Gelrite-Gellan Gum Biopolymer Films Reinforced with AKBA derivative (SK-C5) for Biodegradable Food Packaging Applications
1Natural and Medical Sciences Research Center, University of Nizwa, Birkat Al Mauz Nizwa, Oman.
2School of Health Science, University of Petroleum and Energy Studies, Dehradun, India.
Corresponding Author Email: sbsaurabhbhatia@gmail.com
DOI : http://dx.doi.org/10.12944/CRNFSJ.14.1.19
ABSTRACT:The current study explores the fabrication and characterization of gelrite gellan gum biopolymer films loaded with boswellic acid derivative (1-(4-Methoxyphenyl)-1H-1,2,3-triazol-4-yl) methyl) carbamoyl) 3α‐acetyloxy‐11‐oxo‐urs‐12‐en‐24‐oate (SK-C5). The synthesized SK-C5 was successfully integrated into the gelrite gellan gum films using the casting method. The physicochemical structural properties of the produced films were characterized. The obtained values were measured by using One-way ANOVA was used followed by Fisher’s post hoc test at a 5% significance level. The antioxidant activity of the patterned films improved via increasing the ABTS radical scavenging activity from 14.46% to 33.50% compared to its control sample. The incorporation of SK-C5 also led to an increase in the thickness and haze value of the film samples and a decrease in the transmittance and swelling index of the film samples. The functional group between the polymer and SK-C5 was observed by using FTIR spectroscopy. The mechanical properties of the films did not change significantly. Moreover, the surface hydrophobicity of the film surface was increased. The results indicate that boswellic acid derivatives-loaded biopolymer-based packaging material possess great a potential as active food packaging.
KEYWORDS:Antioxidant; Boswellic Acid; Biopolymer; Food Packaging; Hydrophobic Films
Introduction
Food packaging is a fundamental component of the global supply chain as it determines quality and safety during the final stages of industrial food processing. It plays an essential role in protecting food products from physical damage, contamination, and spoilage while providing safety and hygiene.1 Due to their non-biodegradable nature and their hazardous effects on human health and the environment, plastic-based packaging materials are highly criticized.2 Natural polymers, or biopolymers, are derived from living organisms are considered as sustainable alternatives to petroleum-based plastics. Their extensive use is due to their biodegradability, renewability, and availability.3
Gellan gum is a high-molecular-weight anionic water soluble polysaccharide produced by the aerobic fermentation of Sphingomonas elodea (formerly Pseudomonas elodea) culture. The U.S. Food and Drug Administration has approved its use in food products.4 Gellan gum has potential in packaging material due to its outstanding biocompatibility, safety, solubility, and biodegradability. However, pure gellan gum films have relatively poor mechanical strength, limited barrier properties, and weak antibacterial activity, which restrict their commercial use.5
Biodegradable active packaging has been studied extensively for their potential to extend the shelf life of the food. The growing trend for sustainable and eco-friendly packaging has led to breakthroughs in biobased materials.6 Most of the active packaging materials focused on either absorbing or releasing specific substances using the materials. These include oxygen, ethylene, humidity, carbon dioxide, and taste or smell. In addition, some packaging systems are designed to release carbon dioxide, antimicrobial agents, antioxidants, and flavors, to enhance product quality and extend the shelf life of the product.7 Yildiz et al. reported that the critic acid acts as a cross-linker in chitosan and chick pea flour biopolymer films and also improved the functional characteristics of the developed films.8 Luzi et al. reported that the incorporation of gallic acid in PVA-based biopolymer films improved the antioxidant activity of the fabricated samples.9 Zhou, X. et al. reported that the polyphenols isolated from tea significantly improved the antioxidant and barrier properties of the chitosan and bacterial cellulose films.10 Pastor et al. reported that the addition of fraction of propolis significantly improved the antioxidant activity chitosan-based films.11 Plant extracts have been proven to be rich sources of phytochemicals that can improve the functional properties of biopolymer-based packing films. Using new phytochemicals obtained from natural plant materials active packaging can be designed to protects the texture, color, aroma, and flavor of food products and increases the functional characteristics of food.12 Boswellic acid is a pentacyclic triterpene derived from the aromatic gum resin present in the barks of the Boswellia serrata Roxb. Tree.13
Boswellic acids, including β-boswellic acid (β-BA), 3-O-acetyl-β-boswellic acid (β-ABA), 11-keto-β-boswellic acid (β-KBA), and 3-O-acetyl-11-keto-β-boswellic acid (β-AKBA, 1) (see Fig. 1), derived from the gum resin of Boswellia sacra, are potential alternatives to existing anti-cancer drugs and have significant pharmacological effects.14
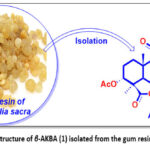 |
Figure 1: Chemical structure of β-AKBA (1) isolated from the gum resin of Boswellia sacra. |
The primary objective of this study was to assess the effects of the addition of boswellic acid derivative on the physical, chemical, and antioxidant properties of gelrite gellan gum-based biopolymer films.
Materials and Methods
Materials
The Gelrite Gellan Gum (72394) was purchased from Sisco Research Laboratories Pvt. Ltd, Maharashtra, Inda, and the glycerol was bought from BDH Laboratory Supplies in Dorset, UK.
All reagents sourced in Germany were obtained from Sigma-Aldrich. The silica gel utilized for column chromatography had a mesh size of 100–200. High-resolution electrospray ionization mass spectra (HR-ESI-MS) was obtained using the Agilent 6530 LC Q-TOF instrument. The 1H and 13C NMR spectra were obtained with CDCl3 as the solvent. A Bruker NMR spectrometer operating at 600 MHz was used for 1H and 150 MHz for 13C. pre-coated silica gel plates F254 were employed for thin layer chromatography (TLC), and spot visualization was conducted using UV light and I2 stain. The organic extracts were thoroughly dried, and pure compound solutions were also dried accordingly.
General procedure for the synthesis of (Prop-2-yn-1-ylcarbamoyl) 3α‐acetyloxy‐11‐oxo‐urs‐12‐en‐24‐oate (3)
A solution of β-AKBA (1) (1.0 equiv.) mixed with HATU (1.2 equiv.) in anhydrous DMF was combined with propargylamine (2) (1.1 equiv.) and DIPEA (2.5 equiv.). This mixture was stirred at room temperature for 10 to 12 hours, and further examined by TLC. After completion, the resultant product was extracted with ethyl acetate, dried over MgSO₄, and concentrated under reduced pressure. The crude product was purified by silica gel column chromatography using n-hexane/ethyl acetate (85:15 v/v) as the eluent. Recrystallization from methanol produced the final compound 3 as light green crystals with a yield of 94% and a melting point of 196 to 198 °C. Structural characterization was confirmed through spectroscopic analysis, including ¹H NMR, ¹³C NMR, and HRMS, which validated the molecular formula C₃₅H₅₂NO₄, featuring a molecular ion peak at m/z 550.39124 ([M+H]⁺).
General procedure for the synthesis of (1-(4-Methoxyphenyl)-1H-1,2,3-triazol-4-yl) methyl) carbamoyl) 3α‐acetyloxy‐11‐oxo‐urs‐12‐en‐24‐oate (5) (SK-C5)
A solution containing compound 3 (1.0 equiv.) and 4-methoxyphenyl azide (4) (1.2 equiv.) was prepared in 10 mL of acetonitrile. Copper(I) iodide (2.0 equiv.) and triethylamine (3.0 equiv.) were then added. The mixture was stirred at room temperature for three hours, with its progress monitored by thin-layer chromatography (TLC). Once the reaction was complete, ethyl acetate (30 mL) was added, followed by a saturated aqueous solution of ammonium chloride (15 mL). The aqueous layer subjected to extraction with ethyl acetate (3 × 30 mL), and the combined organic extracts followed by washing with brine (20 mL). After drying with anhydrous magnesium sulfate, the solution was filtered and concentrated under reduced pressure. The crude product was purified through flash column chromatography using a n-hexane/ethyl acetate mixture (75:25), resulting in the 1H-1,2,3-triazole analogue of β-AKBA (5), obtained as a gummy white solid with a yield of 96%. Its structure was confirmed by spectroscopic methods, including ¹H NMR, ¹³C NMR, and HRMS, which affirmed the molecular formula C42H59N₄O5 and identified a [M+H]⁺ peak at 699.44668 (calculated 699.44682).
Films fabrication
To fabricate the biopolymer films traditional casting method was used. In this method, a 1g of gelrite gellan gum was mixed in 100 mL of distilled water to make 1% (w/w) gelrite gellan gum solution. The solution was placed on a magnetic stirrer with continuous stirring at 300 rpm for almost 3 hours at 30 °C. A 0.5% of glycerol (v/w according to relative to total polymer content) added to the resultant in the solution. After the formation of homogenous mixture, the solution was separated into two beakers. The control solution without SK-C5 was labelled as GA1. Parallelly, 0.1% of SK-C5 was mixed in the other beaker and was labelled as GA2, as shown in Table 1. After the complete homogenization of solution, the 20 mL solution from each beaker was poured into 90 x 15 mm petri plates. The film-forming solution was subjected to drying at 25 °C for 48 hours. After drying, the fabricated films were carefully peeled off and subjected to further analysis.
Table 1: The composition ratios of the components used to fabricate edible films.
|
Sample Code |
Polymer | Additive | Glycerol |
| GA1 | Gelrite Gellan Gum (1%) | – |
Gly (0.5%) |
|
GA2 |
Gelrite Gellan Gum (1%) | SK-C5 (0.1%) |
Gly (0.5%) |
Thickness
The thickness of the bilayer film samples was measured from different regions of each fabricated sample by using Digital micrometer (Dequmont digital vernier caliper (ROHS NORM 2011/65/EU)). The film sample were measured at five different regions and average value was calculated.
Transmittance and Haze Value
The light transmittance (T) and Haze value (H) of the fabricated bilayer bioactive film samples were determined by using a Haze meter model YH1200 from Guangdong Sanenshi Technology Co., Ltd. China. The measurements were taken in triplicates and average value was expressed in percentage (%).
Color analysis
The color parameters were measured by using a portable digital chromameter (model CR-410) by Konica Minolta, INC, Japan. The color of packaging material is usually determined using three parameters: L*, a*, and b* (CIELAB color parameters). Where, the higher L* value determines the lightness, lower L* value determines darkness, negative a* value determines greenish hue, positive a* value determines reddish hue, negative b* value determines blue color, and positive b* value determines yellow color. The analysis was done against a white standard white background. The overall color difference (∆E) was measured by using the equation reported in our previous studies. Three different reading were taken from three different sites of each sample and average value was determined.
Mechanical properties
The mechanical properties such as tensile strength (TS), elongation at break (EB) and puncture resistance was measured in triplicate by using a texture analyzer model TA.XTplus C from Stable Micro Systems (TA Instruments), Godalming, UK. The values were measured by using ASTM D882 method. First, the film samples were cut into 7 x 60 mm dimension strips. These strips were placed in between the jaws of probe (named Tensile Grips) and the analysis was done by using Exponent Software. The mean values of film samples were expressed in MPa for TS and % for EB.
Antioxidant assessment
The current study followed the standard procedure per the ABTS cation scavenging assay described by Re et al.15 In this study, 5 mg of film sample was immersed in 1.95 mL methanolic DPPH solution. The absorbance of the solution was then measured at 517 nm using a UV–Vis spectrophotometer. The antioxidant activity was expressed in percentage (%) and was determined by using the formula:
![]()
Ac denotes the absorbance of the control, and At represents the absorbance of the film samples.
Water Contact Angle (WCA)
The surface properties such as water contact angle (hydrophobic or hydrophilic) was determined by using goniometer (OCA11, Data Physics Instruments). A film strip was cut into 2 x 2 cm dimension and placed on a flat surface and a 1µL water droplet was dropped on the surface of the strip. The WCA was determined by using dpiMAX software. The readings were taken in triplicates. The mean values were determined and WCA expressed in degree (°).
Swelling Index (SI)
Erdem et al.16 methodologies were used to measure the swelling index of GA biopolymer film samples. In the method, a square film strip of 2 x 2 cm2 was cut for both samples. Initial weight of both samples was recorded as W1. Both the samples were immersed in a deionized water for 2 mins at a temperature of 25 °C. After 2 mins, the excess water was removed through blotting by using a filter paper. The final weight of both samples was recorded was W2. Following equation was used to measure the SI of the sample and the values were recorded in percentage (%).
![]()
Morphological properties
The scanning electron microscopy (SEM) was used to check the morphological characteristics of bilayer bioactive films. For this, SEM model JSM6510LA from Jeol, Japan was used. To generate micrographs, the SEM was set at 20kV voltage. For surface area and for cross-sectional area x300 magnification was used. The aluminium stubs with double sided adhesive tapes were used to prepare the film samples. The samples were coated with a thin layer of gold prior to imaging.
FTIR spectrometry
The functional groups present in developed biopolymer films were determined by using FTIR spectroscopy. An InfraRed Bruker Tensor 37 Spectrometer from Ettlingen, Germany was used to obtain the spectra of each film sample. The 4000–400 cm⁻1 range with a resolution of 4 cm−1 was used. The scans were further processed by using Origin Pro software (1.5 2024).
Statistical analysis
The mean values and standard errors from experiments conducted in triplicate were calculated. One-way ANOVA was used followed by Fisher’s post hoc test at a 5% significance level to measure the values.
Results
Synthesis of 1H-1,2,3-triazole analogue of β-AKBA (5)
The synthesis of 1H-1,2,3-triazole analogue of β-AKBA (5) is represented in Fig. 2. The natural compound of β-AKBA (1) was obtained from Boswellia sacra gum resin. This extracted compound contains a free carboxylic acid group at C-24. The HATU coupling agent was used to initiate a reaction between the extracted compound (1) and propargyl anime (2). This resulted in the formation of intermediate compound (3) with the yield of 94%. Moreover, the reaction between compound (3) with 4-methoxyphenyl azide (4) in the presence of copper iodide (Cul), triethylamine (Et3N) as the base, and acetonitrile as the solvent results in the synthesis of 1H-1,2,3-triazole analogue of β-AKBA (5). This reaction is carried out at room temperature for 3 hours. This achieves a high yield of 96%.14
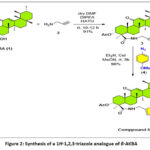 |
Figure 2: Synthesis of a 1H-1,2,3-triazole analogue of β-AKBA |
Visual Characteristics
Fig. 3 demonstrates the visual analysis of fabricated films. Both the samples demonstrated a homogenous and uniform structure. The GA1 sample demonstrated a transparent and glossy surface. However, the GA1 sample was somehow difficult to peel off. The GA2 sample was easy to peel off the petri plate. It demonstrated a cloudy and opaque appearance. Furthermore, the GA2 sample demonstrated superior thickness, rigidity, flexibility, and stiffness than GA1 sample..
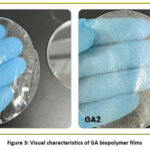 |
Figure 3: Visual characteristics of GA biopolymer films |
Thickness
Table 2 demonstrates the thickness of GA biopolymer films. There was a significant increase in the thickness from 0.07±0.01 mm to 0.10±0.01mm of fabricated films. The GA1 film sample showed a lower thickness of 0.07±0.01 mm. However, the GA2 film sample demonstrated a higher thickness of 0.10±0.01 mm.
Transmittance and Haze Value
The haze value of GA film samples significantly increased from 19.02% (GA1) to 48.24% (GA2), as shown in Table 2. The light transmittance of the GA biopolymer films showed a significant decrease. The GA1 showed the highest transmittance value of 92.10%. The GA2 film sample showed the lowest value of transmittance (88.14%).
Table 2: Thickness, Transmittance, and Haze Value of biopolymer films.
|
Sample Code |
Thickness (mm) | Transmittance (%) | Haze Value (%) |
|
GA1 |
0.07±0.01b | 92.1±0.66a |
19.02±1.66b |
| GA2 | 0.10±0.01a | 88.14±0.16b |
48.24±1.84a |
*Values within a column labeled with distinct letters (such as a and b) indicate significant variances (p < 0.05).
Mechanical attributes
Table 3 demonstrates the mechanical attributes of biopolymer films loaded with SK-C5. The TS of the GA1 film sample was slightly higher than that of GA2. In contrast, the elongation at break EAB of the GA2 film sample was slightly greater value than that of the GA1 film sample. However, neither the TS nor the EAB exhibited any significant decrease in the control film samples or the SK-C5-loaded film samples.
Table 3: Mechanical properties (Tensile strength and Elongation at break) of biopolymer films.
|
Sample Code |
Tensile Strength (MPa) | Elongation at Break (%) |
| GA1 | 0.03±0.01a |
95.45±9.3a |
|
GA2 |
0.02±0.005a |
97.25±5.75a |
*Values within a column labeled with distinct letters (such as a and b) indicate significant variances (p < 0.05).
Color characteristics of the films
The GA1 film samples showed a significantly greater L value (1.70±0.07) when compared to GA2 (1.05±0.28), as shown in Table 4. The negative a* values of both samples indicate the presence of green color in the polymer matrix. The positive value b* value indicates the presence of yellowish hue in the polymer matrix. The significant decrease in overall change in color (ΔE) of the film samples was observed.
Table 4: Color analysis of biopolymer films.
|
Sample Code |
L | a* | b* | ΔE |
| GA1 | 1.70±0.07a | -0.11±0.01a | 0.24±0.03b |
1.72±0.07a |
|
GA2 |
1.05±0.28b | -0.12±0.02a | 0.43±0.01a |
1.14±0.27b |
*Values within a column labeled with distinct letters (such as a and b) indicate significant variances (p < 0.05).
Antioxidant assessment
The ABTS radical scavenging activity of GA biopolymer films is shown in Fig. 4. A significant increase from 14.46% to 33.50% in ABTS radical scavenging activity of GA biopolymer films was observed. The higher ABTS radical scavenging activity in the GA2 film sample was observed.
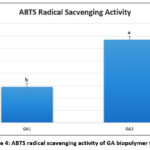 |
Figure 4: ABTS radical scavenging activity of GA biopolymer films. |
Morphological properties
Fig. 5 demonstrated the morphological characteristics of the fabricated biopolymer films. The control sample (GA1) demonstrated smooth, homogenous, and compact surface no visible cracks or pores were observed. The GA2 sample revealed a slightly rough and irregular surface with micro-aggregates on the surface of the fabricated films. The cross-sectional area of the GA1 sample exhibited a dense uniform internal structure. The SK-C5 loaded sample (GA2) demonstrated a more heterogenous internal structure with uneven layers and small voids as well as ridgesacross the cross-sectional areas.
 |
Figure 5: Morphological characteristics of GA biopolymer films. |
Water Contact Angle (WCA)
Table 5 and Fig. 6 demonstrates the water contact angle (WCA) of GA biopolymer films. The WCA contact angle significantly increased with the addition of boswellic acid derivative in the film matrix. The lower WCA of 55.38° was observed in GA1 sample. The GA2 sample demonstrated significantly higher WCA of 73.74°. Table 5: Water contact angle (WCA) and Swelling Index (SI) of GA biopolymer films.
Table 5: Water contact angle (WCA) and Swelling index (SI) of biopolymer films.
|
Sample Code |
WCA (°) | Swelling Index (%) |
| GA1 | 55.38±6.94b |
25.10±1.58a |
|
GA2 |
73.74±1.96a |
21.59±1.43b |
*Values within a column labeled with distinct letters (such as a and b) indicate significant variances (p < 0.05).
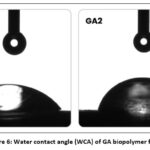 |
Figure 6: Water contact angle (WCA) of GA biopolymer films. |
Swelling Index (SI)
Table 5 demonstrates the swelling index (SI) of GA biopolymer films. A significant decrease in the swelling index (25.10% to 21.59%) of the fabricated samples was observed. The GA1 sample demonstrated the highest SI. The addition of SK-C5 in the polymer matrix significantly reduced the swelling index of the fabricated samples.
FTIR Spectroscopy
Fig. 7 demonstrates the FTIR spectrogram of GA biopolymer films. A broad spectrum was observed at 3296 cm⁻¹. Another peak was observed at 2925 cm⁻¹. The absorption band observed at 1610 cm⁻¹, 404 cm⁻¹, 1029 cm⁻¹, 918 cm⁻¹, and 480 cm⁻¹. The addition of SK-C5 in the polymer matrix demonstrated a significant change in the spectra of the fabricated samples. The band was observed in between 3278 cm⁻¹ and 3882 cm⁻¹. The absorption bands at 1618 cm⁻¹, 1398 cm⁻¹ and 1240 cm⁻¹ were also observed.
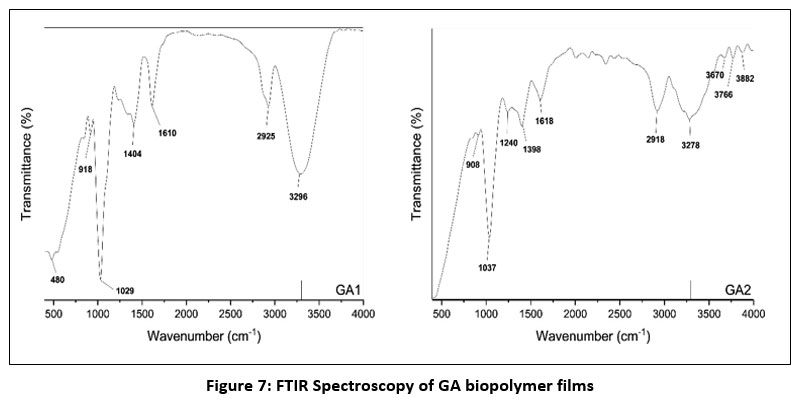 |
Figure 7: FTIR Spectroscopy of GA biopolymer films |
Discussion
The difficulty in peeling the GA1 sample might be attributed to the polymer characteristics The cloudy and opaque appearance of the GA2 sample might be attributed to the presence of SK-C5. The incorporation of a SK-C5 into the polymer matrix resulted in increased fragility compared to other formulations.
The increase in the thickness of GA2 film samples might be attributed to the addition of boswellic acid derivative in the polymer film matrix. Zinna O et al. reported that the incorporation of bioactive components and protein hydrolysates in alginate-based films increased the thickness of the fabricated samples.15 Gautam, S. et al. reported that the sodium carboxymethyl cellulose (CMCNa)-based biopolymer crosslinked with citric acid and loaded with bioactive components significantly increased the thickness of developed films.16
Transmittance measures the fraction of light that passes through a material, with higher values indicating greater light transmission. The haze of a film is the proportion of normally incident-transmitted light that scatters at an angle greater than 2.5° from the direct transmission path.17 The results indicates that the control film sample are more transparent. This might be attributed to formation of more homogenous and less dense polymer matrix. However, the addition of boswellic acid derivative resulted in increase in the scattering of light within the polymer matrix. That decreases the transparency of fabricated films. Bharti, S. K. et al. reported that the haze value of carrageenan and M. esculenta starch based biocomposite films loaded with caraway essential oil increased significantly.18
The performance of a packaging material is determined by the mechanical properties such as tensile strength (TS) and elongation at break (EAB). These properties determined the suitability of packaging material for various manufacturing processes, including printing, lamination, and packaging. These attributes also plays an important role in ensuring the durability of packaging material during transport, handling, and storage.20 A non-significant change in the mechanical properties of the films was observed. This might be due to the addition of SK-C5 in the film matrix having a minor impact on the mechanical properties of fabricated biopolymer films. Thakhiew, W. et al. reported that the addition of green tea extract up to a certain limit did not significantly alter the mechanical properties such as tensile strength and elongation at break of the fabricated films.21 Sukoco, A., et al. developed a double-layered biopolymer film composed of modified zein-gum arabica complex loaded with fish oil and guava leaf extract. They found that the addition of guava leaf extract did not significantly alter the mechanical properties of the fabricated samples.22
The L value represents the lightness of the GA biopolymer films. The lower L value of the GA2 film sample suggests that the film sample became less transparent due to the addition of the boswellic acid derivative. The a* value signifies the red-green color, with a positive a* indicates redness and a negative a* indicates greenness. The increase in negative a* value in GA2 suggests that the incorporation of boswellic acid derivative contributed to the increase in greenish tint of the GA2 sample. The b* value signifies the yellow-blue color, with a positive b* indicates yellow and a negative b* indicates blue color. The significant increase in positive b* value indicates that the incorporation of boswellic acid derivative significantly contributed to the increase in yellowish color of the fabricated films. The significant decrease in overall change in color (ΔE) of the film samples was observed. Choi I, et al. reported the change in color of the biopolymer films with the addition of apple skin particles in the polymer matrix.23
The increase in ABTS radical scavenging activity might be attributed to the presence of boswellic acid derivative in the film matrix. Al-Saban et al. reported that the extract of Boswellia sacra possesses a significant antioxidant property that can lead to increase in the radical scavenging activity.24 Moreover, the GA1 film sample lacks boswellic acid derivatives and exhibits lower antioxidant activity. This indicates that the gelrite gellan gum25 and glycerol26 alone did not possess strong antioxidant properties. Pereira JF, et al. reported that the addition of vitamin C and catuaba extract increased the radical scavenging activity of the developed film samples.27 Chaari, M. et al. reported an increase in the antioxidant activity of the gelatin and sodium alginate biocomposite films loaded with the beet root peel extract.28
The smooth and homogenous structure of GA1 sample indicates uniform polymer chains distribution and effective plasticization by glycerol. The slight rough structure of GA2 sample might be attributed to the partial aggregation of SK-C5 within the polymer matrix. Ng, J. at al. observed that the addition of palm kernel oil nano emulsion in gellan gum resulted in the formation of micro-aggregates on the surface of the developed films.29 In another study by Flores, Z. et al. also found the micro-aggregates on the surface of the chitosan-based films loaded with carvacrol content.30
The low WCA of GA1 sample suggests that it is more hydrophilic in nature. This allows the water droplet to spread more easily on the film surface. This might be attributed to the hydrophilic nature of both gelrite gellan gum and glycerol. The higher WCA of GA2 sample suggests that the incorporation of boswellic acid derivative enhanced the hydrophobic characteristics of the fabricated films. Abdullah JAA, et al. also found the increase in water contact of the film reinforced with green synthesized ZnO nanoparticles.31
The higher SI of GA1 sample might be attributed to the hydrophilic nature of gelrite gellan gum and glycerol. Both contains abundant hydroxyl groups and are capable of forming hydrogen bonds with molecules of water. Furthermore, the glycerol acts as a plasticizer and increases the free volume within the polymer matrix that facilitates the penetration of water. This results in greater swelling index. The decrease of SI of GA2 sample might be due to the interactions between SK-C5 and polymer chains that limits the polymer chain mobility and reduces the availability of the hydrophilic sites for the uptake of water. A study reported by Karimi Khorrami N, et al. reported the significant decrease in swelling index of the alginate film samples loaded with nanostructured lipid carriers.32
The peak at 3296 cm-1 might be attributed to the O-H stretching vibration that indicates an extensive hydrogen bonding between the hydroxyl groups of glycerol and gelrite gellan gum.33 Another peak was observed at 2925 cm-1 corresponds to the aliphatic C-H stretching vibrations.34 The absorption band observed at 1610 cm-1 might be associated to the asymmetric stretching of carboxylate (-COO–) groups.35 This indicates the polysaccharide backbone of gelrite gellan gum.36 The band at 1404 cm-1 corresponds to the symmetric stretching of carboxylate ions.37 The band at 1029 cm-1 is attributed to the C-O-C stretching vibration of glycoside linkages.38 The band at 918 cm-1 represent the C-H out of plane bending vibrations and the peak at 480 cm-1 corresponds to the skeletal vibrations.39 The band that represents the O-H stretching shifted towards a high wavenumber of 3278 cm-1 – 3882 cm-1. This suggests that the addition of SK-C5 improved the hydrogen bonding interactions between the polymer and β-AKBA analogue. The band at 1618 cm-1 represents the carboxylate stretching. However, this shift was minor compared to the GA1 sample. The bands at 1398 cm-1 and 1240 cm-1 corresponds to the symmetric carboxylate stretching and C-O stretching vibrations.40
Conclusion
In conclusion, the gelrite gellan gum biopolymer films loaded with (1-(4-Methoxyphenyl)-1H-1,2,3-triazol-4-yl)methyl) carbamoyl) 3α‐acetyloxy‐11‐oxo‐urs‐12‐en‐24‐oate (SK-C5) was fabricated using traditional casting method. The boswellic acid derivative-loaded film demonstrated significantly higher antioxidant activity, decrease the swelling index, and improved surface hydrophobicity. The increase in haze value and decrease in light transmittance indicates that the addition of SK-C5 increased the light-scattering properties by altering the polymer matrix. The FTIR spectroscopy was used to determine the interaction between the functional groups of polymer matrix and SK-C5. These findings highlight the potential application of boswellic acid derivative in sustainable biopolymer films. This contributes to the innovations in food packaging applications and reducing plastic pollution. Future research should concentrate on the long-term stability, real-time food applications, thermal stability, barrier properties, surface free energy, release kinetics, and antimicrobial efficacy of these films.
Acknowledgement
The Authors are thankful to the Natural and Medical Sciences Research Center, University of Nizwa, Oman, for providing research facilities to conduct the current study.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Talha Shireen Khan: Conceptualization, Methodology, Data Collection, Analysis, Writing – Original Draft.
- Ahmed Al-Harrasi: Supervision, Project Administration
- Saurabh Bhatia: Visualization, Supervision, Project Administration, Writing – Review and Editing
References
- Petkoska AT, Daniloski D, D’Cunha NM, Naumovski N, Broach AT. Edible packaging: Sustainable solutions and novel trends in food packaging. Food Res Int. 2021;140:109981.
CrossRef - Wohner B, Pauer E, Heinrich V, Tacker M. Packaging-related food losses and waste: An overview of drivers and issues. Sustainability. 2019;11(1):264.
CrossRef - Adeyeye O, Sadiku ER, Babu Reddy A, et al. The use of biopolymers in food packaging. Green Biopolymers and Their Nanocomposites. 2019;137-158.
CrossRef - Hishamuddin NI, Razali MH, Mat Amin KA. Application of gellan gum biopolymer in biomedical applications: A review. Makara J Sci. 2022;26(1):2.
CrossRef - Chen F, Chang X, Xu H, Fu X, Ding S, Wang R. Gellan gum-based functional films integrated with bacterial cellulose and nano-TiO2/CuO improve the shelf life of fresh-cut pepper. Food Packag Shelf Life. 2023;38:101103.
CrossRef - Jiang Y, Zhang Y, Deng Y. Latest advances in active materials for food packaging and their application. MDPI. 2023;4055.
CrossRef - Vermeiren L, Devlieghere F, van Beest M, de Kruijf N, Debevere J. Developments in the active packaging of foods. Trends Food Sci Technol. 1999;10(3):77-86.
CrossRef - Westlake JR, Laabei M, Jiang Y, et al. Vanillin cross-linked chitosan film with controlled release of green tea polyphenols for active food packaging. ACS Food Sci Technol. 2023;3(10):1680-1693.
CrossRef - Luzi F, Pannucci E, Santi L, et al. Gallic acid and quercetin as intelligent and active ingredients in poly(vinyl alcohol) films for food packaging. Polymers. 2019;11(12):1999.
CrossRef - Zhou X, Liu X, Wang Q, et al. Antimicrobial and antioxidant films formed by bacterial cellulose, chitosan and tea polyphenol – Shelf life extension of grass carp. Food Packag Shelf Life. 2022;33:100866.
CrossRef - Pastor C, González-Martínez C, Cháfer M, Chiralt A. Improving antioxidant properties of chitosan films by incorporating extracted fractions of propolis. J Renew Mater. 2024;12(12):1949.
CrossRef - Aswathy K, Ninan G. Exploring the role of phytochemical additives in enhancing the properties of gelatin-chitosan films: A review. Fishery Technol. 61(3).
- Narasagoudr SS, Hegde VG, Vanjeri VN, Chougale RB, Masti SP. Influence of boswellic acid on physical, structural and morphological properties of poly(vinyl alcohol) films. Chem Data Collect. 2020;27:100370.
CrossRef - Avula SK, Rehman NU, Khan F, et al. New 1H-1,2,3-triazole analogues of boswellic acid are potential anti-breast cancer agents. J Mol Struct. 2025;1319:139447.
CrossRef - Gautam S, Lapcik L, Lapcikova B, Repka D, Szyk-Warszyńska L. Physicochemical characterisation of polysaccharide films with embedded bioactive substances. Foods. 2023;12(24):4454.
CrossRef - Andreassen E, Larsen Å, Nord-Varhaug K, Skar M, Øysæd H. Haze of polyethylene films—effects of material parameters and clarifying agents. Polym Eng Sci. 2002;42(5):1082-1097.
CrossRef - Bharti S, Pathak V, Alam T, et al. Starch bio-based composite active edible film functionalized with Carum carvi L. essential oil: antimicrobial, rheological, physic-mechanical and optical attributes. J Food Sci Technol. 2022;59(2):456-466.
CrossRef - Balaban P, Puška A. Influence of packaging process on mechanical characteristics of the flexible packaging materials. Adv Eng Lett. 2022;1:65-70.
CrossRef - Thakhiew W, Devahastin S, Soponronnarit S. Physical and mechanical properties of chitosan films as affected by drying methods and addition of antimicrobial agent. J Food Eng. 2013;119(1):140-149.
CrossRef - Sukoco A, Yamamoto Y, Harada H, Hashimoto A, Yoshino T. Fish oil-containing edible films with active film incorporated with extract of Psidium guajava leaves: Preparation and characterization of double-layered edible film. F1000Res. 2024;13:816.
CrossRef - Choi I, Chang Y, Shin S-H, et al. Development of biopolymer composite films using a microfluidization technique for carboxymethylcellulose and apple skin particles. Int J Mol Sci. 2017;18(6):1278.
CrossRef - Al-Saban A, Maad AH, Al-Sham AS, Saad HM. Antimicrobial and antioxidant properties of Boswellia sacra resin: extraction, evaluation, and formulation into a topical cream for dermatological applications. Front Med. 2025;12:1664265.
CrossRef - Dodi G, Sabau RE, Crețu BE-B, Gardikiotis I. Exploring the antioxidant potential of gellan and guar gums in wound healing. Pharmaceutics. 2023;15(8):2152.
CrossRef - Lim MW, Quan Tang Y, Aroua MK, Gew LT. Glycerol extraction of bioactive compounds from Thanaka (Hesperethusa crenulata) bark through LCMS profiling and their antioxidant properties. ACS Omega. 2024;9(12):14388-14405.
CrossRef - Pereira JF, Lonni AAG, Mali S. Development of biopolymeric films with addition of vitamin C and catuaba extract as natural antioxidants. Prep Biochem Biotechnol. 2022;52(1):1-10.
CrossRef - Chaari M, Elhadef K, Akermi S, et al. Novel active food packaging films based on gelatin-sodium alginate containing beetroot peel extract. Antioxidants. 2022;11(11):2095.
CrossRef - Ng J, Lee SY, Thoo YY. Gellan gum film incorporated with palm kernel oil nanoemulsion for enhanced barrier properties. Food Biophys. 2025;20(2):1-14.
CrossRef - Flores Z, San-Martin D, Beldarraín-Iznaga T, Leiva-Vega J, Villalobos-Carvajal R. Effect of homogenization method and carvacrol content on microstructural and physical properties of chitosan-based films. Foods. 2021;10(1):141.
CrossRef - Abdullah JAA, Jiménez-Rosado M, Guerrero A, Romero A. Biopolymer-based films reinforced with green synthesized zinc oxide nanoparticles. Polymers. 2022;14(23):5202.
CrossRef - Karimi Khorrami N, Radi M, Amiri S, McClements DJ. Fabrication and characterization of alginate-based films functionalized with nanostructured lipid carriers. Int J Biol Macromol. 2021;182:373-384.
CrossRef - Hong T, Yin J-Y, Nie S-P, Xie M-Y. Applications of infrared spectroscopy in polysaccharide structural analysis: Progress, challenge and perspective. Food Chem X. 2021;12:100168.
CrossRef - Lu Y, Zhao X, Fang S. Characterization, antimicrobial properties and coatings application of gellan gum oxidized with hydrogen peroxide. Foods. 2019;8:31.
CrossRef - Dirpan A, Ainani AF, Deliana Y, et al. Novel pomelo peel pectin-chitosan bilayer films with gallic acid and CaCl2: Fabrication, characterization, and enhanced antimicrobial-mechanical properties. J Agric Food Res. 2025;24:102441.
CrossRef - Ng JY, Obuobi S, Chua ML, et al. Biomimicry of microbial polysaccharide hydrogels for tissue engineering and regenerative medicine – A review. Carbohydr Polym. 2020;241:116345.
CrossRef - Nikonenko N, Buslov D, Sushko N, Zhbankov R. Investigation of stretching vibrations of glycosidic linkages in disaccharides and polysaccharides with use of IR spectra deconvolution. Biopolymers. 2000;57:257-262.
CrossRef - Mehta M, Dureja H, Garg M. Development and optimization of boswellic acid-loaded proniosomal gel. Drug Deliv. 2016;23:1-10.
CrossRef













