Combined Postharvest Use of Melatonin and Salicylic Acid Extended Shelf Life of Giant Kew Pineapples
Department of Horticulture, Aromatic and Medicinal Plants, Mizoram University, Aizawl, India
Corresponding Author Email: debashismandal1982@gmail.com
Download this article as:
ABSTRACT:Pineapple is an excellent, juicy, nutrient-rich tropical fruit of Bromeliaceae family; generally, has a short shelf life and undergoes significant loss during post-harvest handling. Therefore, the research trial was conducted with two proven plant growth regulators viz. melatonin and salicylic acid, which individually been used for controlling ripening and delaying senescence in many fruits, however, having scanty information on combined application and implication in shelf-life management of pineapple. Thus, the present study involved nine postharvest treatments viz. T1: Melatonin (MT) @ 0.1 mM, T2: MT@ 0.5 mM, T3: Salicylic Acid (SA) @ 5 mM, T4: SA@ 9 mM, T5: MT@ 0.1 mM+ SA@ 5mM, T6: MT@ 0.1 mM+ SA@ 9mM, T7: MT@ 0.5 mM+ SA@ 5mM, T8: MT@ 0.5 mM+ SA@ 9mM, T9: Control ( Sterile water dipped) with three replications following complete randomized design to evaluate the effectiveness on shelf life and quality of pineapple (cv. Giant Kew) fruits during storage at ambient condition (average temperature: 22-270C and relative humidity 60-80%). Results revealed that fruits treated with MT@ 0.5 mM+ SA@ 9mM (T8) had considerably low weight loss (14.58%) with high fruit firmness (59.84 Ncm-2), good crown condition (score: 2.00 ), appealing skin colour (L: 85.78, a: 8.22, b: 30.96) with high TSS (15.59 0Brix), retention of ascorbic acid (15.34 mg 100g-1), flavonoids (4.21 mg QE 100g-1) and antioxidant activity (58.49 % inhibition DPPH) at 12DAS. This treatment had controlled the ethylene gas release (10.78 µl kg-1 h-1) and found to control fruit decay (16.67%) with maximum shelf life (16.33 days). Thus, present study concluded that combined application of MT@ 0.5 mM+ SA@ 9mM can be a potential postharvest treatment for shelf-life extension and quality maintenance in ambiently stored pineapple.
KEYWORDS:Antioxidant; Ethylene; Melatonin; Pineapple; Salicylic acid; Shelf life
Introduction
Pineapples are attractive, refreshing, rich in vitamins, minerals, organic acids, sugars, fiber and array of phytochemicals and economically important tropical fruit of Bromeliaceae family, profusely cultivated in countries like Indonesia, Costa Rica, Philippines, China, Brazil, India, Nigeria, Thailand, Colombia and Mexico etc.1 Pineapples are excellent dessert fruit and are importantly used in juice and other processing industries. Pineapple is a unique non-climacteric fruit which undergoes change in skin colour even after harvest,2 however, during ambient condition fruits rapidly lost surface moisture and face desiccation.3 Besides, ambient storage of pineapple accelerates respiration and other ripening induced metabolic changes and cause drop in soluble solids, sugars and firmness.2 On the other hand, cold storage of pineapple is also challenging as to contribute higher internal browning and chilling injury.4 Rather, controlled atmospheric storage at around 10-120C had reported to better maintain the fruit shelf life.5 But India and other developing countries importantly engaged in pineapple growing, frequently maintain the grower to consumer supply chain in ambient condition, where fruits use to face high spoilage and short shelf life.6
Melatonin, which was first isolated from the pineal glands of animal in 1958, later was identified and isolated from plants too, in 1995.7 Melatonin is a natural product, work as a signaling molecule derived from tryptophan, known as N-acetyl-5-methoxy tryptamine.8 It is having multiple responses in plants which includes different growth and developmental regulations, increment in stress tolerance and antioxidant enzymatic activity; trapping of reactive oxygen species and repair of oxidized protein, thus regulating delaying of senescence.9 Besides, it also impact endogenous activity of abscisic acid, indole acetic acid, ethylene and help in regulation of post-harvest ripening and storage.10 Melatonin is reported to delay ripening in banana,11 plum,12 mango,10 and delayed senescence in pear,13 grapes,14 citrus;15 both in climacteric and non-climacteric fruits. Moreover, reduction in post-harvest disease occurrence was reported in strawberries,16 peaches,17 and kiwifruit etc.18
Salicylic acid which is an endogenous phenolic compound, was naturally isolated from willow bark19 and act as plant growth regulator for stress regulation, modulation of respiration, photosynthesis, transpiration, ripening and senescence.20 It helps in suppressing ethylene production, influence antioxidant enzymes, maintain fruit firmness and increase disease resistance capacity.21 It is reported to use for increasing postharvest shelf life in banana,22 orange,23 grape,24 lemon25 etc. by delaying senescence and controlling postharvest diseases.
Pineapple fruits treated with melatonin have been reported to extend shelf life while maintaining physico-chemical qualities.26 Besides, postharvest use of salicylic acid was reported for influencing quality of stored pineapple while delaying senescence.27 However, combined use of both signal molecules i.e. melatonin and salicylic acid is not yet researched for evaluating impact on storage of pineapple at ambient condition. Thus, this research trial was attempted for evaluating the impact of melatonin and salicylic acid on shelf life and postharvest quality of pineapple (cv. Giant Kew) during storage at ambient condition.
Materials and Methods
Fruit source and treatments
Fully mature, green-coloured pineapple fruits cv. Giant Kew were harvested along with crown; individually with sickle, from the farmer’s field of Sialhawk village, Champhai district, Mizoram and brought to the Post-Harvest Technology Laboratory, Department of Horticulture, Aromatic and Medicinal Plants, Mizoram University, Aizawl, India for further treatments and research uses. Best quality fruits of uniform maturity, free from blemishes, diseases, pests or decayed; were selected for the treatments. Fruits were then washed thoroughly in running tap water and air dried at ambient condition followed by running through UV-C (λ=254 nm; 1.6 kJ m-2) conveyor disinfection unit (Elixir Technologies, Bengaluru, India) for 60 seconds.
Nine different treatments i.e. T1: Melatonin (MT, 98% TLC; Sigma Aldrich) @ 0.1 mM, T2: MT@ 0.5 mM, T3: Salicylic Acid (SA, 99% ACS; Sigma Aldrich) @ 5 mM, T4: SA@ 9 mM, T5: MT@ 0.1 mM+ SA@ 5mM, T6: MT@ 0.1 mM+ SA@ 9mM, T7: MT@ 0.5 mM+ SA@ 5mM, T8: MT@ 0.5 mM+ SA@ 9mM, T9: Control ( Sterile water dipped) were used in the present study with three replications. Fruits for melatonin treatments were dipped in MT solutions (at two concentrations viz. 0.1 mM and 0.5 mM, respectively) for 10 minutes, whereas for salicylic acid treatments fruits were immersed in SA solution of two different concentrations i.e. 5 mM and 9mM for 10 minutes, respectively. For combined treatments fruits were initially dipped in respective MT solutions followed by air drying with handheld air blaster and subsequently dipping in SA solutions, while fruits under control dipped for 10 minutes in water and air dried. All the fruits were stored in ambient condition (average temperature: 22-270C and relative humidity 60-80%) at the laboratory and different physical and biochemical parameters were recorded at 4 days interval.
Determination of fruit physical parameters
Fruit weight was determined by digital balance (Kern PCB, Model: Z742836, Germany) and fruit length and diameter were measured with digital slide caliper (Mitutoyo, Model: 500-505-10, Japan) and physiological weight loss (PLW) and percentage decrease in fruit length (PDFL) and diameter (PDFD) was calculated based on the given formula:
![]()
Where, FW= Fruit weight, FL= Fruit length, FD= Fruit diameter.
Firmness of the fruit was measured (at apical, mid and basal region of fruit) with digital handheld penetrometer (PCE Instruments, Model: PCE-PTR 200N, UK); whereas fruit external skin colour and flesh colour was measured in CIELAB [L* (lightness), a* (red-green), b* (yellow-blue)] with digital handheld colour meter (Konica Minolta, Singapore); colour chart was developed with corresponding L,a,b value using NIX Color Sensor software. Visual score for pineapple crown condition and flesh translucency were determined by using the score chart;6 for crown condition, a) good, fresh and green: 1; b) good with slightly yellow at tip: 2; c) moderate, dry tips and yellowing: 3; d) Bad, dry tips and more yellowing: 4 and e) severe yellow: 5, whereas for flesh translucency, a) 100% opaque:1; b) opaque with slight translucent (less than 50%): 2; c) opaque with moderate translucent (more than 50%): 3 and d) 100% translucent: 4. Carbon-di-oxide (CO2) gas was measured for respiration rate of the stored pineapple with the help of handheld digital gas analyzer (Q check, Model: E2M316, Elixir Technologies, Bengaluru, India) whereas, ethylene (C2H4) gas released from the stored fruit was measured with handheld portable ethylene analyzer (Ethan Meter, Bioconservacion, Spain). Single fruit was stored in a 5L airtight glass jar and hermetically sealed along with placing silicone rubber membrane on the top and kept for 2 hours and later hypodermic needle was inserted through silicone rubber membrane to collect 1ml gas and used for determination of CO2 and C2H4 via respective gas analyzer.28
Determination of fruit biochemical parameters
TSS content of the stored fruits was recorded with digital handheld refractometer (Hanna Instruments, Model: HI96801, Romania) with temperature correction. Titratable acidity, total sugar and reducing sugar content was measured following the standard protocol of AOAC.29 TSS with titratable acidity ratio was calculated by dividing TSS value with titratable acidity percentage. Ascorbic acid content of the fruits was measured by titrating with 2,6 Dichlorophenol indophenol dye.29,30 Total carbohydrate was measured by following anthrone method and total phenol was determined using Folin-Ciocalteau reagent; expressed as catechol equivalent.31 Total flavonoid content of the fruit extract was analyzed colorimetric method using Quercetin as standard.32 Antioxidant activity of the stored fruit was measured using methanolic extract for DPPH assay.33 Percent inhibition of DPPH was calculated using the given formula:
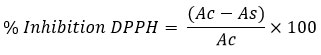
Where, Ac= Control absorbance; As=Sample absorbance.
Determination of fruit decay and shelf life
The number of fruit decayed was recorded based on the visual observation and decay percentage for a particular treatment at 12 DAS was calculated through diving number of fruit decayed up to 12 days with total number of fruits stored under that treatment.22 Shelf life of the fruits were determined based on the percentage of fruit decay, external appearance of crown condition; flesh translucency, counting days after harvest to the day with maximum visual appearance and physico-chemical qualities.34
Statistical analysis
Statistical analysis of the data was done using SPSS (Version 22) software for calculating ANOVA by following the method of Complete Randomized Design (CRD) having nine treatments and three replications; with six fruits per replication and Duncan’s Multiple Range Test (DMRT) was performed for separation of means.35
Results
Percentage of physiological weight loss, decrease in fruit length and fruit diameter
Fruit length, fruit diameter and weight of the ambiently stored pineapple gradually reduced and caused reasonable increment in the respective percentages. Physiological weight loss ranged between 4.67 % to 8.23 % at 4 DAS; got increased and ranged between 14.58% to 27.71% at 12 DAS (Table 1). Sole application of MT and SA showed moderately high weight loss (MT @0.1 mM: 16.85 %, MT@0.5mM:19.94%; SA @5mM: 15.92% and SA@9mM: 18.31%) at 12 DAS. However, fruits treated with MT 0.5 mM + SA 9 mM (T8) had minimum weight loss (14.58%) compared control (27.71 %) at 12 DAS. Similarly, the percentage decrease in fruit length ranged between 0.25% to 3.74% and fruit diameter ranged between 0.23% to 3.24% at 4 DAS, which increased and ranged between 1.76% to 7.84 % and 2.98% to 8.82%, respectively. After 12 days of ambient storage, contrasting with control (T9), fruits treated with MT 0.5 mM + SA 9 mM (T8) recorded lowest percentage decrease in fruit length (1.76%) and fruit diameter (2.98%).
Fruit firmness
Firmness of the stored pineapple fruits markedly reduced during the period of storage. Fruit firmness ranged between 49.56 to 70.32 (N cm-2) at 4DAS, 41.52 to 68.73 (N cm-2) at 8DAS and 35.42 to 59.84 (N cm-2) at 12 DAS (Table1). Although after 12 days of storage, it was recorded that sole application in lower concentrations of MT 0.1mM and SA 5 mM had maintained the fruit firmness (49.58 and 54.28 N cm-2, respectively), but highest firmness (59.84 N cm-2) was recorded in case of the fruits treated with MT 0.5 mM + SA 9 mM (T8) followed by the fruits treated with MT 0.1mM+ SA 5 mM (58.32 N cm-2; T5) compared with control (35.42 N cm-2, T9).
Table 1: Percentage weight loss, decrease in length, diameter and firmness of pineapples during storage
| Treatments | Weight Loss (%) | Length Decrease (%) | Diameter Decrease (%) | Firmness (Ncm-2) | ||||||||
| 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | |
| T1: Melatonin (MT) 0.1mM | 6.73 bcd |
10.76 ab |
16.85 abcd |
0.96c | 1.56d | 2.98c | 1.01c | 2.87 bc |
3.84 abc |
65.43 cd |
60.37 de |
49.58 de |
| T2: MT 0.5mM | 7.18 cd |
12.98c | 19.94d | 2.07e | 2.24f | 4.86e | 1.68e | 3.84d | 5.04d | 59.78b | 49.58b | 43.41b |
| T3: Salicylic Acid (SA) 5 mM | 5.78 abc |
9.23a | 15.92 ab |
0.41 ab |
1.12 bc |
2.65 bc |
0.64b | 2.26 ab |
3.52 ab |
66.84 de |
63.61 ef |
54.28f |
| T4: SA 9 mM | 7.04 bcd |
12.75 bc |
18.32 cd |
1.68 de |
1.97 ef |
3.78d | 1.34d | 3.34 cd |
4.85 cd |
61.58 bc |
56.34c | 45.32 bc |
| T5: MT 0.1mM+ SA 5 mM | 5.23 ab |
9.65a | 15.67 ab |
0.25a | 0.96b | 2.12 ab |
0.57b | 2.09a | 3.15a | 72.20f | 65.48 fg |
58.32g |
| T6: MT 0.1 mM + SA 9 mM | 6.84 bcd |
12.34 bc |
17.58 bcd |
1.23 cd |
1.73 de |
3.24 cd |
1.15 cd |
2.95 bc |
4.56 bcd |
63.45 bcd |
58.63 cd |
47.64 cd |
| T7: MT 0.5 mM + SA 5 mM | 6.27 abc |
10.12a | 16.38 abc |
0.82 bc |
1.42 cd |
1.99 ab |
0.96c | 2.18 ab |
3.34a | 66.52 de |
61.25 de |
50.78e |
| T8: MT 0.5 mM + SA 9 mM | 4.67a | 8.94a | 14.58a | 0.27a | 0.35a | 1.76a | 0.23a | 1.82a | 2.98a | 70.32 ef |
68.73g | 59.84g |
| T9: Control (water dipping) | 8.23d | 15.91d | 27.71e | 3.74f | 4.29g | 7.84f | 3.24f | 6.76e | 8.82e | 49.56a | 41.52a | 35.42a |
| S Em (±) | 0.572 | 0.667 | 0.701 | 0.176 | 0.125 | 0.236 | 0.104 | 0.243 | 0.360 | 1.399 | 1.172 | 0.806 |
| CD at 5% | 0.810 | 0.945 | 0.992 | 0.249 | 0.177 | 0.334 | 0.147 | 0.343 | 0.509 | 1.980 | 1.660 | 1.141 |
DAS = Days After Storage, Means followed by the same letters do not differ significantly at 5% level of probability.
Score for flesh translucency and crown condition
Ambiently stored pineapple fruits’ flesh was 100% opaque (average score < 1.80) for melatonin and/or salicylic acid treated compared with control (opaque with less than 50% translucent; average score: 2.20) at 4 DAS (Table 2). After 8 days of storage, fruits at control were having flesh opaque with more than 50% translucent (average score: 3.20) compared with others, where flesh remained 100% opaque except fruits at T2, T4 and T6 (average score: 2.00 to 2.40). Even after 12 days of ambient storage fruits treated with MT 0.5 mM + SA 9 mM (T8) remained 100% opaque and had minimum flesh translucency score (average score: 1.40) compared with control (average score: 3.40).
At 4 DAS, fruits treated with MT 0.5 mM + SA 9 mM (T8) had minimum crown condition score (average score: 1.20) compared with control (average score: 3.00; moderately dry tip & yellowing). However, at 8 DAS, fruits treated with MT 0.1 mM + SA 5 mM (T5) had minimum crown condition score (average score: 1.60; good, fresh and green) and at 12 DAS, fruits at T8 had minimum score (average score: 2.00; good with slightly yellow at tip) compared with control (average score: 3.20; moderate, dry tips and yellowing and 4.20; bad, dry tips and more yellowing, respectively).
Skin and flesh colour
After 12 days of storage, among sole application treatments, skin colour of the pineapple fruits at T1 (MT 0.1mM) was light orange (L:66.47, a:19.77, b:57.34) and T3 (SA 5mM) was orange (L:73.41, a:17.88, b:47.57), whereas, combined application at T8 (MT 0.5 mM + SA 9 mM) was pale orange (L:85.78, a:8.22, b:30.96), contrasting with control (T9), where it was recorded dark orange (L:52.54, a:30.43, b:40.63) . Similarly, the most intensified flesh colour (dark golden yellow; L: 57.04, a:5.91, b:49.35) was recorded in case of the fruits at control (T9) compared to T8, where flesh colour of the fruits was whitish (L:88.25, a:0.86, b:21.22) [Table: 2, Fig. 1(a), 1(b)].
Table 2: Average score for flesh translucency, crown condition and skin, flesh colour of pineapples at storage
| Treatments | Average score for flesh translucency | Average score for crown condition | Skin Colour at 12 DAS | Flesh Colour at 12 DAS | ||||||||
| 4DAS | 8DAS | 4DAS | 8DAS | 4DAS | 8DAS | L | a | b | L | a | b | |
| T1: Melatonin (MT) 0.1mM | 1.60 | 1.80 | 2.00 | 1.80 | 2.20 | 2.60 | 66.47 | 19.77 | 57.34 | 71.14 | 1.06 | 47.75 |
| T2: MT 0.5mM | 1.80 | 2.40 | 2.60 | 2.40 | 2.80 | 3.40 | 49.75 | 36.68 | 49.4 | 78.28 | 6.51 | 45.77 |
| T3: Salicylic Acid (SA) 5 mM | 1.40 | 1.60 | 1.80 | 1.60 | 1.80 | 2.40 | 73.41 | 17.88 | 47.57 | 73.21 | 0.96 | 32.27 |
| T4: SA 9 mM | 1.80 | 2.20 | 2.40 | 2.40 | 2.60 | 3.20 | 58.01 | 45.91 | 42.19 | 58.61 | 4.32 | 49.28 |
| T5: MT 0.1mM+ SA 5 mM | 1.20 | 1.40 | 1.60 | 1.40 | 1.60 | 2.20 | 80.84 | 16.63 | 50.48 | 81.87 | -0.27 | 52.31 |
| T6: MT 0.1 mM + SA 9 mM | 1.80 | 2.00 | 2.20 | 2.20 | 2.40 | 2.80 | 77.11 | 15.03 | 49.12 | 79.26 | 1.27 | 45.29 |
| T7: MT 0.5 mM + SA 5 mM | 1.60 | 1.80 | 1.80 | 1.80 | 2.00 | 2.40 | 73.21 | 16.83 | 58.35 | 78.73 | 0.87 | 36.55 |
| T8: MT 0.5 mM + SA 9 mM | 1.00 | 1.20 | 1.40 | 1.20 | 1.80 | 2.00 | 85.78 | 8.22 | 30.96 | 88.25 | 0.86 | 21.22 |
| T9: Control (water dipping) | 2.20 | 3.20 | 3.40 | 3.00 | 3.20 | 4.20 | 52.54 | 30.43 | 40.63 | 57.04 | 5.91 | 49.35 |
| S Em (±) | – | – | – | – | – | – | – | – | – | – | – | – |
| CD at 5% | – | – | – | – | – | – | – | – | – | – | – | – |
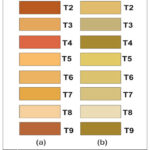 |
Figure 1: (a) Skin Colour, (b) Flesh Colour at 12 DAS under different postharvest treatments |
CO2 and C2H4 gas released
Release of CO2 gas from the stored pineapple gradually increased across all the treatments during the period of storage. At 4 DAS, the maximum amount of CO2 (6.04 ml kg-1 h-1) was released from the fruits at control (T9) compared with the fruits treated with MT 0.5 mM + SA 9 mM (4.33 ml kg-1 h-1, T8). At 12 DAS, among sole applications, fruits treated with SA 5mM had released comparatively lower amount of CO2 (7.87 ml kg-1 h-1) though among all the treatments minimum amount of CO2 (6.87 ml kg-1 h-1) was released from the fruits combinedly treated with MT 0.5 mM + SA 9 mM (T8) followed by the fruits treated with MT 0.1mM+ SA 5 mM (7.38 ml kg-1 h-1, T5) compared with control (12.96 ml kg-1 h-1). Besides, the rate of ethylene gas released from the ambiently stored pineapple increased simultaneously like carbon-di-oxide gas. It was recorded that the release rate of C2H4 from stored pineapple ranged between 6.56-8.25 µl kg-1 h-1 at 4 DAS, which increased and ranged between 8.89 to 9.94 µl kg-1 h-1 at 8 DAS and finally ranged between 10.78-13.39 µl kg-1 h-1 at 12 DAS (Table 3). After 12 days of storage of pineapple at ambient condition, fruits treated with SA 5 mM had lower ethylene released (11.58 µl kg-1 h-1) among the sole application of MT or SA, however, combined application of MT 0.5 mM + SA 9 mM (T8) had the minimum (10.78 µl kg-1 h-1) rate of C2H4 released compared with control (13.39 µl kg-1 h-1).
Table 3: Release of carbon-di-oxide and ethylene gas, total soluble solids (TSS), titratable acidity content of stored pineapples
| Treatments | CO2 (ml kg-1 h-1) | Ethylene (µl kg-1 h-1) | TSS (0 Brix) | Titratable Acidity (%) | ||||||||
| 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | |
| T1: Melatonin (MT) 0.1mM | 4.95 bcd |
6.87 abc |
8.49 cd |
7.42 bcd |
9.51 bcde |
11.69 bcd |
14.81 bc |
15.37b | 12.65 cd |
0.82 bcd |
0.92 abc |
0.98 abc |
| T2: MT 0.5mM | 5.49 de |
8.51d | 8.89d | 7.75 de |
9.85 de |
12.13d | 15.35c | 14.68b | 11.14b | 0.89 de |
0.98 bc |
1.09 cd |
| T3: Salicylic Acid (SA) 5 mM | 4.65 abc |
6.42 ab |
7.87 bc |
6.89 abc |
9.24 abc |
11.58 bc |
14.02 abc |
15.87 bc |
12.84d | 0.76 abc |
0.86 ab |
0.94 abc |
| T4: SA 9 mM | 5.34d | 7.74 cd |
8.81 cd |
7.69 de |
9.78 de |
11.89 cd |
14.97 bc |
14.88b | 11.25b | 0.87 cde |
0.97 bc |
1.05 bcd |
| T5: MT 0.1mM+ SA 5 mM | 4.54 ab |
6.21a | 7.38 ab |
6.74 ab |
9.06 ab |
11.34b | 13.92 ab |
16.98c | 13.78d | 0.72 ab |
0.82 ab |
0.89 ab |
| T6: MT 0.1 mM + SA 9 mM | 5.19 cd |
7.27 bc |
8.69 cd |
7.53 cde |
9.58 cde |
11.67 bc |
14.92 bc |
15.26b | 11.48 bc |
0.85 cd |
0.95 abc |
1.02 bcd |
| T7: MT 0.5 mM + SA 5 mM | 4.69 abc |
6.65 ab |
8.12 bcd |
7.11 abcd |
9.36 abcd |
11.45 bc |
14.11abc | 15.68b | 12.78 cd |
0.79 bcd |
0.89 abc |
0.96 abc |
| T8: MT 0.5 mM + SA 9 mM | 4.33a | 5.87a | 6.87a | 6.56a | 8.89a | 10.78a | 13.45a | 17.19c | 15.59e | 0.67a | 0.78a | 0.85a |
| T9: Control (water dipping) | 6.04e | 11.91e | 12.96e | 8.25e | 9.94e | 13.39e | 16.91d | 11.78a | 9.46a | 0.98e | 1.07c | 1.18d |
| S Em (±) | 0.170 | 0.314 | 0.297 | 0.217 | 0.152 | 0.143 | 0.414 | 0.392 | 0.424 | 0.034 | 0.055 | 0.049 |
| CD at 5% | 0.241 | 0.444 | 0.421 | 0.307 | 0.215 | 0.203 | 0.586 | 0.555 | 0.600 | 0.049 | 0.077 | 0.070 |
DAS = Days After Storage, Means followed by the same letters do not differ significantly at 5% level of probability.
TSS, titratable acidity and TSS:acid ratio
Four days of ambient storage increased the total soluble solids (TSS) contents of the pineapple; maximum for fruits at control (16.91 0Brix) compared with the fruits treated with MT 0.5 mM + SA 9 mM (T8:13.45 0Brix). At 8 DAS, fruits at T8 recorded the maximum TSS (17.19 0Brix) followed by T5 (16.98 0Brix) compared with control (11.78 0Brix). After 12 days of storage, among sole treatments, fruits at T3 had high TSS (12.84 0Brix), whereas, among combined treatments, fruit at T8 had reasonably high TSS (15.59 0Brix) whereas, it was found minimum at control (9.46 0Brix). Fruit acidity increased during the period of storage (Table 3). At 4 DAS it ranged between 0.67% to 0.98%; which increased and ranged between 0.78% to 1.07% at 8 DAS and 0.85% to 1.18% at 12 DAS. At 12 DAS, minimum titratable acidity (0.85%) of the fruit was recorded in T8 (MT 0.5 mM + SA 9 mM) followed by T5 (0.89%) compared with control (1.18%). Similarly, TSS:acid ration at 12 DAS was recorded maximum (18.34) in T8 followed by T5 (15.48) compared with control (8.02) (Table 4).
Total sugar, reducing sugar, total carbohydrate
At 4 DAS total sugar content was recorded maximum (13.78%) in control, whereas it was found minimum (10.78%) in T8 (MT 0.5 mM + SA 9 mM). At 8 DAS and 12 DAS; content of total sugar of stored pineapple was minimum in control (10.08%; 7.56%, respectively) whereas for sole application treatments of MT or SA, high total sugar (14.28% and 12.18%, respectivley) was recorded in T3 (SA 5 mM), however it was recorded maximum (15.17% and 13.17%, respectively) in case of the fruits treated combinedly with MT 0.5 mM + SA 9 mM (T8). At 12 DAS, reducing sugar and total carbohydrate content was recorded minimum (5.67% and 3.15 %, respectively) in case of the fruits at control (T9) whereas, it was recorded maximum (10.08% and 7.21%, respectively) at T8 (Table 4).
Table 4: TSS: acid ratio, total sugar, reducing sugar and total carbohydrate content of pineapples at storage
| Treatments | TSS:acid ratio | Total Sugar (%) | Reducing Sugar (%) | Total Carbohydrate (%) | ||||||||
| 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | |
| T1: Melatonin (MT) 0.1mM | 18.06 ab |
16.71 cd |
12.91c | 12.26 bcd |
13.78 bcde |
11.25 cd |
10.34 abc |
10.67 bcd |
8.85 cd |
4.89b | 7.71 cd |
6.15cd |
| T2: MT 0.5mM | 17.25 ab |
14.98b | 10.22b | 13.02 de |
12.62b | 9.53b | 11.34 cd |
10.07b | 7.16b | 6.78c | 5.31b | 4.71b |
| T3: Salicylic Acid (SA) 5 mM | 18.45 ab |
18.45e | 13.66c | 11.78 abc |
14.28 de |
12.18 de |
9.91 ab |
11.32 cd |
9.56 de |
4.38 ab |
7.92 cd |
6.34 cd |
| T4: SA 9 mM | 17.21 a |
15.34 bc |
10.71b | 12.84 cde |
12.78 bc |
9.78b | 10.87 bc |
10.23 bc |
7.32b | 6.42c | 6.79c | 5.72 bc |
| T5: MT 0.1mM+ SA 5 mM | 19.33 bc |
20.71f | 15.48d | 11.23 ab |
14.65 de |
12.56 de |
9.78 ab |
11.56d | 9.78 de |
4.11 ab |
8.56d | 6.78 cd |
| T6: MT 0.1 mM + SA 9 mM | 17.55a | 16.06 bc |
11.25b | 12.67 cde |
13.56 bcd |
10.12 bc |
10.56 bc |
10.62 bcd |
8.12 bc |
6.15c | 7.37 cd |
6.04 cd |
| T7: MT 0.5 mM + SA 5 mM | 17.86a | 17.62 de |
13.31c | 11.95 bcd |
14.09 cde |
11.84 de |
10.18 abc |
10.78 bcd |
9.12 cde |
4.57 ab |
7.85 cd |
6.21 cd |
| T8: MT 0.5 mM + SA 9 mM | 20.07c | 22.04f | 18.34e | 10.78a | 15.17e | 13.17e | 9.24a | 12.78e | 10.08e | 3.49a | 8.72d | 7.21d |
| T9: Control (water dipping) | 17.26a | 11.01a | 8.02a | 13.78e | 10.08a | 7.56a | 12.14d | 8.24a | 5.67a | 7.32c | 3.85a | 3.15a |
| S Em (±) | 0.408 | 0.486 | 0.472 | 0.350 | 0.431 | 0.450 | 0.368 | 0.345 | 0.363 | 0.391 | 0.406 | 0.377 |
| CD at 5% | 0.577 | 0.688 | 0.669 | 0.495 | 0.610 | 0.638 | 0.522 | 0.489 | 0.514 | 0.553 | 0.575 | 0.534 |
DAS = Days After Storage, Means followed by the same letters do not differ significantly at 5% level of probability.
Ascorbic acid and total phenol
Stored pineapple fruits gradually lost its ascorbic acid content. At 4 DAS, fruit ascorbic acid ranged between 13.75 to 22.78 mg 100g-1; which was reduced and found to range between 11.23 and 19.23 mg 100g-1 at 8 DAS; while finally ranged between 7.02 and 15.34 mg 100g-1 at 12 DAS (Table 5). Minimum ascorbic acid content (7.02 mg 100g-1) of stored pineapple was recorded in control (T9) whereas, maximum (15.34 mg 100g-1) was recorded from the fruits applied with MT 0.5 mM + SA 9 mM (T8) followed by T3 (14.32 mg 100g-1 ) at 12 DAS. In parity with the trend of ascorbic acid content, total phenol content of the stored pineapple fruits reduced drastically during storage. Total phenol content was ranged between 34.76 and 68.79 mg CE 100g-1 at 4 DAS; later found to decrease and ranged between 11.23 and 49.78 mg CE 100g-1 at 12 DAS. At 12 DAS, among individual application treatments of MT or SA, fruits at T3 (SA 5 mM) had high phenol (42.17 mg CE 100g-1), though maximum total phenol content (49.78 mg CE 100g-1) of the fruits was recorded in combined application at T8 (MT 0.5 mM + SA 9 mM) followed by T5 (47.32 mg CE 100g-1) compared with control (11.23 mg CE 100g-1).
Total flavonoids and antioxidant activity
Total flavonoids content of the stored pineapple gradually reduced in ambient storage. Total flavonoids content at 4 DAS was recorded maximum (5.45 mg QE100g-1) in case of the fruits treated with MT 0.5 mM + SA 9 mM (T8) followed by the fruits treated with MT 0.5 mM + SA 5 mM (T7: 5.19 mg QE100g-1) among combined treatments whereas, SA at 5mM (T3: 5.11 mg QE100g-1) recorded high flavonoids among MT or SA sole treatments in stored fruits compared with control (2.56 mg QE100g-1) (Table 5). At 12 DAS, it was measured maximum in T8 (4.21 mg QE100g-1) whereas, minimum in T9 (1.87 mg QE100g-1). After 12 days of ambient storage, highest antioxidant activity (58.49 % inhibition DPPH) was found in fruits treated with MT 0.5 mM + SA 9 mM (T8) followed by fruits treated with MT 0.1 mM + SA 5 mM (T5: 52.34% inhibition DPPH) in combined treatments and fruits treated with SA at 5mM (T3: 48.76% inhibition DPPH) among sole treatments with either MT or SA compared with control (T9: 21.19 % inhibition DPPH).
Table 5: Ascorbic acid, total phenol, total flavonoids content and antioxidant activity of pineapples during storage
| Treatments | Ascorbic Acid (mg 100 g-1) | Total Phenol (mg CE/100g) | Total Flavonoids (mg QE/100g) | Antioxidant activity (%inhibition DPPH) | ||||||
| 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | 4DAS | 8DAS | 12DAS | 12 DAS | |
| T1: Melatonin (MT) 0.1mM | 18.85bc | 16.37bc | 11.98bcd | 58.46cd | 43.97cd | 36.47c | 4.24cd | 3.12ab | 3.18c | 41.78c |
| T2: MT 0.5mM | 16.84b | 13.67ab | 10.53b | 51.24b | 34.67b | 28.78b | 3.29b | 2.56ab | 2.08ab | 36.24b |
| T3: Salicylic Acid (SA) 5 mM | 19.78bcd | 18.32c | 14.32cd | 61.28d | 47.54de | 42.17d | 5.11e | 4.21cd | 3.78de | 48.76d |
| T4: SA 9 mM | 17.28b | 15.17bc | 11.14bc | 56.02c | 37.48b | 30.65b | 3.78bc | 2.86ab | 2.17ab | 38.69bc |
| T5: MT 0.1mM+ SA 5 mM | 21.35cd | 18.56c | 14.78cd | 65.32e | 52.37ef | 47.32e | 5.23e | 4.78d | 4.09e | 52.34e |
| T6: MT 0.1 mM + SA 9 mM | 17.68b | 15.48bc | 11.38bc | 57.32c | 39.65bc | 32.36b | 4.78de | 2.98ab | 2.47b | 40.45c |
| T7: MT 0.5 mM + SA 5 mM | 19.32bc | 17.48bc | 12.56bcd | 59.72cd | 49.32de | 40.04d | 5.19e | 3.56bc | 3.56cd | 46.32d |
| T8: MT 0.5 mM + SA 9 mM | 22.78d | 19.23c | 15.34d | 68.79e | 56.34f | 49.78e | 5.45e | 4.98d | 4.21e | 58.49f |
| T9: Control (water dipping) | 13.75a | 11.23a | 7.02a | 34.76a | 18.98a | 11.23a | 2.56a | 2.13a | 1.87a | 21.19a |
| S Em (±) | 0.986 | 1.227 | 1.141 | 1.181 | 2.047 | 1.149 | 0.234 | 0.323 | 0.154 | 1.088 |
| CD at 5% | 1.396 | 1.737 | 1.616 | 1.673 | 2.897 | 1.627 | 0.331 | 0.458 | 0.218 | 1.540 |
DAS = Days After Storage, CE= Catechin Equivalent, QE= Quercetin Equivalent, DPPH= 2,2-diphenyl-1-picrylhydrazyl, Means followed by the same letters do not differ significantly at 5% level of probability.
Fruit decay and shelf life
Storage of the pineapple at ambient condition had variable incidence of decay across the treatments, however fruits at control had the highest fruit decay (66.67%) whereas it was found less (33.33 %) both in MT 0.1mM and SA 5mM when applied sole, however minimum decay (16.67%) was recorded with combined application at T8 (MT 0.5 mM + SA 9 mM) at 12 DAS. Consequently, it was observed that fruits at control (T9) had minimum shelf life (11.50 days) whereas, shelf life was slightly high at T3(14.17 days) among the sole treatments, although maximum shelf life (16.33 days) of pineapple fruits was found in combined treatment at T8 (MT 0.5 mM + SA 9 mM) followed by the fruits treated with MT 0.1 mM + SA 5 mM (T5: 14.67 days) [Fig. 2].
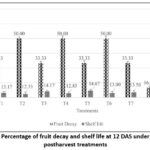 |
Figure 2: Percentage of fruit decay and shelf life at 12 DAS under different postharvest treatments. |
Discussion
Physiological weight loss of the stored pineapple significantly increased across all treatments. It was reported that pineapple fruit stored at ambient condition marked an increment in physiological weight loss due to loss of moisture from fruit surface, transpiration and respiration.34,36 Similarly, fruit length and diameter of stored pineapple reduced due to shrinkage. Scientists reported to have shrinkage of pineapple while storing at 250C.6,36 Present study revealed that pineapple treated with MT 0.5 mM + SA 9 mM (T8) had minimum PLW and least percentage decrease of fruit length and diameter compared with control at 12 DAS. Post harvest use of 1mM MT significantly reduced PLW of the ‘Santa Rosa’ plum.12 Pineapple fruits dipped in 5mM of SA had scored lower PLW and decrease in fruit length and diameter.6 SA @ 2mM checked physiological weight loss in grapes.24 Melatonin reported to play role in delaying of senescence by entrapping reactive oxygen species (ROS) and malondialdehyde (MDA) and maintain firmness by delaying enzymatic softening and maintaining structural integrity with enhanced anti-oxidant activity, thus reduced PLW and fruit shrinkage.37 Further, stomatal closure and reduction in respiration due to SA action had caused lower physiological weight loss in stored fruits.24 Present study revealed that use of MT 0.5 mM + SA 9 mM had maintained the fruit firmness even after 12 DAS compared with control. Melatonin hinders the activity of enzymes like pectin methyl esterase and polygalacturonase which are responsible for breakdown of cell wall and thus prevent degradation of cell wall and maintained firmness of stored fruits.38 SA too had inhibitory role against enzymes like cellulase, lipoxygenase, pectin methyl esterase, polygalacturonase etc. responsible for cell wall degradation, so helps in better maintenance of fruit firmness.20 Average score for flesh translucency and crown condition was found minimum in pineapple fruits treated with MT 0.5 mM + SA 9 mM. It was reported that flesh of pineapple fruits turned from opaque to translucent and contributed higher flesh translucency score during ambient storage due to electrolyte leakage, whereas crown condition deteriorated for senescence, which markedly influenced by the storage temperature.6 Postharvest fruit dipping in MT significantly reduced electrolyte leakage and occurrence of flesh translucency in stored pineapple.26,39 Treatment with SA reported to cause retardation of electrolyte leakage40 and therefore, combined treatment may have caused low score of flesh translucency on dose dependent manner. Anti-senescence role of MT17,18 and SA20,27 may have contributed to better crown quality of the stored pineapple under combined application. External colour of the pineapple fruit turned orange from green while fruit flesh changed from white to deep yellow during storage. These changes happened following the natural ripening phenomenon by chlorophyll breakdown and accumulation of carotenoids.6,26 However, the colour record of peel and flesh as depicted in developed colour chart signified fruits treated with MT@ 0.5 mM along with SA 9 mM had delayed accumulation of colour which may be attributed to the ethylene inhibitory effect of MT10 and SA.20 Besides, storing pineapples in ambient condition caused enhanced respiration and triggered the ethylene gas release. Though, in comparison to control, fruits treated with MT (0.5mM) + SA (9mM) significantly reduced the rate of respiration measured in terms of CO2 released and ethylene production. SA reported to reduce ethylene, the endogenous hormone which influences the enzymatic activity responsible for cell wall hydrolyzation and consequential increment in respiration rate, production by lessening 1-aminocyclopropane-1-carboxylic acid oxidase (ACO) and 1-aminocyclopropane-1-carboxylic acid synthase (ACAS) activity.20,21 While, exogenous application of MT thought to reduce gain of ROS while increasing the antioxidant enzymes and restricting the enzymatic expression of ACAS and ACO, thus subside the ethylene accumulation and enhancement of respiration.7,10 TSS content of the stored pineapple had initial increment up to eight days afterwards declined. Similar observation was recorded in case of pineapple cv. Comte de Paris at ambient storage (250C) where TSS increased up to six days due to ripening and subsequently dropped as of senescence.41 Significantly high TSS was found in MT 0.5 mM + SA 9 mM treated fruits even after 12 days may be because of the delayed ripening and senescence. The observed delay in the evolution of acidity and TSS in pineapples can be related to previous findings in other fruits, where MT and SA were shown to enhance ATP supply, thereby delaying substrate catabolism.42 Post-harvest application of melatonin had delayed ethylene to reach peak and consequently delayed ripening which have resulted in slower accumulation of TSS.10 However, it was found to have increment in titratable acidity during storage of pineapple which largely impacted TSS:acid ratio of the stored fruits. It was reported that there was an increase in titratable acidity in stored pineapple vastly because of accumulation of citric and malic acids due to changes in cell membrane permeability and solute concentration.43 MT+SA treated fruits had better retention of TSS:acid ratio which may be because post-harvest application of melatonin in pineapple reported to cause belated ripening and better retention of TSS: acid ratio.26 Post-harvest use of SA in litchi showed better retention of TSS:acid ratio due to delayed ripening and senescence.44 Total sugar, reducing sugar and total carbohydrate content of the stored pineapple reduced; however fruits treated with MT+SA had better retention of sugars and carbohydrates even after 12 days. It was reported that exogenous application of melatonin in pineapple caused better retention of sugars and carbohydrates during ambient storage.26 SA@ 2mM showed highest amount of sugars retained in grapes after 16 days of storage in ambient condition.24 Prolong storage of pineapple fruits reported to drop in its sugar contents because of senescence.41 MT and SA showed proven anti-senescent effect in many fruits during storage which may be the reason that combined application of these compounds resulted in better sugar and carbohydrate retention.7,10,20,21 Ascorbic acid content was found to decrease during the period of storage. Ascorbic acid oxidase mediated the conversion of L-ascorbic acid to dehydro ascorbic acid may be the reason behind drop of fruit ascorbic acid during storage.45 However, oxidative stress which plays the prevalent role in loss of ascorbic acid, inhibited due to the action of both MT and SA, as previously reported.21,26,46 Storing of pineapple fruits at ambient condition showed significant reduction in total phenol and total flavonoids content along with reduction in antioxidant activity. But fruits treated with MT+SA significantly protected the loss, while treatment with MT 0.5 mM + SA 9 mM resulted highest total phenol and flavonoids content with maximum antioxidant activity at 12DAS. Total phenol content was found to reduce due to polymerization with proteins and for oxidative breakdown which further emanates to form hydroxy methyl furfural and caused loss of flavonoids during storage.47 Accumulation of ROS in ripened fruit at storage caused oxidative damage, which may have impacted the phenolic constituents and antioxidant activity.21 MT and SA both was reported to inhibit accumulation of ROS and therefore protected from oxidative breakdown of phenolic constituents which results in better antioxidative activity.7,10,21,26 Besides, post-harvest use of MT reported to contribute in disease resistance and reduction of decay percentage in multiple fruits during storage.10 Use of MT found to control anthracnose in stored papaya and development of green mold in citrus during storage.38,46 Exogenous application of SA caused inhibition of blue mold development during storage.19 Proven result in control post-harvest decay in fruits like apricot, banana, longan, litchi, grapes etc. was also reported.20,21 Thus, in the present study combined application of MT+SA may have resulted synergistically and particularly in higher concentration had reasonably controlled the postharvest decay in stored pineapple and extended the shelf life. MT and SA were reported to delay senescence, inhibited respiration and ethylene production, controlled oxidative stress while protected from disease occurrence during postharvest condition and extended shelf life in apple,19 banana,11,22 citrus,15,25 grapes,14,24 plum,12 pear13 etc. fruits.
Conclusion
In the present study, when MT or SA was administered individually though had resulted positive influence on postharvest physico-biochemical condition, decay control and shelf life of stored pineapple, however combined use of MT+SA in higher concentrations; found to act synergistically and had significantly reduced physiological weight loss, with maintained fruit firmness and crown condition, retained peel colour and flesh translucency while inhibiting ethylene release and controlled respiration; thus, maintained better biochemical qualities (TSS, TSS:acid ratio, sugars, ascorbic acid) and phenolic constituents (total phenol and total flavonoids) with higher antioxidant activity. Moreover, it controlled fruit decay and resulted in high shelf life. Therefore, use of MT@ 0.5mM along with SA@ 9mM can be a potential postharvest treatment for shelf-life extension and quality maintenance of ambiently stored pineapple.
Acknowledgement
The author would like to thank Mizoram University for granting the Masters and dissertation work and for providing the instrumental facilities to execute the trial.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Debashis Mandal: Conceptualized the study and prepared the manuscript
- Marto Basar: Laboratory analyses and statistical calculations
- Noel Lalhruaitluangi: Laboratory analyses
- Ralte Colney Lalduhsangi: Laboratory analyses
- Agnes Vanlalnghaki Fanai: Laboratory analyses
References
- Crop and livestock products: pineapple. Food and Agriculture Organization of the United Nations. https://www.fao.org/faostat/en/#data/QCL. Published on 2023, Updated on 11.06.2025, Accessed on 18.07.2025.
- Mohd Ali M., Hashim N., Abd Aziz S., et al. Shelf Life Prediction and Kinetics of Quality Changes in Pineapple (Ananas comosus) Varieties at Different Storage Temperatures. Horticulturae. 2022;8(11):992. doi:10.3390/horticulturae8110992
CrossRef - Gomez S., Kuruvila B., Maneesha P.K., et al. Variation in physico-chemical, organoleptic and microbial qualities of intermediate moisture pineapple (Ananas comosus (L.) Merr.) slices during storage. Food Prod Process Nutr. 2022; 4(1): 5. doi:10.1186/s43014-022-00084-2
CrossRef - Dolhaji N.H., Muhammad I.D., Yaakob H., et al. Chilling injury in pineapple fruits: physical quality attributes and antioxidant enzyme activity. Food Res. 2020; 4: 86-95. doi:10.26656/fr.2017.4(S5).004
CrossRef - Paull R.E., Cheng C.C. Postharvest physiology, handling and storage of pineapple. In: The pineapple: botany, production and uses. Wallingford UK: CABI publishing; 2003: 253-279.
CrossRef - Mandal D., Lalremruata, Hazarika T.K., et al. Effect of Post-harvest Treatments on Quality and Shelf Life of Pineapple (Ananas comosus [L.] Merr. ‘Giant Kew’) Fruits at Ambient Storage Condition. Int J Bio-Resour Stress Manag. 2015; 6: 490-496. doi:10.5958/0976-4038.2015.00072.X
CrossRef - Xu T., Chen Y., Kang H. Melatonin Is a Potential Target for Improving Post-Harvest Preservation of Fruits and Vegetables. Front Plant Sci. 2019; 10:1388. doi:10.3389/fpls.2019.01388
CrossRef - Wang Y., Reiter R.J., Chan Z. Phytomelatonin: a universal abiotic stress regulator. J Exp Bot. 2018; 69(5): 963-974. doi:10.1093/jxb/erx473
CrossRef - Pan Y., Xu X., Li L., et al. Melatonin-mediated development and abiotic stress tolerance in plants. Front Plant Sci. 2023;14:1100827. doi:10.3389/fpls.2023.1100827
CrossRef - Ze Y., Gao H., Li T., et al. Insights into the roles of melatonin in maintaining quality and extending shelf life of postharvest fruits. Trends Food Sci Technol. 2021; 109: 569-578. doi:10.1016/j.tifs.2021.01.051
CrossRef - Hu W., Yang H., Tie W., et al. Natural Variation in Banana Varieties Highlights the Role of Melatonin in Postharvest Ripening and Quality. J Agric Food Chem. 2017; 65(46): 9987-9994. doi:10.1021/acs.jafc.7b03354
CrossRef - Bal E. Physicochemical changes in ‘Santa Rosa’ plum fruit treated with melatonin during cold storage. J Food Meas Charact. 2019; 13(3): 1713-1720. doi:10.1007/s11694-019-00088-6
CrossRef - Liu J., Yang J., Zhang H., et al. Melatonin Inhibits Ethylene Synthesis via Nitric Oxide Regulation to Delay Postharvest Senescence in Pears. J Agric Food Chem. 2019; 67(8): 2279-2288. doi:10.1021/acs.jafc.8b06580
CrossRef - Wang L., Luo Z., Yang M., et al. Role of exogenous melatonin in table grapes: First evidence on contribution to the phenolics-oriented response. Food Chem. 2020; 329: 127155. doi:10.1016/j.foodchem.2020.127155
CrossRef - Wang J., Hong M., Feng Y., et al. Effects of exogenous melatonin treatment on fruit quality and ethanol metabolism of’ Aiyuan 38’citrus. Food Fermentation Ind. 2019; 45: 147-154.
- Liu C., Zheng H., Sheng K., et al. Effects of melatonin treatment on the postharvest quality of strawberry fruit. Postharvest Biol Technol. 2018; 139: 47-55. doi:10.1016/j.postharvbio.2018.01.016
CrossRef - Gao H., Zhang Z.K., Chai H.K., et al. Melatonin treatment delays postharvest senescence and regulates reactive oxygen species metabolism in peach fruit. Postharvest Biol Technol. 2016; 118: 103-110. doi:10.1016/j.postharvbio.2016.03.006
- Hu M., Li J., Rao J. Effect of Melatonin on Ripening and Senescence of Postharvest Kiwifruits. Food Sci. 2018; 39 (19): 226-232. doi:10.7506/spkx1002-6630-201819035
- Wang Y., Chen J., Bian W., et al. Control Efficacy of Salicylic Acid Microcapsules against Postharvest Blue Mold in Apple Fruit. Molecules. 2022; 27(22): 8108. doi:10.3390/molecules27228108
CrossRef - Asghari M., Aghdam M.S. Impact of salicylic acid on post-harvest physiology of horticultural crops. Trends Food Sci Technol. 2010; 21(10): 502-509. doi:10.1016/j.tifs.2010.07.009
CrossRef - Chen C., Sun C., Wang Y., et al. The preharvest and postharvest application of salicylic acid and its derivatives on storage of fruit and vegetables: A review. Sci Hortic. 2023; 312: 111858. doi:10.1016/j.scienta.2023.111858
CrossRef - Mandal D., Lalrindika E., Hazarika T.K., et al. Post harvest application of salicylic acid enhanced shelf life and maintained quality of banana cv. Grand Naine at ambient storage. The Bioscan. 2016; 11(1): 265-270.
- Habibi F., Ramezanian A., Guillén F., et al. Blood oranges maintain bioactive compounds and nutritional quality by postharvest treatments with γ-aminobutyric acid, methyl jasmonate or methyl salicylate during cold storage. Food Chem. 2020; 306: 125634. doi:10.1016/j.foodchem.2019.125634
CrossRef - Hazarika T., Marak T. Salicylic acid and oxalic acid in enhancing the quality and extending the shelf life of grape cv. Thompson seedless. Food Sci Technol Int. 2022; 28(6): 463-475. doi:10.1177/10820132211020612
CrossRef - Nikhil, Jawandha S.K., Gill P.P.S., et al. Preservation effect of chitosan and salicylic acid-based composite coatings on the postharvest quality of Lemons (Citrus limon Burm). J Food Meas Charact. 2025; 19(1): 644-655. doi:10.1007/s11694-024-02996-8
CrossRef - Mandal D., Ennio N., Lalhruaitluangi N., et al. Response of melatonin on postharvest qualities and shelf life of pineapple cv. Kew at ambient storage. J Appl Nat Sci. 2024; 16(2): 794-804. doi:10.31018/jans.v16i2.5562
CrossRef - Lu XH, Sun DQ, Mo YW, et al. Effects of post-harvest salicylic acid treatment on fruit quality and anti-oxidant metabolism in pineapple during cold storage. J Hortic Sci Biotechnol. 2010; 85(5): 454-458. doi:10.1080/14620316.2010.11512697
CrossRef - Siddiqui M.W., Homa F., Ayala-Zavala J.F., et al. Postharvest Nitric Oxide Infiltration Reduces Oxidative Metabolism and Prolongs Shelf-Life of Banana. J Plant Growth Regul. 2024; 43(11): 4151-4160. doi:10.1007/s00344-024-11378-9
CrossRef - Official methods of the analysis.15th Ed. Washington DC, USA: Association of Analytical Chemists; 1990.
- Ranganna S. Handbook of Analysis and Quality Control for Fruits and Vegetable Products.2nd New Delhi, India: Tata McGraw Hill Publishing Co. Ltd.; 1997.
- Sadasivam S., Manickam A. Biochemical methods. 2nd New Delhi, India: New Age International (P) Ltd.; 1997.
- Luximon-Ramma A., Bahorun T., Soobrattee M.A., et al. Antioxidant Activities of Phenolic, Proanthocyanidin, and Flavonoid Components in Extracts of Cassia fistula. J Agric Food Chem. 2002; 50(18): 5042-5047. doi:10.1021/jf0201172
CrossRef - Kumari P., Barman K., Patel V.B., et al. Reducing postharvest pericarp browning and preserving health promoting compounds of litchi fruit by combination treatment of salicylic acid and chitosan. Sci Hortic. 2015; 197: 555-563. doi:10.1016/j.scienta.2015.10.017
CrossRef - Mandal D., Chhangte V. Impact of postharvest use of essential oils on quality and shelf life of Indian pineapple. J Postharvest Technol. 2020; 08(3): 96-105. https://www.journals.acspublisher.com/index.php/jpht/ article/view/15336
- Gomez K.A., Gomez A.A. Statistical Procedure for Agricultural Research. 2nd Ed. New York , USA: John Willey and Sons; 1984.
- Techavuthiporn C., Boonyaritthongchai P., Supabvanich S. Physicochemical changes of ‘Phulae’ pineapple fruit treated with short-term anoxia during ambient storage. Food Chem. 2017; 228: 388-393. doi:10.1016/j.foodchem.2017.02.028
CrossRef - Borthakur P., Chinnasamy K., Paramasivam S.K., et al. Exogenous Melatonin as Pre- and Postharvest Application on Quality Attributes, Antioxidant Capacity, and Extension of Shelf Life of Papaya. Horticulturae. 2024;10(10):1099. doi:10.3390/horticulturae10101099
CrossRef - Fan S., Li Q., Feng S., et al. Melatonin Maintains Fruit Quality and Reduces Anthracnose in Postharvest Papaya via Enhancement of Antioxidants and Inhibition of Pathogen Development. Antioxidants. 2022;11(5): 804. doi:10.3390/antiox11050804
CrossRef - Guillén F., Medina-Santamarina J., García-Pastor M.E., et al. Postharvest melatonin treatment delays senescence and increases chilling tolerance in pineapple. LWT. 2022; 169:113989. doi:10.1016/j.lwt.2022.113989
CrossRef - Sangprayoon P., Supapvanich S., Youryon P., et al. Efficiency of salicylic acid or methyl jasmonate immersions on internal browning alleviation and physicochemical quality of Queen pineapple cv. “Sawi” fruit during cold storage. J Food Biochem. 2019; 43(12): e13059. doi:10.1111/jfbc.13059
CrossRef - Hong K, Xu H, Wang J, et al. Quality changes and internal browning developments of summer pineapple fruit during storage at different temperatures. Sci Hortic. 2013; 151: 68-74. doi:10.1016/j.scienta.2012.12.016
CrossRef - Aghdam M.S., Jannatizadeh A., Luo Z., Paliyath G. Ensuring sufficient intracellular ATP supplying and friendly extracellular ATP signaling attenuates stresses, delays senescence and maintains quality in horticultural crops during postharvest life. Trends Food Sci Technol. 2018;76: 67-81. doi:10.1016/j.tifs.2018.04.003
CrossRef - Bartolomé A.P., Rupérez P., Fúster C. Non-Volatile Organic Acids, pH and Titratable Acidity Changes in Pineapple Fruit Slices During Frozen Storage. J Sci Food Agric. 1996;70(4):475-480.doi:10.1002/(SICI)1097-0010(199604)70:4<475::AID- JSFA525>3.0.CO;2-K
CrossRef - Kumar D., Mishra D.S., Chakraborty B., et al. Pericarp browning and quality management of litchi fruit by antioxidants and salicylic acid during ambient storage. J Food Sci Technol. 2013;50(4):797-802. doi:10.1007/s13197-011-0384-2
CrossRef - Choudhary M.L., Dikshit S.N., Shukla N., et al. Evaluation of Guava (Psidium guajava L.) Varieties and Standardization of Recipe for Nectar Preparation. J Hortic Sci. 2008;3(2):161-163. doi:10.24154/jhs.v3i2.581
CrossRef - Lin Y., Fan L., Xia X., et al. Melatonin decreases resistance to postharvest green mold on citrus fruit by scavenging defense-related reactive oxygen species. Postharvest Biol Technol. 2019; 153: 21-30. doi:10.1016/j.postharvbio.2019.03.016
CrossRef - Pipliya S., Kumar S., Srivastav P.P. Impact of cold plasma and thermal treatment on the storage stability and shelf-life of pineapple juice: A comprehensive postharvest quality assessment. Food Phys. 2024;1:100025. doi:10.1016/j.foodp.2024.100025
CrossRef
Abbreviations List
ACO 1-Aminocyclopropane-1-Carboxylic Acid Oxidase
ACAS 1-Aminocyclopropane-1-Carboxylic Acid Synthase
ACS American Chemical Society
ANOVA Analysis of Variance
CE Catechin Equivalent
CRD Complete Randomized Design
DAS Days After Storage
DPPH 2,2-diphenyl-1-picrylhydrazyl
FD Fruit Diameter
FL Fruit Length
FW Fruit Weight
MDA Malondialdehyde
MT Melatonin
PDFD Percentage Decrease in Fruit Diameter
PDFL Percentage Decrease in Fruit Length
PLW Physiological Weight Loss
QE Quercetin Equivalent
ROS Reactive Oxygen Species
SA Salicylic Acid
SPSS Statistical Package for Social Science
TLC Thin Layer Chromatography
TSS Total Soluble Solids
UV-C Ultraviolet C













