In-vivo Efficacy Study on Musa Balbasiana Seeds and Inflorescence for its Hepatoprotective Effect
1Programme of Food Nutrition and Dietetics, Assam down town University, Guwahati, India
2Programme of Biotechnology, Assam down town University, Guwahati, India
3Assam down town University, Guwahati, India
4Department of Home Science, HPB Girls College, Golaghat, India
Corresponding Author Email: ananyakashyap82@gmail.com
Download this article as:
ABSTRACT:Musa balbisiana colla is a widely grown banana having medicinal properties. Having known it holds medicinal value. This present study was done to investigate the hepatoprotective effect of Musa balbasiana seeds and flower parts on hepatocytes damage and associated alteration of antioxidant enzymes in CCl4 model induced hepatotoxicity in experimental animals. Acute hepatotoxicity was induced by intraperitonially on 11th and 12th day of the experiment Adult male Sprague Dawley (SD) rats with body weight between 200 g and 300 g were selected for the study and extracts at dose of 200mg and 400mg seed and flower were administered orally for 14 consecutive days. Hepatotoxicity was determined after 14days of CCl4 administration. The activities of various serum biomarkers aspartate aminotransferase (AST) activity, alanine aminotransferase (ALT) activity, alkaline phosphatase (ALP) activity and lactate dehydrogenase (LDH) activity, levels of enzymatic and non-enzymatic antioxidant (GSH, GST, catalase, Vitamin C, Nitrites) and lipid peroxidation level were estimated in all the rats. Our study showed decrease in all enzymatic and non-enzymatic antioxidant level and a marked increase level of lipid peroxidation in the entire CCl4 induced group compared to normal group. Supplementation of MB seed and flower for 14 days restored the antioxidant levels as well as malondialdehyde level. Increasing the dose i.e 400mg in both seeds and flower extract showed effective activities compared to 200mg administration. Also the biochemical investigations were further supported by histopathological examination. Our results suggested that the MB (Musa balbisiana seeds extract) 400 mg/kg has a significant therapeutic benefit compared to MB flower against the hepatotoxic complications by attenuating oxidative stress and lipid peroxidation. Thus MB seed can have efficacy role as hepatoprotective agent in CCl4 induced liver pathology.
KEYWORDS:Antioxidant; Enzymes; Flower; Hepatoprotective; Musa balbisiana colla; Seed; Serum level
Introduction
Acute liver injury caused by hepatotoxin is known to be one of the major causes of acquired liver pathology.1 Hepatotoxin such as CCl4, toxic dose of paracetamol is responsible for acquired liver injury causing elevation of liver enzymes such as ALT, AST, ALP.2,3 Liver being one of the important organs of gastrointestinal system is susceptible to develop chronic condition such as hepatic fibrosis, liver cirrhosis and hepatocellular carcinoma. Similar effects are caused by CCl4 the most ancient chemical product known to have hepatotoxic effect which is usually used to elucidate the mechanism of hepatotoxic effect such as fatty degeneration, fibrosis, hepatocellular death, and carcinogenicity. CCl4 is activated by cytochrome to form highly reactive trichloromethyl free radical CCl3. This radical can initiate cell damage by binding to cellular molecules such as nucleic acid, protein, lipid resulting in oxidative stress, inflammation, and fatty degeneration, cell injury also subsequently can cause hepatic cancer and cellular apoptosis.4,5 Acute liver disease can be the consequences of various etiologies. Several factors such as viral infections, alcohol abuse, toxic substance and drugs have shown to be harmful factor on liver. To intercept such conditions multidisciplinary approach can be enforced. Modern medicine has come a long way in its management; also, other form of treatment can be outlook for holistic perspective. Such approach focuses on medicinal plants which possess therapeutic properties that can contribute in the management of liver conditions. Musa balbasiana (Family: Musaceae) is a wild type species of banana grown mostly in North Eastern region of India traditionally used in folk medicine. It is a monocotyledons herb locally known as Bhimkol or Athiyakol. All parts of Musa balbasiana hold medicinal value.6 It is a rich source of phenolic compounds and secondary metabolities such as flavanoids, polyphenol, alkaloids, tannins, saponins.7 These compounds reported to possess various health benefit and also strong antioxidant potential. Extract of the seeds, flower, and peel of Musa balbisiana demonstrated antidiabetic properties in streptozotoxin STZ induced diabetic.8 Methanolic extract of Musa balbisiana infloroscenes was also reported to have antioxidative property having beneficial role in treating free radical related problems.9 The various parts of Musa balbasiana are utilised in traditional medicine to treat dysentery, pin worm infection,10 healing cough, antiseptic11 cure hepatitis.12 In the present study we aimed to study the efficacy of Musa balbisiana colla seeds extract in a mouse model of CCL4 induced liver damage for their potent therapeutic significance as hepatoprotective medicine.13
Materials and Methods
Plant material
The Musa balbisiana colla fruit and flower was collected from Guwahati market. The fruits were washed properly and seeds were taken out and flowers were washed and cleaned, later kept for drying in hot air oven at 60 for 48 hrs and then powdered with a grinder. The powder obtained was extracted using ethanol and the extract was then concentrated using magnetic stirrer with hot plate.
Experimental animals
Adult male Sprague Dawley (SD) rats (42 numbers, body weight between 200 g and 300 g) were purchased from M/s Jeeva Life Sciences (Hyderabad, India) and were housed under standard environmental condition with temperature (24 ± 2 ºC), relative humidity (55 ± 15%) with 12h:12h light and dark cycle. The animals were given an unlimited supply of purified water and rodent food. 7 days before to the start of the trial, the animals were acclimated. As directed by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Government of India, the animals were treated with care and painlessly throughout the experimental periods. The experimental protocols related to the animals were finalized by the IAEC, NIPER -Guwahati prior to the beginning of the experiment.
CCl4 induced hepatotoxicity
Carbon tetrachloride-50% v/v in corn oil (1:1) was administered intraperitonially with dose of 2 ml/kg body weight in all the groups (except normal control) to induce acute liver injury in rats. Musa balbisiana seeds and flower extract-200mg and 400mg were administered for 14 consecutive days and CCl4 was administered intraperitoneally on 11th and 12th day of the study. The body weight of every experimental animal was noted at the conclusion of the investigation (i.e on 14th day), animals were lightly anesthetized with isoflurane and blood was collected through retroorbital plexus for preparation of serum.
Protocol design
Animals were randomly distributed into five groups,
Normal control’; animals were neither administered CCl4 to induce liver injury nor administered Musa balbisiana seeds extract (n=6);
CCl4 control; animals were administered CCl4 intraperitonially on 11th and 12th day of the experiment to induce acute liver injury (n=6);
CCl4+MB-200mg; animals were provided orally with Musa balbisiana seeds extract: 200mg for 14 on sequential days and CCl4 was administered intraperitoneally on 11th and 12th day of the study (n=6);
CCl4+MB-400mg; animals received oral administration with Musa balbisiana seeds extract:400mg for 14 on subsequent days and CCl4 was administered intraperitoneally on 11th and 12th day of the study (n=6);
CCl4+Sily-100mg; animals were orally administered with silymarin100mg/kg (suspended: 1% sodium CMC) as reference agent for 14 consecutive days and CCl4 was administered intraperitoneally on 11th and 12th day of the study (n=6).
To assess the efficacy of Musa balbisiana flower extract on carbon tetrachloride (CCl4)-induced liver injury in rats, adult male Sprague Dawley rats (20 numbers, body weight between 200 g and 300 g) were randomized into four groups as discussed below:
Normal control, animals were neither administered CCl4 to induce liver injury nor administered Musa balbisianaseeds extract (n=5);
CCl4 control; animals were administered CCl4 intraperitoneally on the 11th and 12th day of the experiment to induce acute liver injury (n=5);
CCl4+MB-F-200mg; animals were given orally Musa balbisiana flower extract:200mg for 14 on sequential days and CCl4 was administered intraperitoneally on 11th and 12th day of the study (n=5);
CCl4+MB-F-400mg; animals were orally dosed with Musa balbisiana flower extract:400mg for 14 successive days and CCl4 was administered intraperitoneally on 11th and 12th day of the study (n=5);
Harvesting of liver and blood from animals
At the end of the study (i.e., on 14th day), body weight of all experimental animals was recorded, animals were lightly anesthetized with isoflurane and blood was collected through retroorbital plexus for preparation of serum. The central lobe of each liver tissue (n=3 per group) was stored in neutral buffered formalin (10 %) for histopathological studies. Rest of the liver tissues was stored in -80°C freezer for biochemical studies.
Biochemical estimation
The serum levels of lactate dehydrogenase (LDH), alkaline phosphatase (ALP), alanine aminotransferase (ALT) and aspartate aminotransferase (AST) assays was determined using standard reagent kits following the procedure available in the kit. The protein content in the sample was also evaluated using Bicinchoninic acid (BCA)assay kit. Enzymatic antioxidant namely catalase, glutathione S-transferase (GST), superoxide dismutase(SOD) were also assessed. Determination of malodialdehyde(MDA) content, nitrite content, reduced glutathione (GSH) and vitamin C levels were done.
Estimation of total protein content in liver tissue
The protein content in the sample was estimated using Bicinchoninic acid (BCA) assay kit (Sigma-Aldrich). Bovine serum albumin (BSA) was used as reference protein for plotting standard curve; Protein levels were quantified from the standard curve and reported as mg/g of liver tissue weight.
Estimation of malondialdehyde (MDA) contents
Lipid peroxidation in liver tissue was quantified as malondialdehyde (MDA) by its reaction with 2-thiobarbituric acid (TBA). A standard curve prepared from 1,1,3,3-tetramethoxypropane (TMP) was used to calculate MDA levels, expressed as nmol/g wet tissue.
Measurement of nitrite content
Nitrosative stress was assessed by determining nitrite levels in liver tissue homogenates with the Griess reagent. Sodium nitrite was used to construct a standard curve, and sample concentrations were calculated and expressed as mmol/g wet tissue.14
Estimation of GSH concentration
The principle of GSH (glutathione) assay is based on the reduction reaction, where the thiol or sulfhydryl (-SH) group of glutathione (GSH) present in the tissue homogenate reduces 5, 5’-dithiobis-(2-nitrobenzoic acid) (DTNB) to form an anion 2-nitro-5-thiobenzoate (NTB-) per mole of GSH. The formed anion gets converted into a yellow colored dianion (NTB2-) in water at neutral and alkaline pH. The amount of NTB2- produced is quantified by the spectrophotometric method by measuring the absorbance at 412 nm.15 The standard curve was prepared with commercial GSH [Sigma-Aldrich], and GSH content in liver tissue samples was calculated accordingly. Results were expressed as mmol per gram of wet tissue.
Estimation of GST activity
GST activity in tissue samples was measured using 1 mM GSH and 1 mM ‘ethanolic 1-chloro-2,4-dinitrobenzene’ (CDNB) as substrates in a 96-well plate assay. Enzyme activity was calculated using the molar extinction coefficient of ‘CDNB (0.0096 μM⁻¹ cm⁻¹)’ and expressed as ‘nmol of CDNB’ conjugated per minute per mg protein.14
Determination of catalase activity
CAT was calculated using the methodology outlined in previous research.16 The catalase activity of individual sample was calculated by the help of a standard curve, generated by utilizing catalase lyophilized powder (Sigma-Aldrich) and expressed as U/mg protein.
Determination of SOD activity
Superoxide dismutase (SOD) activity in liver tissue was evaluated using a commercial assay kit. The assay was based on a colorimetric method by recording the optical density of the reaction mixture for any color change at 440 nm, and enzyme activity was presented as percentage inhibition.
Determination of Vitamin C level
This assay is based on the copper-dependent oxidation of ascorbic to form two products, namely dehydroascorbic acid and diketogulonic acid. Commercial pure L-Ascorbic acid was used to plot of a reference curve. The concentration of vitamin C in the experimental sample was estimated from this curve of ascorbic acid and was presented as-mg/g of wet tissue.
Histological analysis of the liver
The central lobe of the isolated liver tissue (n=3 from each group) was stored in ‘10% neutral buffered formalin’ for histophatological studies. the liver tissue was cut and sectioned (5µm) and stained with ‘hematoxylin and eosin’ (H&E) to access histological alterations. Similarly, Masson’s trichrome stain was applied to identify collagen accumulation in the liver sections.
Statistical analysis
All the data are presented as mean ± SEM. The statistical significance between the two groups was determined using one-way ANOVA followed by Dunnett’s multiple comparison test’. A result of P< 0.05 is considered statistically significant.
Results
Result of MB seed and flower extract on CCl4 induced alterations in the body weight and liver weight, and liver morphology
On the first and fourteenth days of the investigation, there was no significant (p>0.05) variation in the animals’ body weights among the various experimental groups. The liver weight to body weight ratio was significantly (p<0.05) higher in the CCl4 control group than in the normal control group. MB seed administration at both dosages (200 mg/kg and 400 mg/kg) did not, however, significantly vary (p>0.05) from the group that received CCl4 alone. Similarly, silymarin (100 mg/kg) treatment also did not show notable difference (p> 0.05) compared to the CCl4 alone administered group. Whereas there was no any significant alteration among the groups. No considerable change in the liver weight among the CCl4 induced and MB flower extract treated groups was observed.
Effect of MB seed extract on CCl4 induced changes in total protein contents
There was no significant (p> 0.05) change in the total protein content in the liver tissue of animals among the different experimental groups (Figure 1).
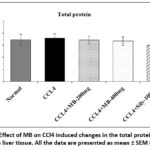 |
Figure 1: Effect of MB on CCl4 induced changes in the total protein content in the liver tissue. All the data are presented as mean ± SEM (n=6). |
Effect of MB seed and flower extract on CCl4- induced changes hepatic biomarkers
The serum concentrations of liver injury biomarkers, AST, LDH, ALT, and ALP levels were estimated to find out the effect of MB on CCl4 induced acute liver necrosis (Figure 2). Levels of AST (Figure 2A, 2A) and ALT (Figure 2B,2B) were significantly higher in the serum of ‘CCl4’ alone administered group in contrast to the normal’ control rats. Administration of MB seeds at 400 mg/kg significantly decreased the AST and ALT levels compared to the CCl4 alone administered group. Treatment with MB flower extract (at both the doses 200 mg/kg and 400 mg/kg) significantly (p< 0.001) decreased the ALT level compared to the CCl4 alone administered group. No statistically significant change in the serum levels of AST activity compared to the CCl4 alone administered group was observed in MB flower extract-400mg. In the present study, there was no significant difference(p> 0.05) in the ALP activity as observed in the serum of normal control, CCl4 control and treatment (MB and silymarin) groups in both seed and flower administration (Figure 2C, 3C). Accordingly, the LDH activity in the CCl4 alone administered group was significantly (p< 0.01) increased compared to that of normal control group (Figure D). Treatment with MB seeds at both doses, the activity of LDH was considerably reduced (p<0.05 at 200 mg/kg and p<0.01 at 400 mg/kg) in comparison to the group that received CCl4 alone. When compared to the group that received CCl4 alone, treatment with silymarin (100 mg/kg) also significant (p<0.05) reduced the activity of LDH.
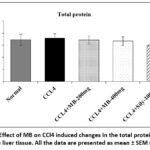 |
Figure 2: Result shows MB seed effect on CCl4- induced changes in the (A) AST activity, (B) ALT activity, (C) ALP activity and (D) LDH activity. |
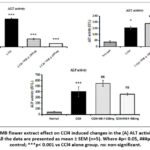 |
Figure 3: Result of MB flower extract effect on CCl4 induced changes in the (A) ALT activity, (B) AST activity, (C) ALP activity. |
Effect of MB seed and MB flower extract on CCl4 induced alterations in liver histology
Liver damage was assessed by H&E staining. Figure 4 displays the representative H&E staining images. The normal control’s photomicrographs revealed hepatocellular histology and an undamaged liver. In CCl4 alone group, liver tissue sections showed extensive hepatocellular degeneration, moderate bile duct hyperplasia, micro and macro cytoplasmic vacuolations and multifocal necrosis with cellular debris; portal areas showed marked chronic inflammatory cells infiltration, loss of architecture and prefibrotic lesions. In CCl4 +MBseeds-200 mg treatment group, liver tissue section showed mild micro and macro cytoplasmic vacuolations, mild bile duct hyperplasia showing approximately 40% protection against CCl4 induced pathological lesions. In CCl4+MB-400mg treatment group, liver tissue section showed minimal micro and macro cytoplasmic vacuolations, minimal bile duct hyperplasia, and minimal multifocal chronic inflammatory cells infiltration showing approximately 55% protection against CCl4 induced pathological lesions. The CCl4 +Silymarin-100mg group showed moderate micro and macro cytoplasmic vacuolations and approximately 62% protection against CCl4 induced pathological lesions.
Similarly, in CCl4 +MB-F-200 mg treatment group, tissue section showed micro and macro cytoplasmic vacuolations with mild protection (10%) compared CCl4 induced pathological lesions. In CCl4+MB-F-400mg treatment group, tissue section showed minimal micro and macro cytoplasmic vacuolations showing moderate protection (20-30%) compared to CCl4 induced pathological lesions.
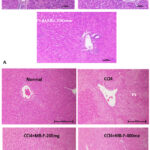 |
Figure 4: (a-b). Hematoxylin and Eosin-stained photomicrographs of liver tissue sections (H&E). Histopathological analysis of the control group’s liver sections. a, b H&E-stained microscopic image of a experimental animal liver tissue from the healthy control group and CCl4 control group. |
Effect of MB seed extract on CCl4 induced accumulation of collagen
Collagen accumulation in the liver tissue was examined using Masson trichrome staining. Figure 5 shows the representative Masson trichrome staining images. Light micrographs from the normal control showed normal collagen distribution around the central vein. In CCl4 group, liver tissue sections showed extensive prefibrotic lesions and multifocal collagen deposition. In CCl4+MB-200 mg group, tissue sections showed moderate prefibrotic lesions and multifocal collagen deposition around the central vein. In CCl4+MB-400 mg, tissue section showed minimal prefibrotic lesions and minimal collagen deposition around the central vein and hepatic parenchyma. In CCl4 +Silymarin-100 mg group, tissue section showed minimal collagen deposition around the central vein and hepatic parenchyma.
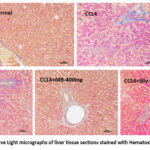 |
Figure 5: Representative Light micrographs of liver tissue sections stained with Hematoxylin and Eosin (H & E). |
Effect of MB seed and flower extract, on CCl4 induced hepatic lipid peroxidative damgae
Malondialdehyde,‘a product of lipid hydroperoxides breakdown, which is considered as an index/measure of lipid peroxidative damage’. Lipid peroxidation was detected by estimating the concentration of MDA in the liver (Figure 9 and 10). In the CCl4 alone administered group, the content of MDA was comparatively (p< 0.01) increased compared to those in the normal control group. The MDA level was significantly (p<0.01) lower in the MB seeds at 400 mg/kg treatment group than in the CCl4 alone administered group, whereas the administration of MB seeds at 200 mg/kg did not show a significant (p>0.05) difference.
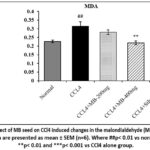 |
Figure 6: Representative Light micrographs of liver tissue sections stained with Hematoxylin and Eosin (H & E). |
Effect of MB seed and flower extract on CCl4 induced changes in nitrite levels in the liver.
The concentration of nitrite was moderately decreased in the CCl4 administered group compared to the normal control group. The inclusion of silymarin (100 mg/kg) (p<0.01) and MB seeds (200 mg/kg and 400 mg/kg) (p<0.05) effectively reversed the nitrite levels’ decrease caused by CCl4 as compared to the group that received CCl4 alone (Figure 7).
In the CCl4control group, nitrite levels were significantly elevated (p < 0.05) relative to the ‘normal control group’ (Figure 8). Treatment with MB flower extract (200 mg/kg) markedly reduced nitrite levels relative to the CCl4 control group, while a moderate reduction was observed in the 400 mg/kg MB-F treated group.
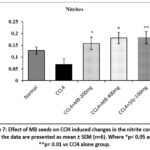 |
Figure 7: Effect of MB seeds on CCl4 induced changes in the nitrite content. All the data are presented as mean ± SEM (n=6). Where *p< 0.05 and **p< 0.01 vs CCl4 alone group. |
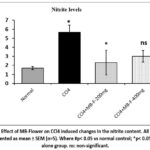 |
Figure 8: Effect of MB-Flower on CCl4 induced changes in the nitrite content. All the data are presented as mean ± SEM (n=5). Where #p< 0.05 vs normal control; *p< 0.05 vs CCl4 alone group. ns: non-significant. |
Result of MB seed and flower extract effect on CCl4 induced changes in non-enzymatic antioxidants in the liver
Role of MB seed extract on Vit C contents
The content of vitamin C in the liver tissue was seen to be significantly (p< 0.001) decreased in the CCl4 alone administered ‘animal group’ compared to the ‘normal control group’. Administration with MB seeds (400 mg/kg) and silymarin (100 mg/kg) markedly (p< 0.001) restored the vitamin C level relative to CCl4 alone received group. In contrast, MB seed extract at 200 mg/kg did not show a significant improvement (p > 0.05) in vitamin C content compared with the CCl4-treated group (Figure 9).
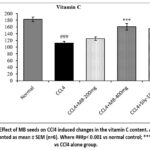 |
Figue 9: Effect of MB seeds on CCl4 induced changes in the vitamin C content. All the data are presented as mean ± SEM (n=6). Where ###p< 0.001 vs normal control; ***p< 0.001 vs CCl4 alone group. |
Effect of MB seed and flower extract on reduced glutathione (GSH) contents
The GSH levels in the liver tissue was comparatively (p< 0.001) decreased in the CCl4 alone infusion group compared to the normal control group. Treatment with MB (400 mg/kg) (p< 0.01) and silymarin (p< 0.001) and MB flower extract (MB-200mg) significantly restored the GSH content in relation to CCl4 alone received group (Fig 10&11). However, treatment with MB seeds (200 mg/kg) did not produce a significant (p> 0.05) difference in the GSH level in relation to the CCl4 alone administered group whereas a moderate elevation (p> 0.05) in the GSH levels were increased in MB-F (400 mg/kg) treated animals.
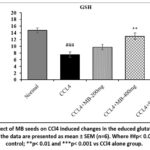 |
Figure 10: Effect of MB seeds on CCl4 induced changes in the educed glutathione (GSH) content. All the data are presented as mean ± SEM (n=6). Where ##p< 0.01 vs normal control; **p< 0.01 and ***p< 0.001 vs CCl4 alone group. |
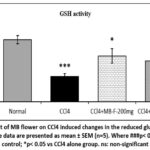 |
Figure 11: Effect of MB flower on CCl4 induced changes in the reduced glutathione (GSH) content. All the data are presented as mean ± SEM (n=5). Where ###p< 0.001 vs normal control; *p< 0.05 vs CCl4 alone group. ns: non-significant. |
Effect of MB seed extract on CCl4 triggered alterations in enzymatic antioxidants in the hepatic tissue
Ameliorative action of MB seed and flower extract on catalase activity
A significant reduction (p< 0.001) in the liver catalase activity was detected in CCl4 only administered group compared to the normal control group. Treatment with MB (400 mg/kg) and silymarin (100 mg/kg) significantly (p< 0.05 for MB 400 mg/kg and p< 0.01 for silymarin 100mg/kg) restored the catalase activity compared to CCl4 alone administered group. However, curation with MB seeds (200 mg/kg) remains no significance (p> 0.05) variation in the catalase activity comparatively to the CCl4 only administered group (Figure 12).
Whereas a marked (p< 0.01) reduction in the catalase activity was obtained in the CCl4 only treated rats when compared to the ‘normal control group’ (Figure 13). Inclusion of MB flower extract at both doses (MB-F-200 mg/kg and MB-F-400 mg/kg) markably (p< 0.05) recovered the catalase action compared to the CCl4 control group.
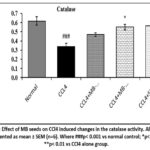 |
Figure 12: Effect of MB seeds on CCl4 induced changes in the catalase activity. All the data are presented as mean ± SEM (n=6). Where ###p< 0.001 vs normal control; *p< 0.05 and **p< 0.01 vs CCl4 alone group. |
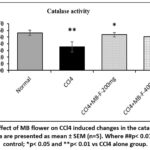 |
Figure 13: Effect of MB flower on CCl4 induced changes in the catalase activity. All the data are presented as mean ± SEM (n=5). Where ##p< 0.01 vs normal control; *p< 0.05 and **p< 0.01 vs CCl4 alone group. |
Effect of MB seed extract on GST activity
The activity of GST was significantly (p< 0.05) decreased in CCl4 alone administered group compared to the normal control group. In comparison to the CCl4 only administered group inclusion with MB seed (400 mg/kg) significantly (p< 0.05) restored the GST activity. Treatment with MB seed (200 mg/kg) and silymarin did not produce a significant (p> 0.05) difference in the GST activity compared to CCl4 alone administered group (Figure 14).
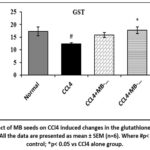 |
Figure 14: Effect of MB seeds on CCl4 induced changes in the glutathione S-transferase (GST) activity. All the data are presented as mean ± SEM (n=6). Where #p< 0.05 vs normal control; *p< 0.05 vs CCl4 alone group. |
Effect of MB seed and flower extract on SOD activity
Liver tissue SOD activity in the CCl4 alone administered group showed significant (p< 0.01) reduction than the activity of normal control group. Treatment with MB seeds (400 mg/kg) and silymarin resulted in a significant (p< 0.05) elevation in the SOD levels compared to CCl4 alone administered group. No significantly alteration in SOD level was seen when treated with MB seeds (200 mg/kg) unlike CCl4 only administered group (Figure 15). Similarly, the SOD activity was significantly decreased in the CCl4 control group as comparing with normal control group (Figure 16). Provision of MB flower extract at both doses (MB-F-200 mg/kg and MB-F-400 mg/kg) significantly (p< 0.05) recovered the SOD activity compared to the CCl4 control group.
 |
Figure 15: Effect of MB seeds on CCl4 induced changes in the superoxide dismutase (SOD) activity. All the data are presented as mean ± SEM (n=6). Where ##p< 0.01 vs normal control; *p< 0.05 vs CCl4 alone group. |
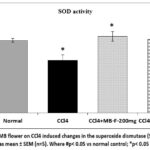 |
Figure 16: Effect of MB flower on CCl4 induced changes in the superoxide dismutase (SOD) activity. All the data are presented as mean ± SEM (n=5). Where #p< 0.05 vs normal control; *p< 0.05 vs CCl4 alone group. |
Discussion
The hepatoprotective activity of MB (Musa balbasiana seeds and flower extract) on CCl4-induced liver injury17,18 was determined by assessing the levels enzymes of liver injury biomarkers, AST, ALT, and LDH activities and the histopathology of liver tissue. One non-specific indicator of tissue turnover, a typical metabolic process, is LDH. It is present in all tissues and acts as a key indicator of tissue damage.19 The level of serum alanine aminotransferase (ALT) level is considered as sensitive and specific indicator of liver cell injury.20 In our study CCl4-induced hepatotoxicity was indicated by marked increase in AST, ALT and LDH activities and also hepatocellular degeneration of liver tissues. Increase in serum level of liver indicates injury to hepatocytes.21 Out of two doses (MB 200 mg/kg and 400 mg/kg), treatment with MB seeds and flower at 400 mg/kg significantly ameliorated the CCl4-induced increase in serum levels of liver injury biomarkers (AST, ALT and LDH) and restored the liver histology compared to the CCl4 alone administered group. Such protective activity of MB seeds and flowers has similar outcomes with literature reported by22 where Phoenix dactylifera L. seed showed improvement against CCl4 induced alterations in liver function parameters.
The levels of non-enzymatic antioxidants, and the activities of enzymatic antioxidants, GST, catalase, and SOD are essential components of our antioxidant defense system, and oxidative stress is indicated by any alterations in their cellular level.23 To analyse the antioxidant defence responses after CCl4 and MB administration, we determined the levels and activities of vitamin C, GSH, GST, catalase, and SOD activity in the liver tissue.GSH level is important to understand oxidative stress, apoptosis, and cell death. GSH is an important antioxidant and an indicator of oxidation by free radicals.24 CCl4 in our study indicated reduction in GSH content which indicates formation of oxidative stress. Vitamin C has a strong reducing ability and plays a vital role as a coenzyme in internal hydroxylation reactions. It serves as an antioxidant and reduces the risk of developing various chronic diseases.25 Glutathione S-transferases (GSTs) detoxify the endogenous and xenobiotic compounds which protect the body from numerous chronic diseases associated with oxidative stress. GST is encoded by a large gene family and is mostly found in mammalian tissues including the liver.26 The protective role of catalase activity can be understood as it catalyzes the decomposition of H2O2 into water and oxygen. It protects cells from free radical causing oxidative injury.27 Superoxide dismutase (SOD) plays a key role in cellular defense by facilitating the dismutation of superoxide radicals yielding oxygen and hydrogen peroxide.28
Liver toxicity is associated with nercrosis and significantly depletion of non- enzymatic antioxidants and a marked increase production of malaondialdehyde.29 In our study CCl4 treatment significantly decreased the potential of these antioxidant defence enzymes in the liver tissue compared to the normal control animals. The CCl₄-alone group showed a substantial increase in hepatic malondialdehyde (MDA) levels, a crucial indicator of lipid peroxidation and oxidative stress30 resulting from lipid hydroperoxide breakdown. Thus oxidative damage occurred in liver can be treated by restoring the activities of these enzymes. Our experimental study comes in line with this activity when treated with CCL4 seeds and flower-400mg the antioxidant defence enymes were restored which was determined by increased level of this enzymes compared to CCL4 alone and the lipid peroxidation were decreased. The involvement of nitric oxide in hepatic injury continues to be disputed. The excess generation of nitric oxide indicates the nitrosative stress, while nitric oxide also required in maintaining liver haemodynamic function.31 Nitrites are stable end-product of nitric oxide metabolism. The liver haemodynamic was disturbed after supplementation of CCL4 which was indicated by marked reduction in nitrite concentration. CCl4 disruption in antioxidant enzymes activities and caused lipid peroxidation in liver tissue has been reported in various literatures.32
Literature confirms that CCl4 induces hepatotoxicity in experimental animals.33 CCl4 induced cause various histological changes such as fatty change, fibrosis, necrosis.34 In this study, we performed H & E staining to access liver injury in a different group of animals. H & E staining is the most widely used diagnostic method in histopathology alteration in the liver tissue. The result of our study has similar hepatocellular histological changes where CCL4 induced showed extensive hepatocellular degeneration, multifocal necrosis, inflammatory cells and prefibrotic lesions. These alterations were depleted after the supply of MB seeds and flower-200mg and MBseeds and flower-400mg in treated liver which showed reduced liver fibrosis, minimal micro and macro cytoplasmic vacoulation, minimal chronic inflammatory cells.35 reported such protective effect on CCL4 causative liver damage where they demonstrated the improvement in histological changes after supplementation of Corrigiolatelephiifolia root methanolic extract in rats.
Conclusion
Findings from the present study provide clear evidence supporting the occurrence of oxidative stress in acute liver injury in CCl4 administered rats. The present findings confirm that liver antioxidants enzymes were significantly restored in liver tissue in MB (Musa balbisiana seeds and flower extract) 400 mg/kg treated animals. Out of the two doses selected, MB 400 mg/kg had superior activity compared to the MB 200 mg/kg. The finding of our study shows potential role of MB seeds and flower in protecting liver against carbon tetrachloride induced oxidative damage in rats and its inhibitory actions can be potent hepatoprotective agent.
Acknowledgement
The authors gratefully acknowledge the Department of Biotechnology (DBT), Government of India, for providing financial support. We also extend our sincere thanks to Assam down town University and NIPER-Guwahati for providing laboratory facilities and institutional support during the course of this study.
Funding Sources
Funding Agency: Department of bio technology, Govt. of India; Grant No: BICL/NER-BPMC/2018/245.
Conflicts of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
Institute animal ethics committee (IAEC), NIPER Guwahati.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Daisy Sharma: Conceptualization, Investigation, Writing – Original Draft, Funding Acquisition
- Pameena Narzary: Data Curation, Writing – Review and Editing, Validation
- Manash Pratim Sarma: Methodology, Formal Analysis, Visualization
- Ananya Kashyap: Resources, Supervision, Project Administration
- Yahia Mazumdar: Review and Editing
References
- Parthasarathy M, & Evan Prince S. The potential effect of phytochemicals and herbal plant remedies for treating drug-induced hepatotoxicity: a review. Molecular Biology Reports. 2021; 48: 4767–4788 . DOI: https://doi.org/10.1007/s11033-021-06444-4
CrossRef - Li J., Chiew A. L., Isbister G. K., et al. Sulfate conjugation may be the key to hepatotoxicity in paracetamol overdose. British Journal of Clinical Pharmacology. 2021 ;87(5): 2392-2396. DOI: https://doi.org/10.1111/bcp.14642
CrossRef - Barghi M, Ashrafi M, Aminlari M, et al. The protective effect of Zataria multiflora Boiss essential oil on CCl₄-induced liver fibrosis in rats. Drug Chem Toxicol. 2021;44(3):229-237. doi:10.1080/01480545.2019.1571502
CrossRef - Weber L. W. D., Boll M., & Stampfl A. Hepatotoxicity and Mechanism of Action of Haloalkanes: Carbon Tetrachloride as a Toxicological Model. Critical Reviews in Toxicology. 2003; 33(2): 105–136. DOI: https://doi.org/10.1080/713611034
CrossRef - Bhakuni G.S., Bedi O., Bariwal J. et al. Animal models of hepatotoxicity. Research. 2016; 65:13–24. DOI: https://doi.org/10.1007/s00011-015-0883-0
CrossRef - Borborah K, Borthakur SK, Tanti B. Musa balbisiana Colla—taxonomy, traditional knowledge, and economic potentialities of the plant in Assam, India. Indian J Tradit Knowl. 2016;15(1):116-120.
- Borah M., & Das S. Antidiabetic, antihyperlipidemic, and antioxidant activities of Musa balbisiana Colla. in Type 1 diabetic rats. Indian Journal of Pharmacology. 2017; 49(1): 71-76. DOI: 10.4103/0253-7613.201030
CrossRef - Gopalan G., Prabha B., Joe A., et al. Screening of Musa balbisiana Colla. seeds for antidiabetic properties and isolation of apiforol, a potential lead, with antidiabetic activity. Journal of the Science of Food and Agriculture. 2019 ;99(5): 2521-2529. DOI: https://doi.org/10.1002/jsfa.9462
CrossRef - Borborah K, Borthakur SK, Tanti B. Musa balbisiana Colla: taxonomy, traditional knowledge, and economic potentialities of the plant in Assam, India. Indian J Tradit Knowl. 2016;15(1):116-120.
- Kalita D., & Deb B. Some folk medicines used by the Sonowal Kacharis tribe of the Brahmaputra valley, Assam. Natural Product Radiance. 2004; 3(40): 240-245.
- Kalita P, Kander CC. Kolakhar—a traditional herbal soda of Assam. J Adv Pharm Res Biosci. 2014;2(5):122-123.
- Borthakur S. K., Choudhury B. T., & Gogoi R. Folklore hepato-protective herbal recipes from Assam in Northeast India. Ethnobotany. 2004; 16:76-82.
- Ohkawa H., Ohishi N., & Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical biochemistry. 1979;95(2): 351-358. DOI: https://doi.org/10.1016/0003-2697(79)90738-3
CrossRef - Sahu B. D., Rentam K. K. R., Putcha U. K., et al.Carnosic acid attenuates renal injury in an experimental model of rat cisplatin-induced nephrotoxicity. Food and Chemical Toxicology. 2011;49(12): 3090-3097. DOI: https://doi.org/10.1016/j.fct.2011.08.018
CrossRef - Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys. 1959;82(1):70-77.
CrossRef - Aebi H. Catalase in Methods of Enzymatic Analysis. 2nd ed. Vol 2. New York, NY: Academic Press; 1974:673-684. doi:10.1016/B978-0-12-091302-2.50032-3
CrossRef - Elbakry MA, El Rabey HA, Elremaly W, et al. The methanolic extract of Moringa oleifera attenuates CCl₄-induced hepato-nephrotoxicity in male rats. Biomed Res. 2019;30(1):23-31.
CrossRef - Geetha S., Jayamurthy P., Pal K., et al. Hepatoprotective effects of sea buckthorn (Hippophaerhamnoides L.) against carbon tetrachloride induced liver injury in rats. Journal of the Science of Food and Agriculture. 2008; 88(9): 1592-1597. DOI: https://doi.org/10.1002/jsfa.3255
CrossRef - Farhana A, Lappin SL. Biochemistry, lactate dehydrogenase. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2023.
- Ozer J., Ratner M., Shaw M., et al The current state of serum biomarkers of hepatotoxicity. Toxicology. 2008; 245(3): 194-205. DOI: https://doi.org/10.1016/j.tox.2007.11.021
CrossRef - Robles-Díaz , Medina-Caliz I., Stephens C., et al Biomarkers in DILI: one more step forward. Frontiers in pharmacology. 2016 ;7: 267. DOI: https://doi.org/10.3389/fphar.2016.00267
CrossRef - Abdelaziz D. H., & Ali S. A. The protective effect of Phoenix dactylifera L. seeds against CCl4-induced hepatotoxicity in rats. Journal of ethnopharmacology. 2014;155(1): 736-743. DOI: https://doi.org/10.1016/j.jep.2014.06.026
CrossRef - Al-Dbass A. M., Al-Daihan S. K., & Bhat R. S. Agaricus blazeiMurill as an efficient hepatoprotective and antioxidant agent against CCl4-induced liver injury in rats. Saudi journal of biological sciences. 2012; 19(3): 303-309. DOI: https://doi.org/10.1016/j.sjbs.2012.03.004
CrossRef - Kaur P., Yousuf S., Ansari M. A., et al. Tellurium-induced dose-dependent impairment of antioxidant status. Biological trace element research. 2003;94(3): 247-258. DOI: https://doi.org/10.1385/BTER:94:3:247
CrossRef - Traber M. G., & Stevens J. F. Vitamins C and E: beneficial effects from a mechanistic perspective. Free Radical Biology and Medicine. 2011;51(5): 1000-1013. DOI: https://doi.org/10.1016/j.freeradbiomed.2011.05.017
CrossRef - Dasari S, Ganjayi MS, Oruganti L, et al. Glutathione S-transferases detoxify endogenous and exogenous toxic agents: a mini-review. J Dairy Vet Anim Res. 2017;5(5):00154.
CrossRef - Chelikani P., Ramana T., & Radhakrishnan T. M. Catalase: a repertoire of unusual features. Indian Journal of Clinical Biochemistry. 2005; 20 (2) : 131-135. DOI: https://doi.org/10.1007/BF02867412
CrossRef - Gottfredsen RH, Larsen UG, Enghild JJ, et al. Hydrogen peroxide induces modifications of human extracellular superoxide dismutase that result in enzyme inhibition. Redox Biol. 2013;1(1):24-31.
CrossRef - Badawi M. A study on the antioxidant activity of rosmarinic acid against carbon tetrachloride–induced liver toxicity in adult male albino rats. Int J Morphol. 2022;3(4):1.
CrossRef - Całyniuk B, Grochowska-Niedworok E, Walkiewicz KW, et al. Malondialdehyde (MDA), a product of lipid peroxidation, as a marker of homeostasis disorders and aging. Ann Acad Med Siles. 2016;70:224-228.
CrossRef - Rassaf T., Ferdinandy P., & Schulz R. Nitrite in organ protection. British journal of pharmacology. 2014;171(1); 1-11. DOI: https://doi.org/10.1111/bph.12291
CrossRef - Alkinani K. B., Ali E. M., Al-Shaikh T. M. et al. Hepatoprotective Effects of (−) Epicatechin in CCl4-Induced Toxicity Model Are Mediated via Modulation of Oxidative Stress Markers in Rats. Evidence-Based Complementary and Alternative Medicine. 2021; 2021(1) : 4655150. DOI: https://doi.org/10.1155/2021/4655150
CrossRef - Zhan, Y. Y., Wang, J. H., Tian, X. et al. Protective effects of seed melon extract on CCl4-induced hepatic fibrosis in mice. Journal of ethnopharmacology. 2016;193: 531-537.DOI: https://doi.org/10.1016/j.jep.2016.10.006
CrossRef - Kim S. H., Cheon H. J., Yun N., et al. Protective effect of a mixture of Aloe vera and Silybum marianum against carbon tetrachloride–induced acute hepatotoxicity and liver fibrosis. Journal of pharmacological sciences. 2009;109(1): 119-127.DOI: https://doi.org/10.1254/jphs.08189FP
CrossRef - Doudach L, El Omari N, Mrabti HN. Hepatoprotective effect of Corrigiola telephiifolia Pourr. root methanolic extract against CCl₄-induced hepatic damage in mice. Biointerface Res Appl Chem. 2021;12(2):2489-2502. doi:10.33263/BRIAC122.24892502
CrossRef
Abbreviations List
2-TBA – 2-thiobarbituric acid
ALT – Amino transferase
ALP – Alkaline phosphatase
AST – Aspartate Amino transferase
BCA – Bicinchoninic acid
BSA – Bovine serum albumin
CAT – Catalase
CCl₄ – Carbon tetrachloride
CDNB – Ethanolic 1-chloro-2,4-dinitrobenzene
CPCSEA – Committee for the Purpose of Control and Supervision of Experiments on Animals
DTNB – 5,5’-dithiobis-(2-nitrobenzoic acid)
GSH – Glutathione
GST – Glutathione S-transferase
IAEC – Institutional Animal Ethics Committee
LDH – Lactate dehydrogenase
MDA – Malodialdehyde
MB – Musa balbisiana
NIPER – National Institute of Pharmaceutical Education and Research
NTB – Anion 2-nitro-5-thiobenzoate
SD – Sprague Dawley
SH – Sulfhydryl
SOD – Superoxide dismutase
STZ – Streptozotoxin
TMP – Tetramethoxypropane













