Enhancing the Nutritional Value of Dried Figs: Reducing Sugar Content through Lactococcus lactic Fermentation
1Department of Food Science, Carthage University, Tunis, Tunisia
2Department of Biological Engineering, Tunisia Private University (ULT), Tunis, Tunisia
3Department of Agraria, Mediterranea University of Reggio Calabria, Reggio Calabria, Italy
4Laboratory of Management and Valorization of Forest Resources, National Research Institute of Water, Forests and Rural Engineering, University of Carthage, Ariana, Tunisia
5Department of Biotechnology, Center of Biotechnology of Borj Cédria, Hammam-Lif, Tunisia
Corresponding Author Email: amgiuffre@unirc.it
Download this article as:
ABSTRACT:Nowadays, the increasing awareness of the importance of a healthy diet has expanded beyond just those who are ill or athletic, affecting the entire population as they recognize the harmful consequences of poor nutrition. It is within this dynamic context that our research is positioned. The primary objective of this research is to enhance the nutritional value of dried figs by preserving their inherent nutritional benefits while reducing their sugar content through lactic fermentation. After conducting comprehensive screenings of various microorganisms, Lactococcus lactis was identified as the optimal bacterial strain due to its efficacy in lowering sugar levels in dried figs. Three fermentation periods (24h, 48h, and 72h) were carefully selected to optimize the process. In addition to assessing sugar content, our study aims to maintain the nutritional benefits of dried figs throughout the fermentation process. Comprehensive characterizations performed before and after fermentation have demonstrated that protein levels and total polyphenols were not significantly impacted by lactic fermentation, apart from a slight increase in the inhibition percentage of antiradical activity. Notably, the sugar content decreased by 31% following the 72-hour fermentation process. The results have clearly demonstrated our capacity to create fermented dried figs that not only possess high nutritional value but also significantly lower sugar content. This approach marks an innovative breakthrough in food science, providing a model for tackling the wider challenges associated with sugar in our diets.
KEYWORDS:Dried Figs; Lactic Fermentation; Nutrition; Polyphenols; Sugar
Introduction
The high prevalence of sugar in most foods remains a significant cause of numerous diseases, highlighting that sugar is not exclusively confined to processed foods but it is also found in natural foods. An example illustrating this nutritional duality is that of dried figs. Although recognized for their antioxidant properties, and abundance of vitamins, fibers, minerals, and proteins, these natural delights present a significant drawback which lies in their high sugar content.
Fruits are naturally abundant in water and sugar, making them highly perishable. To preserve them out of season, sun drying or traditional drying techniques have been used since ancient times.1 This straightforward yet practical method relies on removing water from the fruit’s surface through the natural convection of warm air.
Dried figs are notable in the current market not only for their exceptional sensory qualities but also for their nutritional advantages. Their consumption greatly enhances antioxidant capacity, helping to mitigate damage from free radicals due to their high polyphenol content, particularly flavonoids. Among dried fruits, figs are recognized as the richest source of flavonoids.2 A comparative study has shown that, for the same quantity, dried figs contain more than three times the fiber content of fresh figs.3 This highlights the significance of dried figs in supporting digestion and ensuring healthy intestinal transit. In addition to their fiber content, dried figs are abundant in vitamins, especially vitamin E. This vitamin not only protects against oxidative stress but also plays a crucial role in cellular renewal and regeneration, thereby promoting skin healing.4 Furthermore, dried figs are fundamental for their content in minerals (phosphorus and calcium) which benefit bone health, as well as potassium, which plays a role in regulating blood pressure.5
However, dried figs have a major drawback due to their high sugar content, as indicated by. For instance, 100 g of fresh figs contain around 20 g of total carbohydrates, while the same quantity of dried figs provides 63.87 g of carbohydrates, having almost the same percentage of glucose and fructose as predominant sugars.6 This reality makes this product not recommended for diabetics, overweight individuals, and even cancer patients. Indeed, according to a study,7 cancer cells have a higher sugar consumption than normal cells. Additionally, several studies have established a link between sugar consumption and various conditions, such as Alzheimer’s disease,8 polycystic ovary syndrome,9 and insulin resistance, which is the main cause of type 2 diabetes.10
Fermentation is a crucial process utilized in the food industry, playing an important role in the production and preservation of various food items. During fermentation, yeasts, bacteria and molds convert sugars into gases or alcohol.
This biotechnological method enhances the flavor and texture of foods and improves the shelf life and the nutritional value. Common examples include the production of yogurt, cheese, bread, and beverages such as wine, konbucha and beer.11-14
Lactic fermentation is a process known for preserving and stabilizing products, currently attracts increased attention from researchers due to its added benefits. A recent study has highlighted the positive effects of lactic fermentation on pseudocereals, showing that this process boosts the levels of polyphenol and even B-group vitamins, thanks to the action of lactic bacteria.15
Recently, this lactic fermentation technique has been also employed in fruit juice production, resulting in functional beverages that offer substantial nutritional benefits.16 Therefore, we have opted for the application of this method to reduce sugar levels in dried figs while preserving their nutritional values. This innovative approach aligns with the current trend which emphases the awareness of the benefits of a balanced diet, supporting ongoing choices towards healthier eating habits.
In this context, the aim of our study is to improve the nutritional advantages and the biological properties of dried figs by utilizing lactic fermentation, aiming to produce a dietary food designed for a specific target population.
Materials and Methods
Characterization of Dried Figs
The dried figs used in this study were commercially available “Reine des Graines” figs, which are among the most widely consumed figs and originate from Tunisia. Prior to analysis, the plant matrix underwent thorough washing and was processed through steaming.
Determination of Dry Matter
For the determination of dry matter content, a 5 g sample was subjected to a temperature of 105 °C for 3 hours under atmospheric pressure. The dry matter percentage (%DM) was calculated as follows:
![]()
where:
P0 = Weight of empty container in grams
P1 = Weight of fresh sample with container in grams
P2 = Weight of dry sample with container in grams
Sugar Assay
The colorimetric method, as outlined by Dubois et al.,17 has been employed to assess sugar content.
The plant matrix of dried figs was ground and then subjected to extraction with distilled water for 4 hours. The centrifugation was conducted at 10,000 rpm (20 min), after that the supernatant was recovered.
In glass assay tubes, 1 ml of the sample was placed in an ice bath. Subsequently, 1 ml of a 5% aqueous solution of phenol was added, followed by homogenization. Then, with caution, 5 ml of sulfuric acid H2SO4 (96%) were added. This mixture was vortexed under the hood. After cooling in ice, the tubes were incubated in darkness for 30 minutes, then read using a spectrophotometer (SHIMADZU, Kyoto, Japan) at 485 nm.
Finally, the total sugar contents were determined according to the reference calibration curve made with D-glucose, (y = 11.95x+ 3E-16), (Figure 1).
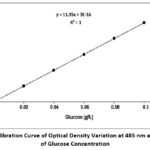 |
Figure 1: Calibration Curve of Optical Density Variation at 485 nm as a Function of Glucose Concentration |
Protein Assay
The Kjeldahl method was employed to determine the protein concentration, primarily involving the measurement of total nitrogen and utilizing a conversion factor to obtain the protein concentration. To carry out this procedure, 5 g ground samples of dried figs were used for the assay.
Following titration, the total nitrogen content was obtained, which was then multiplied by a fixed conversion factor of 6.25 as per literature standards for fruits. This multiplication has allowed us to estimate the precise protein content in this plant matrix.
Mineral Content Assay
Samples weighing 28 g were placed in crucibles in a furnace for 4 hours at 550 °C until white ashes were obtained. Once incinerated, the samples were placed in a desiccator to avoid regaining any moisture. The total ashes content (%) was calculated as follows:
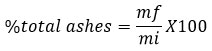
where:
mf: final mass of the sample,
mi: initial mass of the sample.
Total Polyphenol Assay
The polyphenol content was determined following the preparation of extracts from dried figs. Initially, the figs were ground under liquid nitrogen using a mortar and pestle. The resulting powders were then subjected to solid-liquid extraction using 80% ethanol, agitating for a period of 24 hours. The resulting mixture was centrifuged at 10,000 rpm for 20 minutes at 4 °C, allowing the recovery of the supernatant containing polyphenols, which was concentrated by rotary evaporation after filtration.
The total polyphenolic content was performed by spectrophotometry, utilizing the colorimetric method with the Folin-Ciocalteu reagent. In an Eppendorf tube, distilled water and the Folin-Ciocalteu reagent were added to the extract. Subsequently, sodium carbonate (Na2CO3; 7%) was introduced into the reaction mixture, and the volume was adjusted with distilled water. The absorbance was read at 760 nm after incubation at room temperature (90 min).
The quantification of polyphenols in the extracts was performed by referencing a standard curve established with Gallic Acid (0-31.25 μg) (y = 0.0716x). The total phenolic content was expressed as milligrams of gallic acid equivalent per gram of dry extract. The analyses were conducted in triplicate.
Evaluation of Antiradical Activity
The assessment of the antiradical activity of extracts from dried figs, prepared similarly to the method used for total polyphenol evaluation, was conducted based on the reduction of the DPPH radical. This reduction is characterized by the color transition of DPPH from purple to yellow, along with a decrease in optical density (OD) at 515 nm, expressed as the percentage reduction of DPPH. Three DPPH solutions, each with varying concentrations of dried fig extracts, were prepared and incubated for 30 minutes in the dark, followed by OD measurement. Graphically determining the IC50 from the variation in the percentage reduction of DPPH concerning the concentration of the extracts, the IC50 represents the antioxidant concentration needed to inhibit 50% of the DPPH radical, thereby providing a quantitative measure of the antiradical efficiency of the dried fig extracts.
Solid-State Fermentation
In order to ferment dried figs, Lactococcus lactis was selected following preliminary trials that demonstrated its fermentative capability with this substrate. To achieve this, the reactivation of the lyophilized bacterial species is conducted through successive passages in MRS nutrient agar. During the fermentation process, dried figs have served as the sole carbon source for the growth of the lactic acid bacteria.
Inoculum Preparation
The lactic acid bacteria (LAB) are initially inoculated on nutrient agar at 30 °C. Subsequently, 4 to 5 well-isolated bacterial colonies are suspended in nutrient broth (or physiological saline solution at 0.9% NaCl). This suspension is then adjusted to the McFarland standard 0.5 using a spectrophotometer, corresponding to an optical density (OD) read at 625 nm, equivalent to a suspension containing approximately 108 CFU/ml.
Solid-State fermentation
In Petri dishes, 30 g of dried figs were cut into equal pieces with a thickness of 1 cm each to ensure a uniform distribution of the inoculum. Subsequently, the samples were inoculated with 108 CFU/g. The process was conducted at 30 °C for 24h, 48h, and 72h.
The selection of the three fermentation time points was based on previous work on lactic acid fermentation of fruit-based substrates, where similar fermentation frameworks are commonly applied. Several studies have demonstrated that Lactococcus and other lactic acid bacteria show consistent metabolic activity and adaptation patterns when fermenting fruits.18,19 These works provided the scientific basis for designing our fermentation schedule.
The choice of 72 h as the endpoint was guided by practical considerations linked to product quality. Extending fermentation beyond 72 h increases the risk of excessive acidification and undesirable changes in taste and texture, which would negatively affect the sensory properties of dried figs. For this reason, fermentation was stopped at 72 h to avoid altering the characteristic flavor and integrity of the figs.
Comparison between fermented and non-fermented dried figs
Following the fermentation of dried figs over periods of 24h, 48h, and 72h, a comprehensive analysis has been conducted with a dual purpose. Firstly, the evaluation aimed to ascertain the utilization of sugars by the employed lactic acid bacteria, determined through sugar content analyses. Concurrently, careful attention was devoted to ensure that this lactic fermentation has no adverse impact on the inherent nutritional properties of dried figs. To this end, protein, mineral, total polyphenol, and antiradical activity assays have been meticulously repeated after each fermentation interval (24h, 48h, and 72h).
Statistical analysis
For all analyses of both fermented and non-fermented dried figs, three repetitions were used. An Anova test was applied to assess the differences between the control group and the samples that have been exposed to lactic fermentation (24h, 48h, 72h). The significant differences were detected at p < 0.05 and F > Fcritical, the groups were compared by a post-hoc test. The Tukey’s test was used to compare each experimental group with the control group. SPSS 20.0 was used to treat the data.
Results
In the field of microbiology, fermentation plays a key role as a fundamental metabolic process. It is essential for breaking down organic materials to produce energy and various metabolites. Lactic acid bacteria, such as Lactococcus lactis, have played a significant role in this process, particularly within the food industry where they are utilized for fermenting various products. Preliminary trials were conducted to assess the ability of Lactococcus lactis to assimilate the sugar present in dried figs. This preliminary finding provided a promising basis for further exploring the fermentative potential of this bacterium on dried figs and its potential implication in sugar reduction and subsequent health outcomes (Table 1).
Table 1: Comparison between non- fermented and fermented dried figs
|
Analysis |
T0 | SSF 24h | SSF 48h | SSF 72h |
| Humidity (%) | 18a | 18a | 18a |
18a |
|
Sugar assay (g/100 g) |
40.04±0.02a | 29.76±0.1b | 29.15±0.1b | 27.66±0.01c |
| Protein assay (g/100 g) | 2.9±0.03a | 2.9±0.01a | 2.9±0.1a |
2.9±0. 1a |
|
Mineral content assay (g/100 g) |
7.25±0.02a | 4.46±0.03b | 4.46±0.01b | 4.46±0.02b |
| Total Polyphenol (mg GAE/g) | 2.91±0.039b | 2.76±0.02b | 2.83±0.005b |
3±0.01a |
|
Anti-radical air activity: inhibition percentage (%) |
69.4±0.1b | 55.4±0.2c | 62.5±0.02bc | 88±0.12a |
| Anti-radical activity: IC50 (mg/ml) | 4.25±0.23c | 5.40±0.19a | 5.08±0.42b,a |
4.23±0.11c |
Measurements are expressed as mean±standard deviation. Different small letters in the same line show significant differences between samples at a 95% confidence level.
Sugar Analysis
Initially (T0), the sugar content was at 40.04 g/100 g, following lactic fermentation on solid media, sugar levels gradually decreased to 29.76 g/100 g after 24 hours, further dropping to 29.15 g/100 g after 48 hours, and finally stabilizing at 27.66 g/100 g after 72 hours of fermentation, representing a 30.91% decrease in sugar content. A significant decrease (p<0.05) was found in total sugar content in dried figs after fermentation by Lactococcus lactis. This decrease aligns with expectations, given the nature of fermentation processes. This marked reduction underscores the efficiency of fermentation and the bacterium’s adeptness at utilizing sugar as a substrate.
Protein Analysis
Table 1 shows that our matrix contains 2.9% protein. The fermentation process used in our study did not affect this content (p> 0.05), as the results demonstrated that protein levels remained stable at 2.9% after the three fermentation intervals.
Mineral Analysis
Our study revealed that lactic fermentation had a negative effect on mineral content. The results indicated that before fermentation, the ash content, and thus the mineral content, was 7.25%, a result close to those found in certain studies on dried figs.20,21 After 24 hours, 48 hours, and 72 hours of fermentation, the mineral content decreased (p<0.05) and stabilized at 4.46% (Table 1).
Polyphenol Analysis
The obtained results were expressed as milligrams of gallic acid equivalents per gram of dry extract (mg GAE/g). Initially, the matrix of dried figs contained approximately 2.91±0.039 mg GAE/g (Figure 2). A slight and not statistically significant decrease in polyphenol content (p > 0.05) was observed after 24 and 48 hours of fermentation with values reaching 2.76 ± 0.02 mg GAE/g at 24 hours and 2.83 ± 0.005 mg GAE/g at 48 hours. After 72 hours of fermentation, the total polyphenol content showed a significant difference (p < 0.05) reaching 3 ± 0.01 mg GAE/g.
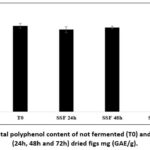 |
Figure 2: Total polyphenol content of not fermented (T0) and fermented (24h, 48h and 72h) dried figs mg (GAE/g). |
Anti-Radical Activity Analysis
Regarding the anti-radical activity tested on our product, it can be observed that at T0, before fermentation, and at a concentration of 6.25 mg/ml of extract used, we observed an average inhibition of 69.4 ± 5%, according to Ouchemoukh et al.2 After the first fermentation period (24 hours), the average inhibition decreased to 55.4 ± 1.5% (p<0.05). Following 48 hours of fermentation, this inhibition rose to 62.5 ± 4.8% (Figure 3), and a significant difference (p<0.05) was observed after 72 hours of fermentation, with the inhibition percentage reaching its maximum at 88 ± 0.2% inhibition for the same extract concentration, and an IC50 of 4.23 ± 0.11 mg/ml.
 |
Figure 3: Evaluation of antiradical activity: inhibition percentage (%) of not fermented (T0) and fermented (24h, 48h and 72h) dried figs. |
Discussion
In the microbiological field, fermentation represents an essential metabolic process involving the degradation of organic substrates to produce energy and metabolites. Lactic acid bacteria, such as Lactococcus lactis, play a prominent role in this mechanism, particularly in the food industry where they are employed to ferment various products. Our study focused on the ability of Lactococcus lactis to ferment dried figs and its implications for sugar reduction, as well as the resulting health benefits.
The results of our research highlighted a significant decrease in sugar content in dried figs following fermentation by Lactococcus lactis. Initially (T0), the sugar content was 40.04 g/100g, results that align with those reported in a study characterizing Tunisian dried figs, where a rate of 35.064 g/100g was found.22 After lactic fermentation on solid medium, the sugar content gradually decreased to 29.76 g/100g after 24 hours, 29.15 g/100g after 48 hours, and finally to 27.66 g/100g after 72 hours of fermentation. A study on lactic fermentation of fig juice also demonstrated similar results after 72 hours of lactic fermentation, indicating a sugar reduction from 32.1 mg/ml to 18.2 mg/ml.23 This decrease in sugar content confirms the efficacy of fermentation and the ability of this bacterium to utilize sugar as a substrate.
Indeed, Lactococcus lactis is a homofermentative lactic acid bacterium that assimilates sugar through the glycolysis pathway. During this process, sugar is degraded into metabolic products such as pyruvate. Some of these products are utilized for bacterial growth and multiplication, while the rest is transformed into lactic acid, which is the main product of lactic fermentation. This metabolic bifurcation, where a fraction of the sugar is allocated to growth and the rest to energy, is crucial for the survival and development of the bacterium.24
The health implications of this reduction in sugar in dried figs are numerous. Decreasing sugar content offers nutritional benefits by reducing the amount of available calories and carbohydrates. In a context where overconsumption of sugar is associated with various chronic diseases, this reduction is of paramount importance. In this regard, a study conducted in France established a direct link between sugar consumption and the risks of cancer, particularly breast and prostate cancers. The results of this study demonstrated that not only added sugars but also natural sugars present in fruits and dried fruits are implicated.25
Furthermore, the by-product of fermentation, lactic acid, is recognized for its beneficial properties for intestinal health. By promoting the balance of intestinal flora and strengthening the intestinal barrier, lactic acid and lactic acid bacteria can contribute to preventing gastrointestinal disorders and combating certain food poisonings.26 Regarding protein content, our results confirmed that lactic fermentation had no impact on protein levels. Before and after the fermentation process, the protein content remained unchanged at 3%. These findings are consistent with those obtained by Nemiche et al.27 in their study on the characterization of Algerian dried figs, where a protein rate of 3.2% was reported. However, our study revealed that lactic fermentation had a negative effect on mineral content. The results indicate that before fermentation, the mineral rate was 7.25%, and after 24h, 48h, and 72h of fermentation, it stabilized at 4.46%. This decrease may be attributed to the assimilation of minerals by lactic acid bacteria, which require the presence of vitamins and minerals for their growth.
Polyphenols are well-known for their nutritional value, particularly in fruits, and figs—whether enjoyed fresh or dried— that stand out as especially rich sources of these beneficial compounds. For this study, the extraction of phenolic compounds using ethanol was chosen as the literature suggests that solid-liquid extraction of fig polyphenols using methanol or ethanol has been effective.3 Our findings align with other studies conducted on figs and dried figs in Tunisia, which revealed a total polyphenol range between 1.21 and 2.77 mg GAE/g.22 Total polyphenol content remained stable throughout the first 48 hours of fermentation, showing no significant variation. A slight but statistically significant increase was observed after 72 hours, rising from 2.91 mg GAE/g at T0 to 3.00 mg GAE/g. Although this increase is minimal, it confirms that lactic fermentation preserves these beneficial compounds, which is a valuable outcome. Moreover, aside from the fact that lactic fermentation did not result in polyphenol degradation, other studies have shown that fermentation can enhance the bioavailability of polyphenols. Ruiz Rodríguez et al.16 demonstrated that fermentation can increase the bioavailability of phenolic compounds in humans. Although fruits are naturally rich in polyphenols, some of these compounds exhibit low intestinal bioavailability due to their structural complexity. Lactic fermentation can promote the production of simpler and more easily absorbable phenolic compounds in the duodenum.28
The increase in anti-radical activity observed in our study is supported by a study on pomegranate juice fermentation with Lactobacillus plantarum, where a 40% increase in anti-radical activity was observed.29 This increase in anti-radical activity is not necessarily accompanied by an increase in total polyphenols, according to Li et al.30 who studied the lactic fermentation effect on apple juice. The results of this study proved that anti-radical activity can increase even in the presence of a decrease in total polyphenols, due to changes in the profile of phenolic compounds induced by fermentation. Indeed, lactic acid bacteria can promote the increase of polyphenols with strong antioxidant power, which can result in a positive balance in terms of anti-radical activity.
In conclusion, the lactic fermentation of dried figs by Lactococcus lactis results in significant modifications in sugar content, mineral content, and anti-radical activity, while maintaining stable protein and polyphenol levels (Figure 4). These changes have potential health benefits, particularly in reducing sugar intake and enhancing antioxidant properties.
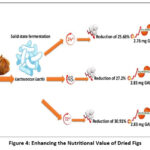 |
Figure 4: Enhancing the Nutritional Value of Dried Figs |
Conclusion
Our study has highlighted the effectiveness of Lactococcus lactis in fermenting dried figs, resulting in a significant reduction in sugar content. This approach holds crucial importance for the valorization of dried figs, as it preserves their nutritional benefits while enhancing their status as a functional food. These findings offer promising prospects for food processing strategies aimed at reducing sugar content, as well as for promoting intestinal health through the presence of lactic acid bacteria. The results also pave the way for future research to explore potential applications of this strategy in the food industry and to deepen our understanding of its effects on human health.It should be noted that this study is preliminary and primarily aimed at evaluating whether lactic acid fermentation could effectively reduce sugar content in dried figs while assessing potential changes in total polyphenols, protein, minerals, and antioxidant activity. Parameters such as pH, lactic acid content, detailed sugar composition, and individual mineral changes were not analyzed in this preliminary study. Future studies will include these measurements to provide a more comprehensive assessment of sugar reduction, nutrient retention, and functional property enhancement in fermented dried figs.
Acknowledgement
This work was conducted within the framework of a scientific collaboration between Prof. Hanene Ghazghazi (National Institute of Research in Rural Engineering, Water and Forests [INRGREF]) and Prof. Angelo Maria Giuffrè (Department of AGRARIA, University of Studies “Mediterranea” of Reggio Calabria, 89124 Reggio Calabria, Italy).
Funding Sources
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not applicable
Author Contributions
- Chrak Charfi: Conceptualization; Software; Formal analysis; Investigation; Resources; Data curation; Writing – original draft; Writing – review and editing.
- Sarra Rafai: Formal analysis; Investigation; Writing – original draft; Writing – review and editing.
- Amani Kahlaoui: Methodology; Writing – original draft
- Angelo Maria Giuffrè: Software; Resources; Writing – Review and Editing; Visualization; Project administration.
- Hanene Ghazghazi: Methodology; Writing – review and editing; Supervision.
- Ines Karkouch: Methodology; Resources; Supervision.
References
- Ouchemoukh S., Hachoud S., Boudraham H., et al. Antioxidant activities of some dried fruits consumed in Algeria. LWT – Food Sci Technol. 2012;49(2):329–332. https://doi.org/10.1016/j.lwt.2012.07.022
CrossRef - Mat Desa W. N., Mohammad M., Fudholi, A. Review of drying technology of fig. Trends in Food Sci Technol. 2019;88:93–103. https://doi.org/10.1016/j.tifs.2019.03.018
CrossRef - Arvaniti O. S., Samaras Y., Gatidou G., et al. Review on fresh and dried figs: Chemical analysis and occurrence of phytochemical compounds, antioxidant capacity and health effects. Food Res Int. 2019;119:44–267. https://doi.org/10.1016/j.foodres.2019.01.055
CrossRef - Zingg J. Vitamin E: Regulatory Role on Signal Transduction. IUBMB Life, 2019;71(4):456–478. https://doi.org/10.1002/iub.1986
CrossRef - Sedaghat S., Rahemi, M. Effects of physio-chemical changes during fruit development on nutritional quality of fig (Ficus carica var. ‘Sabz’) under rain-fed condition. Sci Hort. 2018;237:44–50. https://doi.org/10.1016/j.scienta.2018.04.003
CrossRef - Mahmoudi S., Chawla A. Fig and health. In A. Sarkhosh, A. Yavari, and L. Ferguson (Eds.). (2022). The fig: botany, production and uses (pp. 421–452). CABI. https://doi.org/10.1079/9781789242881.0018
CrossRef - Fadaka A., Ajiboye B., Ojo O., et al. Biology of glucose metabolization in cancer cells. J Oncological Sci. 2017;3(2):45–51. https://doi.org/10.1016/j.jons.2017.06.002
CrossRef - Liu L., Volpe S. L., Ross J. A., et al. Dietary sugar intake and risk of Alzheimer’s disease in older women. Neurosci. 2022;25(11):2302–2313. https://doi.org/10.1080/1028415X.2021.1959099
CrossRef - Xenou M., Gourounti K. Dietary Patterns and Polycystic Ovary Syndrome: a Systematic Review. Mædica, 2021;6(3):516–521. https://doi.org/10.26574/maedica.2020.16.3.516
CrossRef - Galicia-Garcia U., Benito-Vicente A., Jebari S., et al. Pathophysiology of Type 2 Diabetes Mellitus. International J Mol Sci. 2020;21(17):6275. https://doi.org/10.3390/ijms21176275
CrossRef - Giuffré A.M., Zappia C., Capocasale M., et al. Vinegar Production to Valorize Citrus bergamia Eur Food Res Technol. 2019;245(3):667-675. DOI: https://doi.org/10.1007/s00217-018-3189-y
CrossRef - Sidari R., Martorana A., Zappia C., et al. Persistence and effect of a multi-strain starter culture on antioxidant and rheological properties of novel wheat sourdoughs and breads. Foods 2020;9(9):1258. doi:10.3390/foods9091258
CrossRef - Allaith S.A., Abdel-Aziz M.E., Thabit Z.A., et al. Screening and Molecular Identification of Lactic Acid Bacteria Producing β-Glucan in Boza and Cider. Fermentation 2022;8(8) :350. https://doi.org/10.3390/ fermentation8080350
CrossRef - Giuffrè D., Giuffrè A.M. Fermentation Technology and Functional Foods. Frontiers in Bioscience (Elite Ed) 2024;16(1):8. https://doi.org/10.31083/j.fbe1601008
CrossRef - Rollán G. C., Gerez C. L., LeBlanc J. G. Lactic Fermentation as a Strategy to Improve the Nutritional and Functional Values of Pseudocereals. Front Nutr. 2019;6:98. https://www.frontiersin.org/articles/10.3389/fnut.2019.00098
CrossRef - Ruiz Rodríguez L. G., Zamora Gasga V. M., Pescuma M., et al. Fruits and fruit by-products as sources of bioactive compounds. Benefits and trends of lactic acid fermentation in the development of novel fruit-based functional beverages. Food Res Int. 2021;40: https://doi.org/10.1016/j.foodres.2020.109854
CrossRef - DuBois Michel., Gilles K. A., Hamilton J. K et al. Colorimetric Method for Determination of Sugars and Related Substances. Anal Chem. 1956;28(3):350–356. https://doi.org/10.1021/ac60111a017
CrossRef - Li S., Tao Y., Li D., et al. Fermentation of blueberry juices using autochthonous lactic acid bacteria isolated from fruit environment: Fermentation characteristics and evolution of phenolic profiles. 2021;276:130090. https://doi.org/10.1016/j.chemosphere.2021.130090
CrossRef - Maldonado R. R., da Costa Araújo L., da Silva Dariva L. C., et al. Potential application of four types of tropical fruits in lactic fermentation. LWT. 2017 ;86:254–260. https://doi.org/10.1016/j.lwt.2017.08.005
CrossRef - Loumani A., Larbi A. A., Mediani A., et al. Assessment Microbiological and Physicochemical Quality of Figs Dried in Adrar Region, Algeria. J Renew Energ. 2022;151-158. 1109/ICAIGE58321.2023.10346495
CrossRef - Gallali Y. M., Abujnah Y. S., Bannani F. K. Preservation of fruits and vegetables using solar drier: a comparative study of natural and solar drying, III; chemical analysis and sensory evaluation data of the dried samples (grapes, figs, tomatoes and onions). Renew Energy. 2000;19(1):203–212. https://doi.org/10.1016/S0960-1481(99)00032-4
CrossRef - Faleh E., Ghaffari S., Ferchichi A. Polyphenol and soluble Sugars contents of Tunisian Dried Fig. J New Sci. 2015;24. https://www.jnsciences.org/agri-biotech/32-volume-24/122-polyphenol-and-soluble-sugars-contents-of-tunisian-dried-fig.html
- Khezri S., Dehghan P., Mahmoudi R., et al. Fig Juice Fermented with Lactic Acid Bacteria as a Nutraceutical Product. Pharm Sci. 2016;22(4):260–266. https://doi.org/10.15171/PS.2016.40
CrossRef - Abdul Hakim B. N., Xuan N. J., Oslan S. N. H. A Comprehensive Review of Bioactive Compounds from Lactic Acid Bacteria: Potential Functions as Functional Food in Dietetics and the Food Industry. Foods. 2023;2(15) :2850. https://doi.org/10.3390/foods12152850
CrossRef - Debras C., Chazelas E., Srour B., et al. Total and added sugar intakes, sugar types, and cancer risk: results from the prospective NutriNet-Santé cohort. The Am J Clinical Nutr. 2020;112(5):1267–1279. https://doi.org/10.1093/ajcn/ nqaa246
CrossRef - De Filippis F., Pasolli E., Ercolini D. The food-gut axis: lactic acid bacteria and their link to food, the gut microbiome and human health. FEMS Microb Rev. 2020;44(4):454–489. https://doi.org/10.1093/femsre/fuaa015
CrossRef - Nemiche S., Nemiche Sa., Ait Hamadouche N. Caractéristiques physico-chimiques, analyses phytochimiques et activité antioxydante de la Figue sèche commercialisée Ficus carica Phytochem. Biosub. J. 2023;16(2):223-235.
- Lee N.-K., Paik H.-D. Bioconversion Using Lactic Acid Bacteria: Ginsenosides, GABA, and Phenolic Compounds. J Microb Biotechnol. 2017;27(5):869–877. https://doi.org/10.4014/jmb.1612.12005
CrossRef - Pontonio E., Montemurro M., Pinto D., et al. Lactic Acid Fermentation of Pomegranate Juice as a Tool to Improve Antioxidant Activity. Front Microbiol. 2019;10:1550. https://doi.org/10.3389/fmicb.2019.01550
CrossRef - Li Z., Teng J., Lyu Y., et al. Enhanced Antioxidant Activity for Apple Juice Fermented with Lactobacillus plantarum Molecules. 2019;24(1):51. https://doi.org/10.3390/molecules24010051
CrossRef













