Physical and Chemical Stability of Parijoto Fruit Extract (Medinilla speciosa) Nanodispersion in Different pH Solutions
1Department of Food Technology, Faculty of Agricultural Technology, Soegijapranata Catholic University, Semarang, Indonesia.
2Professional Engineer Program, Soegijapranata Catholic University, Semarang, Indonesia.
Corresponding Author Email: kristina@unika.ac.id
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.2.18
ABSTRACT:Parijoto fruit is known for its health benefits due to its rich phytochemical content, particularly anthocyanins. Anthocyanins are natural pigments in many plants that give red, purple, and blue colors. These compounds offer various health benefits, including antioxidant, anticancer, and anti-inflammatory effects. However, anthocyanins are inherently unstable, with their stability impacting factors such as pH, temperature, vitamin C, and oxygen levels. Nanodispersion technology, characterized by its tiny droplet size and clear appearance, can enhance the solubility of active compounds and provides an excellent encapsulation medium for bioactive compounds like anthocyanins. This study aims to assess the stability of parijoto fruit extract nanodispersion solutions under different pH conditions—control (neutral pH), pH 6, and pH 8—over 28 days of storage. Nanodispersion stability will be evaluated based on pH stability, absorbance, % transmittance, total anthocyanin content, encapsulation efficiency, and turbidity. Testing will occur at 7-day intervals, with three replicates for each sample. Collected data will be presented as mean values ± standard deviation and in graphical form. Statistical data analysis will be conducted using SPSS and R Studio software. The study's findings indicate that the control sample (neutral pH without adding H3PO4 or NH4OH) exhibited the best stability. The stability of the control sample (Nk) was indicated by a lower increase in particle size compared to other samples and stable values for the polydispersity index, zeta potential, conductivity, pH, turbidity, transmittance, and encapsulation efficiency. Using NH4OH or H3PO4 to achieve pH 6 and pH 8 conditions led to the degradation of Tween 80, which affected the stability of the nanodispersion during storage.
KEYWORDS:Antioxidant; Nanodispersion; Parijoto Fruit; pH; Stability
Introduction
In the past decade, nanodispersions have become a fascinating area of research in the food industry. This is due to their unique properties, such as their generally transparent appearance and high absorption effectiveness in the body compared to regular emulsions. As a result, nanodispersions offer substantial potential as functional ingredients, especially in food products.1 Nanodispersions are a nanotechnology application derived from stable colloidal systems with kinetic stability created through mixing oil, water, and emulsifiers.2 Nanodispersions can be categorized into several types: oil-in-water (O/W) nanodispersions, water-in-oil (W/O), transparent/light, and colloidal dispersions. Nanodispersions have petite particle sizes, allowing them to effectively deliver hydrophobic bioactive compounds into the body due to their increased surface area.3 However, one of the limitations of nanodispersions in food products, according to Qian et al. (2012) in Zhang et al.,4 is the need for food-grade emulsifiers with high efficacy and without undesirable flavours. Additionally, emulsifiers must be able to stabilize nanodispersions against Ostwald Ripening.
Tween-80 is a surfactant with non-toxic, non-ionic properties and high biocompatibility. In addition, Tween-80 exhibits high bioavailability compared to other emulsifiers. This surfactant is commonly used in the cosmetic, pharmaceutical, and even food industries, particularly as a surfactant in emulsions.3 Surfactants can reduce the interfacial tension between oil and water and form an elastic steric film around the oil droplets. This causes the oil droplets that have combined to be less likely to separate again, resulting in a stable emulsion with improved quality.5
One of the plants from Indonesia that has numerous scientifically proven benefits in functional foods is the Parijoto fruit (Medinilla speciosa). The Parijoto fruit is rich in phytochemical components, including anthocyanins, flavonoids, tannins, saponins, alkaloids, glycosides, and cardenolides.6 The main compound in Parijoto fruit, anthocyanin, has many health benefits, such as reducing the risk of diseases like cardiovascular diseases and diabetes, possessing anti-inflammatory properties, and functioning as an antioxidant.7 However, anthocyanins are highly unstable and susceptible to degradation, influenced by chemical structure, pH, solvent type, storage temperature, light, oxygen, proteins, and metal ions. High temperatures and pH levels and exposure to light can cause anthocyanins to degrade and change colour.8 Therefore, suitable nanoencapsulation techniques, such as nanodispersions, are considered capable of improving the stability, bioavailability, and solubility of lipophilic bioactive compounds and preventing the hydrolysis and oxidation of bioactive compounds, especially anthocyanins found in Parijoto fruit. Thus, the formation of nanodispersions for anthocyanin compounds in Parijoto fruit has significant potential for development in the food sector.
The anthocyanin content in nanodispersions of Parijoto fruit extract is expected to offer potential as a raw material for developing functional foods, particularly in beverage products. However, as mentioned earlier, the instability of anthocyanins requires special treatment to evaluate the characteristics of Parijoto extract nanodispersions when applied as beverage products. Moldovan & David9 reported that low pH contributes to higher anthocyanin stability. Despite this, limited studies have explored the impact of pH variations on the physical and chemical stability of Parijoto extract nanodispersions in real food applications. To bridge this gap, this study evaluates the stability of Parijoto extract nanodispersions under different pH conditions, the physical and chemical characteristics will be analyzed to ensure that Parijoto extract nanodispersions remain stable for various food applications. Therefore, a series of tests on Parijoto extract nanodispersions are needed to assess the effects of adding these nanodispersions to solutions at specific pH levels on their physical and chemical stability.
Materials and Methods
Materials
Fresh Parijoto fruit, 96% ethanol (Merck, Germany), double distilled water (IKA), distilled water, potassium Chloride, Sodium Acetate, Tween 80 (Merck, Germany), citric acid, phosphoric acid, ammonium hydroxide, hydrochloric acid.
Methods
Preparation of Dried Parijoto Extract Sample
The sorted and cleaned Parijoto fruit was weighed to 200 grams. After weighing, steam-blanching was performed on the Parijoto fruit for 3 minutes to inactivate peroxidase enzymes and prevent browning during drying. The Parijoto fruit was then soaked in a 1% citric acid solution for 5 minutes and filtered. This step was done as a pre-treatment to prevent case-hardening. Before drying, the cabinet dryer was sterilized using 96% ethanol to avoid cross-contamination. The Parijoto fruit was dried using a cabinet dryer for 6 hours at a temperature of 70°C. The dried Parijoto fruit was then ground using a grinder for 2 minutes. The Parijoto powder was sieved using a 106 mesh sieve to produce a powder with a homogeneous particle size.
Parijoto Extract Preparation using Ultrasonic Assisted Extraction (UAE)
A total of 20 grams of Parijoto powder sample was mixed with 200 ml of 96% ethanol in each of the five 250 ml Erlenmeyer flasks. The four sample bottles were sonicated using UAE for 30 minutes (45 KHz, 100%, 45°C), followed by shaking for 1 hour at 200 rpm using a shaker. After shaking, each sample was centrifuged at 4000 rpm for 15 minutes. After centrifugation, the supernatant of each sample was separated and filtered using a 0.22 µm membrane filter. The filtrate obtained was evaporated until thickened under vacuum conditions using a rotary vacuum evaporator at a temperature of 35°C for 15 minutes. The remaining residue was then dissolved in double distilled water at a ratio of 1:9 (w/v) in a volumetric flask to make the Parijoto extract stock.
Preparation of Parijoto Extract Nanodispersion
The Parijoto extract nanodispersion was prepared by mixing a surfactant with a high hydrophile-lipophile balance (HLB) with the previously prepared Parijoto extract. The surfactant used in this study was Tween 80 at a concentration of 12%. Using a magnetic stirrer, the surfactant solution was prepared by homogenizing 12 ml of Tween 80 with 78 ml of deionized water. The previously prepared Parijoto extract stock was then diluted with double distilled water to obtain a Parijoto extract concentration of 7.5% (v/v). The Tween 80 surfactant solution (12%) was then mixed with the 7.5% Parijoto extract at a ratio of (0.24:2.76) using a magnetic stirrer until entirely homogeneous. The sample was then sonicated using UAE at 35°C (45 KHz, 60 minutes, 100%). The sample was homogenized using a high-speed centrifuge at 15,000 rpm, 4°C, for 15 minutes to produce a homogeneous nanodispersion.
Preparation of Nanodispersion with Varying pH Conditions
The prepared Parijoto extract nanodispersion was added at 10% to a double-distilled water solution with different pH conditions (pH 6 and 8) using H3PO4 (0.1 N) and/or NH4OH (0.1 N). The samples will then be stored in a refrigerator at approximately 4°C to analyze their stability over 28 days.
Physical Characterization of Parijoto Extract Nanodispersion: Particle Size, Polydispersity Index, Zeta Potential, and Conductivity
The physical characterization of all Parijoto fruit nanodispersion samples was performed using a Zetasizer Pro (Malvern Instruments, Ltd., Malvern) and the ZS Explorer application. The Zetasizer operates based on the principle of dynamic light scattering (DLS). Measurements were conducted at a wavelength of 633 nm and a temperature of 25°C with a detector placed at an angle of 173° to reduce the effects of multiple scattering. Zeta Potential and Conductivity were measured automatically after an equilibration process of 120 seconds at 25°C.
pH Value of Parijoto Extract Nanodispersion
The pH value was measured using a Schott pH meter at room temperature (27 ± 2°C), calibrated with standard pH 4, 7, and 10 buffer solutions. Once the pH value on the meter stabilized, the reading was recorded. Care must be taken when inserting the pH meter electrode into the sample to ensure proper contact with the sample.
Absorbance Value of Parijoto Extract Nanodispersion
To determine the concentration of active compounds or particles in the Parijoto extract nanodispersion, the absorbance of all nanodispersion samples with different pH conditions was analyzed at a wavelength of 500 nm using a UV-Vis spectrophotometer (Shimadzu, Kyoto, Japan) with double distilled water as a blank.7
Total anthocyanin in 7.5% Parijoto Extract
The total anthocyanin content was measured using the differential pH method, a modification of the method by Zahed et al.10 and Adhamatikha and Murtini (2021). pH 1 Potassium Chloride 0.2 M (KCl) buffer solution was prepared by dissolving 1.490 grams of KCl with distilled water in a 100 ml volumetric flask. pH 4.5 Sodium Acetate 0.2 M (CH3COONa) buffer solution was prepared by dissolving 1.640 grams of CH3COONa with distilled water in a 100 ml volumetric flask. A 0.2 N HCl solution was used to adjust the pH of the KCl and CH3COONa buffers to pH 1 and pH 4.5, respectively.
1 ml of 7.5% Parijoto extract was diluted with double distilled water at a ratio of 1:9. The diluted Parijoto extract was then mixed with 1 ml of each buffer solution (pH 1 and pH 4.5). Each sample was vortexed until homogeneous and stored in a dark room for 15 minutes. The incubated samples were measured for absorbance using a UV-Vis spectrophotometer (Shimadzu, Kyoto, Japan) at wavelengths of 520 nm and 700 nm. The absorbance (A) was calculated using the formula:
![]()
The total anthocyanin in the 7.5% Parijoto extract was calculated using the formula:
![]()
Where:
A = absorbance
BM = 449.2 g/mol (Molecular weight of cyanidin-3-glucoside)
Ε = 26,900 L/mol.cm (Molar absorptivity of cyanidin-3-glucoside)
1000 = conversion factor from grams to milligrams
FP = 10 (dilution factor)
L = cuvette path length (1 cm)
Encapsulation Efficiency of Parijoto Extract Nanodispersion
The encapsulation efficiency method for Parijoto extract nanodispersion follows the method applied by Zahed et al.10 To calculate the total anthocyanin (TA) in the nanodispersion sample, 0.2 grams of each sample was dissolved in 2 ml of aquabides, then 18 ml of 50% ethanol was added and filtered with Whatman 42 filter paper. Absorbance was measured at 520 nm and 700 nm at pH 1.0 and pH 4.5 using the formula:
![]()
The TA value in the Parijoto extract nanodispersion sample was calculated using the formula:
![]()
To calculate Surface Anthocyanin (SA), 0.1 grams of the nanodispersion sample was dissolved in 10 ml of 50% ethanol and centrifuged (15°C, 1790 xg, 5 minutes). The supernatant was separated, and the total anthocyanin content was measured using the differential pH method to obtain the SA. Encapsulation efficiency of Parijoto extract nanodispersion was calculated using the formula:
![]()
Where:
TA = Total Anthocyanin in parijoto fruit extract nanodispersion samples
SA = Surface Anthocyanin in parijoto fruit extract nanodispersion samples
Turbidity of Parijoto Extract Nanodispersion
Changes in turbidity can indicate changes in the stability of the emulsion system. Therefore, a turbidity test was conducted on the Parijoto extract nanodispersion. The turbidity test followed the method by Zhang et al.11 The turbidity test was conducted using a Hach 2100Q turbidimeter (Germany). Before testing, the turbidimeter was calibrated using StablCal 20, 100, and 800 NTU calibration solutions. The nanodispersion sample was placed in a glass cuvette, and the turbidity value was recorded in NTU units.
Transmittance Percentage of Parijoto Extract Nanodispersion
To assess the self-emulsification ability of the Parijoto extract nanodispersion samples, the transmittance of all samples was measured by diluting 10 ml of each type of sample into 90 ml of distilled water, following the method by Wiwiek et al.12 The transmittance of the diluted samples was measured using a UV-Vis spectrophotometer (Shimadzu, Kyoto, Japan) at a wavelength of 650 nm.
Statistical Analysis
All collected data will be analyzed using Microsoft Excel. Experimental data will be presented as the mean along with the standard deviation. Data will then be analyzed with SPSS for Windows version 13. The statistical analysis used is the Kruskal-Wallis test to determine significant differences in the stability values of zeta potential, polydispersity index, particle size, and conductivity of the parijoto fruit extract nanodispersion with varying pH conditions during storage. This study also employs R Studio to analyze data for the stability of pH, transmittance, absorbance, turbidity, and encapsulation efficiency of the parijoto fruit extract nanodispersion.
Results
Particle Size and Polydispersity Index
Table 1: Results of Particle Size and Polydispersity Index Analysis
| Sample Code | Particle Size (nm) | Polydispersity Index | ||
| Day-0 | Day-30 | Day-0 | Day-30 | |
| NK | 49.12 ± 1.62 | 52.37 ± 5.99 | 0.24 ± 0.04 | 0.27 ± 0.02 |
| N6 | 76.23 ± 3.79 | 122.58 ± 5.84 | 0.50 ± 0.15 | 0.64 ± 0.03 |
| N8 | 89.76 ± 1.72 | 243.71 ± 36.91 | 0.75 ± 0.09 | 0.78 ± 0.15 |
Sample codes NK, N6, and N8: nanodispersion samples of parijoto extract added to aquabides solution with controlled pH levels of 6 and 8.
Based on the results of particle size stability and polydispersity index analysis shown in Table 1, it was found that all samples experienced an increase in particle size and polydispersity index values during storage. All samples showed an increase in polydispersity index values during storage. The NK sample had the lowest final polydispersity index value of 0.27, indicating that the sample remained in the monodisperse category. Meanwhile, N6 and N8 samples had polydispersity index values of 0.64 and 0.78, respectively, falling into the polydisperse category.
Zeta Potential and Conductivity
Table 2: Results of Zeta Potential and Conductivity Analysis
| Sample Code | Zeta Potential (mV) | Conductivity | ||
| Day-0 | Day-30 | Day-0 | Day-30 | |
| NK | -22.19 ± 2.74 | -20.92 ± 3.46 | 0.08 ± 0.01 | 0.078 ± 0.00715 |
| N6 | -18.34 ± 2.89 | -7.11 ± 0.86 | 0.086 ± 0.01 | 0.033 ± 0.00123 |
| N8 | -19.54 ± 1.94 | 4.29 ± 1.13 | 0.093 ± 0.01 | 0.007 ± 0.00046 |
Sample codes NK, N6, and N8: nanodispersion samples of parijoto extract added to aquabides solution with controlled pH levels of 6 and 8.
Based on the zeta potential stability analysis results in Table 2, it was observed that all NK samples exhibited zeta potential values moving closer to zero.
Meanwhile, the stability analysis results for conductivity values in Table 2 show a decrease in the final conductivity of each sample, with the highest final conductivity found in the NK sample.
Stability of pH Value
Based on Figure 1, a decrease in pH was observed in each sample over the 28-day storage period. The most significant pH change occurred in the pH 8 sample, with an initial pH of 4.89 that decreased to 3.2 on day 28.
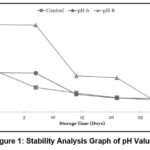 |
Figure 1: Stability Analysis Graph of pH Values |
Stability of Absorbance Value
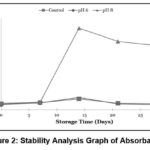 |
Figure 2: Stability Analysis Graph of Absorbance |
Based on the absorbance values observed in the samples over the 28-day storage period in Figure 2, it was found that all samples showed an increase in absorbance, although the increase was not significant.
Stability of Transmittance and Turbidity Values
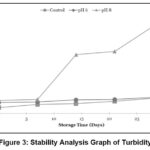 |
Figure 3: Stability Analysis Graph of Turbidity |
Based on the turbidity analysis results shown in Figure 3, all samples experienced an increase in turbidity values during the 28-day storage period, with the most significant increase observed in sample N8. The rate of increase for sample N8 was also higher compared to the rates of turbidity increase in samples Nk and N6.
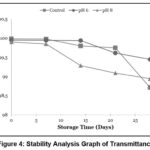 |
Figure 4: Stability Analysis Graph of Transmittance |
Based on the stability analysis results of the transmittance values shown in Figure 4, it was observed that all samples experienced a decrease in transmittance, although the decrease was generally not significant. At the end of the storage period on day 28, sample N8 had the lowest transmittance value compared to the other treatments, while samples N6 and Nk exhibited higher transmittance values.
Stability of Total Anthocyanins, Surface Anthocyanins, and Encapsulation Efficiency
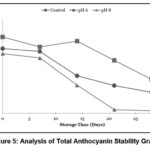 |
Figure 5: Analysis of Total Anthocyanin Stability Graph |
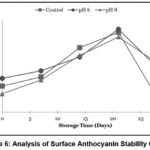 |
Figure 6: Analysis of Surface Anthocyanin Stability Graph |
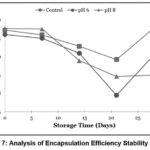 |
Figure 7; Analysis of Encapsulation Efficiency Stability Graph |
Based on the analysis of encapsulation efficiency in Figure 7, it was observed that there was a fluctuating change in encapsulation efficiency values, showing a decreasing trend across all treatments. The encapsulation efficiency in this study is closely related to the total anthocyanin and surface anthocyanin values. As shown in Figure 5, the total anthocyanin values for each treatment experienced a decline. The analysis of surface anthocyanin in this study, as depicted in Figure 6, indicated that surface anthocyanin values increased until the 21st day of storage, after which a decline was observed in the subsequent storage period.
Discussion
Particle size in nanodispersions can reflect the characteristics of the system. The smaller the particle size of a nanodispersion, the larger its surface area, leading to increased bioavailability of the encapsulated active compounds due to more effective interactions with the surrounding medium. Nanodispersions typically have a particle size range of 10–200 nm. Smaller particle sizes indicate higher emulsion stability, as the likelihood of aggregation and coalescence is reduced. Additionally, particle size is closely related to the polydispersity index, which indicates the homogeneity of the emulsion system.13 The increase in particle size during storage may occur due to several factors, including particle aggregation, surfactant degradation, and pH changes in the formulation. Surfactant degradation will cause the surfactant to lose its ability to maintain repulsive forces between particles, while pH changes during storage can alter the charge on the particle surface. These factors can lead to particle aggregation, where smaller particles combine to form larger particles.14 The particle size values obtained in this study may be attributed to the use of Tween 80 as a surfactant in the Parijoto Nanodispersion. Tween 80 is a non-ionic surfactant with a unique structure compared to other types of Tween surfactants. Tween 80 has a single unsaturated hydrocarbon chain (C18:1), which provides greater flexibility to the nanodispersion. Its mode of action involves reducing surface tension at the nanodispersion droplets, enabling the formation of smaller and more stable droplets.15
In terms of the variation in pH solution conditions, the addition of Parijoto nanodispersion to solutions with varying pH levels follows this particle size trend: NK ≤ N6 ≤ N8. The smaller particle size at the NK and N6 samples can be attributed to Tween 80, which acts as a surfactant at its most optimal pH range. A study by Sahumena et al.16 demonstrated that Tween 80, as a surfactant in Vitamin E nanodispersion using virgin coconut oil and olive oil, exhibited the highest stability at pH 6.19. Tween 80 may degrade more rapidly at higher pH levels, reducing its ability to maintain the system’s surface tension. However, based on the particle size analysis, all samples are still classified as nanodispersions, as their sizes remain below 1000 nm, by the theory by.17
The polydispersity index (PDI) describes the size distribution of particles in a nanodispersion, with smaller PDI values indicating a more homogeneous particle size distribution. When the particle size distribution is uniform or monodisperse, the likelihood of droplet coalescence in the emulsion system decreases. Furthermore, a low PDI suggests that all droplets in the nanodispersion system have a high surface area due to the uniform particle size distribution, leading to enhanced interaction between the active compounds within the nanodispersion and the surrounding medium.13 A higher polydispersity index value indicates that the particles have increasingly irregular size and distribution.18 According to previously mentioned theory, the surfactant Tween 80 used in this study works most optimally when the formulation’s pH is close to neutral. Surfactant degradation also occurs more rapidly at basic pH levels. When both of these factors are present, there is an increased potential for particle aggregation in the nanodispersion. Particle aggregation results in particles with uneven sizes and increasingly irregular distribution. Based on this, the particle size and polydispersity index measurements in this study are consistent with the theory, showing that particle size stability and polydispersity index values are better in the control sample or under neutral pH conditions.
Zeta potential measures the strength of the electrical charge present on the surface of nanodispersion particles. A higher Zeta potential value (whether positive or negative) indicates that there is sufficient repulsive force between charged emulsion particles to prevent particle aggregation. An emulsion is considered to have high stability when its Zeta potential is greater than +30 mV or less than -30 mV. Zeta potential is often linked to conductivity, though the relationship is not always linear. Conductivity is closely related to the ability of a solution to conduct electrical charges. A higher conductivity value generally indicates that an emulsion system has a high number of ions in its continuous phase.
Conductivity can also influence the interaction of encapsulated active compounds and the release properties of these compounds into the surrounding environment. However, high conductivity actually indicates instability in the nanodispersion system.13 This instability is caused by the high ion content in the continuous phase, which reduces the Zeta potential. Consequently, the repulsive forces between particles decrease, leading to particle aggregation. According to Handayani,19 using non-ionic surfactants like Tween 80, as used in this study, tends to decrease the zeta potential value of the formulation. As mentioned earlier, Tween 80 is a non-ionic surfactant, meaning the surface of the nanodispersion droplets lacks charge, resulting in weak repulsive forces between particles.15,3 Zeta potential measurement involves measuring the speed of charged particles moving toward an oppositely charged electrode. According to the theory by Olii,20 which states that a good nanodispersion has a zeta potential value near -30 mV or +30 mV, it was found in this study that the control pH sample (NK) maintained fairly good zeta potential stability; meanwhile, N6 and N8 samples shows a lower zeta potential value.
The addition of NH4OH and H3PO4 to obtain pH 6 and pH 8 has the potential to affect zeta potential values. Tween 80 degradation is known to occur more rapidly in acidic and basic conditions. Degraded Tween 80 during storage may release free fatty acids from its structure, contributing to an increase in H+ ions in the formulation. According to Lowry,21 an increase in H+ ions in the medium can cause changes in particle surface charge, weakening the repulsive forces between particles in the formulation and lowering the zeta potential value.
A decrease in conductivity indicates more limited ion movement within the system, potentially triggered by an increase in particle size within the system. Tween 80 degradation is also known to increase Van der Waals forces between particles. Greater Van der Waals forces between particles encourage particles to aggregate and form larger particles. Particle size changes also affect the conductivity value in the formulation, as larger particle sizes reduce the space available for ion movement. This reduces the system’s ability to conduct electricity between two points. Supported by the previously mentioned theory that Tween 80 degradation occurs more rapidly in acidic and basic conditions, the conductivity stability analysis results in this study align with the finding that the control sample NK exhibited the best conductivity stability.14,22
Nanodispersions can be formulated to remain stable across various pH conditions depending on the composition of the materials and the type of surfactant used. Research by Teo et al.23 indicates that nanodispersions using Tween surfactants have a pH range from 2 to 10 and show the highest stability at pH 4.5 to 5. Anthocyanins are pigments found in various fruits, including the parijoto fruit. Anthocyanins can influence the color stability and pH value of a solution. This is because anthocyanins will undergo protonation at low pH, leading to solutions interacting with anthocyanins having higher hydrogen ion (H+) concentrations.24 Measuring pH values during storage is important because pH is a key stability attribute for nanodispersions; chemical reactions during storage may alter the final product quality, indicated by changes in pH. 25
The pH of a nanodispersion can be influenced by several factors, including the amount of extract used, the characteristics of the added extract, and the type of surfactant used.26 Parijoto fruit extract is known to have a very low pH of approximately 2, so using it in a nanodispersion formulation will result in a low pH for the nanodispersion. The decrease in pH during storage observed in all samples may be caused by anthocyanin degradation in the solution and the structural degradation of Tween 80 used in this study. Tween 80 is stable in neutral pH formulations, but under acidic conditions with an abundance of H+ ions, or under basic conditions with an abundance of OH- ions, hydrolysis of ester bonds within the polysorbate structure can occur, releasing free fatty acids from its structure. Acidic hydrolysis involves protonation of the carbonyl carbon structure, while basic hydrolysis occurs when OH- ions attack the carbonyl carbon structure.14 In this study, the abundance of H+ ions is supported by the use of H3PO4, while OH- ions are provided by NH4OH. To achieve the pH 8 solution variation, more NH4OH was added compared to the amount used to reach pH 6, increasing the OH- concentration in the medium. The release of free fatty acids from the Tween 80 structure during storage can lead to a pH decrease in the formulation due to the addition of H+ ions from the carboxyl groups of free fatty acids, which dissociate in solution into H+ ions and R-COO- anions.27
The absorbance value of nanodispersions is influenced by various factors, primarily the physicochemical characteristics of the nanodispersion system itself. In an oil-in-water nanodispersion, small particles are formed by the dispersed oil phase in the water phase with the aid of surfactants or emulsifiers. Interactions between particles and surfactants can affect the characteristics of the emulsion system. The stability of nanodispersions can be influenced by environmental conditions, such as temperature, pH, and the presence of oxygen, which may trigger oxidative reactions that damage the nanodispersion structure. Changes in absorbance values can indicate occurrences of agglomeration, coalescence, or particle degradation within the emulsion system.28
The purpose of measuring absorbance in this study was to assess particle size within the formulation. According to Mekuye,29 there is a relationship between absorbance values and particle size. An increase in absorbance indicates that the particles in the formulation are becoming smaller, as smaller particles have a larger surface area, which leads to greater light absorption in the formulation. However, the absorbance measurements in this study are somewhat inconsistent with this theory, as absorbance values increased despite particle size analysis on day 30 indicating that all samples experienced an increase in particle size. Generally, an increase in particle size should result in decreased absorbance, not the opposite. High particle concentrations and poor particle distribution in the sample may have disrupted accurate absorbance measurements using the UV-Vis spectrophotometer.30
Turbidity measurements of the sample, taken with a turbidimeter, are used to evaluate the formulation’s stability against phenomena such as flocculation, creaming, or coalescence. Turbidity values also reflect the cloudiness of a solution; higher turbidity values indicate a more turbid solution, and lower values indicate increased clarity. According to Uluata et al.,31 the increase in turbidity values can be associated with an increase in particle size during storage. As particle size increases and the sample becomes cloudier, less light is scattered, leading to higher turbidity values. This finding is consistent with the particle size analysis results at the end of the storage period, which indicated that all samples experienced an increase in size.
The transmittance value of a sample indicates its clarity. Stephanie (2015) states that a transmittance value approaching 100% means the sample is increasingly clear. Higher transmittance values generally suggest smaller droplet sizes within the sample. Smaller particles allow light to pass through more easily, resulting in higher transmittance values. Aquabidest is used as a reference because it is free from particles, allowing light to pass through completely, resulting in a transmittance value of 100%.32 Nanodispersions are known to appear clear, with transmittance values typically ranging between 90 and 100%. The decrease in transmittance was noted to occur after a storage period of 7 days. As previously mentioned, transmittance values can indicate particle size within the formulation. The analysis of transmittance values in this study was found to be inversely related to the final particle sizes for each treatment. The N8 treatment, with the lowest transmittance value, had the largest final particle size, while sample Nk, with the highest final transmittance value, had the smallest final particle size.
The increase in particle size within the samples resulted in increased turbidity and decreased transmittance values. This phenomenon occurred due to the degradation of the surfactant Tween 80, leading to a loss of its ability to maintain inter-particle tension and resulting in particle aggregation. This is related to the earlier explanation that the presence of abundant H⁺ and OH⁻ in the formulation would lead to greater degradation of Tween 80. When degraded, Tween 80 loses its ability to stabilize particles, leading to particle aggregation phenomena, which are triggered by the thinning or even loss of the steric layer around the particles, making them more susceptible to aggregation.33 At the end of this study, it was noted that the lowest transmittance value was found in sample N8. However, the transmittance values for all samples remained within a good range for nanodispersions by day 28 of storage.
Anthocyanins found in parijoto fruit are known to have various health benefits. However, research conducted by Khoo34 has shown that anthocyanins have very low stability and their compounds are prone to degradation. Several factors influence the stability of anthocyanins, including pH, temperature, storage conditions, light, oxygen, and the presence of enzymes, proteins, and metal ions, which can disrupt anthocyanin stability. Anthocyanins are highly sensitive to the pH of their environment. Under low pH conditions, specifically at pH <3, anthocyanins exist in the flavilium cation form, resulting in red or purple coloration. As the pH increases above 4 and proton transfer reactions occur, anthocyanins form colorless hemiketal compounds. Degradation of anthocyanins is known to occur when the pH exceeds 7.35 To prevent degradation of anthocyanins due to environmental influences, encapsulation treatments can be applied. Encapsulation works by trapping bioactive compounds such as anthocyanins within a matrix, thereby reducing their degradation rate. Reports indicate that encapsulation can effectively prevent interactions between active compounds and their environment.36
As shown in Figure 5, the total anthocyanin values for each treatment experienced a decline. This reduction in total anthocyanin may be attributed to the degradation of anthocyanin structures into their aglycone form, known as anthocyanidin. According to Oktavi,37 the aglycone form of anthocyanin is known to have lower stability, leading to color changes. The decrease in surface anthocyanin on the 21st day may have been caused by the degradation of anthocyanin compounds in the samples, leading to them no longer being detected as anthocyanins, as the UV-Vis spectrophotometer could not detect their presence at the used wavelength.38 This study revealed that the highest rate of decline in encapsulation efficiency occurred in sample N8, attributed to Tween 80 losing its ability to maintain the stability of the anthocyanin nanodispersion in the formulation, which in turn reduced its encapsulation efficiency. As previously mentioned, the addition of NH4OH and H3PO4 introduces additional H+ and OH- ions into the formulation, which triggers the degradation of Tween 80, resulting in its loss of ability to stabilize the formulation’s particles.13 Consequently, the highest rate of decline in encapsulation efficiency was observed in sample N8, followed by samples N6 and Nk.
Conclusion
The control pH condition, without the addition of acidic or basic solutions, provided the best stability, as indicated by the stability analysis of particle size, zeta potential, polydispersity index, conductivity, pH value, turbidity, % transmittance, absorbance, and encapsulation efficiency over the 28-day storage period. Variations in pH conditions of 6 and 8 in the nanodispersion solution led to faster degradation of Tween 80 during the 28-day storage, resulting in reduced stability of the samples compared to those in the control treatment. Future research could explore the effects of pH modulation on other stabilizing agents, as well as investigate alternative surfactants or formulations that may improve the robustness of nanodispersions under varied storage conditions.
Acknowledgement
This study endeavour was completed by teamwork, which is gratefully welcomed. Sincere gratitude is extended to the research adviser for their tremendous advice, assistance, and knowledge during the investigation. Colleagues and other researchers are also thanked for their support and collaboration, which helped make this study a success.
Funding Sources
Sincere appreciation is given to the Ministry of Research and Higher Education for providing financial support for this study under the Fundamental Research Grant 2024, number 0459/E5/PG.02.00/2024. Additionally, gratitude is given to the research partners for their important contributions.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study. All data generated or analyzed during this study are included in this published article. Additional supplementary data may be made available upon reasonable request by contacting the corresponding author via email.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Materials from Other Sources
Not Applicable
Author Contributions
- Victoria Kristina Ananingsih: Conceptualization, Funding Acquisition, Writing – Review & Editing, Supervision.
- Callista Angelique Marcellino: Methodology, Data Collection, Analysis, Visualization, Writing – Original Draft, Review, Editing.
- Kathleen Felicia Sakoco: Data Collection, Analysis, Visualization, Writing – Original Draft.
- Yohanes Alan Sarsita: Conceptualization, Funding Acquisition, Methodology, Supervision, Writing – Review.
- Novita Ika Putri: Conceptualization, Visualization, Supervision, Writing – Review.
- Alberta Rika Pratiwi: Supervision, Writing – Review.
- Bernadeta Soedarini: Supervision, Writing – Review.
- Florentinus Budi Setiawan : Supervision, Writing – Review
References
- Algahtani, M.S., Ahmad, M.Z., Ahmad, J. Investigation of factors influencing formation of nanoemulsion by spontaneous emulsification: impact on droplet size, polydispersity index, and stability. Bioengineering. 2012; 9(8): 384. https://doi.org/10.3390/bioengineering9080384
CrossRef - Amilia, A. N., Rosita, N., Yusuf, H. Efek Penambahan Kolesterol dan Tween 80 terhadap Karakteristik dan Stabilitas Fisik Liposom Hydrolyzed Collagen: The Effect of Adding Cholesterol and Tween 80 on the Physical Characteristics and Stability of Hydrolyzed Collagen Liposomes. Jurnal Sains dan Kesehatan. 2024; 6(1): 164-171. https://doi.org/10.25026/jsk.v6i1.2291
CrossRef - Sabbah, M., Esposito, M., Pierro, P.D., et al. Insight into zeta potential measurements in biopolymer film preparation. Biotechnol. Biomater. 2016; 6(2): 2-4. http://dx.doi.org/10.4172/2155-952X.1000e126
CrossRef - Zhang, J., Bing, L., & Reineccius, G. A. Formation, optical property and stability of orange oil nanoemulsions stabilized by Quallija saponins. LWT – Food Science and Technology. 2015; 64(2): 1063–1070. https://doi.org/10.1016/j.lwt.2015.07.034
CrossRef - Zhang, L., Liu, A., Wang, W., et al. Characterization of microemulsion nanofilms based on Tilapia fish skin gelatine and ZnO nanoparticles incorporated with ginger essential oil: Meat packaging application. International Journal of Food Science & Technology. 2017; 52(7): 1670-1679. https://doi.org/10.1111/ijfs.13441
CrossRef - Nafi’ah, L.N. AKTIVITAS FARMAKOLOGI TANAMAN PARIJOTO (Medinilla speciosa). Jurnal Riset Rumpun Ilmu Kesehatan. 2022; 1(1); 09-18. https://doi.org/10.55606/jurrikes.v1i1.172
CrossRef - Sa’adah, N.N., Indiani, A.M., Nurhayati, et al. Bioprospecting of parijoto fruit extract (Medinilla speciosa) as antioxidant and immunostimulant: Phagocytosis activity of macrophage cells. In AIP Conference Proceedings. 2020; 2260(1). https://doi.org/10.1063/5.0016435
CrossRef - Ifadah, R.A., Wiratara, P.R.W., Afgani, C.A. Ulasan ilmiah: antosianin dan manfaatnya untuk kesehatan. Jurnal Teknologi Pengolahan Pertanian. 2022; 3(2). https://doi.org/10.35308/jtpp.v3i2.4450
CrossRef - Moldovan, B., David, L. Influence of different sweeteners on the stability of anthocyanins from cornelian cherry juice. Foods. 2020; 9(9): 1266. https://doi.org/10.3390/foods9091266
CrossRef - Zahed, N., Esmaeilzadeh Kenari, R., Farahmandfar, R. Effect of different extraction methods on antioxidant properties and encapsulation efficiency of anthocyanin of pomegranate peel. Food Science & Nutrition, 2023; 11(7): 3780-3787. https://doi.org/10.1002/fsn3.3362
CrossRef - Zhang, J., Bing, L., Reineccius, G. A. Formation, optical property and stability of orange oil nanoemulsions stabilized by Quallija saponins. LWT – Food Science and Technology. 2015; 64(2): 1063–1070. https://doi.org/10.1016/j.lwt.2015.07.034
CrossRef - Wiwiek, I. A., Martodihardjo, S., Budiana, I. G. M. N. Preparation and In-Vitro characterization of Self-Nano emulsifying system of C-Phenylcalix-[4]-Resorcinaryl Octacinnamate and C-Methylcalix-[4]-Resorcinaryl Octabenzoate as ultraviolet absorbers. Bali Medical Journal. 2017; 6(3): 569-577. https://doi.org/10.15562/bmj.v6i3.699
CrossRef - Algahtani, M.S., Ahmad, M.Z., Ahmad, J. Investigation of factors influencing formation of nanoemulsion by spontaneous emulsification: impact on droplet size, polydispersity index, and stability. Bioengineering. 2022; 9(8): 384. https://doi.org/10.3390/bioengineering9080384
CrossRef - Dwivedi, M., Buske, J., Haemmerling, F., et al. Acidic and alkaline hydrolysis of polysorbates under aqueous conditions: towards understanding polysorbate degradation in biopharmaceutical formulations. European Journal of Pharmaceutical Sciences. 2020; 144: 105211. https://doi.org/10.1016/j.ejps.2019.105211
CrossRef - Guttoff, M., Saberi, A.H. and McClements, D.J., 2015. Formation of vitamin D nanoemulsion-based delivery systems by spontaneous emulsification: factors affecting particle size and stability. Food chemistry. 2015; 171: 117-122. https://doi.org/10.1016/j.foodchem.2014.08.087
CrossRef - Sahumena, M.H., Mabilla, S.Y., Ningsih, S.R., Adjeng, A.N.T., Aswan, M. and Nisa, M., 2020. Preparation and Evaluation of Physical Characteristics of Vitamin E Nanoemulsion using virgin coconut Oil (VCO) and olive oil as oil phase with variation Concentration of tween 80 Surfactant. Research Journal of Pharmacy and Technology. 2020; 13(7): 3232-3236. http://dx.doi.org/10.5958/0974-360X.2020.00572.7
CrossRef - Muchlisyiyah, J., Laeliocattleya, R. A., Putri, W. D. R. Kimia Fisik Pangan. Universitas Brawijaya Press. 2017. ISBN 978-602-432-427-8.
- Prasetyo, B. E., Maruhawa, S. M., Laila, L. Formulation and physical evaluation of castor oil-based nanoemulsion for diclofenac sodium delivery system. Research Journal of Pharmacy and Technology. 2018; 11(9): 3861-3865. https://doi.org/10.5958/0974-360X.2018.00707.2
CrossRef - Handayani, F. S., Nugroho, B. H., Munawiroh, S. Z. Optimasi formulasi nanodispersi minyak biji anggur energi rendah dengan d-optimal mixture design (DMD). Jurnal Ilmiah Farmasi. 2018; 14(1): 17-34. https://doi.org/10.20885/jif.vol14.iss1.art03
CrossRef - Olii, A. T., Pamudji, J. S., Mudhakir, D., et al. Pengembangan, Evaluasi, dan Uji Aktivitas Antiinflamasi Akut Sediaan Nanoemulsi Spontan Minyak Jintan Hitam. Jurnal Farmasi Indonesia. 2014; 7(2): 78. https://doi.org/10.35617/JFI.V7I2.219
- Lowry, G. V., Hill, R. J., Harper, S., et al. Guidance to improve the scientific value of zeta-potential measurements in nanoEHS. Environmental Science: Nano. 2016; 3(5): 953-965. https://doi.org/10.1039/C6EN00136J
CrossRef - Wei, Q., Yang, Q., Gao, W. Analysis of particle size distribution and electro-kinetics of submicron particles in shear-thinning liquids. Colloid and Polymer Science. 2024; 302(2): 173-181. https://doi.org/10.1007/s00396-023-05189-9
CrossRef - Teo, A., Goh, K. K., Wen, J., et al. Physicochemical properties of whey protein, lactoferrin and Tween 20 stabilized nanodispersions: Effect of temperature, pH and salt. Food chemistry. 2016; 197: 297-306. https://doi.org/10.1016/j.foodchem.2015.10.086
CrossRef - Torskangerpoll, K.. Andersen, Ø.M. Colour stability of anthocyanins in aqueous solutions at various pH values. Food Chemistry. 2005; 89(3): 427-440. https://doi.org/10.1016/j.foodchem.2004.03.002
CrossRef - Bernardi, D. S., Pereira, T. A., Maciel, N. R., et al. Formation and stability of oil-in-water nanoemulsions containing rice bran oil: in vitro and in vivo assessments. Journal of nanobiotechnology. 2011; 9: 1-9. https://doi.org/10.1186/1477-3155-9-44
CrossRef - Son, H. Y., Lee, M. S., Chang, E., et al. Formulation and characterization of quercetin-loaded oil in water nanoemulsion and evaluation of hypocholesterolemic activity in rats. Nutrients. 2019; 11(2): 244. https://doi.org/10.3390/nu11020244
CrossRef - Honemann, M. N., Wendler, J., Graf, T., et al. Monitoring polysorbate hydrolysis in biopharmaceuticals using a QC-ready free fatty acid quantification method. Journal of Chromatography B. 2019; 1116: 1-8.https://doi.org/10.1016/j.jchromb.2019.03.030
CrossRef - McClements, D.J., Rao, J., Food-grade nanoemulsions: formulation, fabrication, properties, performance, biological fate, and potential toxicity. Critical reviews in food science and nutrition. 2011; 51(4): 285-330. https://doi.org/10.1080/10408398.2011.559558
CrossRef - Mekuye, B. The impact of size on the optical properties of silver nanoparticles based on dielectric function. Journal of Nanomaterials and Nanostructures. 2023. https://doi.org/10.5772/intechopen.113976
CrossRef - Kandi, S., Charles, A.L. Statistical comparative study between the conventional DPPH spectrophotometric and dropping DPPH analytical method without spectrophotometer: Evaluation for the advancement of antioxidant activity analysis. Food chemistry. 2019; 287: 338-345 .https://doi.org/10.1016/j.foodchem.2019.02.110
CrossRef - Uluata, S., Decker, E. A., McClements, D. J. Optimization of nanoemulsion fabrication using microfluidization: role of surfactant concentration on formation and stability. Food biophysics. 2016; 11: 52-59. https://doi.org/10.1007/s11483-015-9416-1
CrossRef - Listyorini, N. M. D., Wijayanti, N. L. P. D., Astuti, K. W. Optimasi pembuatan nanoemulsi virgin coconut oil. Jurnal Kimia. 2018; 12(1): 8-12. https://doi.org/10.24843/JCHEM.2018.v12.i01.p02
CrossRef - Amilia, A. N., Rosita, N.,Yusuf, H. Efek Penambahan Kolesterol dan Tween 80 terhadap Karakteristik dan Stabilitas Fisik Liposom Hydrolyzed Collagen: The Effect of Adding Cholesterol and Tween 80 on the Physical Characteristics and Stability of Hydrolyzed Collagen Liposomes. Jurnal Sains dan Kesehatan. 2024; 6(1): 164-171. https://doi.org/10.25026/jsk.v6i1.2291
CrossRef - Khoo, H. E., Azlan, A., Tang, S. T., Lim, S. M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food & nutrition research. 2017. https://doi.org/10.1080/16546628.2017.1361779
CrossRef - Sharma, R. J., Gupta, R. C., Singh, S., et al. Stability of anthocyanins-and anthocyanidins-enriched extracts, and formulations of fruit pulp of Eugenia jambolana (‘jamun’). Food Chemistry. 2016; 190: 808-817. https://doi.org/10.1016/j.foodchem.2015.06.029
CrossRef - Marcillo-Parra, V., Tupuna-Yerovi, D. S., González, Z., Ruales, J. Encapsulation of bioactive compounds from fruit and vegetable by-products for food application–A review. Trends in Food Science & Technology. 2021; 116: 11-23. https://doi.org/10.1016/j.tifs.2021.07.009
CrossRef - Oktavi, R. A., Cahyono, B., Suzery, M. Enkapsulasi Ekstrak Antosianin Dari Bunga Rosela (Hibiscus sabdariffa L.) dengan Variasi Penyalut Akta Kimia Indonesia.2020; 5(2): 86-101. https://doi.org/10.12962/j25493736.v5i2.7841
CrossRef - Priska, M., Peni, N., Carvallo, L., Ngapa, Y. D. Antosianin dan pemanfaatannya. Cakra Kimia (Indonesian E-Journal of Applied Chemistry). 2018; 6(2): 79-97. https://ojs.unud.ac.id/index.php/cakra/article/view/46629













