Optimization of The Replacement of Alcoholic Sugars with Sucrose for Actinidia Deliciosa Under Osmo-Drying Conditions by Measuring Physichochemical Characteristic Using the Rsm Method
Department of Chemical Engineering, Sathyabama Institute of Science and Technology, Chennai, India.
Corresponding Author Email: sathish.chemical@sathyabama.ac.in
DOI : http://dx.doi.org/10.12944/CRNFSJ.14.1.7
ABSTRACT:This research explores the effectiveness osmo-convective drying approaches for kiwi fruit (Actinidia deliciosa) with a focus on preserving nutritional integrity and enhancing quality attributes. Drying is a critical preservation method to reduce moisture content, inhibit microbial growth, and extend the shelf life of perishable fruits like kiwi. The methodology involved an initial osmotic pre-treatment for one hour using a honey salt solution to promote water loss and solid gain, followed by convective drying at 60 °C. Three osmotic agents erythritol, sorbitol, and sucrose were evaluated, each at 50% (w/w) concentration, to assess their effects on drying performance and product quality. Key parameters measured included water loss, solid gain during osmotic treatment, changes in moisture content, and internal temperature of the fruit during drying. Moisture content dropped significantly from 83% to below 20% within two hours of drying. The dried kiwi retained approximately 78% of its original vitamin C content and 70% of total phenolics, indicating effective preservation of key bioactive compounds. Texture analysis showed a 25% increase in firmness, supported by microstructural observations, while color measurements indicated minimal browning, preserving visual appeal. The combined osmo-convective drying process proved efficient in producing dried kiwi slices with improved nutritional value, texture, and appearance, demonstrating its potential as a viable preservation technology. Across all treatments, the dried kiwifruit showed clear differences in quality. Erythritol proved most effective, producing the highest water loss and the lowest water activity (0.25), which greatly improves shelf stability. Color changes were relatively small in sucrose and erythritol samples, while sorbitol caused more noticeable darkening. Sorbitol, however, helped retain the most phenolic compounds, including chlorogenic and caffeic acids. The Midilli model closely matched the drying behavior, and the high effective diffusivity value (2.78 × 10⁻¹⁰ m²/s) confirmed rapid moisture removal. Overall, each osmotic agent influenced quality in distinct and meaningful ways.
KEYWORDS:Kiwi; Nutritional Value; Optimization; Osmo-Convective Drying; Osmotic Solutions
Introduction
Global fruit demand is rising, making long-distance distribution challenging due to the high perishability of fresh produce. Drying extends shelf life, reduces packaging needs, and cuts transport costs by lowering weight and volume.1 Among various methods, osmo-convective drying—a two-step process involving osmotic dehydration followed by convective hot-air drying—is particularly effective for preserving nutrients, color, flavor, and texture while reducing enzymatic browning.2
The efficiency of osmotic dehydration hinges on parameters like the type of osmotic agent, solution concentration, temperature, and immersion time. Traditionally, sugars such as sucrose, glucose, and fructose are used for fruits.3 However, the growing consumer demand for healthier, low-sugar foods has spurred the search for alternative osmotic agents.4 In this context, sugar alcohols (polyols) like sorbitol and erythritol present a promising option due to their low caloric content, low digestibility, and minimal impact on blood sugar levels.5
Kiwifruit (Actinidia deliciosa) is an ideal candidate for this investigation due to its rich nutritional profile, high levels of vitamin C, and valuable phytonutrients. However, its delicate nature makes it highly prone to spoilage, underscoring the need for effective preservation techniques like osmo-convective drying.6
While previous studies have explored osmotic dehydration of kiwifruit, they have predominantly relied on traditional sugars. Although some research exists on polyols for other fruits, a direct and systematic comparison of sucrose, sorbitol, and erythritol in a combined osmo-convective drying process for kiwifruit is lacking.7 Key knowledge gaps remain regarding how these sugar alcohols mechanistically compare to sucrose in terms of Drying kinetics (moisture loss, solid gain, drying rate), final product quality (color stability, texture, water activity) and nutrient preservation.
Osmotic dehydration operates at low temperatures, limiting nutrient loss and browning while enhancing shelf life. However, drawbacks include taste alterations due to acidity loss and sugar coating, which may require rinsing.8 Typically, sugars are employed as osmotic agents for fruits, while salts are commonly used for vegetables.9
Traditionally, sugars like sucrose, glucose, and fructose are used, but sugar alcohols such as sorbitol and erythritol are becoming popular because they are low in calories.10 Erythritol stands out for speeding up water removal, lowering water activity, and improving shelf life, while sorbitol helps maintain a firmer texture.11 Sorbitol-treated fruits retained 15% more polyphenols and exhibited less shrinkage and browning compared to sucrose-treated ones.12 When combined with hot-air drying, this approach keeps the fruit’s color, texture, and antioxidants.13 Despite these advances, knowledge gaps remain regarding the optimal osmotic agents for kiwifruit, their effects on drying kinetics and nutrient preservation, and the comprehensive quality outcomes of osmo-convective drying processes under varied conditions.14 On comparing the sucrose and sorbitol at 50°C and 50°Brix for 3 hours, sucrose led to a higher initial WL (48 g/100g) than sorbitol (42 g/100g). However, sorbitol-pretreated samples had a 20% faster convective drying rate in the second stage.15
To address these gaps by systematically evaluating the use of sugar alcohols as alternatives to traditional sucrose in osmotic solutions, paired with convective drying, to improve drying performance and the nutritional and physicochemical quality of dried kiwifruit.16
In this research, the primary goal of this research is to pioneer the application of sorbitol and erythritol in the osmo-convective drying process for kiwifruit, an approach that has not been previously reported. We hypothesize that these polyols will serve as effective alternatives to sucrose, improving drying efficiency and enhancing the nutritional and physicochemical quality of the final dried product. This study will systematically compare the performance of these osmotic agents by analyzing drying kinetics, nutrient content, and key quality attributes to validate their innovative use.
Materials and Methods
Sourcing and Selection
Fresh kiwifruit (Actinidia deliciosa) were procured from a local market in Chennai. Fruits were selected based on uniform size, shape, and color, and the absence of physical damage or microbial decay.
Preparation
The fruits were washed in potable water to remove surface dirt and air-dried. The outer fuzzy skin was peeled using a stainless-steel peeler. The ends were trimmed, and the fruits were sliced into uniform discs or sectors of a specific thickness (10mm)
Initial Moisture Content
The initial moisture content was determined using the AOAC method.17 which is typically the oven drying method. Specifically, a representative sample of fresh kiwi pulp (approx. 5g) was placed in a pre-weighed petri dish and dried in a hot air oven at 105°C until a constant weight was achieved.
Osmotic Solution Preparation
Three osmotic solutions were prepared. Sucrose, Erythritol, and Sorbitol, each at 50% (w/w) concentration. This was done by dissolving 500g of the solute in 500g of distilled water with mild heating and stirring to ensure complete dissolution. The solutions were allowed to cool to room temperature before use.18
Osmotic Dehydration (OD) Process
A known weight of kiwi slices was placed in a food-grade mesh bag and fully immersed in a known volume of the osmotic solution (1:4 fruit-to-solution ratio) within a sealed container. The containers were placed on a laboratory orbital shaker to ensure continuous, uniform agitation. The osmotic treatment was conducted for a predetermined time. At each interval, samples were removed from the solution, gently blotted with absorbent paper to remove surface liquid, and weighed immediately.19 A laboratory-scale tray dryer with controlled temperature and air velocity was used. The osmotically pre-treated kiwi slices from each group (sucrose, erythritol, sorbitol) were spread in a single layer on drying trays. They were dried at three different temperatures: 40°C, 50°C, and 60°C. The trays were removed at fixed time intervals, weighed. The drying process was tracked by recording the sample weights at fixed time intervals. This procedure was repeated for each temperature setting to ensure consistency.20
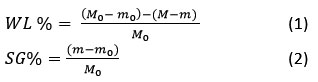
In this content, Mo is the starting mass of the fresh fruit before undergoing osmotic treatment (in grams). M represents the mass of the kiwi at a specific time t during the osmotic process (in grams). m refers to the dry weight of the kiwi at the same time t, while mo is the dry weight of the fresh fruit before treatment (in grams). The percentage of water loss (%WL) was calculated based on the initial weight of the fresh kiwi and represented the total water removed during osmotic dehydration. Similarly, the percentage of solid gain (%SG) reflected the proportion of solids taken up by the kiwi relative to its original weight.
Kinetics Measurement
The application of sugar alcohols was significant in accelerating dehydration and improving drying kinetics compared to sucrose, with sorbitol exhibiting the shortest drying time and highest solid gain. Final product quality assessment included color measurement, water activity (aw), and bioactive compound analysis (carotenoids and polyphenols).21
Product Quality Assessment
The quality assessment of dried products was carried out by analysing several parameters, including colour variation, water activity, carotenoid concentration, and polyphenol content. Colour measurements were performed using a Konica Minolta CR400 colorimeter. To evaluate water activity (aw), both fresh and dried samples were analysed using the LabMaster-aw Standard instrument.22 For dried samples, the aw measurement was taken only after a minimum of 24 hours of equilibration in a desiccator to ensure uniform moisture distribution post-drying. The quantification of caffeic acid and chlorogenic acid was carried out in C18 column, and their concentrations were subsequently analyzed via reverse-phase high-performance liquid chromatography coupled with diode-array detection (RP-HPLC-DAD).23 The analysis was carried out with chromatograms monitored at a wavelength of 454 nm.
Optimization of Process parameters
The regression model included four linear terms (A, B, C, D), their corresponding quadratic components (A², B², C², D²), six two-way interaction terms (AB, AC, AD, BC, BD, CD), and a block term. A second-order polynomial model was assessed using ANOVA at a 5% significance level to evaluate how well it fit the experimental data. The model’s validity was confirmed by comparing the calculated F-value with the critical value from standard statistical tables; when the calculated F exceeded the critical value, the null hypothesis was rejected, indicating that at least one regression coefficient had a meaningful impact on the outcome and to study the effects of various process variables on the responses, a Central Composite Design (CCD) was employed, incorporating four independent factors at three levels each: concentration of the osmotic solution, temperature of the drying air, osmotic bath temperature, and treatment time. The primary response variables analysed were mass reduction, solid gain, and water loss during the osmotic dehydration process.24
Results
Initial Moisture Content Analysis
Figure 1 illustrates the variation in moisture content of kiwi slices during osmotic dehydration in different hypertonic solutions. It is evident that the initial 30 minutes of treatment were the most efficient, showing the greatest reduction in water.25
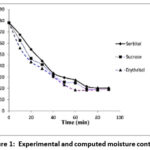 |
Figure 1: Experimental and computed moisture content |
Analysis of the experimental findings showed that samples treated with the three osmotic solutions yielded nearly comparable results with respect to both solid gain and water loss, indicating that the sugars behaved in a similar manner during prolonged dehydration. Among the solutions, erythritol consistently produced slightly higher values for these parameters. This outcome can be explained by erythritol’s relatively high osmotic pressure, a result of its molecular properties, even when used at moderate concentrations. Osmotic dehydration functions through the migration of water from the fruit into the external solution, driven by osmotic pressure gradients.26 Compared with other sugars at the same concentration, erythritol solutions generally exhibit lower viscosity, which enhances mass transfer and accelerates water removal from the fruit. Furthermore, erythritol may interact with plant cell components in a way that modifies the semi-permeable nature of the cell wall, thus enabling more efficient exchange of water and solutes. Previous studies also indicate that polyols such as erythritol are particularly effective at establishing osmotic gradients due to their higher permeability. Notably, the most substantial water loss occurred within the first 30 minutes of treatment, during which the fruit maintained its structural stability. An effective osmotic medium should ideally promote moderate solute uptake in order to preserve both the texture and sensory attributes of the fruit, preventing excessive sweetness or crystallization. Smaller molecules penetrate the cell wall and membrane interfaces more readily, while stronger osmotic pressure differences not only enhance water removal but also increase solute absorption within the tissue.27
Erythritol tends to be the best choice for osmoconvective drying of kiwifruit due to highest osmotic pressure which aids the faster water loss, Lower viscosity for the better mass transfer, mild sweetness, low calories and good texture retention.
Table 1: ANOVA table
|
Source |
Sum of Squares | df | Mean Square | F-value | p-value | |
|
Model |
461.71 | 14 | 32.98 | 46171.21 | < 0.0001 | significant |
| A-Osmotic solution temperature | 6.83 | 1 | 6.83 | 9557.33 | < 0.0001 | |
|
B-Osmotic solution concentration |
3.53 | 1 | 3.53 | 4937.33 | < 0.0001 | |
| C-Immersion time | 3.84 | 1 | 3.84 | 5376.00 |
< 0.0001 |
|
|
D-Drying temperature |
0.8067 | 1 | 0.8067 | 1129.33 | < 0.0001 | |
| AB | 0.0100 | 1 | 0.0100 | 14.00 |
0.0022 |
|
|
AC |
5.06 | 1 | 5.06 | 7087.50 | < 0.0001 | |
| AD | 2.72 | 1 | 2.72 | 3811.50 |
< 0.0001 |
|
|
BC |
10.56 | 1 | 10.56 | 14787.50 | < 0.0001 | |
| BD | 9.92 | 1 | 9.92 | 13891.50 |
< 0.0001 |
|
|
CD |
2.89 | 1 | 2.89 | 4046.00 | < 0.0001 | |
| A² | 348.04 | 1 | 348.04 | 4.873E+05 | < 0.0001 | |
|
B² |
61.33 | 1 | 61.33 | 85867.30 | < 0.0001 | |
| C² | 27.26 | 1 | 27.26 | 38163.24 |
< 0.0001 |
|
|
D² |
119.94 | 1 | 119.94 | 1.679E+05 | < 0.0001 | |
| Residual | 0.0100 | 14 | 0.0007 | Predicted R² |
0.9999 |
 |
Figure 2a: Variable interactions impacting water loss using RSM-CCD |
 |
Figure 2b: Variable interactions impacting solid gain using RSM-CCD |
Discussion
The regression model was constructed with four main linear factors (A, B, C, D), their corresponding quadratic components (A², B², C², D²), and six pairwise interaction effects (AB, AC, AD, BC, BD, CD), along with one block term, as outlined. This second-order empirical model was assessed through analysis of variance (ANOVA) at the 5% significance level. The statistical adequacy of the model was verified using the F-test. The nutrition details of the fresh and osmo-convective dried fruit were provided in Table 2. Generally, when the computed F-value surpasses the critical (tabulated) F-value, the null hypothesis stating that all regression coefficients are equal to zero is rejected. To explore how process variables, affect mass reduction, solid gain, and water loss during osmotic dehydration, a Central Composite Design (CCD) was applied, considering four factors at three levels. The coded levels of the independent factors are reflected, while the trial-based runs and their corresponding responses are summarized in Table 1. The influence of each variable on water loss is illustrated through contour plots in Figure 2. These plots clearly indicate that solute concentration, solution temperature, drying temperature, and treatment duration significantly affected water loss. In particular, maximum water loss and solid gain was achieved under the following optimized conditions: 55% solute concentration in the osmotic medium, solution temperature of 45 °C, treatment time of 120 minutes, and drying temperature of 50 °C results that are consistent with findings reported in earlier studies. The observed increase in water loss is mainly linked to the stronger osmotic gradient created by raising the solute concentration up to 55%. This highlights that even minor changes in concentration can substantially boost moisture migration.28 A more concentrated solution exerts higher osmotic pressure, thereby enhancing the driving force for water transfer from the fruit. Beyond a certain concentration, however, water loss began to decline, as shown by the convex nature of the response curve. This reduction could be explained by greater solute uptake within the fruit tissue and diminished osmotic flow across the semi-permeable cell membranes. Alongside solute concentration, both temperature and treatment time had a pronounced effect on water removal. Elevated temperatures can modify membrane permeability, facilitating faster water transport, while also lowering the viscosity of the osmotic medium. The reduction in viscosity supports internal convective movement, preventing localized dilution near the fruit surface and maintaining a strong osmotic gradient, thereby improving dehydration efficiency.29
The regression equation is
Water loss = 25.47 + 0.517A + 0.385B + 0.4C + 0.183D – 0.025AB – 0.56AC – 0.4125AD – 0.8125BC – 0.7875BD – 0.425CD – 3.66A2 – 1.54B2 – 1.02C2 – 2.15D2
Solid Gain = 7.8 – 0.1333A + 0.147B + 0.45C + 0.2583D + 0.1125AB – 0.125AC + 0.5125AD + 0.25BC – 0.3375 BD – 0.425CD – 1.29A2 – 1.234B2 – 1.08C2 – 1.18D2
The R² value showed excellent agreement with the Adjusted R² of 1.000, with a difference far below the threshold of 0.2, reflecting strong consistency of the model. To evaluate goodness-of-fit and adequacy, both (R²) and the adjusted R² were analysed. The R² values exceeded 0.99, meaning that more than 99% of the variability in the observed data was successfully explained by the model. Moreover, the adjusted R² values were nearly identical to the corresponding R² values, further supporting the robustness and statistical validity of the model.30
Table 2: Comparative Nutritional Test Results and osmotic agent properties
| Parameters | Units per 100g | Fresh kiwi | Dried kiwi |
| Energy | kcal | 56 | 61 |
| Carbohydrate | g | 13.2 | 14.6 |
| Total fat | g | 0.46 | 0.52 |
| Protein | g | 1.08 | 1.14 |
| Sodium as Na | mg | 2.5 | 3 |
| Calcium | mg | 26 | 34 |
| Magnesium | mg | 14.3 | 17 |
| Potassium | mg | 304 | 312 |
| Vitamin C | mg | 90 | 92.7 |
Drying kinetics
Based on the above characteristic curve the kinetic equation for drying of kiwi is given by Newton, Henderson and Pabis, Logarithmic, Midilli.
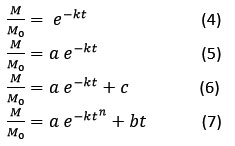
Here, M/Mₒ represents the moisture ratio, t denotes the drying time, M and Mₒ is the time based moisture content and initial dry basis moisture. The parameter k refers to the drying rate constant, while n, a, b, and c are empirical model constants. Results indicated that higher temperatures accelerated the rate of moisture removal, which in turn significantly shortened the overall drying duration.31
 |
Figure 3: Drying kinetics (a) Newton model (b) Henderson and Pabis model (c) Logarithmic model (d) Midilli model |
Analytical Error Analysis
In order to determine the most appropriate models for interpreting the adsorption data, different error functions must be examined. These functions indicate how closely each model matches the experimental results. Among them, the coefficient of determination (R²) is widely used in adsorption research to evaluate the fit of both isotherm and kinetic models. A model demonstrating a higher R² value along with lower error function values is regarded as providing a more accurate representation.32
The R² indicates how much of the variation in the dependent variable is accounted for by the independent variables in a regression model. MSE, is calculated by averaging the squared differences between actual and predicted values. When the square root of the MSE is taken, it yields the (RMSE), which provides insight into the typical magnitude of prediction errors. Likewise, MAD is calculated by taking the average of the absolute differences between the actual and predicted values. Meanwhile, the (MAPE) represents these absolute differences as a percentage of the actual values, averaged across all observations.
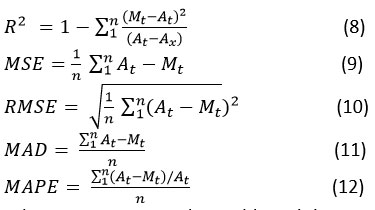
Where Aₜ represents the trial-based data, Mₜ is the model data, Aₓ denotes the average of the experimental values, and n is the total number of experiments. In choosing the most suitable isotherm model, preference is generally given to the one showing the maximum coefficient of determination (R²) along with the lowest error function values which was tabulated in Table 3. However, relying solely on R² is not sufficient to determine the best fit, since each model is associated with at least one minimum error value.33,34 Based on its highest R² value and lowest error estimates, the Midilli model was identified as the best fit.35,36
Table 3: Model parameters and error analysis
| Model | Parameters | R2 | MSE | RMSE | MAD | MAPE | Remarks |
| Newton | k = 0.0048 | 0.976 | 0.1075 | 0.038 | 0.2833 | 0.6425 | Good but not best |
| Henderson & Pabis | a = 1.045
k = 0.0053 |
0.986 | 1.355 | 0.035 | 1.123 | 0.8356 | Very good |
| Logarithmic | a = 0.920
k = 0.0185 c = 0.0456 |
0.955 | 0.0132 | 0.024 | 0.333 | 0.1788 | Least accurate |
| Midilli | a = 1.000,
k = 0.0031 n = 1.282 b = 0.00001 |
0.998 | 0.0096 | 0.013 | 0.09033 | 0.0720 | Best fit |
Modeling Effective Diffusivity
In osmoconvective drying, kiwi fruit slices are typically subjected to osmotic dehydration followed by convective drying. The effective diffusivity (𝐷eff) during this procedure can be estimated using Fick’s second law, which describes moisture diffusion in porous media.37,38 Effective diffusivities is a vital parameter for modeling and optimizing the osmoconvective drying process of kiwi fruit.39 Understanding and accurately estimating Deff allows for better control over drying time and product quality. Experimental studies provide valuable insights into the impact of various pretreatment methods on moisture diffusion rates, aiding in the development of more efficient drying protocols.
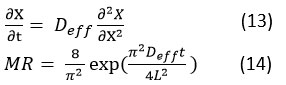
Plotting ln(MR) vs t gives a straight line with slope k. From the slope Deff was calculated.40
![]()
Where: X is the Moisture content (wet basis), 𝑡 is time, x is the position within the sample.
The effective diffusivity can be determined by fitting experimental drying data to this equation and calculating the slope of the logarithmic moisture ratio versus time plot. An effective diffusivity of 2.78×10−10 m2/s was observed, indicating a moisture removal rate.41
Effects on Microstructure
To assess how various osmotic treatments affect mass transfer at the cellular level, scanning electron microscopy (SEM) was used to examine kiwi samples from both untreated and osmotically treated groups.42 High-magnification images were taken of the fruit surfaces that had been in direct contact with the osmotic solutions.43 The observations indicated that osmotic dehydration caused significant collapse of the cell walls and disruption of the overall cell structure. After two hours of treatment, the internal structure appeared uniform across samples, with cell walls and intracellular spaces becoming difficult to distinguish due to surface coverage by sugars from the osmotic solution.44,45 This structural collapse occurs as water is rapidly removed from vacuoles, placing inward stress on the water-filled cells and leading to their breakdown.46 The phenomenon is especially evident in the initial phase of osmotic dehydration, when the osmotic gradient is steepest and cells still contain a highwater content.47 Comparatively, micrographs taken after one hour of treatment showed clearer cell boundaries and more pronounced intercellular spaces.48 This may be attributed to partial pectin solubilization in the middle lamella and increased porosity linked to the lower acidity of the solution at that point.49 As dehydration continued, cells nearer the fruit’s surface showed progressive structural collapse under osmotic stress, as confirmed by the SEM images taken at the two-hour mark.50
 |
Figure4: SEM images of kiwi samples under control and treated conditions: (a) control at 1 h; (b) treated sample at 1 h; (c) control at 2 h; (d) treated sample at 2 h. |
Product Quality Characteristics
One important factor influencing the microbiological safety and shelf life of dried kiwifruit products is water activity (aw). The removal of free water and the uptake of osmotic solutes, which bind water molecules and decrease their availability, cause aw to drastically drop during osmotic dehydration. 0.4±0.1 for sucrose, 0.32±0.1 for sorbitol, and 0.25±0.1 for erythritol were the results of the aw analysis of the dried samples. In contrast to kiwifruits treated with sucrose, those treated with sugar alcohols such as erythritol and sorbitol exhibit somewhat lower aw values in the dried product, indicating better moisture binding and water removal effectiveness. These sugar alcohols improve moisture management and speed up drying kinetics, which enhances microbial stability. The overall color change of the samples was expressed as ∆E.
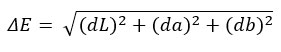
Following calibration under a standard illuminant, three coordinates—L* for lightness/darkness, a* and b* for redness/greenness, and yellowness/blueness, respectively—that objectively describe color were measured. Fresh kiwi had the lowest value for the lightness parameter (L*) (46.3±0.71), which rose significantly in dried samples (48.2±1.5). In terms of the a* parameter (red–green chromaticity), fresh kiwi had a negative value (−5.1 ± 1.23), which is consistent with its natural green color, whereas the dried sample had 5.6 ± 1.2, which may have been caused by pigment transformation or concentration effects during drying. The dried sample had a b* parameter of 11.2 ± 2.2, while the fresh kiwi showed 14.3 ± 2.6. Lastly, there was no baseline comparison for color difference (ΔE) in fresh kiwi fruit. The color change shows the respective ΔE values of 11.318 indicating a moderate color change while using erythritol as an osmotic agent. Similarly, ΔE = 25.16 for sorbitol and ΔE = 9.78 for sucrose.
The amount of chlorogenic acid (µg/mL) in both fresh and dried kiwi fruits varied greatly depending on the temperature and drying technique. The dried sample had 12.00 µg/mL of sucrose, while the fresh kiwi had 1.9 µg/mL. Sorbitol is 14.92 µg/mL, while erythritol is 13.39 µg/mL. According to these findings, drying at 50°C preserved chlorogenic acid the best, whereas higher temperatures significantly degraded it. Sorbitol had the highest caffeic acid content at 0.67 µg/mL, while fresh kiwi had the lowest at 0.15 µg/mL. Erythritol yielded 0.41 µg/mL, while sucrose retained 0.52 µg/mL. All methods showed a significant decrease in caffeic acid content at higher temperatures. The removal of moisture and the intensification of fruit components during heating may have contributed to the concentration of caffeic acid and chlorogenic acid in dried kiwi fruit, which is higher than in fresh fruit.50
Conclusion
The findings of the study indicated that the most active phase of osmotic dehydration in kiwi slices occurred within the first 30 minutes of treatment. The extent of water loss (WL) and solid gain (SG) varied depending on the osmotic agent used, with sucrose demonstrating the most immediate effect. However, erythritol yielded the highest overall dehydration efficiency. High Solid Gain, less water to remove within the shorter drying time. From an industrial engineering perspective, Erythritol is the best choice as it saves time and energy. These results suggest that sugar alcohols may serve as promising alternatives to traditional sucrose, potentially enhancing the quality of dried products. When drying was carried out under optimized conditions, the Midilli model was found to most accurately represent the drying behaviour. A relatively high effective moisture diffusivity of 2.78 × 10⁻¹⁰ m²/s and low activation energy, calculated using Fick’s law, supported the conclusion that the drying process was both rapid and energy-efficient. Additionally, it improved the preservation of caffeic and chlorogenic acids and produced the overall color change (ΔE = 11.38), suggesting that it may be able to preserve the nutritional value and sensory appeal of dried kiwi fruit. With the exception of erythritol, which better maintained the fruit’s appearance, the majority of osmotic agents resulted in some degree of color degradation in terms of the finished product’s visual quality. The optimization of process parameters were analysed through CCD, reveals maximum water loss and solid gain was achieved under the following optimized conditions: 55% solute concentration in the osmotic medium, solution temperature of 45 °C, treatment time of 120 minutes, and drying temperature of 50 °C. The validity of the optimized model was confirmed through confirmation experiments. The close agreement between the predicted values and the actual experimental results (p > 0.05), with a low percentage error validates the robustness of the CCD model. Additionally, microstructural analysis revealed that osmotic treatment led to plasmolysis and changes to the cellular surface architecture in both treated and untreated fruit tissues. According to the study, sugar alcohols can provide a higher-quality dry product and be a better substitute for regular sugar (sucrose). Future research should optimize hybrid pre-treatment methods like Ultrasound-Microwave Assisted OD for better nutrient retention and efficiency. It should explore new osmotic agents and anti-browning additives to improve preservation and functionality. Developing kinetic models to predict moisture loss, nutrient degradation, and quality changes is essential for scaling. Life Cycle Assessment and economic analysis are needed to ensure industrial sustainability and profitability. Studies on nutrient bioavailability and extending applications to different kiwifruit varieties and product forms will broaden the technology’s impact.
Acknowledgement
We are grateful to the management of Sathyabama Institute of Science and Technology for providing the research facilities that greatly assisted in this work.
Funding Sources
The author(s) received no financial support for the research, authorship, or publication of this article.
Conflicts of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Jayasathya Ezhilkumar: Conceptualization, Methodology, Writing – Original Draft.
- Sathish Sundararaman: Visualization, Supervision, Project Administration, Writing – Review and Editing.
- Venkat Vignesh Seenuvasan: Data Collection, Analysis, Resources
References
- Araújo Ald, Pena R. Combined pulsed vacuum osmotic dehydration and convective air-drying process of jambolan fruits. Foods. 2023; 12: 1785. doi: 10.3390/foods12091785
CrossRef - Araújo AL, Pena RS. Influence of process conditions on the mass transfer of osmotically dehydrated jambolan fruits. Food Sci. Technol. 2022; 42: e37520.
CrossRef - Abhishek Rana, Atul Dhiman, Satish Kumar, et al. Osmosonication for dehydration of fruits and vegetables: Mechanistic understanding, mathematical models and comprehensive applications in processing. Trends in Food Science & Technology. 2024; 104688. doi: https://doi.org/10.1016/j.tifs.2024.104688
CrossRef - Barathiraja R, Thirumal P, Thirumalaikumarasamy D, et al. Investigation of drying kinetics and qualities of turkey berry in fluidized bed dryer. Today Proc. 2021; 46: 7711–7718.
CrossRef - Bao T, Hao X, Shishir MRI, et al. Cold plasma: An emerging pretreatment technology for the drying of jujube slices. Food Chem. 2021; 337: 127783.
CrossRef - Bialik M, Wiktor A, Witrowa-Rajchert D, et al. The influence of osmotic dehydration conditions on drying kinetics and total carotenoid content of kiwiberry (actinidia arguta). J. Food Eng. 2020; 16: 20180328.
CrossRef - Bhat TA, Hussain SZ, Wani SM, et al. The impact of different drying methods on antioxidant activity, polyphenols, vitamin C and rehydration characteristics of Kiwifruit. Food Bioscience. 2022; 10182. doi: https://doi.org/10.1016/j.fbio.2022.101821
CrossRef - Araújo AL, Pena RS. Influence of process conditions on the mass transfer of osmotically dehydrated jambolan fruits. Food Sci. Technol. 2022; 42: e37520.
CrossRef - Alexandra Mari, Parisouli DN & Krokida M. Exploring Osmotic Dehydration for Food Preservation: Methods, Modelling, and Modern Applications. 2024; 13, 2783. doi: https://doi.org/10.3390/foods13172783
CrossRef - Antonio M, Rosa R, Roberta P, et al. Evolution of Quality Parameters and Bioactivity of Actinidia chinensis cv. Sungold (Kiwifruit) Slices Subjected to Different Drying Conditions Storage for 4 Months. Foods, 2024; 13, 2100. doi: https://doi.org/10.3390/ foods13132100
CrossRef - Akharume F, Smith A, Sivanandan L, et al. Recent Progress on Osmo-convective Dehydration of Fruits. Journal of Food Science & Technology, 2020; 4:9. doi: 10.25177/JFST.4.9.RA.613
CrossRef - Ali Asghari, Assana Zongo P, Freddy Osse E, et al. Review of osmotic dehydration: Promising technologies forenhancing products’ attributes, opportunities, andchallenges for the food industries. Compr Rev Food Sci Food Saf, 2024; 23: e13346. doi: 10.1111/1541-4337.13346
CrossRef - Carmo JRD, Corrêa JLG, de Santos AAL, et al. Use of Osmotic Dehydration Assisted by Ultrasound to Obtain Dried Mango Slices Enriched With Isomaltulose. Journal of Food Science. 2025; 90:e70223. doi: https://doi.org/10.1111/1750-3841.70223
CrossRef - Cichowska-Bogusz J, Figiel A, Carbonell-Barrachina AA, et al. Physicochemical properties of dried apple slices: Impact of osmo-dehydration, sonication, and drying methods. 2020; 25: 1078. doi: https://doi.org/10.3390/molecules25051078
CrossRef - Clemente G, Femenia A, Llavata B, et al. Combined Effect of Airborne Ultrasound and Temperature On the Drying Kinetics and Quality Properties of Kiwifruit (Actinidia Deliciosa). Food and Bioprocess Technology, 2024; 17:440–451. doi: https://doi.org/10.1007/s11947-023-03138-6
CrossRef - Das HK, Satapathy S, Panda BS. Drying behaviour of osmo-convective drying of carrot slices and quality characteristics of dehydrated products. Food Process. Technol. 2020; 11 (5): 1–5.
- De Lima CA, da Silva RMM, Gomes JP, et al. Evaluation of physical-chemical changes in banana peel under osmo-convective drying conditions. Agric. Stud. 2020; 8 (3): 238–251. doi: https://doi.org/10.5296/jas.v8i3.16347
- Eze JI, Agbo EA. Osmotic dehydration in food processing: A Review. J. Nutr. Food Saf. 2025; 17 (4): 76–86.
- Fakhreddin Salehi, Rana Cheraghi & Majid Rasouli. Mass transfer analysis and kinetic modeling of ultrasound‑assisted osmotic dehydration of kiwifruit slices. Scientific reports. 2023; 13:11859. doi: https://doi.org/10.1038/s41598-023-39146-x
CrossRef - Chandra A, Kumar S, Tarafdar A, et al. Ultrasonic and osmotic pretreatments followed by convective and vacuum drying of papaya slices. Sci. Food Agric. 2020; 101: 2264–2272.
CrossRef - Gamlath J, Satapathy S, Das HK. Evaluation of mass transfer during osmo-convective drying of benincasa hispida using regressional-desirability method. Microbiol. Biotechnol. Food Sci. 2020; 9 (6): 1218–1223. doi: https://doi.org/10.15414/jmbfs.2020.9.6.1218-1223
- Kroehnke J, Szadzińska J, Radziejewska-Kubzdela E, et al. Osmotic dehydration and convective drying of kiwifruit (Actinidia deliciosa) – The influence of ultrasound on process kinetics and product quality. Ultrason Sonochem. 2021; 71:105377. doi: 10.1016/j.ultsonch.2020.105377.
CrossRef - Junqueira JRJ, Corrêa JLG, Mendonça KS, et al. Modelling mass transfer during osmotic dehydration of different vegetable structures under vacuum conditions. Food Sci. Technol. 2021; 41: 439–448.
CrossRef - Kadir N, Yeasmen N, Bhuiyan MHR, et al. Osmotic and convective hot air drying of sweet gourd. Food Science and Biotechnology. 2023; 33(5): 1269. doi: 10.1007/s10068‑023‑01399‑7
CrossRef - Kalantari D, Salami M. Recent progress on osmo-convective dehydration of fruits. Food Sci. Nutr. Res. 2020; 3 (3): 221–232.
- Khan MG, Ahmad F, Zaidi S, et al. Effect of ultrasound‑assisted osmotic dehydration pre‑treatment on tray drying of musk melon cubes. Food Biophysics. 2025; 20: 83. doi: 10.1007/s11483‑025‑09973‑8
CrossRef - Khmelev V, Shalunov A, Terentiev S, et al. Experimental and computer study of the mechanism and identification of conditions for energy-efficient removal of moisture from materials under ultrasonic exposure. PLoS One. 2024; 19(5): e0302585. doi: 10.1371/journal.pone.0302585.
CrossRef - Kręcisz M, Kolniak-Ostek J, Stępień B, et al. Bio-compounds, antioxidant activity, and phenolic content of broccoli after impregnation with beetroot juice. 2025; 30(10): 2143. doi: 10.3390/molecules30102143.
CrossRef - Kowalska H, Marzec A, Kowalska J, et al. The use of a hybrid drying method with pre-osmotic treatment in strawberry bio-snack technology. J. Food Eng. 2020; 16: 20180318.
CrossRef - Macedo LL, Corrêa JLG, Vimercati WC, et al. The impact of using vacuum and isomaltulose as an osmotic agent on mass exchange during osmotic dehydration and their effects on qualitative parameters of strawberries. Food Process Eng.2022; 45: e14057.
CrossRef - Nandane AS, Kulkarni DV, Pujari PP. Ultrasonic and osmotic pretreatments followed by convective and vacuum drying of papaya slices. Food Process. Preserv. 2020; 44(12): e14951. doi:10.1111/jfpp.14951
CrossRef - Małgorzata Nowacka, Magdalena Dadan 1 & Urszula Tylewicz. Current Applications of Ultrasound in Fruit and Vegetables Osmotic Dehydration Processes. Sci. 2021; 11, 1269. doi: https://doi.org/10.3390/app11031269
CrossRef - Ostermeier R, Parniakov O, Töpfl S, et al. Applicability of pulsed electric field (PEF) pre-treatment for a convective two-step drying process. 2020; 9: 512.
CrossRef - Pandiselvam R, Aydar AY, Kutlu N, et al. Individual and interactive effect of ultrasound pre-treatment on drying kinetics and biochemical qualities of food: A critical review. Ultrason Sonochem. 2023; 92:106261. doi: 10.1016/j.ultsonch.2022.106261.
CrossRef - Pavkov I, Radojčin M, Stamenković Z, et al. Effects of osmotic dehydration on the hot air drying of apricot halves: Drying kinetics, mass transfer, and shrinkage. Processes. 2021; 9(2): 202. doi:10.3390/pr9020202
CrossRef - Petikirige C, et al. Stability of fructooligosaccharides in convectively dried fruits after initial osmoconcentration. Food and Bioprocess Technology 2023; 16: 2511‑ doi: 10.1007/s11947‑023‑03084‑3
CrossRef - Prithani R, Dash KK. Mass transfer modelling in ultrasound assisted osmotic dehydration of kiwi fruit. Innovative Food Science & Emerging Technologies. 2020; 102407. doi: https://doi.org/10.1016/j.ifset.2020.102407
CrossRef - Matos CMS, de Jesus MS, da Silva ADS, et al. Effect of osmotic dehydration on physico‑chemical characteristics, bioactive compounds and volatiles profile of diospyros kaki subjected to different drying methods. 2025; 14(10): 1727. doi: 10.3390/foods14101727
CrossRef - Radojˇcin M, Pavkov I, Bursa´c Kovaˇcevi´c D, et al. Effect of selected drying Methods and emerging drying intensification technologies on the quality of dried fruit: A Review. 2021; 9: 132. doi: 10.3390/pr9010132
CrossRef - Silva RV, de Souza MSM, de Souza PA. Application of osmoconvective dehydration for the technological use of banana bark. Soc. Dev. 2020; 9 (8): e643985334. doi: 10.33448/rsd-v9i8.5334
- Adeyeye SAO, Ashaolu TJ, Babu AS. Food drying: A review. Agricultural Reviews. 2022; 1(8): 10. doi: 10.18805/ag.R- 2537
CrossRef - Tan S, Miao Y, Zhou C, et al. Effects of hot air drying on drying kinetics and anthocyanin degradation of blood-flesh peach. 2022; 11: 1596.
CrossRef - Tefera AT, Srzednicki G, Craske J. Effects of osmotic dehydration on the hot air drying of apricot halves: drying kinetics, mass transfer, and shrinkage. 2021; 9 (11): 1941. doi: 10.3390/pr9111941
CrossRef - Thamkaew G, Galindo FG. Influence of pulsed and moderate electric field protocols on the reversible permeabilization and drying of thai basil leaves. Food Sci. Emerg. 2020; 64: 102430.
CrossRef - Tri Yulni, Waqif Agusta, Jayanegara A, et al. Unveiling the Influence of Osmotic Pretreatment on Dried Fruit Characteristics: A Meta-Analysis Approach. Nutr. Food Sci. 2024; 29(2):178-189. doi: https://doi.org/10.3746/pnf.2024.29.2.178
CrossRef - Tylewicz U, Mannozzi C, Castagnini JM, et al. Application of PEF and OD assisted drying for Kiwifruit waste valorisation. Innovative food science & emerging technologies. 2022; 102952. doi: https://doi.org/10.1016/j.fset.2022.102952
CrossRef - Wiktor A, Hrycak B, Jasi ´nski M, et al. Impact of atmospheric pressure microwave plasma treatment on quality of selected spices. Sci. 2020; 10: 6815.
CrossRef - Wojtyś A, Pietrzyk S, Grzesińska K, et al. Ultrasound-Assisted Osmotic Dehydration of Apples in Xylitol Solution: Effects on Kinetics, Physicochemical Properties and Antioxidant Activity. Molecules. 2025; 30(11):2304. doi: 10.3390/molecules30112304.
CrossRef - Zhou YH, Vidyarthi SK, Zhong CS, et al. Cold plasma enhances drying and colour, rehydration ratio and polyphenols of wolfberry via microstructure and ultrastructure alteration. 2020; 134: 110173.
CrossRef - Kasapoglu, M. Z. Innovative drying methods of kiwi fruit: Effects on phenolic content, antioxidant activity, and microstructural properties. Italian Journal of Food Science. 2025; 37(3): 427-436. https://doi.org/10.15586/ijfs.v37i3.2997
CrossRef
Abbreviations List
ANOVA – Analysis of Variance
MAD – Mean Absolute Deviation
MAPE – Mean Absolute Percentage Error
MSE – Mean Squared Error
OD – Osmotic dehydration
R2 – Coefficient of determination
RMSE – Root Mean Square Error
SG – Solid gain
WL – Water loss
WR – Weight reduction













