Effect of UV Radiation on Ergosterol Synthesis in Agaricus bisporus and its Hepatoprotective Potential
1Public University of Santo Domingo de los Tsáchilas - UPSDT, km 28, Santo Domingo - Quevedo road, Ecuador
2Department of Interdisciplinary Studies (Health Sciences), National University of Chimborazo, Riobamba, Ecuador
3Department of Life Sciences and Agriculture, University of the Armed Forces (ESPE), Sangolquí, Ecuador
4Department of Industrial and Production Sciences, State Technical University of Quevedo, Quevedo, Ecuador
5Department of Applied Sciences to Industry, National University of Cuyo, San Rafael, Argentina
6Department not specified, Pontifical Catholic University of Ecuador, Quito, Ecuador
*Corresponding Author Email: jaldas@puce.edu.ec
DOI : http://dx.doi.org/10.12944/CRNFSJ.14.1.21
ABSTRACT:Vitamin D deficiency is a widespread public health issue linked to increased risk of autoimmune diseases. This study explored the use of mushroom stem flour, a by-product of the mushroom industry, as a functional ingredient enriched with ergosterol, a vitamin D2 precursor, and evaluated its safety and therapeutic potential. Different UV irradiation methods (UVA, UVB, and UVC) were applied to optimize ergosterol production, considering environmental variables such as exposure time, temperature, and humidity. UVB300 irradiation led to a 70% increase in ergosterol concentration compared to natural sunlight exposure (4.52 ± 0.1 µg/g p.s. vs. 2.9 ± 0.4 µg/g p.s., p<0.05), with peak levels (23.07 ± 2.14 µg/g p.s.) observed at 30 °C and 76.3% humidity. Safety was confirmed through acute and subchronic toxicity studies in rats receiving daily oral doses of 5,000 mg/kg body weight, showing no adverse clinical, biochemical, or behavioral effects over 8 weeks. In a liver injury model, supplementation with the enriched product significantly reduced ALT levels (from 6700 ± 200 UI in the control group to 2900 ± 170 UI in the UVB-treated group) and lowered the incidence of severe liver damage from 82% ± 5% to 38% ± 4%. Statistical analysis was performed using one-way ANOVA followed by Fisher’s protected LSD test (SPSS for Windows, version 10), with significance set at p<0.0533. These findings demonstrate that UVB-irradiated mushroom stem flour is a safe, natural source of vitamin D2 with notable hepatoprotective and immunomodulatory effects, supporting its potential application as a functional supplement for immune and liver health.
KEYWORDS:Agaricus bisporus; Antioxidant; Dietary supplements; Ergosterol; Vitamin-D2
Introduction
Edible mushrooms, such as Agaricus bisporus, have gained increasing attention due to their nutritional value and health benefits, particularly their high content of bioactive compounds.¹ Among these compounds, ergosterol is notable for its ability to convert into vitamin D₂ when exposed to ultraviolet (UV) radiation.2,3 Consequently, mushrooms represent a natural dietary source of this essential vitamin, which plays a crucial role in numerous biological functions.4,5
The growing global demand for mushrooms has led to a progressive accumulation of agro-industrial waste, creating significant environmental challenges.6,7 To address this issue, several studies have explored the valorization of mushroom byproducts for the production of biofuels, chitin, chitosan, β-glucans, and vitamin D₂ through UV irradiation.8-10 In addition to being a valuable source of protein, dietary fiber, minerals, and B-complex vitamins, mushrooms contain terpenoids, polyphenols, ergothioneine, and polysaccharides such as β-glucans.11 Notably, ergosterol accounts for approximately 90% of the total sterol content in Agaricus bisporus, underscoring its importance as a precursor of vitamin D₂.2,12
Ergosterol concentrations in species such as Agaricus bisporus, Pleurotus ostreatus, and Lentinula edodes range from 2,290 to 6,200 μg/g, while conversion to vitamin D₂ following UV irradiation can reach levels between 25.9 and 742 μg/g.13 However, a substantial proportion of total mushroom production—estimated at 25% to 30%—is discarded due to commercial standards that exclude cut stems, irregular shapes, and other aesthetic defects.14,15
Although cultivated mushrooms do not synthesize vitamin D during growth because of limited sunlight exposure, their high ergosterol content allows for efficient vitamin D₂ formation following UV treatment.16 This process is analogous to vitamin D₃ synthesis in human skin upon sunlight exposure and involves photochemical and thermal reactions that optimize vitamin D₂ production in mushrooms.17 Furthermore, UV-B irradiation not only enhances vitamin D₂ levels in Agaricus bisporus but also improves antioxidant capacity and other bioactive properties, supporting the development of functional foods with potential benefits for human health.18
Interest in the hepatoprotective effects of edible mushrooms has increased in recent years. Studies by Shi et al.19 and Téllez and Mateos20 using animal models demonstrated that Agaricus bisporus extracts can reduce hepatic oxidative stress, suggesting a protective role against liver disorders such as cirrhosis and hepatitis. Additionally, vitamin D has been extensively studied for its immunomodulatory properties.21,22 Holick23 identified vitamin D deficiency as a global public health concern and linked it to increased susceptibility to various diseases, including liver-related conditions.24,25
The growing recognition of vitamin D’s role in immune system regulation has driven the development of novel therapeutic strategies based on natural bioactive compounds. In this context, the food industry is increasingly focused on incorporating these compounds into nutraceutical formulations with targeted health benefits. Therefore, the present study evaluated the effect of UV radiation on ergosterol conversion in Agaricus bisporus and its hepatoprotective potential, highlighting the use of mushroom industry byproducts as a sustainable source of vitamin D₂ to address deficiency and enhance immune response in the population
Materials and Methods
Shall start as a continuation to introduction on the same page. All-important materials used along with their source shall be mentioned. The main methods used shall be briefly described, citing references.1
Raw material
Mushroom (Agaricus bisporus) residues and byproducts from the edible mushroom industry were used. These residues included stems and defective mushrooms that could not be marketed, as well as waste generated during product preparation for both fresh consumption and canned processing.
Ultraviolet treatment
A UV treatment was applied to increase ergosterol content. A specialized chamber was designed to establish an efficient irradiation dose. The irradiation source used was the UVP-LMS-38 EL Series model (UVP Analytik Jena LLC, Jena, Germany), which emits at three wavelengths: 365 nm, 302 nm, and 254 nm. The UV treatment was applied directly to the fresh byproduct, and different samples were prepared, adjusting factors such as irradiation time, source distance, and optimal wavelength. Once the optimal conditions were determined, samples were placed in the chamber at the appropriate distance and orientation. Exposure time was controlled using a stopwatch, and after completion, samples were repositioned for uniform irradiation.
Sample preparation
After UV treatment, the mushroom residues were dried at 40 ºC, soil, and sieved using a 125 µm mesh, resulting in a fine powder referred to as Irradiated Mushroom Stem Flour (HTCHi).
Ergosterol quantification by HPLC
Sample preparation and ergosterol content analysis were conducted at the Microanalysis Service of CITIUS – Celestino Mutis (University of Seville, Spain) following the method described by Shao et al. (2010),26 with slight modifications, using high-performance liquid chromatography (HPLC). Ergosterol levels were quantified in all treated samples. Briefly, 200 mg of sample was mixed with
mL of 2N KOH/MeOH to saponify the tissue and release esterified ergosterol. The mixture was stirred for 1 hour at 80 °C, cooled, and then 3 mL of distilled water was added. Extraction was performed three times using 3 mL of ethyl ether. The extract was washed with water until neutral and dried under a nitrogen stream. The ergosterol-containing extract was dissolved in 1 mL of chloroform and filtered through a 0.45 µm syringe filter. A 20 µL aliquot was injected into an HPLC system equipped with a Merck Chromolith RP18e column (100 x 4.6 mm), with the UV detector set at 282 nm. Ergosterol was eluted at a 1 mL/min flow rate using a gradient of water (A) and methanol (B), with the following conditions: 80-100% B in 20 min, 100% B until 34 min, 100-80% B at 37 min, and 5 min at initial conditions. Quantification was performed by comparing peak areas using cholecalciferol as an internal standard.27
Lyophilization
HTCHi was dispensed into Falcon tubes and weighed to determine dry weight by gravimetry. Samples were then frozen at -80 ºC for 24 hours. A Telstar Cryodos lyophilizer was used for vacuum dehydration over 48 hours, stabilizing the product for extended storage.
HTCHi toxicity
Toxicity studies were conducted using male Wistar rats weighing 150-200 g, provided by the Animal Facility Service of the University of Seville (Espartinas, Seville). All procedures followed the ethical guidelines of the Animal Experimentation Ethics Committee of the University of Seville, Spain. The study involved administering the product for 15 days, with an extension up to 8 weeks, during which biochemical, hematological, and coagulation parameters were evaluated.
Effect of vitamin D2 on liver disease
To assess the effect of the treatment on liver damage, rats with chronic hepatitis were used. Blood samples were collected, and serum was separated to analyze liver enzymes, including serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT). Measurements were taken before and after treatment with the obtained product. All procedures were conducted at the Faculty of Pharmacy, University of Seville, Spain.
Statistical analysis
Data are presented as mean ± standard deviation (SD) and were analyzed using one-way ANOVA, followed by Fisher’s protected LSD test for multiple group comparisons. All statistical analyses were performed using SPSS for Windows, version 10 (SPSS, Inc.), with a significance level of p<0.05.28
Results
Ergosterol Content in Different Treatments
As shown in Table 1, HTCH_ST exhibited an ergosterol concentration of 0.8 ± 0.2 µg/g p.s., a low value due to the lack of UV radiation exposure during mushroom cultivation. Compared to sunlight irradiation (HTCH_Luz, 2.9 ± 0.4 µg/g p.s), artificial UV radiation treatments resulted in a higher ergosterol content, with HTCH_UVB300 displaying the highest value (4.52 ± 0.1 µg/g p.s.), followed by HTCH_UVC256 (4.43 ± 0.3 µg/g p.s.) and HTCH_UVA362 (4.31 ± 0.2 µg/g p.s.). Although no significant differences were observed among the UV irradiation types, an overall increase in ergosterol concentration was evident compared to sunlight exposure. Additionally, in HTCH_UVC256, slight browning was observed after 30 minutes of exposure. These results suggest that UVB radiation at 300 nm is the most effective method for maximizing ergosterol content, a key precursor of vitamin D2.
Table 1: Ergosterol content in the different treatments.
|
Treatment |
Ergosterol content in A. bisporus (µg/g p.s.) |
| HTCHST |
0,8 ± 0,2 * |
|
HTCHLuz |
2,9 ± 0,4 |
| HTCHUVA362 |
4,31 ± 0,2 * |
|
HTCHUVB300 |
4,52 ± 0,1 * |
| HTCHUVC256 |
4,43 ± 0,3 * |
Note: Significance relative to irradiation under sunlight. *p<0.05, **p<0.01, ***p<0.001
HTCHST: Mushroom Stem Flour without UV treatment; HTCHUVA362: Mushroom Stem Flour with UV 362 nm treatment; HTCHUVB300: Mushroom Stem Flour with UV 300 nm treatment; HTCHUVC256: Mushroom Stem Flour with UV 256 nm treatment and additionally, a sample exposed to sunlight was included; HTCHLuz: Mushroom Stem Flour with sunlight.
Quantification of ergosterol as a function of environmental factors during the fermentation process
Ergosterol values showed significant variations depending on time, humidity, and temperature (Table 2). Over time, ergosterol content increased progressively, reaching its maximum value at 30 minutes (12.7±0.42 µg/g p.s..), then stabilizing. In terms of humidity, the highest ergosterol values were recorded at 79.8% humidity (16.03±1.4 µg/g p.s..), suggesting that this level favors ergosterol accumulation. Regarding temperature, ergosterol levels increased steadily, peaking at 23.07±2.14 µg/g p.s. at 30°C, followed by a progressive decline with further temperature increases. These results indicate that ergosterol synthesis is highly dependent on fermentation conditions, with optimal values observed at 30°C and 76.3% humidity.
Table 2: Quantification of ergosterol as a function of time, humidity, and temperature during the fermentation process.
|
Test Variables/ Measurement Parameter |
Time (min) | 10 | 20 | 25 | 30 | 35 | 40 | 45 | 50 | 55 | 60 |
| ergosterol µg/g p.s. |
5.9± 0.56* |
7.1± 0.45* |
8.9± 0.27* |
12.7± 0.42* |
10.7± 0.57* |
12.2± 0.61* |
12.3± 0.58* |
12.3± 0.64* |
12.1± 0.52* |
12.2± |
|
|
Humidity (%) |
55 |
61 | 63.5 | 73.5 | 74.7 | 76.3 | 79.8 | 83.7 |
85.7 |
92.8 |
|
| ergosterol µg/g p.s. | 8.10± 1.9* |
8.76± 2.0* |
9.04± 1.7* |
13.15± 1.9* |
13.57± 1.3* |
13.95± 1.3* |
16.03± 1.4* |
11.33± 0.7* |
11.27± 0.8* |
11.41± |
|
|
Temperature (°C) |
10 | 15 | 20 | 25 | 30 | 35 | 40 | 50 | 55 | 60 | |
| ergosterol µg/g p.s. | 7.63± 1.13* |
10.03± 1.21* |
12.83± 1.00* |
13.90± 1.55* |
23.07± 2.14* |
16.10 ±2.1* |
13.24 ±1.21* |
7.12 ±1.15* |
4.27± 1.42* |
4.15± |
Note: Significance with respect to irradiation under sunlight. *p<0.05, **p<0.01, ***p<0.001
Effect of administering a dose of a. bisporus enriched with vitamin D2 in rats
As shown in Table 3, the administration of A. bisporus enriched with Vitamin D2 at a dose of 5,000 mg/kg body weight did not cause any adverse effects or anomalies, nor were any clinical signs of toxicity observed in the rats throughout the 14-day study. Additionally, factors such as body weight and diet were considered to ensure they did not influence the study’s objective. Once it was confirmed that no adverse effects or toxicity were present, the administration of this product was extended for a prolonged period of up to 8 weeks to determine whether a longer exposure would lead to toxicity. Various parameters, including biochemical, hematological, and coagulation markers, as well as behavioral anomalies, were analyzed. The results once again demonstrated the absence of toxicity, as these factors are crucial for assessing health risks.
Table 3: Effect of administration of the dose of A. bisporus enriched with Vitamin-D2 in rats.
|
Parameter |
After the administration of 5,000 mg/kg body weight | |||
| 8 hours | 12 hours | 24 hours |
14 hours |
|
|
Skin and Fur |
Normal | Normal | Normal | Normal |
| Eyes | Normal | Normal | Normal |
Normal |
|
Mucosa |
Normal | Normal | Normal | Normal |
| Behavioral Patterns | Normal | Normal | Normal |
Normal |
|
Salivation |
Normal | Normal | Normal | Normal |
| Lethargy | Normal | Normal | Normal |
Normal |
Note: * A daily dose of 5,000 IU/kg of body weight was administered for 14 days.
Effect of A bisporus on liver diseases
Figure 1 shows alanine aminotransferase (ALT) levels under different experimental conditions. The control group exhibited the highest ALT levels (approximately 6700 ± 200 UI), indicating greater liver damage. Supplementation with vitamin D2 significantly reduced these levels to approximately 3000 ± 150 UI. Similarly, treatment with HTCH under standard conditions (HTCH ST) resulted in a reduction to around 3200 ± 180 UI. The most notable decrease was observed in the group treated with HTCH and exposed to UVB at 35°C, with values of approximately 2900 ± 170 UI. These findings demonstrate a hepatoprotective effect of both vitamin D2 and HTCH, with a potential synergistic effect in the presence of UVB radiation.
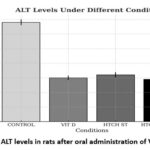 |
Figure 1: ALT levels in rats after oral administration of Vitamin D2 |
Figure 2 shows the percentage of severe liver injury in different experimental groups. The control group had the highest incidence of severe liver injury (82% ± 5%), while vitamin D administration reduced this value to 60% ± 7%. Treatments with standard HTCH (HTCH ST) and HTCH with UVB exposure at 35 °C showed an even greater decrease in liver injury, with values of 50% ± 6 and 38% ± 4, respectively. These results indicate a protective effect of vitamin D2 and, to a greater extent, of HTCH treatments, especially under UVB exposure conditions, on severe liver damage.
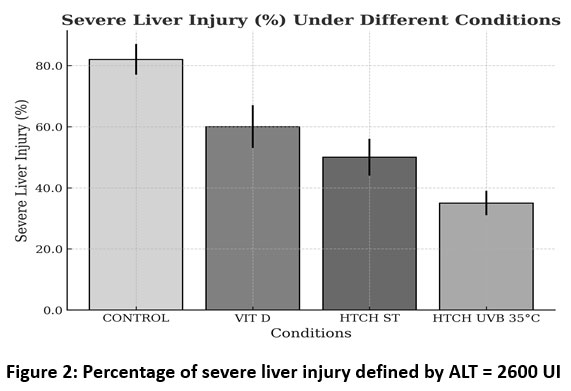 |
Figure 2: Percentage of severe liver injury defined by ALT = 2600 UI |
Discussion
Ergosterol content in the different treatments
The results of this study indicate that the irradiation of the byproduct of Agaricus bisporus with artificial light (HTCHUVB300) increases its ergosterol concentration by 70% compared to natural irradiation (HTCHLuz). These findings align with previous studies that have analyzed the ergosterol content in different parts of A. bisporus, where results showed that the distribution of ergosterol was significantly different in the various tissues of the mushrooms, with the highest content in the gills (11.02 mg/g) and the lowest content in the stems (4.4 mg/g) under artificial irradiation conditions.29 On the other hand, the ergosterol content in non-irradiated A. bisporus is virtually nil or very low, similar to the results obtained in this study.3
Ergosterol quantification based on environmental factors during the fermentation process
The vitamin D content in mushrooms exposed to UV radiation varies according to multiple factors, including the mushroom species, the intensity and duration of exposure, the surface exposed (whole or sliced), and the amount of light absorbed.30 It was observed that the ergosterol concentration reached its peak at 30 minutes, stabilizing afterward, in agreement with previous studies conducted in Germany, where 100 g of sliced Agaricus bisporus produced 17.5 μg of ergosterol after 15 minutes of exposure and 32.5 μg after 30 minutes.31 In terms of humidity, the highest ergosterol content was recorded at 79.8%, suggesting that this level favors cell membrane stability and compound synthesis, in line with research highlighting the importance of water availability in sterol accumulation in fungi.32 Regarding temperature, the maximum ergosterol content was detected at 30 °C; however, excessive thermal increase could intensify oxidation, induce cellular stress, and affect compound biosynthesis. Similar results were reported by Villares et al. (2014).33 These findings highlight the need for precise control of fermentation conditions to optimize ergosterol production in edible mushrooms.
Effect of the administration of Agaricus bisporus enriched with Vitamin D2 in rats
The absence of toxicity observed in this study at a dose of 5,000 mg/kg aligns with previous research indicating that vitamin D₂ does not produce adverse effects in animals, even at similar or higher doses.34 DeClementi and Sobczak (2018) evaluated the effects of high doses of vitamin D in animal models and reported that doses lower than 10,000 mg/kg do not cause acute toxicity or alterations in biochemical or hematological parameters, supporting the safety of vitamin D₂ when administered in moderate amounts over extended periods. This makes it a viable ingredient for the development of industrial products, such as functional foods and nutraceuticals.35 In this context, mushrooms exposed to UVB radiation represent an effective source of vitamin D₂ to improve the nutritional status of the human body, with bioavailability comparable to vitamin D₂ supplements. Additionally, certain foods such as fatty fish (5.7 μg per serving) and eggs (7.1 μg per serving) naturally contain significant amounts of this vitamin.22
Effect of Agaricus bisporus on liver diseases
This study demonstrated that the consumption of Agaricus bisporus enriched with vitamin D₂ can contribute to the relief of immune-mediated hepatitis. To evaluate this effect, the levels of alanine aminotransferase (ALT), a hepatic enzyme whose presence in the blood significantly increases in cases of liver damage, were measured. ALT is primarily found in the liver and is released when there is damage to hepatocytes. In rats with induced liver damage, a decrease in these enzymes was observed after the administration of A. bisporus enriched with vitamin D₂, suggesting that its immunomodulatory effect is efficient in reducing liver inflammation. These results indicate that the combination of bioactive compounds present in A. bisporus and vitamin D₂ generates a synergistic effect in the improvement of immune-mediated hepatitis.
Vitamin D is a molecule with immunomodulatory and anti-inflammatory properties, playing key roles in regulating the immune system, preventing autoimmune diseases, and cancer, among other pathologies affecting human health. Its synthesis in the body occurs through two main pathways: the first, via hydroxylation in the liver and kidneys, facilitating calcium absorption and mineral homeostasis; and the second, through the action of immune cells that produce 1,25-dihydroxyvitamin D in response to an infection, contributing to immune regulation.21,36 Furthermore, the consumption of UVB-exposed mushrooms represents an alternative dietary source of vitamin D, which could help meet the daily intake recommendations, which range from 400 to 1,000 IU/day (10–25 μg/day) for an average adult.3
Several studies have linked vitamin D to immune response modulation, including macrophage activation against infections and the regulation of proliferation and antibody production by B lymphocytes. Its deficiency has been associated with an increased risk of acute hepatitis and other extrahepatic manifestations, with an inverse correlation observed between serum vitamin D levels and the severity of liver disease. Furthermore, high concentrations of this vitamin have been reported to enhance the sustained virological response in patients with chronic infections. In this regard, the administration of Agaricus bisporus enriched with vitamin D₂ could improve the sensitivity to antiviral therapies in patients with chronic hepatitis, offering a complementary approach to its treatment.2,37
Conclusion
The results showed that UVB irradiation at 300 nm is the most efficient strategy to increase ergosterol content in Agaricus bisporus, especially when the fermentation process is carried out under optimal conditions of 30°C and 79.8% humidity. Additionally, the administration of A. bisporus enriched with vitamin D2 at a dose of 5,000 mg/kg body weight did not cause adverse effects or toxicity in rats, both in the short and long term, supporting its safety for consumption. Furthermore, a significant hepatoprotective effect was observed, evidenced by a reduction in alanine aminotransferase (ALT) levels and the incidence of severe liver damage, suggesting a potential synergistic effect between vitamin D2 and UVB radiation. These findings highlight the potential of biofortified A. bisporus as a viable strategy to improve vitamin D2 intake in human nutrition, with promising applications in the prevention of liver diseases. In this context, it is concluded that byproducts of A. bisporus enriched with vitamin D2 can be used as raw materials for the development of functional foods and nutraceuticals of industrial interest.
Acknowledgement
The authors gratefully acknowledge the University of Seville, Spain, for its institutional support during the development of this research.
Funding Sources
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Anabell Del Rocío Urbina-Salazar: Conceptualization, Methodology, Investigation, Writing – Original Draft.
- Alberto Renato Inca-Torres: Methodology, Data Curation, Formal Analysis, Writing – Review and Editing.
- Karina Paredes Paliz: Investigation, Data Collection, Validation, Writing – Review and Editing.
- Juan Alejandro Neira Mosquera: Formal Analysis, Visualization, Software, Writing – Review and Editing.
- Sungey Naynee Sanchez Llaguno: Investigation, Resources, Data Collection, Writing – Review and Editing.
- Jhonnatan Plácido Aldas Morejón: Conceptualization, Supervision, Project Administration, Writing – Review and Editing.
- Karol Yannela Revilla Escobar: Supervision, Funding Acquisition, Resources, Validation, Writig, Review and Editing.
References
- Atila F, Owaid M, Shariati M. The nutritional and medical benefits of Agaricus bisporus: A review. J Microbiol Biotechnol Food Sci. 2017;7(3). doi:10.15414/jmbfs.2017/18.7.3.281-286
CrossRef - Aliaño-González MJ, Barea-Sepúlveda M, Espada-Bellido E, et al. Ultrasound-assisted extraction of total phenolic compounds and antioxidant activity in mushrooms. Agronomy. 2022;12(8):1812. doi:10.3390/agronomy12081812
CrossRef - Jiang Q, Zhang M, Mujumdar AS. UV induced conversion during drying of ergosterol to vitamin D in various mushrooms. Trends Food Sci Technol. 2020;105:200-210. doi:10.1016/j.tifs.2020.09.011
CrossRef - Hu Z, He B, Ma L, et al. Recent advances in ergosterol biosynthesis and regulation mechanisms in Saccharomyces cerevisiae. Indian J Microbiol. 2017;4(3):270-277. doi:10.1007/s12088-017-0657-1
CrossRef - Huang BF, Pan XD, Zhang JS, et al. Determination of vitamins D₂ and D₃ in edible fungus by reversed-phase two-dimensional liquid chromatography. J Food Qual. 2020;2020:1-6. doi:10.1155/2020/8869279
CrossRef - Pan J, Xie Y, Ao N, et al. Synergistic enhancement of methane production from spent mushroom substrate. Biomass Bioenergy. 2021;150:106128. doi:10.1016/j.biombioe.2021.106128
CrossRef - Royse DJ. A global perspective on the high five. In: Proceedings of the 8th International Conference on Mushroom Biology and Mushroom Products. New Delhi, India; 2014.
- Cebin AV, Petravić-Tominac V, Djakovic S, et al. Polysaccharides and antioxidants from culinary-medicinal white button mushroom, Agaricus bisporus (Agaricomycetes), waste biomass. Int J Med Mushrooms. 2018;20(8):797-808. doi:10.1615/IntJMedMushrooms.2018027412
CrossRef - Fadhil A, Mous EF. Antimicrobial activities of chitosan produced from Agaricus bisporus stalks. Plant Arch. 2020;20(1):109-111.
- Papoutsis K, Grasso S, Menon A, et al. Recovery of ergosterol and vitamin D₂ from mushroom waste. Trends Food Sci Technol. 2020;99:351-366. doi:10.1016/j.tifs.2020.03.005
CrossRef - Krishnamoorthi R, Srinivash M, Mahalingam PU, et al. Dietary nutrients in edible mushroom, Agaricus bisporus, and their biological activities. Process Biochem. 2022;121:10-17. doi:10.1016/j.procbio.2022.06.021
CrossRef - Roncero-Ramos I, Delgado-Andrade C. The beneficial role of edible mushrooms in human health. Curr Opin Food Sci. 2017;14:122-128. doi:10.1016/j.cofs.2017.04.002
CrossRef - Kamgang NA, Sun Y, Vittori S, et al. Conversion of ergosterol into vitamin D₂ and other photoisomers in Agaricus bisporus mushrooms under UV-C irradiation. SSRN Electron J. 2022. doi:10.2139/ssrn.4172715
CrossRef - Bilbao-Sainz BS, Chiou T, Williams D, et al. Vitamin D-fortified chitosan films from mushroom waste. Carbohydr Polym. 2017;167:97-104. doi:10.1016/j.carbpol.2017.03.010
CrossRef - Chou WT, Sheih IC, Fang TJ. The applications of polysaccharides from various mushroom wastes as prebiotics in different systems. J Food Sci. 2013;78(7):1041-1048. doi:10.1111/1750-3841.12160
CrossRef - Nölle N, Argyropoulos D, Ambacher S, et al. Vitamin D₂ enrichment in mushrooms by UV-light during drying. LWT Food Sci Technol. 2017;85:400-404. doi:10.1016/j.lwt.2016.11.072
CrossRef - Plum LA, DeLuca HF. The functional metabolism and molecular biology of vitamin D action. Clin Rev Bone Miner Metab. 2009;7:20-41. doi:10.1007/s12018-009-9040-z
CrossRef - Manta B, Sarkisian A, García-Fontana B, et al. Pathophysiology of COVID-19. Odontoestomatología. 2022;24. doi:10.22592/ode2022n39e312
CrossRef - Shi Y, Wang M, Wu L, et al. COVID-19 associated liver injury. Liver Res. 2023;7(3):207-215. doi:10.1016/j.livres.2023.07.001
CrossRef - Téllez L, Mateos RMM. COVID-19 and liver disease. Gastroenterol Hepatol Engl Ed. 2020;43(8):472-480.
CrossRef - Dankers W, Colin EM, van Hamburg JP, Lubberts E. Vitamin D in autoimmunity: molecular mechanisms and therapeutic potential. Front Immunol. 2017;7:697. doi:10.3389/fimmu.2016.00697
CrossRef - Greiller CL, Martineau AR. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients. 2015;7(6):4240-4270. doi:10.3390/nu7064240
CrossRef - Holick MF. The vitamin D deficiency pandemic: approaches for diagnosis, treatment and prevention. Rev Endocr Metab Disord. 2017;18(2):153-165. doi:10.1007/s11154-017-9424-1
CrossRef - Bouillon R, Marcocci C, Carmeliet G, et al. Skeletal and extraskeletal actions of vitamin D: current evidence and outstanding questions. Endocr Rev. 2019;40(4):1109-1151. doi:10.1210/er.2018-00126
CrossRef - Infante M, Ricordi C, Sanchez J, et al. Influence of vitamin D on islet autoimmunity and beta-cell function in type 1 diabetes. Nutrients. 2019;11(9):2185. doi:10.3390/nu11092185
CrossRef - Shao S, Hernandez M, Kramer JK, et al. Ergosterol profiles and antioxidant activities of button mushrooms. J Agric Food Chem. 2010;58(22):11616-11625. doi:10.1021/jf102285b
CrossRef - Inca-Torres AR. Valorization of By-Products from the Edible Mushroom Industry: Application for Obtaining High-Value Products [doctoral thesis]. Seville, Spain: University of Seville; 2019.
- Seaman MA, Levin JR, Serlin RC. New developments in pairwise multiple comparisons. Psychol Bull. 1991;110:577-586. doi:10.1037/0033-2909.110.3.577
CrossRef - Jasinghe V, Perera C. Ultraviolet irradiation: the generator of vitamin D₂ in edible mushrooms. Food Chem. 2006;95:638-643. doi:10.1016/j.foodchem.2005.01.046
CrossRef - Cardwell G, Bornman J, James A, Black L. A review of mushrooms as a potential source of dietary vitamin D. Nutrients. 2018;10:1498. doi:10.3390/nu10101498
CrossRef - Urbain P, Jakobsen J. Dose-response effect of sunlight on vitamin D₂ production in Agaricus bisporus. J Agric Food Chem. 2015;63:8156. doi:10.1021/acs.jafc.5b02945
CrossRef - Guo Z, Liu X, Wang N, et al. Membrane component ergosterol builds a platform for promoting effector secretion and virulence in Magnaporthe oryzae. New Phytol. 2022;237:930-943. doi:10.1111/nph.18575
CrossRef - Villares A, Mateo-Vivaracho L, García-Lafuente A, et al. Storage temperature and UV-irradiation influence on ergosterol content. Food Chem. 2014;147. doi:10.1016/j.foodchem.2013.09.144
CrossRef - DeClementi C, Sobczak B. Common rodenticide toxicoses in small animals. Vet Clin North Am Small Anim Pract. 2018;48(6):1027-1038. doi:10.1016/j.cvsm.2018.06.006
CrossRef - Cremades-De Molina O, Diaz-Herrero M, Carbonero-Aguilar P, et al. White button mushroom ergothioneine aqueous extracts obtained by the application of enzymes and membrane technology. Food Biosci. 2015;10:42-47. doi:10.1016/j.fbio.2015.01.001
CrossRef - Roupas P, Keogh J, Noakes M, et al. Mushrooms and agaritine: A minireview. J Funct Foods. 2010;2:91-98. doi:10.1016/j.jff.2010.04.003
CrossRef - Chen Y, Zhang J, Ge X, et al. Vitamin D receptor inhibits nuclear factor κB activation by interacting with IκB kinase β protein. J Biol Chem. 2013;288(27):19450-19458. doi:10.1074/jbc.M113.467670
CrossRef













