Development of a Spray-Dried Starter Culture for the Efficient Removal of Off-Odors in Burdock
1College of Life Science and Food Engineering, Huaiyin Institute of Technology, Huaian, China.
2Institute of Agro-Product Processing, Jiangsu Academy of Agricultural Sciences, Nanjing, China.
3Department of Food Science and Chemical Technology, Universidad de Chile, Chile.
4Department of Central Laboratory, Jiangsu Academy Agricultural Sciences, Nanjing, China.
Corresponding Author Email:clisu1@163.com
Download this article as:
ABSTRACT:Burdock is a nutritious vegetable with significant functional properties; however, its characteristic off-odors limit broader commercialization. Our previous research also identified 2-isobutyl-3-methoxypyrazine (IBMP) and 2-sec-butyl-3-methoxypyrazine (IPMP) as the primary off-odor compounds in burdock, and verified that fermentation with Leuconostoc mesenteroides ZN-E could significantly degrade these compounds and improve burdock flavor. Based on this, this study further optimized culture conditions, medium composition and spray-drying processes for this strain to enhance bacterial viability and fermentation performance. The optimal fermentation conditions (32℃, 18 h) and medium composition (3% maltose, 4% soy peptone, 6% carrot juice) were determined by single-factor experiments, wherein carrot juice acted as a growth stimulant due to its rich content of natural sugars, vitamins, and minerals crucial for microbial development. Heat adaptation treatment at 45℃ for 20 minutes significantly improved thermal tolerance, contributing to the reduction of cell mortality during spray drying. Orthogonal tests established optimal drying parameters (inlet temperature 130℃, outlet temperature 80℃, feed rate 0.4 L/h), resulting in a powder with high viable counts. The spray-dried powder demonstrated superior performance by degrading 47.44% of IBMP, a key characteristic component responsible for the undesirable odor in burdock, significantly outperforming liquid cultures (37.22% degradation). The spray-dried powder also received enhanced sensory evaluation scores. This study provides a scalable methodology for producing a stable starter culture capable of efficient burdock off-odor removal, supporting its industrial application. These findings align with recent advances in probiotic starter culture development, where spray-drying has emerged as a cost-effective alternative to freeze-drying for industrial-scale production. The superior performance of spray-dried cultures in off-odor removal, as demonstrated in this study, contributes to the growing body of evidence supporting the application of optimized drying processes in functional food production.
KEYWORDS:Burdock; Growth Stimulant; Heat Adaptation; Lactic Acid Bacteria Fermentation; Off-Odors; Sensory Evaluation; Spray Drying
Introduction
Burdock (Arctium lappa) is recognized for its high nutritional value, being rich in protein, calcium, vitamins A and B, among other nutrients.1,2 It exhibits various biological activities, including antioxidant, anti-inflammatory, anti-diabetic, and anti-cancer properties,1,3 leading to its widespread use as a functional additive in food processing. However, the pronounced off-odors associated with burdock significantly restrict its marketability and application in food products. Lactic acid bacteria (LAB) fermentation has been employed to improve the safety, nutritional quality, organoleptic properties, and shelf-life of vegetables and fruits, including odor enhancement.4,5 For instance, Zhang et al. demonstrated the efficacy of Lactobacillus fermentation in removing off-odors from noni fruit juice while simultaneously enhancing its antioxidant activity.6 Similarly, Guo et al. reported that lactobacillus fermentation facilitated the degradation of amygdalin, offering a simplified method for its reduction.7 Previous research from our laboratory identified 2-isobutyl-3-methoxypyrazine (IBMP) and 2-sec-butyl-3-methoxypyrazine (IPMP) as the primary compounds responsible for burdock’s distinctive off-odors. We further demonstrated that LAB fermentation could ameliorate burdock odor through the degradation of these off-odor compounds and their precursors, alongside the generation of new aldehydes.8 Recent studies have expanded the range of applications for lactic acid fermentation (LAB) in vegetable processing. Controlled fermentation using Lactobacillus plantarum and L. brevis can reduce off-flavours in various root vegetables and enhance their nutritional value. Furthermore, research on Leuconostoc mesenteroides has revealed its ability to produce exopolysaccharides during fermentation, which improves the texture and functionality of the resulting products.
Leuconostoc mesenteroides ZN-E, isolated from homemade fermented Paocai, exhibited the capability to degrade IBMP. To facilitate the industrial application of this strain, its development into a stable starter culture is essential, necessitating the enhancement of viable bacterial counts and fermentation performance.
Currently, the primary methods for preparing direct starter cultures are freeze-drying and spray drying. While freeze-drying offers higher survival rates and superior fermentation performance, it is associated with longer processing cycles, elevated costs, and greater equipment requirements.9 In contrast, spray drying is a unitary process utilizing relatively simple equipment, allowing for the direct spraying of bacterial suspensions,10 thereby reducing capital investment and processing time. The resulting bacterial powder is also easier to transport and exhibits extended shelf-life.11 However, the application of spray drying is challenged by high bacterial mortality rates, low bacterial concentration, and diminished fermentation activity in the final product. The process involves exposure to high temperatures (e.g., outlet temperatures around 80°C and inlet temperatures up to 140°C).12,13 Although the exposure time within the drying chamber is brief, bacteria are highly susceptible to dehydration and thermal stress, leading to significant cell death.14,15 High-cell-density fermentation is a common technical means to improve the survival rate of bacteria after spray drying: Wan et al. achieved viable counts of 10¹⁰ CFU/mL in Lactobacillus acidophilus through high-density fermentation, with spray-dried powders retaining 10⁸ CFU/g.16 Heat shock pretreatment is another effective method to enhance bacterial thermotolerance: Khem et al. observed that heat adaptation of Lactobacillus paracasei at 52℃ for 15 minutes prior to spray drying increased survival rates by 30–70 times and improved activity 18-fold,17 so as to improve the activity of the bacteria after spray drying. Khem et al. found that the survival rate of the bacterium increased when the strain of Lactobacillus paracasei was subjected to heat-excited treatment (52℃, 15 min) followed by heat treatment (60℃), the survival rate of the bacterium increased by 30 ~ 70 times, and the activity of the bacterium increased by 18 times after spray drying.18 Optimization of process parameters, such as feed rate, inlet, and outlet temperatures, also plays a critical role in maximizing viability, as demonstrated by Xiong et al.19
In this study, we optimized the fermentation medium, culture conditions, heat adaptation treatment, and spray-drying parameters for Leuconostoc mesenteroides ZN-E based on viable cell count, fermentation activity, sensory evaluation, and IBMP removal efficiency. This research validates the fermentation characteristics of L. mesenteroides ZN-E and provides experimental evidence for the development of a direct starter culture for odor modification in burdock products, thereby supporting its industrial application.
Materials and Methods
Bacterial Strain and Materials
Leuconostoc mesenteroides ZN-E was previously isolated from fermented Paocai and maintained at the Institute of Agricultural Product Processing, Jiangsu Academy of Agricultural Sciences (Nanjing, China).
Standard MRS (de Man, Rogosa and Sharpe) broth was prepared containing (g/L): glucose (20.0), peptone (10.0), beef extract (10.0), yeast extract (5.0), sodium acetate (5.0), dipotassium hydrogen phosphate (2.0), diammonium hydrogen citrate (2.0), manganese sulfate (0.25), magnesium sulfate (0.58), and Tween-80 (1 mL/L). For the screening of carbon and nitrogen sources in microbial medium, the single-factor replacement method is a widely used technical approach in existing literature:19,20 in this study, for the “Carbon source-optimized MRS”, the glucose component in the standard MRS formulation was replaced with individual carbon sources (each at 2% w/v), including glucose, fructose, sucrose, lactose, D-galactose, and maltose, while the concentrations of all other components remained unchanged. For the “Nitrogen source-optimized MRS”, the nitrogen source mixture (peptone (10.0 g/L), beef extract (10.0 g/L), yeast extract (5.0 g/L)) in the standard MRS was replaced with individual nitrogen sources (each at 2% w/v), including peptone, tryptone, soybean peptone, beef extract, ammonium sulfate, and whey protein powder, with all other components retained at their original concentrations.
Chemicals, including sodium acetate, dipotassium hydrogen phosphate, diammonium hydrogen citrate, manganese sulfate, magnesium sulfate, and Tween-80, were procured from Sigma-Aldrich (Shanghai, China). Carbon and nitrogen source compounds (glucose, fructose, sucrose, lactose, D-galactose, maltose, peptone, tryptone, soybean peptone, beef extract, ammonium sulfate, whey protein powder) were obtained from Shanghai Yuanye Biotechnology Co., Ltd. (Shanghai, China). Fresh vegetables (tomato, carrot, oyster mushroom, corn, pumpkin, cucumber, white radish, cabbage) were acquired from local supermarkets (Suguo and Box Horse, Nanjing, China). All solvents and reagents were of analytical grade.
Single-Factor Optimization of Culture Conditions
The Leuconostoc mesenteroides ZN-E stored at -80 ℃ in the laboratory was streaked into MRS solid medium, and cultured at 37 ℃ for 24 to 48h for activation. After two generations of activation, the bacterial liquid was inoculated into MRS liquid medium for expansion at a rate of 2% (v/v).
Cryopreserved L. mesenteroides ZN-E was streaked onto MRS agar and incubated at 37°C for 24–48 h for reactivation. After two sequential subcultures, the bacterium was inoculated at 2% (v/v) into MRS broth for further cultivation.
In the single-factor optimization experiment of medium composition, the medium used for each batch of fermentation was 100 mL.
Determination of Optimal Culture Time and Temperature
For growth curve analysis, L. mesenteroides ZN-E was inoculated (2% v/v) into MRS broth and incubated at 37°C. Optical densities at 600 nm (OD600) and pH were monitored at 2 h intervals over 24 h using a UV-Vis spectrophotometer (Beijing Puxi General Instrument Co., Ltd., China) and a pH meter (METTLER TOLEDO Instruments Ltd., Shanghai, China), respectively. Uninoculated MRS broth served as the blank control. OD600 and pH values were plotted against incubation time to generate growth and acidification curves.
To determine the optimal growth temperature, activated ZN-E was inoculated into fresh MRS broth and incubated at 15, 20, 28, 32, 37, and 42°C for 18 h, after which OD600 was measured.
Single-Factor Optimization of Medium Composition
In the single-factor optimization of medium composition, the medium used for each batch of fermentation was 100 mL.
Carbon Source Selection and Optimization
On the UMRS liquid medium, glucose, fructose, sucrose, lactose, D-galactose, and maltose were used as carbon sources with the addition amount of 2%. The nitrogen source was added according to the formula in MRS medium. The bacterial liquid was inoculated at 2% (v/v) and cultured at 32°C for 18 h. Afterward, the OD600nm of the fermentation liquid was measured.20,21
The growth of ZN-E strain was affected by different carbon sources content. Based on the influence of carbon sources, a single-factor experiment on the selected carbon sources’ concentrations was conducted, with concentrations of 1%, 2%, 3%, 4%, 5%, and 6%, and other components remained unchanged.
Nitrogen Source Selection and Optimization
Cultures were inoculated in Nitrogen source-optimized MRS with 2% (v/v) ZN-E and incubated at 32°C for 18 h, followed by OD₆₀₀ measurement.
The concentration of the selected nitrogen source was then optimized at 1%, 2%, 3%, 4%, 5%, and 6% (w/v).
Evaluation of Growth Factors
Vegetable juice is often used as a natural growth factor for LAB culture in existing research, and the addition amount is generally 5%-10% (w/v),22-24 with Standard MRS as control. Cultures were incubated at 32°C for 18 h, and OD₆₀₀ was measured.
The concentration of the selected vegetable juice was subsequently optimized at 1%, 2%, 3%, 4%, 5%, 6%, 8%, and 10% (v/v).
Orthogonal Experimental Design for Medium Optimization
Orthogonal test L9 (34) was used to analyze three parameters of spray drying process, including outlet temperature ( 60, 70, 80 ℃),25 inlet temperature (130, 150, 170 ),25 and feed rate ( 0.4, 0.6, 0.8 L/h),13 to detect the influence of the determination of viable bacteria in ZN-E milk powder, so as to screen out the best spray drying conditions. Other medium components were maintained as in standard MRS. In the L9 orthogonal design for medium optimization (Table 1) and spray drying (Table 1), factor D (designated as the “Blank column” in the original table) is formally defined as a dummy variable. This dummy variable serves dual purposes: first, to estimate the experimental error inherent in the test system, and second, to verify the reliability and robustness of the orthogonal test results by accounting for unforeseen or uncontrolled minor variations that may influence the response parameter.
Table 1: Orthogonal Test Results for Optimization Of Spray Drying Conditions For Strain ZN-E.
|
Experiment numbers |
Output temperature(℃) | Inlet temperature (℃) | Feed rate (L/h) | Blank column |
Lg [Viable count (CFU/g)] |
|
1 |
1 (60) | 1 (130) | 1 (0.4) | 1 | 10.090 |
| 2 | 1 | 2 (150) | 2 (0.6) | 2 |
10.127 |
|
3 |
1 | 3 (170) | 3 (0.8) | 3 | 9.841 |
| 4 | 2 (70) | 1 | 2 | 3 |
10.061 |
|
5 |
2 | 2 | 3 | 1 | 9.878 |
| 6 | 2 | 3 | 1 | 2 |
9.770 |
|
7 |
3 (80) | 1 | 3 | 2 | 10.423 |
| 8 | 3 | 2 | 1 | 3 |
10.337 |
|
9 |
3 | 3 | 2 | 1 | 9.822 |
| `k1 | 10.019 | 10.191 | 10.066 |
|
|
|
`k2 |
9.903 | 10.114 | 10.003 |
|
|
|
`k3 |
10.194 | 9.811 |
10.047 |
||
|
Range |
0.291 | 0.380 | 0.063 | ||
| Better level | A3 | B1 | C1 |
|
The data was expressed as mean ± standard deviation (SD, n = 3).
‘`k1’ means the average value of OD600nm corresponding to the same column number 1; ‘`k2’means the average value of OD600nm corresponding to the same column number 2; ‘`k3’means the average value of OD600nm corresponding to the same column number 3.
‘A3’ means the outlet temperature was 80 ℃; ‘B1’ means the Inlet temperature was 130 ℃; ‘C1’ means the feed rate was 0.4 L/h.
Heat Adaptation Treatment
Heat Resistance Assessment: L. mesenteroides ZN-E cultures were subjected to thermal challenge in a water bath at 50, 55, 60, 65, 70, and 80°C. Viable counts were determined via plate counting at 5-minute intervals to establish thermal death curves and identify the sub-lethal temperature range for heat shock pretreatment. The plate counting method for determining bacterial viable counts refers to the national standard method of food microbiology detection, and the colony counting is carried out after 24-48h of culture at the optimal temperature of the strain.19
Within this range, heat shock treatments were applied for 20 minutes at various temperatures, ensuring a viability loss not exceeding one log cycle. Subsequently, cultures were exposed to a lethal heat treatment (70°C, 5 min). Viable counts and survival rates were determined and compared to a control group receiving no heat shock pretreatment. The selection of 45°C as the optimal heat adaptation temperature was based on the identification of the sub-lethal temperature range from thermal death curves, which indicated that this temperature provided sufficient stress to induce thermotolerance without causing excessive cell mortality. The 20-minute duration was determined through systematic optimization, as shorter durations (10 minutes) provided insufficient adaptation, while longer durations (>30 minutes) led to cumulative thermal damage. This optimal condition (45°C, 20 min) resulted in a survival rate of 74.32% after lethal heat challenge, representing a significant improvement over non-adapted controls. The heat adaptation protocol was designed based on established methodologies in the literature, with modifications specific to L. mesenteroides ZN-E based on its unique thermal tolerance characteristics.
Heat adaptation optimization: Using the selected heat shock temperature, the duration of heat adaptation was further optimized by applying treatments for 10, 20, 30, 50, 80, and 120 minutes, followed by the lethal heat challenge (70°C, 5 min).
Orthogonal Experimental Design for Spray Drying
Bacterial cells from the optimized cultures were harvested via centrifugation, resuspended in sterile skim milk (10% w/v), and subsequently subjected to spray drying (Y-1912301448 Spray Drying Machine, Komei Intelligent Equipment (Nanjing) Co., Ltd.,Nanjing, China.). We have also supplemented the final solids content of the feed slurry (15% w/w) for the spray-drying process, with each single factor totaling 500ml. An L₉(3⁴) orthogonal test was employed to evaluate three spray-drying parameters: outlet temperature (A: 60, 70, 80°C), 13,26 inlet temperature (B: 130, 150, 170°C),27 and feed rate (C: 0.4, 0.6, 0.8 L/h).13 The primary response was the viable count in the resulting powder (CFU/g).
Water Content Measurements
To obtain absolutely dried samples, spray-dried powders were placed on glass plates and dried at 105 ◦C for 4 hours. The weights of each sample were recorded prior to and following the drying process, which were then used to calculate the water content. The water content is expressed as (mass of water / total mass of powder) × 100 (%). This gravimetric method for determining water content is a classic method in food and microbial powder detection, with high accuracy and good repeatability.24
Survival Rate and Process Yield Measurements
The survival rate was expressed as the live cells after drying × 100/live cells before drying. The process yield was calculated as the dry powder mass divided by the total solids fed, multiplied by 100%. All experiments were carried out in triplicate.
Analysis of Fermentation Performance
L. mesenteroides ZN-E was cultivated in both the optimized medium (from orthogonal tests) and the standard MRS medium. Cultures were spray-dried using the optimized parameters, and viable counts were determined pre- and post-drying.
pH Measurement
Spray-dried powder and liquid culture of ZN-E (inoculated at 2.65 × 10¹⁰ CFU/g) were used to ferment burdock. The pH of the fermented burdock was monitored using a pH meter (METTLER TOLEDO).
Sensory Evaluation
A comparative sensory analysis was conducted by ten experienced panelists (mixed gender) to evaluate flavor differences between burdock fermented with ZN-E liquid culture and spray-dried powder. A 10-point hedonic scale was used (1 = least preferred, 10 = most preferred), as described by X. Zhang et al.28 The sensory evaluation panelists were trained according to the standard of sensory evaluation of food flavor, and the evaluation environment was maintained at 25±2°C with no peculiar smell.28
IBMP Quantification
ZN-E powder and liquid culture were inoculated (10% v/v) into MRS medium containing burdock and incubated at 32°C for 2 days. IBMP content was analyzed by HS-SPME-GC-MS.29 Quantification was performed using an IBMP standard curve (y = 9×10⁻⁹x + 0.4477, R² = 0.9892, where y is IBMP concentration in ng/mL and x is the peak area in Ab*s). Data processing utilized Xcalibur software.30
Statistical Analysis
All experiments were performed in at least triplicate. Data are presented as mean ± standard deviation (SD). Graphical representations were generated using GraphPad Prism 8.0.2, and statistical analysis (ANOVA with post-hoc tests, significance level P < 0.05) was performed using OriginPro (version 2022b).
Results
Optimization of Culture Conditions
Optimal Culture Time
The growth of lactic acid bacteria (LAB) typically follows a characteristic pattern comprising four distinct phases: lag, logarithmic (exponential), stationary, and decline phases. As illustrated in Fig. 1A, Leuconostoc mesenteroides ZN-E exhibited a short lag phase (0-1 h), during which the cells adapted to the new environment by synthesizing essential enzymes and metabolites, with minimal sugar consumption and no significant increase in optical density (OD). This was followed by an exponential growth phase from 1 to 18 h, characterized by rapid cell division and a significant increase in biomass. During this phase, the cells metabolized available nutrients efficiently, leading to a steady decrease in pH due to lactic acid production (Fig.1B). The growth kinetics during this phase can be described by the Logistic model, which accounts for the self-inhibition effect as cell density increases.
Concurrently, the pH decreased steadily from an initial value of approximately 6.5 to below 4.0, stabilizing after 18 h (Fig.1B), which correlated with the accumulation of lactic acid and other metabolic by-products. The stabilization of pH and the cessation of growth indicated the entry into the stationary phase, where the rate of cell division balanced the rate of cell death due to nutrient depletion and acid accumulation. The maximum lactic acid concentration reached during this phase was critical for determining the optimal fermentation time, as it directly influenced the product quality and preservation.
The optimal culture time for L. mesenteroides ZN-E was determined to be approximately 18-20 h, based on the intersection of the growth curve and acid production curve.
Optimal Culture Temperature
Temperature significantly influences bacterial growth. As shown in Fig. 1C, for the strain Leuconostoc mesenteroides ZN-E, the cell density (measured as OD600) increased with temperature until reaching a maximum at 32℃, which was significantly higher than that at other tested temperatures (P < 0.05).
Optimization of Medium Composition
Carbon Source Selection and Concentration Optimization
Among the evaluated carbon sources (Fig. 2A), maltose supported the highest microbial growth (OD600 = 1.926), followed by sucrose (OD600= 1.836, P < 0.05), while lactose resulted in the lowest cell density (OD600= 0.146); thus, maltose was selected for further optimization. Subsequent optimization revealed that cell density increased with maltose concentration up to 3% (Fig. 2B), with no significant difference (P > 0.05) observed between 2%, 3%, and 4% maltose; higher concentrations beyond this range led to growth inhibition due to elevated osmolarity, confirming 3% maltose as the optimal concentration.
Nitrogen Source Selection and Concentration Optimization
In the optimization of nitrogen sources for Leuconostoc mesenteroides, organic nitrogen sources proved superior for ZN-E growth, with soybean peptone yielding the highest cell density and significantly outperforming other sources (P < 0.05) (Fig. 2C). Beef extract ranked as the second most effective nitrogen source. Concentration tests revealed that 4% soybean peptone supported maximum growth, while higher concentrations were detrimental, presumably due to osmotic stress, thus establishing 4% as the optimal concentration (Fig. 2D).
Growth Factor Selection and Optimization
Adding nutrient proliferation factors (i.e., fruit/vegetable juices) allows Leuconostoc mesenteroides to acquire essential growth nutrients, effectively boosting its growth rate. In this study, eight vegetable juices—tomato, carrot, oyster mushroom, corn, pumpkin, cucumber, white radish, and cabbage juices—were tested as nutritional factors to assess their impacts on the growth of strain ZN-E. As shown in Fig. 2E, supplementing MRS basal medium with these juices led to distinct changes in cell density: carrot juice exerted the strongest proliferation effect on ZN-E, achieving an OD600 value of 1.939 (compared to 1.735 in the MRS basal medium alone), followed by tomato juice (OD600 = 1.825, P < 0.05). Fig. 2F further indicated that cell density increased gradually with higher carrot juice addition, but the promoting effect plateaued beyond a certain concentration; considering economic costs, 6% was determined as the optimal addition level.
Orthogonal Experiment for Medium Formulation
An L9(34) orthogonal test assessed the synergistic effects of key factors (maltose, soy peptone, carrot juice). Results (Table 2) indicated that the order of influence on ZN-E growth was: B (soy peptone) > A (maltose) > C (carrot juice). The optimal medium formulation was determined to be A2B2C3: 30 g/L maltose, 40 g/L soy peptone, and 60 mL/L carrot juice.
Table 2: Orthogonal Test Results of ZN-E Strain Medium Condition Optimization.
|
Experiment numbers |
Maltose (g/L) | Soy peptone (g/L) | Carrot juice (mL/L) | Blank column | OD600nm |
| 1 | 1 (20) | 1 (30) | 1 (40) | 1 |
1.781 |
|
2 |
1 | 2 (40) | 2 (50) | 2 | 1.902 |
| 3 | 1 | 3 (50) | 3 (60) | 3 |
1.865 |
|
4 |
2 (30) | 1 | 2 | 3 | 1.760 |
| 5 | 2 | 2 | 3 | 1 |
2.087 |
|
6 |
2 | 3 | 1 | 2 | 1.851 |
| 7 | 3 (40) | 1 | 3 | 2 |
1.749 |
|
8 |
3 | 2 | 1 | 3 | 1.870 |
| 9 | 3 | 3 | 2 | 1 |
1.887 |
|
`k1 |
1.849 | 1.763 | 1.834 | ||
| `k2 | 1.899 | 1.953 |
1.850 |
||
|
`k3 |
1.835 | 1.868 | 1.900 | ||
| Range | 0.064 | 0.190 |
0.066 |
||
|
Better level |
A2 | B2 |
C3 |
The data was expressed as mean ± standard deviation (SD, n = 3).
‘`k1’means the average value of OD600nm corresponding to the same column number 1; ‘`k2’means the average value of OD600nm corresponding to the same column number 2; ‘`k3’means the average value of OD600nm corresponding to the same column number 3.
‘A2’ means the content of maltose was 30 g/L; ‘B2’ means the content of soy peptone was 40 g/L; ‘C3’ means the content of carrot juice was 60 mL/L.
Heat Adaptation Treatment
Heat shock at sub-lethal temperatures is a well-documented strategy to enhance microbial tolerance to subsequent lethal thermal stress. As shown in Fig. 3 (A and B), the viable count and survival rate exhibited peaks at 20 minutes (2.16×1010 CFU/mL and 74.32%, respectively), followed by a decline with prolonged exposure. Therefore, the optimal heat adaptation conditions were determined as 45℃ for 20 minutes.
Optimization of Spray-Drying Parameters
The range analysis of the orthogonal test (Table 1) demonstrated that the influence of spray-drying parameters on the survival of Leuconostoc mesenteroides ZN-E decreased in the order: inlet temperature (B) > outlet temperature (A) > feed rate (C). The optimal parameter combination, derived from the mean values, was determined to be A3B1C1, which corresponds to an outlet temperature of 80℃, an inlet temperature of 130℃, and a feed rate of 0.4 L/h.
Under the optimal spray-drying conditions, the process yield was 51.8%. The viable count of the resulting powder reached 2.65 × 1010 CFU/g. Based on the initial cell concentration of 2.16 × 1010 CFU/mL in the feed suspension, the survival yield was calculated to be 63.5%. The water content of the dried powders was below 5% (4.74%).
Fermentation Performance Evaluation
Fermentation Activity (Acidification)
The pH of fermented burdock decreased progressively over time, consistent with acid production by the microorganisms.31 Initially, the pH in samples fermented with the spray-dried bacterial powder was higher than in those inoculated with the liquid culture (Fig. 4A), suggesting a potential lag in metabolic reactivation. Nevertheless, the acidification rates became comparable as fermentation proceeded, indicating that the final fermentative activity of the spray-dried powder was restored to a level similar to or even surpassing that of the liquid culture.
Sensory Evaluation
Sensory scores for burdock aroma (Fig.4B) were significantly higher (P < 0.05) for samples fermented with the spray-dried powder compared to the liquid culture.
IBMP Removal Efficiency
Gas chromatography-mass spectrometry (GC-MS) analysis indicated that unfermented burdock contains a high level of 2-isobutyl-3-methoxypyrazine (IBMP) at a concentration of 129.263 ng/mL. Fermentation processes significantly reduced IBMP levels: liquid culture fermentation led to a 37.22% degradation, lowering IBMP to 81.148 ng/mL, while fermentation with spray-dried powder achieved a more substantial 47.44% degradation, reducing IBMP to 67.944 ng/mL.
Discussion
Optimization of Culture Conditions
Optimal Culture Time
The optimal culture time for L. mesenteroides ZN-E was determined to be approximately 18-20 h, based on the intersection of the growth curve and acid production curve. This timeframe ensured maximum biomass yield and lactic acid production while avoiding prolonged fermentation that might lead to cell lysis and release of intracellular components that could impair product quality. Consistent with this observation, similar optimal culture time windows were also reflected in the fermentation kinetics of L. mesenteroides E131.32 At 18-20 h, E131 was in the mid-to-late exponential growth phase: its biomass (cell dry mass, CDM) reached 1.8-2.0 g L⁻¹, approaching the maximum yield (~2.11 g CDM L⁻¹) with no cell lysis. Concurrently, acid production was efficient: lactic acid reached 6-7 g L⁻¹ and acetic acid 1.5-2 g L⁻¹, accounting for a large portion of total acid (final ~10.5 g L⁻¹), reflecting active heterofermentative metabolism. Glucose (carbon source) was sufficiently consumed (dropping to 3-4 g L⁻¹) to sustain biomass accumulation and acid synthesis. This consistency-18-20 h enabling near-maximum biomass and efficient acid production without cell impairment-supports the optimality of this timeframe for Leuconostoc mesenteroides strains.
 |
Figure 1: Growth curve and pH value of ZN-E strain at 24h and the effect of temperature on the growth of ZN-E strain. (A) Growth curve of ZN-E strain; (B) The change of pH value during the growth of ZN-E strain. |
Optimal Culture Temperature
This observation aligns with the growth characteristics of Leuconostoc mesenteroides reported in existing literature, this bacterium reported successful activation and cultivation at 30 °C, and 32°C, being slightly above this optimal value, still falls within a thermally favorable range that does not inhibit its growth.33 Additionally, studies34 (focused on the influence of temperature and pH on the bacterium’s growth and bacteriocin production) noted that Leuconostoc mesenteroides achieves optimal growth at around 30°C; while 32°C is not the exact temperature for peak bacteriocin synthesis, it remains a temperature that supports effective cell growth, consistent with the need for balancing growth and metabolic activity. Furthermore, the findings from Drosinos et al., which examined Leuconostoc mesenteroides E131, indicated that the bacterium exhibits good growth at temperatures between 14°C and 25°C (with optimal growth at 25°C for bacteriocin production and 30°C for growth), and 32°C, as a temperature close to its upper growth limit but not exceeding it, further confirms its suitability for sustaining the growth of Leuconostoc mesenteroides strains like ZN-E.
Optimization of Medium Composition
Carbon Source Selection and Concentration Optimization
Notably, sucrose has also been identified as an effective carbon source for cultivating lactic acid bacteria like Leuconostoc mesenteroides, with optimized 3% sucrose conditions enhancing both growth and product formation,35 while the use of glucose and fructose as sole carbon sources has been studied in relation to cold-induced enzyme expression, further illustrating how substrate choice impacts functional gene expression.36 Optimizing carbon source selection for microbial growth is critical for advancing biotechnological processes, as evidenced by studies across diverse microbial systems: Bernardo et al.37 found that Leuconostoc mesenteroides produces optically pure D(-)-lactic acid, indicating that targeting strains with efficient utilization pathways for specific carbon sources can optimize growth and product yield; Liu et al.38 developed a novel selection circuit that couples cell growth to metabolite production using maltose as the sole carbon source, ensuring robust growth and sustaining selection pressure across multiple generations to maintain long-term microbial productivity; and Owoade et al.39 highlighted the necessity of selecting suitable carbon sources (e.g., carbon monoxide) for efficient bioethanol production in bioconversion processes, further emphasizing the role of tailored substrate selection in enhancing microbial growth and bioprocess efficiency. Collectively, these studies demonstrate that the strategic selection and optimization of carbon sources—ranging from traditional sugars (e.g., maltose, sucrose) to unconventional substrates (e.g., carbon monoxide)—are fundamental to boosting microbial growth and metabolite production, and employing innovative selection strategies, mechanistic modeling, and precise media formulation can significantly improve the efficiency and sustainability of microbial bioprocesses, including those involving Leuconostoc mesenteroides.
Nitrogen Source Selection and Concentration Optimization
These findings align with previous studies emphasizing the importance of peptone, including soybean peptone, in enhancing microbial growth and dextran production. For instance, research on dextran production by L. mesenteroides has demonstrated that combining peptone with beef extract under high sucrose conditions significantly boosts dextran yield, underscoring its relevance in industrial fermentation.40 Elevating sucrose concentration to 50 g/L, along with nitrogen sources (peptone and beef extract, each at 25 g/L), boosts dextran production by L. mesenteroides NRRL B-640. In contrast, buffering agents such as K₂HPO₄, while facilitating microbial growth and enzyme release, do not directly affect dextran synthesis.41
Growth Factor Selection and Optimization
Beyond this study, fruit/vegetable juices generally serve as multifunctional growth promoters for L. mesenteroides. In terms of carbon source supply and metabolite stimulation, they provide fermentable sugars (e.g., glucose, fructose) that synergize with supplemented sucrose to enhance growth and polysaccharide synthesis: for example, tomato juice with 15% sucrose supported L. mesenteroides BD1710 in synthesizing 32 g/L dextran (equivalent to yields in chemically defined media) with a peak molecular weight of 6.35×10⁵ Da,42 while carrot juice with ≤15% sucrose enabled complete sugar-to-dextran conversion within 24 h and improved broth consistency (3.5 Pa·sⁿ) and product stability.43 In regulating bioactive substances, juice-borne compounds modulate microbial metabolism and product functionality: fermenting murta (Ugni molinae) berry juice with L. mesenteroides increased total polyphenol content, elevating DPPH/ORAC antioxidant activity and α-glucosidase inhibitory capacity (critical for glucose metabolism regulation),44 and Sanhua plum juice fermented by L. mesenteroides retained higher total phenols and anthocyanins than other lactic acid bacteria, preserving juice color and bioactivity. Additionally, in optimizing the growth environment, these juices supply complex nutrients matching L. mesenteroides’ demands for growth factors and amino acids: carrot-isolated L. mesenteroides strains (C2, C7) showed high survival under simulated gastrointestinal conditions and inhibited pathogens (e.g., Salmonella, Listeria) when cultured in vegetable-derived media,45 and fermented carrot juice maintained a stable pH of 3.8 with minimal sedimentation during 3 weeks of cold storage, addressing raw juice quality defects.43 Genomic data on L. mesenteroides (e.g., strain ATCC 8293; DOE Joint Genome Institute, 2018) further supports its ability to utilize nutrients from fruit/vegetable juices, highlighting these natural matrices as key to enhancing its growth and metabolic performance.
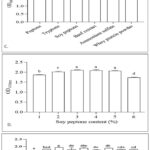 |
Figure 2: Effects of different carbon and nitrogen sources and vegetable juice on ZN-E strain. (A) Effects of different carbon sources on ZN-E strain; (B) Effects of maltose content on ZN-E strain. |
Heat Adaptation Treatment
Heat shock at sub-lethal temperatures is a well-documented strategy to enhance microbial tolerance to subsequent lethal thermal stress, primarily through the induction of heat shock proteins (HSPs)—chaperones that stabilize cellular proteins, repair denaturation, and maintain membrane integrity under stress.46 This phenomenon is particularly relevant for Leuconostoc mesenteroides, a lactic acid bacterium widely used in food fermentation (e.g., sourdough, vegetable fermentation), where it often encounters fluctuating thermal conditions during processing.
Notably, the efficacy of heat shock depends strongly on the choice of sub-lethal temperature and duration, as these parameters dictate the magnitude of stress adaptation without causing irreversible cell damage. Survival after lethal heat challenge is a key indicator of acquired thermotolerance.47 As shown in Fig. 3 (A and B), the viable count and survival rate exhibited peaks at 20 minutes (2.16×10¹⁰ CFU/mL and 74.32%, respectively), followed by a decline with prolonged exposure. Therefore, the optimal heat adaptation conditions were determined as 45°C for 20 minutes. This aligns with studies on Lactobacillus paracasei, where heat adaptation at 44°C was optimal.48
Gardiner et al.49 demonstrated that probiotic strains such as Lactobacillus paracasei exhibit increased survival rates during heat treatment and spray drying, implying that adaptive responses may be harnessed to enhance microbial resilience. Furthermore, Ardanareswari et al.50 investigated the resistance of Lactobacillus paracasei SNP2 to heat shock and spray drying, finding that heat adaptation and pH adjustments could improve microbial survival, which aligns with the concept that sub-lethal heat exposure primes organisms for subsequent thermal challenges.
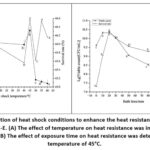 |
Figure 3: Optimization of heat shock conditions to enhance the heat resistance of Leuconostoc mesenteroides ZN-E. (A) The effect of temperature on heat resistance was investigated after a 20-minute shock. (B) The effect of exposure time on heat resistance was determined at a shock temperature of 45°C. |
Optimization of Spray-Drying Parameters
Optimizing spray-drying conditions is paramount for preserving the viability of probiotic strains like Leuconostoc mesenteroides in functional food and pharmaceutical applications. As noted by Castro-Muñoz et al.,51 meticulous adjustment of drying parameters is essential to enhance the stability of probiotics. This is supported by research on other lactobacilli; for instance, spray-drying of L. rhamnosustypically employs inlet temperatures of 130–150°C, while a formulation containing L. plantarumin almond milk powder uses temperatures as high as 170–190°C, as highlighted in a PMC review.52 However, excessively high temperatures can be detrimental. The inactivation mechanisms for lactic acid starter cultures are significantly influenced by drying parameters,53 particularly temperature and duration, which can induce cell death through damage to cell membranes and vital components if not properly controlled.
Further insights from inactivation studies54 confirm that high temperatures, especially during prolonged secondary drying phases, can severely compromise the viability of Leuconostoc mesenteroides, underscoring the critical need for precise temperature management. Additionally, the development of effective instant microbial starters55 emphasizes that carrier media composition, drying temperature, and subsequent storage conditions are intrinsically linked parameters that collectively determine the survival and efficacy of Leuconostoc mesenteroides in final dried products.
Fermentation Performance Evaluation
Fermentation Activity (Acidification)
This recovery of acidogenic capacity can be attributed to the physiological adaptation of the bacteria post-drying. Although spray drying alters the physicochemical properties of Leuconostoc mesenteroides—such as membrane fatty acid composition, which can initially impair survival and metabolic function—the bacteria can regain activity under suitable fermentation conditions.56 The use of protective agents during drying is critical in this regard, as they mitigate cellular damage and help preserve the viability necessary for post-rehydration metabolic performance, including acid production.56,57
Further supporting this, studies confirm that Leuconostoc mesenteroides retains the ability to produce lactic acid after spray drying, though the final acid yield is influenced by drying parameters and the protective matrix used.58,59 Factors such as fermentation pH and the presence of encapsulation materials also play a significant role in stabilizing the bacteria and modulating acid synthesis during fermentation.60,61 Moreover, statistical optimization approaches have been employed to enhance related metabolic activities, such as dextran production, indicating that drying parameters can be fine-tuned to better preserve acidogenic activity.40 Nonetheless, the specific interplay between spray-drying conditions and ultimate acid production levels warrants further investigation.
 |
Figure 4: Fermentation activity, flavor sensory evaluation and IMBP removal ability analysis of Leuconostoc mesenteroides ZN-E spray-dried bacterial powder. (A) Results of fermentation activity of Leuconostoc mesenteroides ZN-E spray drying powder; |
Sensory Evaluation
This improvement aligns with findings that LAB fermentation can generate favorable flavor compounds and inhibit off-odor formation.62 Research indicates that Leuconostoc mesenteroides contributes significantly to flavor enhancement through the production of volatile compounds during fermentation.63 Its metabolic capabilities, including the synthesis of aroma compounds, are essential for improving the sensory qualities of fermented products. The application of spray drying has been shown to influence these metabolic activities, potentially affecting the bacteria’s ability to produce desirable aroma compounds. For instance, studies on other lactic acid bacteria suggest that drying processes can impact their metabolic functions, including aroma production, by inducing stress responses or altering cell viability.58
Furthermore, the preservation of aroma-producing capacity post-spray drying is crucial for maintaining the quality of fermented products. Leuconostoc mesenteroides has been utilized in various fermentations, such as kimchi and kefir, where its role in flavor development is well documented.58,64 The ability of spray-dried cultures to retain their functional properties, including aroma synthesis, directly influences the sensory profile of the final product. Some studies highlight that certain strains of Leuconostoc mesenteroides can be enhanced for lactic acid tolerance and aroma production through specific processing techniques, suggesting that optimized spray-drying conditions could preserve or even improve these traits.58
In addition, the metabolic network of Leuconostoc mesenteroides, which underpins its aroma-producing capacity, can be affected by processing methods. The production of volatile compounds, such as dextran and other flavor-active metabolites, is integral to its fermentation performance.40 Ensuring that spray drying does not compromise these metabolic pathways is essential for harnessing the full aroma potential of this bacterium in industrial applications.
Overall, while spray drying offers practical benefits for the application of Leuconostoc mesenteroides in fermentation, its impact on aroma production depends on the preservation of metabolic activity and cell viability. Optimizing drying parameters to maintain the bacteria’s functional integrity is vital for ensuring that the aroma-producing capacity remains effective in fermented products.58,63 Future research should focus on refining spray-drying techniques to maximize the retention of aroma-related metabolic functions in Leuconostoc mesenteroides, thereby enhancing the sensory qualities of fermented foods and beverages.
IBMP Removal Efficiency
Gas chromatography-mass spectrometry (GC-MS) analysis has indicated that unfermented burdock contains a high level of 2-isobutyl-3-methoxypyrazine (IBMP)—a volatile organic compound (VOC) of the pyrazine family linked to undesirable “green” or “earthy” off-flavors in products like coffee and wine—at a concentration of 129.263 ng/mL.65,66 Fermentation processes significantly reduced IBMP levels: liquid culture fermentation led to a 37.22% degradation, lowering IBMP to 81.148 ng/mL, while fermentation with spray-dried powder achieved a more substantial 47.44% degradation, reducing IBMP to 67.944 ng/mL. This demonstrates that the spray-dried powder was approximately 1.3 times more effective in removing IBMP, confirming the efficacy of lactic acid bacteria (LAB) fermentation in degrading undesirable volatiles, as previously reported in other plant substrates67,68 found that all plant-derived Leuconostoc strains were able to degrade hexanal and 2- pentylfuran below the detection limit within 18 h of fermentation on soybean substrates.
Leuconostoc mesenteroides, a common LAB species involved in diverse fermentation processes (e.g., coffee and plant-based food production), plays a key role in metabolizing sugars and shaping flavor profiles, making it a promising candidate for mitigating off-flavors. While direct evidence of IBMP degradation by L. mesenteroides remains limited, its capacity to reduce structurally similar off-flavor compounds supports its potential for IBMP metabolism. In the fermentation of plant-based milks, L. mesenteroides notably diminished green, grassy off-flavors—often associated with compounds like IBMP—through metabolic activities including aldehyde reduction and ester synthesis.69 Additionally, L. mesenteroides exhibits efficacy in degrading aldehydes (e.g., hexanal, nonanal) and heterocyclic compounds (e.g., pyrazines) via enzymes such as alcohol dehydrogenases (ADH), aldehyde dehydrogenases (ALDH), and aldehyde reductases; these enzymes catalyze the conversion of offensive aldehydes/ketones into less odorous alcohols or acids, which may indirectly contribute to IBMP breakdown.70 Its enzymatic repertoire also includes β-glucosidases and proteases, which further modify volatile profiles by cleaving glycosidic bonds or facilitating redox reactions, enhancing its ability to improve sensory quality.70,71
Notably, controlled fermentation with L. mesenteroides is critical for producing high-quality fermented foods (e.g., lafun) free of off-flavor compounds like IBMP, as it may inhibit the microbial or biochemical pathways responsible for generating such undesirable VOCs.71 In coffee fermentation, L. mesenteroides is part of the microbial community in wet and semi-dry processes, where it influences aroma by regulating VOC production and degradation—including pyrazines, which are also relevant to IBMP metabolism.72 In selenium-enriched Cardamine violifolia pickle fermentation, L. mesenteroides further demonstrated its role in modifying volatile profiles, reducing unpleasant compounds and enriching desirable aromas, which aligns with its broader capacity to enhance sensory attributes.73
Despite these promising findings, future targeted studies are needed to explicitly quantify IBMP degradation by L. mesenteroides, elucidate the specific metabolic pathways involved, and determine the kinetics of IBMP breakdown. Such research will strengthen the application of L. mesenteroides in controlled fermentation processes to consistently improve the sensory quality of plant-based and fermented food products.
Conclusion
This study successfully developed a scalable process for producing a spray-dried starter culture of Leuconostoc mesenteroides ZN-E with high viability and functional activity, aimed at mitigating off-odors in burdock-based products. Optimal fermentation conditions and medium composition were established to support robust bacterial growth. A heat adaptation strategy was employed to significantly enhance thermotolerance during subsequent processing. Through orthogonal optimization of spray-drying parameters, a starter culture powder with high viable counts was obtained. The spray-dried powder exhibited superior performance compared to liquid cultures, achieving a higher degradation rate of the key off-odor compound IBMP and receiving better sensory evaluation scores. This work establishes an industrially relevant and cost-effective approach for producing stable starter cultures, offering a practical solution for improving the flavor quality of burdock products.
Based on the research findings, the following future research directions are proposed: 1) Optimize the types and ratios of carrier materials (e.g., maltodextrin, gum arabic, skim milk) to further improve the survival rate of L. mesenteroides ZN-E during spray drying and storage; 2) Evaluate the long-term storage stability of the spray-dried starter culture under different conditions (e.g., temperature, humidity, packaging materials) and establish the optimal storage scheme; 3) Extend the application of this spray-dried starter culture to other root vegetables with off-odor problems (e.g., radish, cruciferous vegetables) to verify its universal applicability; 4) Carry out scale-up production experiments to optimize the industrial production process and reduce production costs; 5) Use multi-omics technologies (e.g., transcriptomics, metabolomics) to elucidate the molecular mechanism of L. mesenteroides ZN-E degrading IBMP; 6) Explore micro/nanoencapsulation technologies to coat the spray-dried powder, further enhance cell viability and environmental stress resistance, and promote the commercial development of high-quality fermented vegetable products.
Acknowledgement
We thank all the authors for their rigorous efforts in experimental design, data analysis, and manuscript writing and revision, which made the completion of this research possible. Thank all the authors for their contributions to this research.
Funding Sources
This work was supported by the Jiangsu Agricultural Science and Technology Innovation Fund (CX(22)3062).
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable.
Author Contributions
- Qingyun Bai: Writing – Review and Editing.
- Wenjing Zhou: Writing – Review and Editing,Writing – Original Draft.
- Roberto Lemus: Mondaca: Writing – Review & Editing, Data Curation.
- Wei Han: Formal Analysis.
- Jin Feng: Formal Analysis.
- Ying Li: Formal Analysis.
- Li Cui: Conceptualization, Funding Acquisition.
References
- Zhang X, Herrera-Balandrano DD, Huang W, et al. Comparison of nutritional and nutraceutical properties of burdock roots cultivated in Fengxian and Peixian of China. Foods. 2021;10(9):2095. doi:10.3390/foods10092095
CrossRef - Yosri N, Alsharif SM, Xiao J, et al. Arctium lappa (burdock): insights from ethnopharmacology potential, chemical constituents, clinical studies, pharmacological utility and nanomedicine. Biomed Pharmacother. 2023;158:114104. doi:10.1016/j.biopha.2023.114104
CrossRef - Ha MS, Lee JH, Jeong WM, et al. The combined intervention of aqua exercise and burdock extract synergistically improved arterial stiffness: a randomized, double-blind, controlled trial. Metabolites. 2022;12(10):970. doi:10.3390/metabo12100970
CrossRef - Yang J, Ji Y, Park H, et al. Selection of functional lactic acid bacteria as starter cultures for the fermentation of Korean leek (Allium tuberosum Rottler ex Sprengel). Int J Food Microbiol. 2014;191:164-171. doi:10.1016/j.ijfoodmicro.2014.09.008
CrossRef - Tangyu M, Muller J, Bolten CJ, et al. Fermentation of plant-based milk alternatives for improved flavour and nutritional value. Appl Microbiol Biotechnol. 2019;103:9263-9275. doi:10.1007/s00253-019-10131-7
CrossRef - Zhang C, Chen X, Guo X, et al. A novel strategy for improving the antioxidant, iridoid, and flavor properties of noni (Morinda citrifolia L.) fruit juice by lactic acid bacteria fermentation. LWT. 2023;184:115075. doi:10.1016/j.lwt.2023.115075
CrossRef - Guo M, Kong Q, Wang W, et al. Biotransformation of amygdalin by lactic acid bacteria fermentation. Process Biochem. 2023;132:221-227. doi:10.1016/j.procbio.2023.06.017
CrossRef - Cui L, Zhao X, Zhang D, et al. Isolation and identification of lactic acid bacteria and their effects on the off-odor of burdocks. J Agric Food Chem. 2023;71(19):7485-7494. doi:10.1021/acs.jafc.3c01234
CrossRef - Burton JP, Tannock GW. Properties of porcine and yogurt lactobacilli in relation to lactose intolerance. J Dairy Sci. 1997;80(10):2318-2324. doi:10.3168/jds.S0022-0302(97)76179-9
CrossRef - Homayoonfal M, Malekjani N, Baeghbali V, et al. Optimization of spray drying process parameters for food bioactive ingredients. Crit Rev Food Sci Nutr. 2024;64(17):5631-5671. doi:10.1080/10408398.2023.2187654
CrossRef - Sosnik A, Seremeta KP. Advantages and challenges of the spray-drying technology for the production of pure drug particles and drug-loaded polymeric carriers. Adv Colloid Interface Sci. 2015;223:40-54. doi:10.1016/j.cis.2015.05.003
CrossRef - Broeckx G, Kiekens S, Jokicevic K, et al. Effects of initial cell concentration, growth phase, and process parameters on the viability of Lactobacillus rhamnosus GG after spray drying. Dry Technol. 2020;38(11):1474-1492. doi:10.1080/07373937.2019.1659943
CrossRef - Tan LL, Mahotra M, Chan SY, et al. In situ alginate crosslinking during spray-drying of lactobacilli probiotics promotes gastrointestinal-targeted delivery. Carbohydr Polym. 2022;286:119279. doi:10.1016/j.carbpol.2022.119279
CrossRef - Picot A, Lacroix C. Effects of micronization on viability and thermotolerance of probiotic freeze-dried cultures. Int Dairy J. 2003;13(6):455-462. doi:10.1016/S0958-6946(03)00042-7
CrossRef - Garre E, Raginel F, Palacios A, et al. Oxidative stress responses and lipid peroxidation damage are induced during dehydration in the production of dry active wine yeasts. Int J Food Microbiol. 2010;136(3):295-303. doi:10.1016/j.ijfoodmicro.2009.10.020
CrossRef - Hongbing W. Screening and optimizing of Lactobacillus bulgaricus and Streptococcus thermophilus wort enrichment medium. Food Ferment Ind. 2006;32(6):51-54.
- Yang H, Huang P, Hao L, et al. Enhancing viability of dried lactic acid bacteria prepared by freeze drying and spray drying via heat preadaptation. Food Microbiol. 2023;112:104239. doi:10.1016/j.fm.2023.104239
CrossRef - Khem S, Small DM, May BK. The behaviour of whey protein isolate in protecting Lactobacillus plantarum. Food Chem. 2016;190:717-723. doi:10.1016/j.foodchem.2015.06.015
CrossRef - Xiong J, Wang YY, Yousaf M, et al. Improvement of physicochemical properties and flavour of pickled radish through the use of a direct-vat set starter consisting of Lactiplantibacillus plantarum and Leuconostoc mesenteroides. Int J Food Sci Technol. 2023;58(12):6272-6284. doi:10.1111/ijfs.16256
CrossRef - Ahsan T, Liang C, Yu S, et al. Screening and optimization of fermentation medium for Bacillus velezensis BP-1 and its biocontrol effects against Peyronellaea arachidicola. Appl Sci. 2023;13(8):4653. doi:10.3390/app13084653
CrossRef - Xie W, Li X, Xu H, et al. Optimization of heterotrophic culture conditions for the microalgae Euglena gracilis to produce proteins. Mar Drugs. 2023;21(10):519. doi:10.3390/md21100519
CrossRef - Venkateswarulu T, Prabhakar KV, Kumar RB. Optimization of nutritional components of medium by response surface methodology for enhanced production of lactase. 3 Biotech. 2017;7(3):202. doi:10.1007/s13205-017-0847-0
CrossRef - Cui S, Zhu D, Mao B, et al. Rapid evaluation of optimal growth substrates and improvement of industrial production of Bifidobacterium adolescentis based on the automatic feedback feeding method. LWT. 2021;143:110960. doi:10.1016/j.lwt.2021.110960
CrossRef - Zhang H, Chen P, Xu L, et al. Construction of cordycepin high-production strain and optimization of culture conditions. Curr Microbiol. 2023;80(1):12. doi:10.1007/s00284-022-03076-4
CrossRef - Guo Q, Li S, Tang J, et al. Microencapsulation of Lactobacillus plantarum by spray drying: protective effects during simulated food processing, gastrointestinal conditions, and in kefir. Int J Biol Macromol. 2022;194:539-545. doi:10.1016/j.ijbiomac.2021.11.173
CrossRef - Vassaux A, Rannou M, Peers S, et al. Impact of the purification process on the spray-drying performances of the three families of lipopeptide biosurfactant produced by Bacillus subtilis. Front Bioeng Biotechnol. 2021;9:815337. doi:10.3389/fbioe.2021.815337
CrossRef - Nie S, Li L, Wang Y, et al. Discrimination and characterization of volatile organic compound fingerprints during sea bass (Lateolabrax japonicus) fermentation by combining GC-IMS and GC-MS. Food Biosci. 2022;50:102048. doi:10.1016/j.fbio.2022.102048
CrossRef - Zhang X, Guan H, Zhao Q, et al. Effect of thermal treatment on the flavor quality of Chinese spicy cabbage. Food Control. 2023;144:109338. doi:10.1016/j.foodcont.2022.109338
CrossRef - Kafkas E, Cabaroglu T, Selli S, et al. Identification of volatile aroma compounds of strawberry wine using solid-phase microextraction techniques coupled with gas chromatography-mass spectrometry. Flavour Fragr J. 2006;21(1):68-71. doi:10.1002/ffj.1558
CrossRef - Ryona I, Pan BS, Intrigliolo DS, et al. Effects of cluster light exposure on 3-isobutyl-2-methoxypyrazine accumulation and degradation patterns in red wine grapes (Vitis vinifera L. cv. Cabernet Franc). J Agric Food Chem. 2008;56(22):10838-10846. doi:10.1021/jf801677t
CrossRef - Dan T, Ren W, Liu Y, et al. Volatile flavor compounds profile and fermentation characteristics of milk fermented by Lactobacillus delbrueckii subsp. bulgaricus. Front Microbiol. 2019;10:2183. doi:10.3389/fmicb.2019.02183
CrossRef - Drosinos EH, Mataragas M, Nasis P, et al. Growth and bacteriocin production kinetics of Leuconostoc mesenteroides E131. J Appl Microbiol. 2005;99(6):1314-1323. doi:10.1111/j.1365-2672.2005.02745.x
CrossRef - Pan L, Wang Q, Qu L, et al. Pilot-scale production of exopolysaccharide from Leuconostoc pseudomesenteroides XG5 and its application in set yogurt. J Dairy Sci. 2022;105(2):1072-1083. doi:10.3168/jds.2021-20936
CrossRef - Krier F, Revol-Junelles AM, Germain P. Influence of temperature and pH on production of two bacteriocins by Leuconostoc mesenteroides subsp. mesenteroides FR52 during batch fermentation. Appl Microbiol Biotechnol. 1998;50(3):359-363. doi:10.1007/s002530051300
CrossRef - Vasyliuk O, Garmasheva I, Skrotskyi S, et al. The influence of the conditions of cultivation of lactic acid bacteria on their antagonistic activity against some plant pathogenic bacteria. Plant Direct. 2025;9(9):e70101. doi:10.1002/pld3.70101
CrossRef - Fernandez de Ullivarri M, Buttimer C, Wijman J, et al. Cold induced expression of a novel levansucrase gene sacB1 enhances exopolysaccharide production and stress resilience in Leuconostoc mesenteroides. Sci Rep. 2025;15(1):22980. doi:10.1038/s41598-025-22980-3
CrossRef - Bernardo MP, Coelho LF, de Lima CJB, et al. Isolation and characterization of bacterial producers of optically pure D(-) and L(+) lactic acid. Afr J Microbiol Res. 2013;7(21):2618-2628.
CrossRef - Liu SD, Wu YN, Wang TM, et al. Maltose utilization as a novel selection strategy for continuous evolution of microbes with enhanced metabolite production. ACS Synth Biol. 2017;6(12):2326-2338. doi:10.1021/acssynbio.7b00227
CrossRef - Owoade A, Alshami AS, Sparling R, et al. Optimization of process parameters and medium components for carbon monoxide bioconversion with Clostridium autoethanogenum. Biofuels Bioprod Bioref. 2024;18(6):1940-1951. doi:10.1002/bbb.2589
CrossRef - Yáñez-Fernández J, Herrera Ovando MG, Patlán Ramírez L, et al. Factorial design to optimize dextran production by the native strain Leuconostoc mesenteroides SF3. ACS Omega. 2021;6(46):31203-31210. doi:10.1021/acsomega.1c04678
CrossRef - Majumder A, Bhandari S, Purama RK, et al. Enhanced production of a novel dextran from Leuconostoc mesenteroides NRRL B-640 by response surface methodology. Ann Microbiol. 2009;59(2):309-315. doi:10.1007/BF03178367
CrossRef - Han J, Hang F, Guo B, et al. Dextran synthesized by Leuconostoc mesenteroides BD1710 in tomato juice supplemented with sucrose. Carbohydr Polym. 2014;112:556-562. doi:10.1016/j.carbpol.2014.06.033
CrossRef - Jo SJ, Oh SM, Jang EK, et al. Physicochemical properties of carrot juice fermented by Leuconostoc mesenteroides SM. J Korean Soc Food Sci Nutr. 2008;37(2):210-216. doi:10.3746/jkfn.2008.37.2.210
CrossRef - Escobar-Beiza N, Pérez-Correa JR, Franco W. Fermentation of murta (Ugni molinae) juice: effect on antioxidant activity and control of enzymes associated with glucose assimilation. Int J Mol Sci. 2023;24(20):15678. doi:10.3390/ijms242015678
CrossRef - Schifano E, Tomassini A, Preziosi A, et al. Leuconostoc mesenteroides strains isolated from carrots show probiotic features. Microorganisms. 2021;9(11):2290. doi:10.3390/microorganisms9112290
CrossRef - Kimpel JA, Nagao RT, Goekjian VH, et al. Regulation of the heat-shock response in soybean seedlings. Plant Physiol. 1990;94(3):988-995. doi:10.1104/pp.94.3.988
CrossRef - Desmond C, Stanton C, Fitzgerald GF, et al. Environmental adaptation of probiotic lactobacilli towards improvement of performance during spray drying. Int Dairy J. 2001;11(10):801-808. doi:10.1016/S0958-6946(01)00115-9
CrossRef - Ardanareswari K, Utami T, Rahayu ES. Effect of heat adaptation and pH adjustment on the survival of spray-dried Lactobacillus paracasei SNP2. Br Food J. 2017;119(10):2267-2276. doi:10.1108/BFJ-02-2017-0080
CrossRef - Paéz R, Lavari L, Vinderola G, et al. Effect of heat treatment and spray drying on lactobacilli viability and resistance to simulated gastrointestinal digestion. Food Res Int. 2012;48(2):748-754. doi:10.1016/j.foodres.2012.06.020
CrossRef - Nunes GL, Motta MH, Cichoski AJ, et al. Encapsulation of Lactobacillus acidophilus La-5 and Bifidobacterium Bb-12 by spray drying and evaluation of its resistance in simulated gastrointestinal conditions, thermal treatments and storage conditions. Cienc Rural. 2018;48(6):e20170711. doi:10.1590/0103-8478cr20170711
CrossRef - Fonseca MT, Vital AC, Silva MB, et al. Improving the stability of spray-dried probiotic acerola juice: a study on hydrocolloids’ efficacy and process variables. Food Bioprod Process. 2024;147:209-218. doi:10.1016/j.fbp.2024.05.004
CrossRef - Kiepś J, Dembczyński R. Current trends in the production of probiotic formulations. Foods. 2022;11(15):2330. doi:10.3390/foods11152330
CrossRef - Santivarangkna C, Kulozik U, Foerst P. Inactivation mechanisms of lactic acid starter cultures preserved by drying processes. J Appl Microbiol. 2008;105(1):1-13. doi:10.1111/j.1365-2672.2008.03744.x
CrossRef - Kresnowati MTAP, Bindar Y, Rahmina F. Development of instant microbial starter for production of fermented cassava flour: effect of vacuum drying temperature, carrier media, and storage temperature. In: Proceedings of the 3rd International Conference on Industrial Biotechnology. Bandung Institute of Technology; 2018.
CrossRef - Liu J, Xie S, Xu M, et al. Screening the protective agents able to improve the survival of lactic acid bacteria strains subjected to spray drying using several key enzymes responsible for carbohydrate utilization. Microorganisms. 2024;12(6):1094. doi:10.3390/microorganisms12061094
CrossRef - Vermelho AB, Moreira JV, Junior AN, et al. Microorganisms, drying, and preservation processes in the baking industry. Preprints. 2023;2023050123. doi:10.20944/preprints202305.0123.v1
CrossRef - Farag MA, Jomaa SA, El-Wahed AA, et al. The many faces of kefir fermented dairy products: quality characteristics, flavour chemistry, nutritional value, health benefits, and safety. Nutrients. 2020;12(2):346. doi:10.3390/nu12020346
CrossRef - de Almeida KV, Zanetti VC, Camelo-Silva C, et al. Powdered water kefir: effect of spray drying and lyophilization on physical, physicochemical, and microbiological properties. Food Chem Adv. 2024;5:100759. doi:10.1016/j.focha.2024.100759
CrossRef - Garcia C, Guerin M, Souidi K, et al. Lactic fermented fruit or vegetable juices: past, present and future. Beverages. 2020;6(1):8. doi:10.3390/beverages6010008
CrossRef - Vera Peña MY, Rodriguez Rodriguez WL. Effect of pH on the growth of three lactic acid bacteria strains isolated from sour cream. Rev Investig Vet Peru. 2020;31(3):e17879.
- Cai Z, Ruan Y, He J, et al. Effects of microbial fermentation on the flavor of cured duck legs. Poult Sci. 2020;99(9):4642-4652. doi:10.1016/j.psj.2020.06.014
CrossRef - Al-Kharousi ZS. Highlighting lactic acid bacteria in beverages: diversity, fermentation, challenges, and future perspectives. Foods. 2025;14(12):2043. doi:10.3390/foods14122043
CrossRef - Wätjen AP, De Vero L, Carmona EN, et al. Leuconostoc performance in soy-based fermentations: survival, acidification, sugar metabolism, and flavor comparisons. Food Microbiol. 2023;115:104337. doi:10.1016/j.fm.2023.104337
CrossRef - Poltronieri P, Rossi F. Challenges in specialty coffee processing and quality assurance. Challenges. 2016;7(2):19. doi:10.3390/challe7020019
CrossRef - Kumar Verma D, Thyab Gddoa Al-Sahlany S, Kareem Niamah A, et al. Recent trends in microbial flavour compounds: a review on chemistry, synthesis mechanism and their application in food. Saudi J Biol Sci. 2022;29(3):1565-1576. doi:10.1016/j.sjbs.2021.11.034
CrossRef - Sugahara H, Kato S, Nagayama K, et al. Heterofermentative lactic acid bacteria such as Limosilactobacillus as a strong inhibitor of aldehyde compounds in plant-based milk alternatives. Front Sustain Food Syst. 2022;6:965986. doi:10.3389/fsufs.2022.965986
CrossRef - Tangyu M, Fritz M, Tan JP, et al. Flavour by design: food-grade lactic acid bacteria improve the volatile aroma spectrum of oat milk, sunflower seed milk, pea milk, and faba milk towards improved flavour and sensory perception. Microb Cell Fact. 2023;22(1):133. doi:10.1186/s12934-023-02132-7
CrossRef - Molina GES, Ras G, Barone G, et al. Multiphasic and mixture lactic acid bacteria screening approach for the removal of antinutrients and off-flavors present in a pea, oat and potato blend. Food Res Int. 2024;197:115200. doi:10.1016/j.foodres.2024.115200
CrossRef - Fawole AO, Karatzas KA, Parker JK, et al. Impact of selected starters and cassava varieties on the proximate, rheological, and volatile profiles of lafun. Foods. 2025;14(4):660. doi:10.3390/foods14040660
CrossRef - Elhalis H, Cox J, Zhao J. Coffee fermentation: expedition from traditional to controlled process and perspectives for industrialization. Appl Food Res. 2023;3(1):100253. doi:10.1016/j.afres.2023.100253
CrossRef - Gong J, Rao S, Liu X, et al. Comparative study on selenium and volatile compounds in selenium-enriched Cardamine violifolia pickles fermented by three distinct methods. Fermentation. 2024;10(12):632. doi:10.3390/fermentation10120632
CrossRef













