Sun , Yang Y., Wang C., et al. Epigallocatechin-3-gallate at the nanoscale: a new strategy for cancer treatment. Pharm Biol. 2024;62(1):676-690.
CrossRef - World Health Organization-Global cancer burden growing, amidst mounting need for services-2024. Accessed on: https://www.who.int/news/item/01-02-2024-global-cancer-burden-growing–amidst-mounting-need-for-services (Accessed on June 20th 2025).
- Prathap , Kirubha S., Rajan A. T., et al. The increasing prevalence of cancer in the elderly: An investigation of epidemiological trends. Aging Med (Milton). 2024;7(4):516-527.
CrossRef - Li L., Shan T., Zhang D., Ma F. Nowcasting and forecasting global aging and cancer burden: analysis of data from the GLOBOCAN and Global Burden of Disease Study. J Natl Cancer Cent. 2024;4(3):223-232.
CrossRef - Sung H., Ferlay J., Siegel R. L., et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209-249.
CrossRef - Hesso , Kayyali R., Zacharias L., et al. Cancer care pathways across seven countries in Europe: What are the current obstacles? And how can artificial intelligence help? J Cancer Policy. 2024; 39:100457.
CrossRef - ICMR-NCDIR Cancer Burden Report-2021. https://www.icmr.gov.in/icmrobject/custom_data/ 1702892908_press_release_cancer_report_ncdir.pdf. (Accessed on June 20th 2025).
- Khanna , Sharma P., Budukh A., et al. Rural-urban disparity in cancer burden and care: findings from an Indian cancer registry. BMC Cancer. 2024;24(1):308.
CrossRef - Kulothungan , Ramamoorthy T., Sarveswaran G., et al. Association of tobacco use and cancer incidence in India: A systematic review and meta-analysis. JCO Glob Oncol. 2024; 10: e2400152.
CrossRef - Taefehshokr , Parhizkar A., Hayati S., et al. Cancer immunotherapy: Challenges and limitations. Pathol Res Pract. 2022; 229:153723.
CrossRef - Leng G., Duan B., Liu J., et al. The advancements and prospective developments in anti-tumor targeted therapy. 2024; 56:101024.
CrossRef - Zafar , Khatoon S., Khan M. J., et al. Advancements and limitations in traditional anti-cancer therapies: a comprehensive review of surgery, chemotherapy, radiation therapy, and hormonal therapy. Discov Oncol. 2025; 16(1):607.
CrossRef - Liu P., Zheng C. C., Huang Y. N., et al. Molecular mechanisms of chemo- and radiotherapy resistance and the potential implications for cancer treatment. MedComm (2020). 2021; 2(3):315-340.
CrossRef - Siegel R. L., Miller K. D., Wagle N. S., et al. Cancer statistics, 2023. CA Cancer J Clin. 2023; 73(1):17-48.
CrossRef - Obidiro , Battogtokh G., Akala E. O. Triple Negative Breast Cancer Treatment Options and Limitations: Future Outlook. Pharmaceutics. 2023; 15(7):1796.
CrossRef - Chintamaneni K., Pindiprolu S. K. S. S., Swain S. S., et al. Conquering chemoresistance in pancreatic cancer: Exploring novel drug therapies and delivery approaches amidst desmoplasia and hypoxia. Cancer Lett. 2024; 588:216782.
CrossRef - Dogra K., Prakash A., Gupta S., Gupta M. Prognostic significance and molecular classification of triple negative breast cancer: A systematic review. Eur J Breast Health. 2025; 21(2):101-114.
CrossRef - Zhong , Li Y., Xiong L., et al. Small molecules in targeted cancer therapy: Advances, challenges, and future perspectives. Signal Transduct Target Ther. 2021; 6(1): 201
CrossRef - Ponte G. S., Pavan I. C. B., Mancini M. C. S., et al. The hallmarks of flavonoids in cancer. Molecules. 2021; 26(7):2029.
CrossRef - Mohi-Ud-Din , Mir R. H., Sabreen S., et al. Recent insights into therapeutic potential of plant-derived flavonoids against cancer. Anticancer Agents Med Chem. 2022; 22(20):3343-3369.
CrossRef - Sahoo A., Swain S. S., Singh S. R., et al. Anti-inflammatory and immunomodulatory phytochemicals for management of oral lichen planus: A multi-omics system biology and experimental assessment. Chem Biodivers. 2025: e00662.
CrossRef - Newman D. J., Cragg G. M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J Nat Prod. 2020; 83(3):770-803.
CrossRef - Swain S. S., Sahoo S. K. Piperlongumine and its derivatives against cancer: A recent update and future prospective. Arch Pharm (Weinheim). 2024; 357(7): e2300768.
CrossRef - Rizvi A. A., Einstein G. P., Tulp O. L., et al. Introduction to traditional medicine and their role in prevention and treatment of emerging and re-emerging diseases. Biomolecules. 2022; 12(10):1442.
CrossRef - Sulaiman , George B. P., Balachandran I., et al. Cancer and traditional medicine: An integrative approach. Pharmaceuticals (Basel). 2025; 18(5):644.
CrossRef - Dutt , Garg V., Khatri N., et al. Phytochemicals in anticancer drug development. Anticancer Agents Med Chem. 2019; 19(2):172-183.
CrossRef - Grover , Thakur K., Bhardwaj M., et al. Phytotherapeutics in cancer: From potential drug candidates to clinical translation. Curr Top Med Chem. 2024; 24(12):1050-1074.
CrossRef - Pandey P., Lakhanpal S., Mahmood D., et al. An updated review summarizing the anticancer potential of flavonoids via targeting NF-kB pathway. Front Pharmacol. 2025; 15:1513422.
CrossRef - Swain S. S., Padhy R. N. Isolation of ESBL-producing gram-negative bacteria and in silico inhibition of ESBLs by flavonoids. J Taibah Univ Med Sci, 2016;11(3), 217-229.
CrossRef - Swain S. S., Singh S. R., Sahoo A., et al. Anti-HIV-drug and phyto-flavonoid combination against SARS-CoV-2: A molecular docking-simulation base assessment. J Biomol Struct Dyn. 2022; 40(14):6463-6476.
CrossRef - Mutha E., Tatiya A. U., Surana S. J. Flavonoids as natural phenolic compounds and their role in therapeutics: An overview. Futur J Pharm Sci. 2021;7(1):25.
CrossRef - Smeu A., Marcovici I., Dehelean C. A., et al. Flavonoids and flavonoid-based nanopharmaceuticals as promising therapeutic strategies for colorectal cancer-an updated literature review. Pharmaceuticals (Basel). 2025; 18(2):231.
CrossRef - Zhang W., Hu J. J., Fu R. Q., et al. Flavonoids inhibit cell proliferation and induce apoptosis and autophagy through downregulation of PI3Kγ mediated PI3K/AKT/mTOR/p70S6K/ULK signaling pathway in human breast cancer cells. Sci Rep. 2018; 8(1):11255.
CrossRef - De Luna C. F., Ferreira W. A. S., Casseb S. M. M., et al. Anticancer potential of flavonoids: An overview with an emphasis on tangeretin. Pharmaceuticals (Basel). 2023; 16(9):1229.
CrossRef - Yan , Qi M., Li P., et al. Apigenin in cancer therapy: Anti-cancer effects and mechanisms of action. Cell Biosci. 2017; 7:50.
CrossRef - Imran , Rauf A., Abu-Izneid T., et al. Luteolin, a flavonoid, as an anticancer agent: A review. Biomed Pharmacother. 2019; 112:108612.
CrossRef - Mir A., Dar A., Hamid L., et al. Flavonoids as promising molecules in the cancer therapy: An insight. Curr Res Pharmacol Drug Discov. 2023; 6:100167.
CrossRef - Silva-Pinto A., de Pontes J. T. C., Aguilar-Morón B., et al. Phytochemical insights into flavonoids in cancer: Mechanisms, therapeutic potential, and the case of quercetin. Heliyon. 2025; 11(4): e42682.
CrossRef - Guo H., Ding H., Tang X., et al. Quercetin induces pro-apoptotic autophagy via SIRT1/AMPK signalling pathway in human lung cancer cell lines A549 and H1299 in vitro. Thorac Cancer. 2021; 12(9):1415-1422
CrossRef - Wang W., Yuan X., Mu J., et al. Quercetin induces MGMT+glioblastoma cells apoptosis via dual inhibition of Wnt3a/β-Catenin and Akt/NF-κB signaling pathways. Phytomedicine. 2023; 118:154933.
CrossRef - Chen T., Wang J., Li M., et al. Genistein inhibits proliferation and metastasis in human cervical cancer cells through the focal adhesion kinase signaling pathway: A network pharmacology-based in vitro study in HeLa cells. Molecules. 2023; 28(4):1919.
CrossRef - Wu H. T., Lin J., Liu Y. E., et al. Luteolin suppresses androgen receptor-positive triple-negative breast cancer cell proliferation and metastasis by epigenetic regulation of MMP9 expression via the AKT/mTOR signalling pathway. Phytomedicine. 2021; 81:153437.
CrossRef - Maszczyk , Banach K., Rok J., et al. Evaluation of possible neobavaisoflavone chemosensitizing properties towards doxorubicin and etoposide in SW1783 Anaplastic Astrocytoma Cells. Cells. 2023; 12(4):593.
CrossRef - Liu , Ji P., Liu B., et al. Apigenin enhances the cisplatin cytotoxic effect through p53-modulated apoptosis. Oncol Lett. 2017; 13(2):1024-1030.
CrossRef - Qi, Ding Z., Yao Y., et al. Apigenin induces apoptosis and counteracts cisplatin-induced chemoresistance via Mcl-1 in ovarian cancer cells. Exp Ther Med. 2020; 20(2):1329-1336.
CrossRef - Papachristou , Anninou N., Koukoulis G., et al. Differential effects of cisplatin combined with the flavonoid apigenin on HepG2, Hep3B, and Huh7 liver cancer cell lines. Mutat Res Genet Toxicol Environ Mutagen. 2021; 866:503352.
CrossRef - Staedler , Idrizi E., Kenzaoui B. H., et al. Drug combinations with quercetin: doxorubicin plus quercetin in human breast cancer cells. Cancer Chemother Pharmacol. 2011; 68(5):1161-72.
CrossRef - Liu , Li R., Qian J., et al. Combination therapy of doxorubicin and quercetin on multidrug-resistant breast cancer and their sequential delivery by reduction-sensitive hyaluronic acid-based conjugate/d-α-tocopheryl poly(ethylene glycol) 1000 succinate mixed micelles. Mol Pharm. 2020; 17(4):1415-1427.
CrossRef - Almohammad Aljabr B., Zihlif M., Abu-Dahab R., et al. Effect of quercetin on doxorubicin cytotoxicity in sensitive and resistant human MCF7 breast cancer cell lines. Biomed Rep. 2024; 20(4):58.
CrossRef - Asnaashari , Amjad E., Sokouti B. Synergistic effects of flavonoids and paclitaxel in cancer treatment: a systematic review. Cancer Cell Int. 2023; 23(1):211.
CrossRef - Farhan , Rizvi A., Aatif M., et al. Current Understanding of Flavonoids in Cancer Therapy and Prevention. Metabolites. 2023; 13(4):481.
CrossRef - Yang G, Xing J, Aikemu B, et al. Kaempferol exhibits a synergistic effect with doxorubicin to inhibit proliferation, migration, and invasion of liver cancer. Oncol Rep. 2021; 45(4):32.
CrossRef - Erdogan , Doganlar O., Doganlar Z. B., et al. Naringin sensitizes human prostate cancer cells to paclitaxel therapy. Prostate Int. 2018; 6(4):126-135.
CrossRef - Ya F., Li Q., Wang D., et al. Cyanidin-3-o-β-glucoside induces megakaryocyte apoptosis via PI3K/Akt- and MAPKs-mediated inhibition of NF-κB signalling. Thromb Haemost. 2018; 118(7):1215-1229.
CrossRef - Li J., Wang T., Liu P., et al. Hesperetin ameliorates hepatic oxidative stress and inflammation viathe PI3K/AKT-Nrf2-ARE pathway in oleic acid-induced HepG2 cells and a rat model of high-fat diet-induced NAFLD. Food Funct. 2021; 12(9):3898-3918.
CrossRef - Sharifi-Rad M., Pezzani R., Redaelli M., et al. Preclinical pharmacological activities of epigallocatechin-3-gallate in signaling pathways: An update on cancer. Molecules. 2020; 25(3):467.
CrossRef - Li , Xie E., Sun S., et al. Flavonoids for gastrointestinal tract local and associated systemic effects: A review of clinical trials and future perspectives. J Adv Res. 2025: S2090-1232(25)00033-5.
- Hu , Luo Y., Yang J., et al. Botanical flavonoids: Efficacy, absorption, metabolism and advanced pharmaceutical technology for improving bioavailability. Molecules. 2025;30(5):1184.
CrossRef - Yuan , Guo Y., Pu F., et al. Opportunities and challenges in enhancing the bioavailability and bioactivity of dietary flavonoids: A novel delivery system perspective. Food Chem. 2024;430:137115.
CrossRef - Thilakarathna S. H., Rupasinghe P. Flavonoid bioavailability and attempts for bioavailability enhancement. Nutrients. 2013;5(9):3367-87.
CrossRef - Li , Ning J., Wang Y., et al. Drug interaction study of flavonoids toward CYP3A4 and their quantitative structure activity relationship (QSAR) analysis for predicting potential effects. Toxicol Lett. 2018;294:27-36.
CrossRef - Tang Z., Zhang Q. The potential toxic side effects of flavonoids. 2021;46(2):357-366. https://doi.org/10.32604/biocell.2022.015958.
CrossRef - Swain S. S., Rout S. S., Sahoo A., et al. Antituberculosis, antioxidant and cytotoxicity profiles of quercetin: A systematic and cost-effective in silicoand in vitro Nat Prod Res. 2022;36(18):4763-4767.
CrossRef - Cunha , Daniel-da-Silva A. L., Oliveira H. Drug delivery systems and flavonoids: Current knowledge in melanoma treatment and future perspectives. Micromachines (Basel). 2022;13(11):1838.
CrossRef - Li , Shen Q., Feng L., et al. A nanoscale natural drug delivery system for targeted drug delivery against ovarian cancer: action mechanism, application enlightenment and future potential. Front Immunol. 2024; 15:1427573.
CrossRef - Gaikwad S., Morade Y. Y., Kothule A. M., et al. Overview of phytosomes in treating cancer: Advancement, challenges, and future outlook. Heliyon. 2023; 9(6):e16561.
CrossRef - Baksi , Singh D. P., Borse S. P., et al. In vitro and in vivo anticancer efficacy potential of Quercetin loaded polymeric nanoparticles. Biomed Pharmacother. 2018; 106:1513-1526.
CrossRef - Rashedi , Ghorbani Haghjo A., Mesgari Abbasi M., et al. Anti-tumor Effect of Quercetin Loaded Chitosan Nanoparticles on Induced Colon Cancer in Wistar Rats. Adv Pharm Bull. 2019; 9(3):409-415.
CrossRef - Jangid K., Solanki R., Patel S., et al. Genistein encapsulated inulin-stearic acid bioconjugate nanoparticles: Formulation development, characterization and anticancer activity. Int J Biol Macromol. 2022; 206:213-221.
CrossRef - Farabegoli F., Granja A., Magalhães J., et al. Epigallocatechin-3-gallate delivered in nanoparticles increases cytotoxicity in three breast carcinoma cell lines. ACS Omega. 2022;7(46):41872-41881.
CrossRef - Kazmi I,. Al-Abbasi F. A., Nadeem M. S., et al. Formulation, Optimization and Evaluation of Luteolin-Loaded Topical Nanoparticulate Delivery System for the Skin Cancer. Pharmaceutics. 2021; 13(11):1749.
CrossRef - Bonilla-Vidal , Świtalska M., Espina M., et al. Dually Active Apigenin-Loaded Nanostructured Lipid Carriers for Cancer Treatment. Int J Nanomedicine. 2023; 18:6979-6997.
CrossRef - Oršolić , Jazvinšćak Jembrek M. Potential strategies for overcoming drug resistance pathways using propolis and its polyphenolic/flavonoid compounds in combination with chemotherapy and radiotherapy. Nutrients. 2024; 16(21):3741.
CrossRef - Zhou C., Huang Y., Nie S., et al. Biological effects and mechanisms of fisetin in cancer: a promising anti-cancer agent. Eur J Med Res. 2023; 28(1):297.
CrossRef - Naseri A roadmap to establish a comprehensive platform for sustainable manufacturing of natural products in yeast. Nat Commun. 2023; 14(1):1916.
CrossRef - Lyu , Lyu Y., Yu H., et al. Biotechnological advances for improving natural pigment production: a state-of-the-art review. Bioresour Bioprocess. 2022; 9(1): 8. https://doi.org/10.1186/s40643-022-00497-4.
CrossRef - Yang J., Pi C., Wang G. Inhibition of PI3K/Akt/mTOR pathway by apigenin induces apoptosis and autophagy in hepatocellular carcinoma cells. Biomed Pharmacother. 2018; 103:699-707.
CrossRef - Chandrashekar N., Pandi A. Baicalein: A review on its anti-cancer effects and mechanisms in lung carcinoma. J Food Biochem. 2022; 46(9): e14230.
CrossRef - Bie B., Sun J., Guo Y., et al. Baicalein: A review of its anti-cancer effects and mechanisms in hepatocellular carcinoma. Biomed Pharmacother. 2017; 93:1285-1291.
CrossRef - Lai X., Li Y., Gao M. Biochanin A regulates the growth and migration of NSCLC through suppressing the VEGF/VEGFR2 signalling pathway. Oncol Res. 2018. doi: 10.3727/096504018X15321979274728.
CrossRef - Wang D., Zheng C., Chen B., et al. Biochanin A induces apoptosis in MCF-7 breast cancer cells through mitochondrial pathway and Pi3K/AKT inhibition. Cell Biochem Funct. 2024;42(8):e70014.
CrossRef - Lirdprapamongkol K., Sakurai H., Abdelhamed S., et al. A flavonoid chrysin suppresses hypoxic survival and metastatic growth of mouse breast cancer cells. Oncol Rep. 2013; 30(5):2357-64.
CrossRef - Xia Y., Lian S., Khoi P. N., et al. Chrysin inhibits cell invasion by inhibition of Recepteur dorigine Nantais via suppressing early growth response-1 and NF-κB transcription factor activities in gastric cancer cells. Int J Oncol. 2015; 46(4):1835-43.
CrossRef - Chen D., Yuan M., Ye Q., et al. Cyanidin-3-O-glucoside inhibits epithelial-to-mesenchymal transition, and migration and invasion of breast cancer cells by upregulating KLF4. Food Nutr Res. 2020; 64. doi:10.29219/fnr.v64.4240.
CrossRef - Kwon J. Y., Lee K. W., Kim J. E., et al. Delphinidin suppresses ultraviolet B-induced cyclooxygenases-2 expression through inhibition of MAPKK4 and PI-3 kinase. Carcinogenesis. 2009; 30(11):1932-40.
CrossRef - Sun S., Xu K., Yan M., et al. Delphinidin induces autophagic flux blockage and apoptosis by inhibiting both multidrug resistance gene 1 and DEAD-box helicase 17 expressions in liver cancer cells. J Pharm Pharmacol. 2023; 75(2):253-263.
CrossRef - Zhao F., Hong X., Li D., et al. Diosmetin induces apoptosis in ovarian cancer cells by activating reactive oxygen species and inhibiting the Nrf2 pathway. Med Oncol. 2021; 38(7):78.
CrossRef - Pan Z., Tan Z., Li H., et al. Diosmetin induces apoptosis and protective autophagy in human gastric cancer HGC-27 cells via the PI3K/Akt/FoxO1 and MAPK/JNK pathways. Med Oncol. 2023; 40(11):319.
CrossRef - Singh B. N., Shankar S., Srivastava R. K. Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications. Biochem Pharmacol. 2011; 82(12):1807-21.
CrossRef - Zhong W. F., Wang X. H., Pan B., et al. Eupatilin induces human renal cancer cell apoptosis via ROS-mediated MAPK and PI3K/AKT signaling pathways. Oncol Lett. 2016; 12(4):2894-2899.
CrossRef - Wang X., Zhu Y., Zhu L., et al. Eupatilin inhibits the proliferation of human esophageal cancer TE1 cells by targeting the Akt‑GSK3β and MAPK/ERK signaling cascades. Oncol Rep. 2018; 39(6):2942-2950.
CrossRef - Xiao Y., Liu Y., Gao Z., Li X., et al. Fisetin inhibits the proliferation, migration and invasion of pancreatic cancer by targeting PI3K/AKT/mTOR signaling. Aging (Albany NY). 2021; 13(22): 24753-24767.
CrossRef - Pavese J. M., Farmer R. L., Bergan R. C. Inhibition of cancer cell invasion and metastasis by genistein. Cancer Metastasis Rev. 2010; 29(3):465-82.
CrossRef - Lu Q., Lai Y., Zhang H., et al. Hesperetin inhibits TGF-β1-induced migration and invasion of triple negative breast cancer MDA-MB-231 cells via suppressing Fyn/Paxillin/RhoA pathway. Integr Cancer Ther. 2022; 21:15347354221086900.
CrossRef - Kim J. E., Lee D. E., Lee K. W., et al. Isorhamnetin suppresses skin cancer through direct inhibition of MEK1 and PI3-K. Cancer Prev Res (Phila). 2011; 4(4):582-91.
CrossRef - Zhang P., Sun Y., Shi L., et al. Effect of isorhamnetin on carbonic anhydrase IX expression and tumorigenesis of bladder cancer by activating PPARγ/PTEN/AKT pathway. Tissue Cell. 2023; 82:102048.
CrossRef - Amjad E., Sokouti B., Asnaashari S. A systematic review of anti-cancer roles and mechanisms of kaempferol as a natural compound. Cancer Cell Int. 2022; 22(1):260.
CrossRef - Choi E. Y., Han E. J., Jeon S. J., et al. Kaempferol inhibits cervical cancer cells by inducing apoptosis and autophagy viainactivation of the PI3K/AKT/mTOR signaling pathway. Anticancer Res. 2024; 44(7):2961-2972.
CrossRef - Arjsri P., Srisawad K., Umsumarng S., et al. Anti-inflammatory and anti-migratory effects of morin on non-small-cell lung cancer metastasis via inhibition of NLRP3/MAPK signaling pathway. Biomolecules. 2025; 15(1):103.
CrossRef - Maharjan S., Lee M. G., Lee K. S., et al. Morin overcomes doxorubicin resistance in human breast cancer by inducing DNA damage and modulating the LKB1/AMPK/mTORC1 signaling pathway. Biofactors. 2025; 51(1): e2112.
CrossRef - Feng J., Chen X., Wang Y., et al. Myricetin inhibits proliferation and induces apoptosis and cell cycle arrest in gastric cancer cells. Mol Cell Biochem. 2015; 408(1-2):163-70.
CrossRef - Sharma P., Khan M. A., Najmi A. K., et al. Myricetin-induced apoptosis in triple-negative breast cancer cells through inhibition of the PI3K/Akt/mTOR pathway. Med Oncol. 2022; 39(12):248.
CrossRef - Motallebi M., Bhia M., Rajani H. F., et al. Naringenin: A potential flavonoid phytochemical for cancer therapy. Life Sci. 2022; 305:120752.
CrossRef - Chang T. M., Chi M. C., Chiang Y. C., et al. Promotion of ROS-mediated apoptosis, G2/M arrest, and autophagy by naringenin in non-small cell lung cancer. Int J Biol Sc 2024; 20(3):1093-1109.
CrossRef - Guo J., Jin G., Hu Y., et al. Wogonin restrains the malignant progression of lung cancer through modulating MMP1 and PI3K/AKT signalling pathway. Protein Pept Lett. 2023; 30(1):25-34.
CrossRef - Satari A., Ghasemi S., Habtemariam S., et al. Rutin: A Flavonoid as an Effective Sensitizer for Anticancer Therapy; Insights into Multifaceted Mechanisms and Applicability for Combination Therapy. Evid Based Complement Alternat Med. 2021; 2021:9913179.
CrossRef - Li Q., Xu D., Gu Z., et al. Rutin restrains the growth and metastasis of mouse breast cancer cells by regulating the microRNA-129-1-3p-mediated calcium signaling pathway. J Biochem Mol Toxicol. 2021; 35(7):e22794.
CrossRef - Surichan S., Arroo R. R., Tsatsakis A. M., et al. Tangeretin inhibits the proliferation of human breast cancer cells via CYP1A1/CYP1B1 enzyme induction and CYP1A1/CYP1B1-mediated metabolism to the product 4′ hydroxy tangeretin. Toxicol In Vitro. 2018; 50:274-284.
CrossRef - Gv V., Ranganathan P. Jr, Palati S. Tangeretin’s Anti-apoptotic signalling mechanisms in oral cancer cells: In vitro anti-cancer activity. Cureus. 2023; 15(10): e47452.
- Liu Y., Lu L., Cheng P., et al. Wogonin inhibits colorectal cancer proliferation and epithelial mesenchymal transformation by suppressing phosphorylation in the AKT Pathway. Am J Chin Med. 2024; 52(4):1155-1172.
CrossRef
Abbreviations List
COX2 Cyclooxygenase-2
DNMT DNA Methyltransferase
EGCG Epigallocatechin-3-Gallate
EGFR Epidermal Growth Factor Receptor
EMT Epithelial–Mesenchymal Transition
FDA Food and Drug Administration
GLOBOCAN Global Cancer Observatory
HCC Hepatocellular Carcinoma
HDAC Histone Deacetylase
MAPK Mitogen-Activated Protein Kinase
MMPs Metalloproteinases
MMP-2/9 Matrix Metalloproteinase-2/9
mTOR Mammalian Target of Rapamycin
NF-κB Nuclear Factor Kappa B
PI3K/Akt Phosphoinositide 3-Kinase/ Protein Kinase B
STAT3 Signal Transducer and Activator of Transcription 3
TCM Traditional Chinese Medicine
TGF-β Transforming Growth Factor-Beta
VEGF Vascular Endothelial Growth Factor
WHO World Health Organization
5-FU 5-Fluorouracil













