Relationship Between Rapeseed Oil Intake and Inflammatory Cytokines: Results from NHANES 2003–2006 and Mendelian Randomization
1Department of Cardiology, Taihe Hospital, Hubei University of Medicine, Shiyan, China.
2Oil Crops Research Institute, Chinese Academy of Agricultural Sciences, Wuhan, China.
3Department of Cardiology, Zhongnan Hospital of Wuhan University, Wuhan, China.
4Institute of Myocardial Injury and Repair, Wuhan University, Wuhan, China.
Corresponding Author E-mail: 13035287850@163.com
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.3.29
ABSTRACT:Rapeseed oil (RO) has been extensively studied for its possible advantages in enhancing physical health. However, questions regarding its influence on inflammation and its suitability for incorporation into the diets of individuals with medical needs and those suffering from clinical conditions remain critical areas that require exploration. By utilizing the NHANES (National Health and Nutrition Examination Survey) database, researchers constructed models that accounted for various factors, including race, age, marital status, BMI, and caloric intake. The application of a two-sample Mendelian randomization (MR) technique enabled the identification of causal relationships informed by data from current genome-wide association studies. This examination included many methodologies, including random effects calculation, effect estimate, inverse variance weighted, MR-Egger, weighted median, simple mode, and weighted mode method. To reinforce prior findings, RO was administered to mice in experimental settings. The expression of inflammatory factors in the RO group significantly decreased. An analysis of the NHANES survey data demonstrated a negative relationship between RO consumption and CRP (C-reactive protein) levels among adults in the United States, with the β value of RO's 95% confidence interval being -0.08(-0.15, -0.01), aligning with outcomes from MR Analysis and subsequent animal research. The identified link between RO intake and diminished inflammation indicates the potential role of RO in moderating inflammatory responses, thereby highlighting the significance of RO in dietary choices for enhanced health outcomes.
KEYWORDS:CRP; Inflammatory Cytokines; Mendelian Randomization; NHANES; Rapeseed Oil
Introduction
Over the course of the past decades, rapeseed has developed into an important oilseed crop on a worldwide basis. At the moment, rapeseed oil (RO) is the third largest vegetable oil in terms of volume, following in the footsteps of palm oil and soybean oil.1 A total of 38 million metric tons of RO were produced worldwide, with Europe providing 63% and Canada contributing 31%.2 Rapeseed oil is one of the oils that is used the most commonly in the United States, falling behind only soybean oil in terms of its frequency of use.3
To ensure that the body is able to react effectively to tissue injury or infection, it is necessary for immune cells, inflammatory cells, and the chemicals that are associated with them to interact in a manner that is precisely regulated. Chronic inflammation, on the other hand, is defined by a prolonged, dysregulated, and maladaptive response that demonstrates the presence of continuous active inflammation, tissue damage, and ineffective attempts to cure the damage. Chronic inflammation, often known as non-resolving inflammation, is linked to a spectrum of chronic diseases, including coronary artery disease, obesity, malignancy, inflammatory bowel disease, autoimmune dysregulation and diabetes.4-9
Anti-inflammatory medications are commonly utilized for the treatment of these conditions. However, a significant number of these treatments possess side effects influenced by the dosage, and none are suitable for primary preventive measures, which considerably limits their application.10 Conversely, both one’s way of life and the surrounding environment are known to have a substantial impact on the inflammatory processes that occur. Among environmental factors, diet emerges as an essential component in modulating inflammation. There has been a significant advancement in understanding the influence of dietary selections on health, specifically through the modulation of inflammation by particular nutrients or dietary elements.11 Recent developments have resulted in a substantially more accurate understanding of this relationship. When it comes to lowering inflammation, there is some evidence to suggest that dietary methods that encourage a larger consumption of vegetables and oil may be beneficial.12-14 RO possesses the following characteristics: a low concentration of saturated fatty acids (SFAs) (7%), abundant monounsaturated fatty acids (MUFAs) and polyunsaturated fatty acids (PUFAs), including 61% oleic acid, 21% linoleic acid, and 11% alpha-linolenic acid (ALA),15-17 phytosterols (from 0.53% to 0.97%), and vitamin E (from 700 to 1,200 parts per million).18,19 It has been demonstrated via research that each of these components has anti-inflammatory properties.
Owing to the partial and contested20-22 data regarding the impacts of RO on inflammatory processes, this study investigates the association between RO and inflammation through an analysis of data from the National Health and Nutrition Examination Survey (NHANES) and utilizing Mendelian randomization (MR) techniques, and verifies it through experiments on mice.
Materials and Methods
NHANES study design and data sources
The NHANES functions as a biennial survey that provides a nationally representative overview of the population in the United States. Initiated in 1999, it has been carried out annually, assessing approximately 5,000 individuals each year. The collected data includes demographic, socioeconomic, dietary, and health-related information acquired through interviews and physical examinations conducted at participants’ homes using mobile examination units. This data collection program, sanctioned by the Ethics Review Board of the NCHS, necessitates obtaining signed consent from all participants following comprehensive information distribution.23 Researchers can utilize the available data, analytical techniques, and research instruments to aid in replicating the findings or the methodologies employed. The complete NHANES study design and dataset are accessible via https://www.cdc.gov/nchs/nhanes/.
Data extraction was conducted using the NHANES database, which included 20,470 participants, covering the period from 2003 to 2006. The following criteria were established for exclusion: elimination of records that did not have information on oil response or oil consumption (14,995 cases), absence of CRP data (375 cases), exclusion of female participants (2,715 cases), and data lacking additional covariates (159 cases). In total, 2,226 people satisfied the eligibility requirements and were incorporated into the study, as stated in Figure 1.
The study employed published studies and partnerships that provide publicly available aggregate data. All original investigations had permission from the appropriate ethical review board, and subjects provided informed consent. Moreover, the research did not utilize data at the individual level. Consequently, it is not required to obtain approval from a new ethics review committee.
CRP definition
The primary focus of this research was on CRP. Blood samples from participants of all genders, aged three years and older, were gathered, preserved, and sent to the University of Washington in Seattle, Washington.24 The measurement of CRP levels utilized the latex-enhanced nephelometry method (https://wwwn.cdc.gov/nchs/nhanes/).
MR analysis data sources
The schematic representation of the MR design is illustrated in Figure 3 (created with BioRender.com). MR is predicated on three principal assumptions: (1) that the genetic factors designated as instrumental variables (IVs) demonstrate a strong correlation with the exposure; (2) that genetic variation is independent of any potential confounding variables; and (3) that genetic variations affect the results solely via the exposure.25 This research employed summary-level data derived from well-established genome-wide association studies (GWAS) that investigated the connections between RO and 41 distinct circulating inflammatory regulators. By pinpointing genetic variants associated with RO, the study deduced causal relationships between RO and each individual circulating inflammatory factor.
The datasets that were used in this MR Analysis were obtained from summary GWAS data that was available to the general audience. The data on RO was gathered from the Open GWAS database maintained by the European Union. This data had 63,464 controls and 1,485 cases of European descent. In the current study, information was provided on inflammatory cytokines, including specifics on the genetic variant relationships for 41 cytokines and growth factors among 8,293 individuals of Finnish heritage.26 An explanation of the significance of ethical issues may be found above.
Selection of genetic instruments
SNPS related to exposure and with a significance threshold (p < 5×10-5) were used as IVs. These genetic variants were subsequently refined using linkage disequilibrium (LD) distance threshold of 10,000 kb and r2 < 0.001 to confirm their independence.27 Only those IVs with an F-statistic exceeding 10 were included.28,29 Subsequently, palindromic SNPs were eliminated.
Statistical analyses for MR
We adopt five methods for analysis: inverse variance weighting (IVW),30 weighted median,31 MR-Egger,32 and MR pleiotropic residuals and outliers (MR-PRESSO).33 Each of these methods works under different assumptions and deals with IVs that show horizontal pleiotropic effects. In the IVW approach, each SNP is treated as a valid genetic instrument, and the technique is based on the premise that pleiotropy does not exist.30 For the weighted median method to be applicable, a minimum of fifty percent of the instrumental variables must be available.31 It is possible for the MR-Egger approach to properly anticipate causal linkages even in circumstances in which all IVs are considered to be erroneous.32 By doing global testing, the MR-PRESSO approach identifies possible outliers in the instrumental variable, and then it provides unbiased estimates of causality by excluding the outliers that have been detected.33
The findings are presented as Odds Ratios (OR) along with their corresponding 95% confidence intervals (CI). All statistical analyses were conducted in R (version 4.3.2) using the TwoSampleMR and MR-PRESSO packages.
Animals and Procedures
In this study, six-week-old male mice with the ApoE−/− gene deficiency were employed. For the research, the mice were randomly assigned to two distinct groups: one group (n = 6) received a high-fat diet (HFD), while the other group (n = 6) was provided with a diet where the oil was substituted with RO. Blood samples were then collected from each mouse after a duration of 12 weeks on their respective diets.
Measurement of cytokine concentrations
The cytokines assessed included Eotaxin, IL-1B, and IL-2RA. Mouse serum cytokine levels were determined using an ELISA Kit (Thermo Fisher Scientific, Inc., Waltham, MA, USA; COIBO BIO Inc., Shanghai, China) following the guidelines provided by the manufacturer.
Statistics for ELISA experiments
The experimental outcomes were represented as mean ± standard deviation (SD). Data analysis was conducted using GraphPad Prism (California, USA). A student’s t-test was utilized to compare the two groups and identify statistical differences between the groups.
Results
Baseline characteristics of participants
After removing cases with incomplete covariate information, the total number was 2980. Data on fundamental population covariates, including BMI, kcal, and CRP, were found to be absent for some participants. Ultimately, the study included 2623 individuals. Within the analyzed group, the average age (mean (SD)) was 50.92 (18.85) years, comprising 1489 males (53.4%) and 1302 females (46.6%). Table 1 delineates the features of these people.
Table 1: Characteristics of participants according to CO intake NHANES, 2003-2006.
|
Characteristic |
Overall, N = 2226 (100%) | Other oils, N = 1466 (61%) | CO, N = 760 (39%) | P -value |
| Age (years), median (SD) | 37.5 (21.4) | 36.6 (20.9) | 38.9 (22.1) |
0.052 |
|
Race |
0.012 | |||
| 1 | 513 (9.2%) | 383 (11%) | 130 (5.6%) |
|
|
2 |
63 (2.8%) | 49 (3.4%) | 14 (1.8%) | |
| 3 | 1,650 (88%) | 1,034 (85%) | 616 (93%) |
|
|
Marital status (%) |
1,006 (57%) | 614 (55%) | 392 (59%) | 0.13 |
| CRP mg/L, median (SD) | 0.28 (0.75) | 0.31 (0.91) | 0.24 (0.38) |
>0.9 |
|
Diabetes (%) |
194 (9.3%) | 114 (8.5%) | 80 (11%) | 0.2 |
| Dyslipidemia (%) | 984 (53%) | 621 (50%) | 363 (57%) |
0.067 |
|
Hypertension (%) |
585 (30%) | 357 (28%) | 228 (34%) | 0.039 |
| Calorie(kcal/day) | 2,467 (860) | 2,466 (868) | 2,468 (847) |
>0.9 |
|
BMI (kg/m²), median (SD) |
26 (6) | 26 (6) | 27 (7) |
0.005 |
Race:1, Mexican American; 2, Other Hispanic; 3, Non-Hispanic White, Non-Hispanic Black, Other Race-Including Multi-Racial. SD, standard deviation.
Association of RO with CRP
The association between RO and CRP is illustrated in Figure 2. Within the model, when controlling for race, age, and marital status, BMI, kcal, and RO exhibited a significant negative correlation with CRP, with the β value of RO’s 95% confidence interval being -0.08(-0.15, -0.01).
 |
Figure 1: Flowchart detailing the procedure for identifying suitable participants for inclusion in the study. |
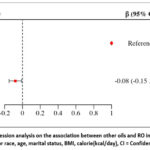 |
Figure 2: Linear regression analysis on the association between other oils and RO in the male subgroup. Adjusted for race, age, marital status, BMI, calorie(kcal/day), CI = Confidence Interval. |
The causal impact of RO on different inflammatory cytokines
The outcomes of all MR analyses are illustrated in Figure 4, which presents the p-values for every MR analysis method employed. Subsequently, we identified the positive outcomes of the MR analysis founded on the significant p-value obtained from the IVW. Notably, IL_1B, IL_2RA, and EOTAXIN exhibited significant negative correlations. Furthermore, the direction of the test results aligned consistently across all five methods, bolstering confidence in the actual causality.
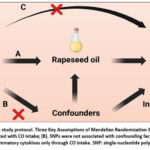 |
Figure 3: MR analysis study protocol. Three Key Assumptions of Mendelian Randomization Study. |
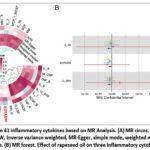 |
Figure 4: Effect of CO on 41 inflammatory cytokines based on MR Analysis. |
To evaluate the strength of the causal association between RO and CRP, a leave-one-out sensitivity analysis was performed for every SNP. Our findings revealed that the outcomes consistently aligned on one side of the zero line, regardless of the absence of any SNPs (Figure 5A). In addition, we illustrated the effect sizes of both IVW and MR-Egger, which appeared to match the significance that was found (Figure 5B). Furthermore, during the entirety of the MR Tests, the patterns were generally constant and statistically significant (Figure 5C). In conclusion, the funnel plot analysis revealed that the individual SNPs were distributed in a symmetrical manner on both sides of the vertical line in the IVW and MR-Egger assessments (Figure 5D).
 |
Figure 5: Figure presents a sensitivity analysis pertaining to the causal relationships between RO and CRP. |
In the MR study examining the association between RO and several inflammatory cytokines, we found SNPs associated with RO using the GWAS data. We employed many analytical techniques, including the IVW approach, MR-Egger method, weighted median methodology, and weighted mode analysis. The results indicated a possible correlation between RO and a diminished risk of EOTAXIN (OR: 0.125, 95% CI: 0.020, 0.778, p = 2.6e−02), IL_1B (OR: 0.028, 95% CI: 0.002, 0.507, p = 1.6e−02), and IL_2RA (OR: 0.055, 95% CI: 0.004, 0.818, p = 3.5e−02) while revealing no significant causal relationships between RO and other inflammatory cytokines (Figure 5). The Cochran’s Q test revealed an absence of heterogeneity (CO-EOTAXIN, Q = 20.527, p = 0.610; CO-IL_1B, Q = 24.024, p = 0.403; CO-IL_2RA, Q = 10.425, p = 0.988). Additionally, we conducted an evaluation of horizontal pleiotropy, which revealed no indication of directional pleiotropy (CO-EOTAXIN, intercept = 0.014, p = 0.787; CO-IL_1B, intercept = -0.020, p = 0.820; CO-IL_2RA, intercept = -0.047, p = 0.552). The MR-PRESSO global test demonstrated no horizontal pleiotropy effects (CO-EOTAXIN, p = 0.671; CO-IL_1B, p = 0.334; CO-IL_2RA, p = 0.998), so affirming that our MR results were unaffected by horizontal pleiotropy.
Validation of ELISA-based NHANES and MR analysis
Compared with the HFD group, serum EOTAXIN IL_1B, IL_2RA, and CRP were reduced in the RO group, validating the NHANES and MR analysis results (Figure 6).
 |
Figure 6: ELISA validation results in mice. (A) Staining of atherosclerotic lesions on the aorta by Oil red O in group HFD. (B) Staining of atherosclerotic lesions on the aorta by Oil Red O in group RO. |
Discussion
NHANES collected data from typical samples from multiple perspectives. What is unique is that through interviews and physical examinations, it involves information such as the basic population information, socio-economic conditions, diet and health, medical care, and physiological measurements of the samples. The findings obtained from the NHANES are utilized to identify disease risk factors as well as to assess the prevalence of significant health conditions, as well as to assess the relationship between nutritional status and health and disease prevention. They can also be applied in epidemiological and health science research, all of which serve the health of the nation.23 After adjusting the RO in the NHANES for race, age, marital status, BMI, and Calorie, a negative correlation between RO intake and CRP was obtained. The data of MR is derived from reliable research.34 The anti-inflammatory effect of RO was supported by the inverse correlation between RO and EOTAXIN, IL_1B, and IL_2RA by MR Analysis with 41 inflammatory cytokines, which were further validated in mice.
Abundant scientific research endorses the use of rapeseed oil, which not only positively impacts lipid levels but also serves as a crucial dietary element for enhancing health. The intake of rapeseed oil influences several other disease risk biomarkers through its effects on biological processes. According to the findings, rapeseed oil can significantly lower total and LDL cholesterol levels when compared to other dietary fat sources, along with additional benefits such as elevated tocopherol levels and better insulin sensitivity.35 RO has a controversial relationship with inflammation. A research project assigned adult male participants diagnosed with Type 2 Diabetes (T2D) to receive rapeseed oil enhanced with gam-oryzanol over a period of 12 weeks for laboratory assessment. In the ORZO group, significant reductions were observed in serum hs-CRP, MDA, IL-6, and IFN-γ levels, while serum IL_1B exhibited a slight decrease.36 A study examining the impact of RO on women with type 2 diabetes revealed a substantial reduction in CRP levels among the rapeseed oil group.37 The biological mechanisms that link RO to inflammation remain elusive. The high-carbohydrate diet based on rapeseed oil as a lipid source does not prevent hepatic fatty acid accumulation and inflammation.21 The results of our analysis, combined with previous studies, provide more support for the ameliorative effects of RO on inflammation.
RO is rich in oleic acid, vitamin E, and polyphenols. These ingredients play an important role in anti-inflammation.38-40 RO contains 61% oleic acid,35 and the anti-inflammatory effects of RO have been reported in many kinds of literature, such as a meta-analysis study that qualified 31 randomized controlled trials out of 719 identified studies involving 1634 participants. The results showed that supplementation of oleic acid significantly reduced C-reactive protein.41 Studies show that oleic acid can both effectively inhibit the growth of harmful bacteria linked to bacterial vaginosis and promote the growth of beneficial lactobacillus in the vaginal environment, presenting a new strategy for addressing this common gynecological issue.42 It is possible that oleic acid has the ability to directly control the production and activity of antioxidant enzymes. Additionally, its anti-inflammatory qualities may be attributed to the modulation of cytokine signaling, by suppressing pro-inflammatory mediators and enhancing anti-inflammatory ones. Additionally, oleic acid has the potential to modulate the production of microRNA, which can result in a beneficial anti-inflammatory impact.38 The tocopherols in RO (700-1,200 ppm) are one of the most potent nutrients known to regulate immune function.35 Studies have shown that vitamin E has unique antioxidant and anti-inflammatory properties that inhibit pro-inflammatory signal transduction and may help fight inflammation-related diseases.39 Polyphenols have received much attention due to their anti-inflammatory properties.40 and have shown significant alleviating effects on intestinal inflammation by acting on TLR4/NF-κB signalling pathway.43 The abundant anti-inflammatory elements in RO may significantly contribute to its anti-inflammatory effects.
Our research has several benefits. To guarantee the integrity of our results, we included suitable weights and modifications for confounding variables in our study. Second, this study is a large sample study using the NHANES and MR Databases. The results of our MR Analysis may provide more reliable conclusions because the MR Analysis is resistant to confounding and reverse causation. Third, our study examined the relationship between RO and CRP in American populations of different races, ages, marital status, BMI, and calories. This study aims to evaluate the validity of findings across variables, including ethnicity and marital status, in contrast to earlier studies that focused solely on certain groups. Fourth, the anti-inflammatory effect of RO was verified in mice, providing data support for the results of a large sample size.
However, limitations are inevitable. Firstly, the cross-sectional design of this study precludes a detailed exploration of the underlying mechanisms. In addition, as the data is self-reported, there may be deviations in accuracy. More in-depth randomized controlled trials are needed to conduct mechanism research in cells, animals, and humans, and further high-quality prospective studies should be carried out to obtain more definite results.
Conclusion
NHANES studies conducted in cross-section have indicated an inverse relationship between RO and CRP among adults in the United States. Similar results were obtained through MR Analysis, including EOTAXIN, IL_1B, and IL_2RA, and were verified by ELISA detection of the serum of the mouse model. These findings provide a theoretical basis and technical guidance for people’s future choice of edible oil. To verify these discoveries and explore the impact of RO consumption on inflammatory responses, forthcoming prospective research is warranted, and large-scale population cohort studies containing more information (such as region, race, diet, etc.) are needed.
Acknowledgement
The authors express their gratitude to the participants and investigators of the public database NHANES, the IEU Open GWAS project study, etc. The authors also thank Laura Corbin Nicholas Timpson (2020): Cytokines GWAS results in association with inflammatory factors in the American Journal of Human Genetics 100,40-50.
Funding Sources
This study was supported by the National Natural Science Foundation of China (82200974).
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding authors.
Ethics Statement
Approval for all animal experimental protocols was granted by the Ethics Committee, ensuring adherence to the relevant guidelines. All original studies have been approved by the corresponding ethical review board, and the participants have provided informed consent. This trial is registered at the NCHS Ethics Review Board (ERB) with the registration number: NCHS IRB/ERB Protocol #98-12; Protocol #2005-06; Protocol #2005-06.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trail Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Yaxi Xu: Conceptualization, Methodology, Data Curation, Formal Analysis, Writing-Original Draft.
- Ze Chen: Visualization, Supervision. Writing-Review and Editing.
- Xiaorui Duan: Visualization, Supervision. Writing-Review and Editing.
References
- Wittenberger K. USDA Economic Research Service. ERS/USDA Briefing Room–Soybeans and Oil Crops: Canola Seed, Oil, and Meal. 2012;
- GmbH IM. Oil World: Annual Global Analysis of All Major Oilseeds. Oils and oilmeals: supply demand and price outlook. vol 1. 2011.
- Agriculture USDo. Economic Research Service-Canola. Accessed April 2, 2013. Available at: http://www.ers.usda.gov/topics/crops/soybeans-oil-crops/canola.aspx#.UVpT8Vdk2PY.
- Weiss U. Inflammation. Nature. 2008;454(7203):427-427.
CrossRef - Ferguson LR, Laing WA. Chronic inflammation, mutation and human disease. Mutation Research. 2010;690(1-2):1-2.
CrossRef - Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140(6):883-899.
CrossRef - Libby P. Inflammation in atherosclerosis. Nature. Dec 19-26 2002;420(6917):868-74.
CrossRef - Nathan C. Epidemic inflammation: pondering obesity. Molecular Medicine. 2008;14:485-492.
CrossRef - Drouet M, Dubuquoy L, Desreumaux P, Bertin B. Visceral fat and gut inflammation. Nutrition. 2012;28(2):113-117.
CrossRef - Zhang Q, Ren Y, Mo Y, et al. Inhibiting Hv1 channel in peripheral sensory neurons attenuates chronic inflammatory pain and opioid side effects. Cell Research. 2022/05/01 2022;32(5):461-476.
CrossRef - Furman D, Campisi J, Verdin E, et al. Chronic inflammation in the etiology of disease across the life span. Nature Medicine. 2019/12/01 2019;25(12):1822-1832.
CrossRef - Bakker GC, Van Erk MJ, Pellis L, et al. An antiinflammatory dietary mix modulates inflammation and oxidative and metabolic stress in overweight men: a nutrigenomics approach. The American journal of clinical nutrition. 2010;91(4):1044-1059.
CrossRef - Galland L. Diet and inflammation. Nutrition in Clinical Practice. 2010;25(6):634-640.
CrossRef - Xiao S, Yu H, Xie Y, Guo Y, Fan J, Yao W. The anti-inflammatory potential of Cinnamomum camphora (L.) J.Presl essential oil in vitro and in vivo. Journal of Ethnopharmacology. 2021, 267, 113516.
CrossRef - Dupont J, White P, Johnston K, et al. Food safety and health effects of canola oil. Journal of the American College of Nutrition. 1989;8(5):360-375.
CrossRef - Johnson GH, Keast DR, Kris-Etherton PM. Dietary modeling shows that the substitution of canola oil for fats commonly used in the United States would increase compliance with dietary recommendations for fatty acids. Journal of the American Dietetic Association. 2007;107(10):1726-1734.
CrossRef - Matthaus B, Özcan MM, Al Juhaimi F. Some rape/canola seed oils: fatty acid composition and tocopherols. Z Naturforsch C J Biosci. Mar 2016;71(3-4):73-7. doi:10.1515/znc-2016-0003
CrossRef - Gunstone F. Vegetable oils in food technology: composition, properties and uses. John Wiley & Sons; 2011.
CrossRef - Orlovius K. Oilseed rape. Fertilizing for high yield and quality, Bulletin. 2003;16
- Lin L, Allemekinders H, Dansby A, et al. Evidence of health benefits of canola oil. Nutrition reviews. 2013;71(6):370-385.
CrossRef - Antunes MM, Godoy G, Fernandes IdL, et al. The dietary replacement of soybean oil by canola oil does not prevent liver fatty acid accumulation and liver inflammation in mice. Nutrients. 2020;12(12):3667.
CrossRef - Atefi M, Pishdad GR, Faghih S. The effects of canola and olive oils on insulin resistance, inflammation and oxidative stress in women with type 2 diabetes: a randomized and controlled trial. Journal of Diabetes & Metabolic Disorders. 2018;17:85-91.
CrossRef - Zipf G, Chiappa M, Porter KS, Ostchega Y, Lewis BG, Dostal J. National health and nutrition examination survey: plan and operations, 1999-2010. Vital Health Stat 1. Aug 2013;(56):1-37.
- Sun J, Wang W, Li D, et al. Association between C-Reactive protein and periodontitis in an obese population from the NHANES 2009–2010. BMC Oral Health. 2023;23(1):512.
CrossRef - Davies NM, Holmes MV, George DS. Reading Mendelian randomisation studies: a guide, glossary, and checklist for clinicians. Bmj Clinical Research. 2018;362:k601.
CrossRef - Ahola-Olli AV, Würtz P, Havulinna AS, et al. Genome-wide association study identifies 27 loci influencing concentrations of circulating cytokines and growth factors. The American Journal of Human Genetics. 2017;100(1):40-50.
CrossRef - A global reference for human genetic variation. Nature. 2015;526(7571):68-74.
CrossRef - Pierce BL, Ahsan H. Power and instrument strength requirements for Mendelian randomization studies using multiple genetic variants. International Journal of Epidemiology. 2011;40(3):740-52.
CrossRef - Brion MJ, Shakhbazov K, Visscher PM. Calculating statistical power in Mendelian randomization studies. Int J Epidemiol. Oct 2013;42(5):1497-501.
CrossRef - Burgess S, Butterworth A, Thompson SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol. Nov 2013;37(7):658-65.
CrossRef - Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol. May 2016;40(4):304-14.
CrossRef - Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol. Apr 2015;44(2):512-25.
CrossRef - Verbanck M, Chen CY, Neale B, Do R. Publisher Correction: Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. Aug 2018;50(8):1196.
CrossRef - Elsworth B, Lyon M, Alexander T, et al. The MRC IEU OpenGWAS data infrastructure. BioRxiv. 2020:2020.08. 10.244293.
CrossRef - Lin L, Allemekinders H, Dansby A, et al. Evidence of health benefits of canola oil. Nutr Rev. Jun 2013;71(6):370-85.
CrossRef - Nikooyeh B, Zargaraan A, Ebrahimof S, et al. Added γ-oryzanol boosted anti-inflammatory effects of canola oil in adult subjects with type 2 diabetes: a randomized controlled clinical trial. European Journal of Nutrition. 2024;63(2):425-433.
CrossRef - Atefi M, Pishdad GR, Faghih S. The effects of canola and olive oils on insulin resistance, inflammation and oxidative stress in women with type 2 diabetes: a randomized and controlled trial. Journal of Diabetes & Metabolic Disorders. 2018;17(2):85-91.
CrossRef - Santa-María C, López-Enríquez S, Montserrat-de la Paz S, et al. Update on Anti-Inflammatory Molecular Mechanisms Induced by Oleic Acid. Nutrients. Jan 1 2023;15(1).
CrossRef - Jiang Q. Natural forms of vitamin E: metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic Biol Med. Jul 2014;72:76-90.
CrossRef - Natsume M. Polyphenols: Inflammation. Curr Pharm Des. 2018;24(2):191-202.
CrossRef - Wang Q, Liu R, Chang M, Zhang H, Jin Q, Wang X. Dietary oleic acid supplementation and blood inflammatory markers: a systematic review and meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. 2022;62(9):2508-2525.
CrossRef - Zhu M, Frank MW, Radka CD, et al. Vaginal Lactobacillus fatty acid response mechanisms reveal a metabolite-targeted strategy for bacterial vaginosis treatment. Cell. 2024;187(19):5413-5430.e29.
CrossRef - Yu C, Wang D, Yang Z, Wang T. Pharmacological Effects of Polyphenol Phytochemicals on the Intestinal Inflammation via Targeting TLR4/NF-κB Signaling Pathway. Int J Mol Sci. Jun 22 2022;23(13).
CrossRef













