Introduction
Rice is one of major food in the world, and it has some varieties. Appearance-wise, there are pigmented and non-pigmented rice. The characteristics between pigmented and non-pigmented rice are differentiated by the bran. Pigmented rice contains anthocyanins in the aleurone (bran layers) as the pigment colorants, have functions as antioxidants and a higher nutritional content than non-pigmented rice.1 Indonesia has hundreds of rice varieties, both pigmented and non-pigmented. Inpari 24 (red rice) and Jeliteng (black rice) are improved pigmented rice varieties released by the Indonesian Agency for Agricultural Research and Development.2 Saodah (red rice) and Cempo Ireng (black rice) are local rice varieties in Yogyakarta Province.3,4 Pigmented rice contains bioactive compounds such as phenolic acids, flavonoids, anthocyanins, proanthocyanidins, tocopherols, tocotrienols, c-oryzanol and phytic acid.1The bioactive compound composition of pigmented depends on the cultivar (genetic), environment, cultivation practices, postharvest and processing.1
Over the years, aroma has become one of consumer preference for rice.5 Volatile compounds play a key role in aroma formation in rice bran (RB) and are usually identified using gas chromatography-mass spectrometry (GC-MS). The volatile compound of RB consists of esters, alkanes, alcohol, ketones and aldehydes.6 Acid and aldehydes, especially hexanal and nonanal in high amount, are thought to form rancid aroma in RB because of lipid degradation reactions compounds.6,7 Our research have showed that the dominant volatile compounds of black RB were 2-furanmetanol, hexanal, naphthalene, 1R-α -pinene, and 4-ethenyl-2-methoxyphenol, while for non-pigmented RB, they were 2-furanmetanol, nonanal, methyl tetradecanoate, phenol and 4-ethenyl-2-methoxyphenol; they produced burnt, nutty, fatty and pungent aromas.8
Solid-state fermentation (SSF) is one of the fermentation techniques that can increase the content of bioactive components in foods9-11 and is thought to cause the reduction of lipid oxidation in RB. SSF using Rhizopus oryzae resulted in higher total phenolic content and antioxidant activity of Inpari 30 and Cempo Ireng RB; it also reduced the hexanal content due to hydrolases production during the fermentation process, which are responsible for the degradation of polysaccharides, oxidative and extracellular ligninolytic systems.8,12 SSF also increased the content of chlorogenic acid, p-hydroxybenzoic acid and vanillin, which gives the vanilla flavour in the bran.13
Identification of volatile compounds in some varieties of fermented Indonesia RB has been examined using GC-MS.8 The developments of such studies are needed to expand our previous study8 and enhance the knowledge of volatile compounds as well as aroma profiles of Indonesian pigmented RB varieties. Thus, the objective of these studies was to identify the volatile compounds and aroma profiles that are responsible for the flavour attributes of fermented RB from pigmented rice varieties—Saodah, Inpari 24 (red rice), Jeliteng and Cempo Ireng (black rice)—using GC-MS and qualitative descriptive analysis (QDA) methods.
Materials and Methods
Rice Bran Preparation
The samples used in this study were red paddies (Saodah and Inpari 24 varieties) and black paddies (Cempo Ireng and Jeliteng varieties); they were obtained from farmers in Bantul and Sleman Regency, Yogyakarta, Indonesia. RB preparation was done based on previous study3 with modification. Rice paddy was de-hulled using LM 24 to obtain brown rice. All samples were then polished using ICHI N50 resulting RB. RB samples were sterilized using an autoclave at 121oC for 15 minutes and stored at 5oC.The samples were divided into a non-fermented group: Inpari 24RB (Inp24NF), Saodah (SaodahNF), Cempo Ireng (CINF) and Jeliteng (Jeliteng NF), and a fermented group: Inpari 24 RB (Inp24F), Saodah (SaodahF), Cempo Ireng (CIF) and Jeliteng (JelitengF).
RB Fermentation
Rhizopus oligosporus with code 6010 was purchased from the Center for Food and Nutrition Studies, Universitas Gadjah Mada, Yogyakarta, Indonesia. Culture preparation and fermentation method refer to the previous study8 with a slight modification. R. oligosporus was inoculated with the pour plate method on potato dextrose agar (PDA). Fermented and non-fermented RB were dried using a freeze dryer (VirTis, SP SCIENTIFIC BenchTop Pro) for 2 days before further analysis.
Sample Extraction
HS-SPME method was used for sample extraction according to the prior study14 with modification. Briefly, 2cm of DVB/CAR/PDMS, 50/30 μm fibre (SUPELCO Bellefonte, PA USA) with 2,4,6-trimethyl pyridine as internal standard were used for the extraction. Three grams (±0.1 g) of the sample were put in a 22 mL headspace vial and sealed with Septa PTFE/Silicon septum. The sample was immersed in a water bath at 80oC and then extracted with DVB/CAR/PDMS, 50/30 μm fibre for 30 minutes. The fibres were removed from the vial and injected into the GC-MS injector at desorption for 10 minutes at 250oC in splitless mode.
Volatile Compound Identification
The identification of volatile compound in RB was done using GC-MS (GC Agilent Technologies 7890 A, MS Agilent 5975 C with triple exist detector XL EI/CI) that was equipped with a splitless mode injection port at 250oC. DBWax capillary column (30 m×0.25 mm×0.25 μm film thickness; Agilent Technologies) was used with a mass detector (TSQ Quantum XLS). The detector temperature was programmed at initial temperature of 40ºC for 5 minutes, which was then increased to 110ºC with 5ºC/min speed and then increased again to 230ºC at a speed of 8ºC/min; finally, it was maintained for 5 min. Interface area temperature was set at 250ºC. Helium is used as a carrier gas at a rate of 0.8 mL/min.
Evaluation of Aroma Attributes
QDA was used to evaluate the aroma attributes in RBs. The analysis was performed by 10 panellists (7 females and 3 males) who were trained based on ISO 8586-2012. Before evaluation, all panellists supplied an informed consent letter. The training was held 3 times (3 hours/time) with a final evaluation. Qualitative analysis was carried out by focus group discussions to obtain subjective data on the description of the aroma of fermented and non-fermented RB. Eight samples with trivial code consisting of fermented and non-fermented RB powder from four different varieties were presented individually to avoid bias during testing. Three-gram samples were served in odourless glasses at room temperature. The trained panellists provided an assessment of the aroma attributes present in the RB samples. Furthermore, the panellists were asked to inhale the aroma of the sample for 5 seconds and then neutralized it with the aroma of coffee and drink mineral water.15
Data Analysis
The results obtained were processed by principal component analysis (PCA). The PCA results were visualized in the form of a biplot graphic using XLSTAT 2021 software. The correlation of RB volatile compound and aroma attributes from GC-MS and QDA were analysed using Pearson’s correlation with XLSTAT 2021.
Result and Discussion
Volatile Compounds of Rice Bran
A total of 114 compounds were identified from GC–MS analysis in fermented and non-fermented RB; they consisted of 14 aldehydes, 12 ketones, 14 alcohols, 15 hydrocarbons, 8 acids, 23 esters, 9 benzenes, 5 phenols, 6 furans, 2 lactones, 1 monoterpene, 1 sesquiterpen, 1 thiazole, 1 pyrazine and 1 pyridine (Figure 1). The identified volatiles and their relative peak areas in the respective variety are summarized in Table 1.
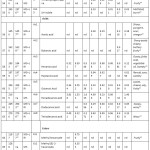 |
Table 1: Volatile Compounds in Various Varieties of Non-fermented RB. |
*MS, mass spectrum match to those NIST/EPA/NIH Mass spectral database; MS+LRI, mass spectrum match to those NIST/EPA/NIH Mass spectral databases and LRI match witch literature value nd: not detected.
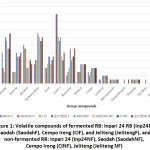 |
Figure 1: Volatile Compounds of Fermented RB: Inpari 24 RB (Inp24F), Saodah (SaodahF), Cempo Ireng (CIF), and Jeliteng (JelitengF), and Non-fermented RB: Inpari 24 (Inp24NF), Saodah (SaodahNF), Cempo Ireng (CINF), Jeliteng (Jeliteng NF) |
Volatile compounds in RB are probably composed of the original compounds, the compound resulting from the Maillard reaction due to the sterilization process and the compound resulting from the fermentation. The original compounds in RB were hexanal; heptanal; octanal; nonanal; benzaldehyde;(3E)-oct-3-en-2-one;2,6,6-trimethyl-2-cyclohexene-1,4-dione; (5Z)-6,10-dimethylundeca-5,9-dien-2-one; 1-(1H-pyrrol-2-yl)ethan-1-one; oct-1-en-3-ol; octan-1-ol; 3,7-dimethylocta-1,6-dien-3-ol; phenylmethanol; 1,2-dimethylbenzene; tetradecane; pentadecane; acetic acid; butanoic acid; hexanoic acid; octanoic acid; phenol; 2-pentylfuran; 2,3-dihydro-1-benzofuran; (3R)-3,4,4-trimethyloxolan-2-one; (4R)-1-Methyl-4-prop-1-en-2-ylcyclohexene (d-limonene) and naphthalene.5,19,49,50
The RBs might be subjected to the Maillard reaction and lipid oxidation might due to heat treatment, which is the sterilization process in this study.19 In the Maillard reaction, the sugar’s reactive carbonyl group combined with the nucleophilic amino group of acid and formed new volatile compounds.51 Reaction between amino acids and carbohydrates in Maillard reaction were reported to form pyrazines such as 2-methyl-5-[(E)-prop-1-enyl]pyrazine, which contributed in roasted and green aroma.52,53 Heat treatment also allegedly formed vanillin and furfural, due to thermal degradation of ferulic acid and sugars, respectively.19 4-ethenyl-2-methoxyphenol was also reported to be derived from thermal decarboxylation of ferulic acid in RB.54
There were 20 volatile compounds that could have been formed during the fermentation process—as they were only found in fermented RB-as shown in Figure 2. They are oct-2-enal, (E)-;(3Z)-pent-3-en-2-one; 6-methyl-3,5-heptadiene-2-one;3-methylbutan-1-ol;butane-2,3-diol; pyridin-3-ylmethanol (nicotinyl alcohol); methyl hexanoate; methyl (E)-2-hexenoate; methyl octanoate; ethyl dodecanoate; methyl pentadecanoate; methyl (9Z)-hexadec-9-enoate; ethyl (E)-hexadec-9-enoate; butyl hexadecanoate; ethyl octadecanoate; methyl (9Z,12Z,15Z)-octadeca-9,12,15-trienoate (methyl linolenate); dihydro-3-hydroxy-4,4-dimethyl-2(3H)-furanone; 2,3-dimethylpyridine and 2-methyl-5-[(E)-prop-1-enyl]pyrazine. This proved that fermentation might produce more volatile compounds in RB.
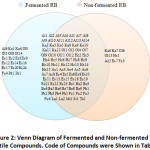 |
Figure 2: Venn Diagram of Fermented and Non-fermented RB Volatile Compounds. Code of Compounds was Shown in Table 1. |
Esters were reported as the major volatiles constituent in fermented RB. The major esters compounds identified in the fermented RB were methyl palmitate, 9-octadecenoic acid, methyl ester, (E)- and ethyl hexadecanoate. Those compounds are responsible for waxy, fatty and oily odor44 and were increasing due to the fermentation process. Esters mostly are formed by esterification between acids and alcohols during fermentation.55 The second most common substances in fermented RB were alcohols. Among them were the contents of butane-2,3-diol, 3-methylbutan-1-ol and phenylmethanol. Butane-2,3-diol is thought to provide a characteristic of buttery and creamy aroma, and is formed from glucose catabolism via the glycolysis pathway.56,57 Phenylmethanol is known to be formed from the reduction of benzoic acid assisted by microorganisms.58 Similar to 3-methyl-3-butanal, phenylmethanol tends to increase in RB treated with fermentation due to the metabolic activity of microbes that form in RB through the glycolysis pathway.
Amyl alcohols (3-methylbutan-1-oland 2-methylbutan-1-ol) have fermented and malt-like odour notes.48 These are also detected in other fermented rice such as makgeolli (Korean rice wine).48 Another alcohol compound that was identified in fermented RB is 2-phenylethanol; it is thought to have a slightly rose floral scent.59 The formation of 2-phenylethanolmight be from hydrolysis of phenylethyl ester and phenylethyl acetal.60
In the group of aldehydes, hexanal, nonanal and benzaldehyde were the compounds with the highest relative peak areas found in fermented RB. These three compounds tend to increase compared to non-fermented RB. Saturated aldehydes such as hexanal and nonanal might be formed by linoleic acid (C18:2) oxidation as one of the main fatty acids in RB. Linoleic acid might be oxidized to form 9-OOOH and 13-OOOH hydrogen peroxides, which are further degraded to form saturated aldehydes such as oct-2-enal and hexanal.61 Hexanal allegedly contributed to grass, tallow and fat aroma; meanwhile, nonanal might have contributed to green, fat and citrus aroma. Benzaldehyde may be the odour-active compounds in RB.6 The content of amino acids in RB such as valine, isoleucine, leucine and phenylalanine can be transformed into Strecker aldehydes, leading to 2-methylpropanal, 2-methylbutanal, 3-methylbutanal and phenylacetaldehyde, which is also the most effective precursor for the production of benzaldehyde.7,62 Benzaldehyde is responsible for giving the nutty and almond aroma.44
In addition, other aldehydes found in fermented rice bran in relatively low peak areas include heptanal, octanal, (2E)-hept-2-enal, oct-2-enal, furfural, decanal, non-2-enal, benzeneacetaldehyde, 3-methylbenzaldehyde and vanillin. Furfural, which provides bread, almond and sweet aroma,44 was derived from the thermal degradation of sugars such as fructose and glucose.63 Unsaturated aldehydes such as (2E)-hept-2-enal, oct-2-enal and non-2-enal are the products of linoleic acid oxidation that provides fat and green, nut and fat aroma, respectively.44,61 Non-2-enal and hexanal were also reported to cause rancid defects in virgin olive oil.64 Benzeneacetaldehyde was found in all varieties of non-fermented RB, while in fermented RB, it was only identified in Jeliteng, and the content tends to increase due to the fermentation process. Benzeneacetaldehyde may be formed from phenylalanine precursor.7 Vanillin, which gives vanilla aroma, originates from lignin degradation in aerobic conditions and might be formed by the thermal degradation of ferulic acid.19
Naphthalene was the highest content of hydrocarbon that was found in RB. The relative peak areas of naphthalene which contributed to camphor wood-like aroma tends to increase due to fermentation. In other prior research, naphthalene was also found to be an odour-active compound in red and black rice.6
Principle Component Analysis (PCA) of Volatile Compounds in RB
The PCA biplot of volatile compounds in fermented and non-fermented RB is shown in Figure 3.
 |
Figure 3: Principal Component Analysis (PCA) biplot of a) Non-fermented RB, b) Fermented RB, c) Fermented and Non-fermented RB. |
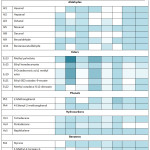 |
Figure 4: Summary of Major Volatile Compounds (ppb) of Inpari 24 Non-fermented (Inp24NF); Inpari 24 Fermented (Inp24F); Saodah Non-fermented (SaodahNF); Saodah Fermented (SaodahF); Cempo Ireng Non-fermented (CINF); Cempo Ireng Fermented (CIF); Jeliteng Non-fermented (JelitengNF); and Jeliteng Fermented (JelitengF). |
PCA was used to analyse the grouping of fermented and non-fermented RB in all varieties and to determine the volatiles characteristic in each group. The data variation (F1 and F2) of non-fermented, fermented and RB groups were 82.15%, 87.08% and 72.11%, respectively (Figure 3). Figure 3a showed that non-fermented RB variety Cempo Ireng, Inpari 24 and Saodah were grouped together with hexadecanoic acid (Ac6), methyl palmitate (Es13), (4R)-1-methyl-4-prop-1-en-2-ylcyclohexene (d-limonene) (Mt1), benzaldehyde (Al9), nonanal (Al5) and octanal (Al3) as the dominant compounds, while Saodah was dominated with ethanol (Ol1). The result of fermented RB is shown in Figure 3b. Saodah and Inpari 24 RB were dominated by methyl palmitate (Es13), 9-octadecenoic acid, methyl ester, (E)- (Es19), butane-2,3-diol (Ol10), ethyl hexadecanoate (Es15) and ethanol (Ol1), while Jeliteng and Cempo Ireng had the higher amount of (4R)-1-methyl-4-prop-1-en-2-ylcyclohexene (d-limonene) (Mt1), hexadecanoic acid (Ac6), 2-methoxyphenol (Ph1) and naphthalene (Hc5).
Cempo Ireng and Jeliteng might be in a group since they have the same pigment: black rice. Figure 3c showed the PCA of fermented and non-fermented RB. Fermented RB of Inpari 24, Saodah and Cempo Ireng were grouped together and their dominant compounds were methyl palmitate (Es13), methyl (E)-octadec-9-enoate (Es19), butane-2,3-diol (Ol10) and ethanol (Ol1) which contribute to oily, waxy, fatty, orris, fruity, creamy, buttery and sweet odours. Meanwhile, fermented Jeliteng was in the same group with all non-fermented RB. This group was characterized by higher amount of (4R)-1-methyl-4-prop-1-en-2-ylcyclohexene (d-limonene) (Mt1), hexadecanoic acid (Ac6), nonanal (Al5), naphthalene (Hc5) and benzaldehyde (Al9), which provided lemon, orange, green, fat, champor wood-like and almond aroma.
The dominant aroma of fermented RB were esters, especially methyl palmitate; ethyl hexadecanoate; 9-octadecenoic acid, methyl ester; ethyl (9Z)-octadec-9-enoate and methyl octadeca-9,12-dienoate (Figure 4). Esters were formed by the esterification of acids and alcohol, and provide fruity and floral notes.55 Therefore, JelitengF had the lowest relative peak areas of esters due to a few numbers of alcohols. The relative peak areas of esters might be affected by sugar content of RB, yeast strains, the temperature of fermentation and aeration.48 Alcohols that dominated the fermented RB were ethanol; 2-methylpropan-1-ol; 3-methylbutan-1-ol; butane-2,3-diol and 2-phenylethanol. Alcohol in fermented RB might be derived by sugar fermentation or amino acids catabolism, so the content of alcohol in fermented RB might be differed by sugar and amino acid availability.65 Other compounds that dominated fermented RBs were hydrocarbons. Tetradecane, pentadecane and naphthalene were the most abundant hydrocarbons contained in fermented RBs. Even so, it might have a little contribution on RB aroma because generally hydrocarbons have high threshold values.66 Acids also became one of main compounds in fermented RB. Prior study stated that acid was the most abundant volatile oil in red and black rice and tends to provide an unpleasant aroma.50 In this study, the relative peak areas of acids were relatively lower than other compounds such as esters and phenols. The differences might be affected by the degree of oxidation.6 RBs used in this study were fresh so the level of oxidation could be minimized and result in a lower level of acids.
QDA of Aroma Attributes
Aroma attributes of fermented and non-fermented RBs from each variety are shown in Figure 5. The QDA result showed that both the fermented and non-fermented RBs had a similar aroma attribute (sweet, caramel, rancid, acid, pungent, fatty, milky, woody, sour, cereal, vanilla, nutty, smokey) except “fermented” aroma, which was only identified in fermented RBs. PCA analysis was used group varieties that had some similarities and matching aromas, based on panellists’ identification.
 |
Figure 5: Aroma Attributes of a) Non-fermented RB and b) Fermented RB by QDA. |
 |
Figure 6: Principal Component Analysis (PCA) Biplot of a) Non-fermented RB, b) Fermented RB, c) Fermented and Non-fermented RB by QDA. |
Both fermented and non-fermented RBs tended to have sweet, acid, vanilla, cereal and caramel aromas as their major attributes. The differences between these RBs were in the number of panellists that recognized the aroma. Aroma attributes of fermented RBs were noticed more by panellists; this might conclude that the intensities of aromas in fermented RBs were higher than non-fermented RBs. The aroma attribute similarities between fermented and non-fermented RBs were allegedly because the RBs used in this study were fresh; thus, they were not dominated by unwanted aroma like pungent and rancid. Sweet, caramel and vanilla aroma might have been derived from the Maillard reaction.52,53
Non-fermented RBs in varieties Jeliteng and Inpari 24 were in the same group with cereal and acid as the dominant aromas, while Saodah and Cempo Ireng were in the same group with the dominant aromas of sweet, vanilla and caramel (Figure 6a). Biplot aroma of fermented RB is shown in Figure 6b. Inpari 24 and Cempo Ireng were in the same group with cereal, acid and sweet as the dominant aromas, while Saodah and Jeliteng were in the same group that were characterized by vanilla and caramel aromas. Figure 6c shows the PCA of fermented and non-fermented RB aromas. Fermented RB varieties Inpari 24, Jeliteng, Cempo Ireng are in the group with non-fermented Inpari 24. These RBs are characterized by acid and cereal aromas. Non-fermented RBs varieties such as Jeliteng, Cempo Ireng and Saodah are in the same group with SaodahF; they were characterized by sweet, vanilla and caramel aromas. Figure 6a shows that non-fermented RB in Jeliteng and Inpari 24 varieties were in the same group with cereal and acid as the dominant aromas, while Saodah and Cempo Ireng were in the same group with the dominant aromas of sweet, vanilla and caramel. Biplot aroma of fermented RBs is shown in Figure 6b. Inpari 24 and Cempo Ireng were in the same group with cereal, acid and sweet as the dominant aromas, while Saodah and Jeliteng were in the same group that were characterized by vanilla and caramel aromas. Figure 6.c shows the PCA of fermented and non-fermented RB aromas. Fermented RBs in Inpari 24, Jeliteng, and Cempo Ireng varieties were the group with non-fermented Inpari 24. These RBs are characterized by acid and cereal aromas. Non-fermented RB varieties such as Jeliteng, Cempo Ireng and Saodah were in the same group with the fermented Saodah; they characterized by sweet, vanilla and caramel aromas.
Pearson’s Correlation of Volatile Compounds and Aroma Profile
The correlation between volatile compounds of RB are identified by GC-MS, and the aroma attributes are identified by QDA (Table 2). It shows that some volatile compounds have positive correlation with aroma description obtained by QDA. Hexanal has positive correlation with grass aroma. This is in accordance with the study44 that stated grass, tallow and fat as the aromas of hexanal. Oct-2-enal has positive correlation with fatty aroma, similar to a previous study’s description.44 The 2-methylpropan-1-ol and 3-methylbutan-1-ol were positively correlated with fermented aroma. It is also in accordance with the description by another previous study.35Acetic acid was described to have sharp, pungent, sour and vinegar aroma35 and the Pearson’s correlation showed that acetic acid correlated with pungent aroma. Ethyl hexadecanoate and ethyl octadecanoate have positive correlation with fatty aroma, while ethyl (9Z)-octadec-9-enoatepositively correlated with acid aroma. 2-methyl-5-[(E)-prop-1-enyl] pyrazine correlated to smokey aroma. These correlations match with the aroma description by previous studies.44,45
Table 2: Pearson’s Correlation between RB Volatile Compounds by GC-MS and Aroma Description by Panelists.
| Variables | Hexanal | 2-0ctenal,(E) | 1-Propanol2-methyl | 1–Butanol, 3–methyl | Acetic acid | Ethyl hexa-decanate | Ethyl stearate | Ethyl oleate | Pyrazine, 2- methyl -5-(1- Prop enyl) -(E) |
| Sweet | -0.035 | 0.232 | 0.003 | -0.160 | -0.177 | 0.165 | 0.189 | 0.163 | -0.567 |
| Caramel | 0.307 | -0.418 | -0.267 | -0.195 | -0.294 | -0.451 | -0.574 | -0.421 | 0.082 |
| Vanilla | -0.274 | -0.549 | -0.353 | -0.221 | 0.002 | -0.526 | -0.570 | -0.541 | -0.114 |
| Grass | 0.895 | 0.519 | 0.278 | 0.135 | -0.003 | 0.452 | 0.424 | 0.503 | 0.424 |
| Milky | -0.384 | -0.257 | -0.152 | -0.156 | 0.142 | -0.194 | -0.087 | -0.244 | -0.087 |
| Fatty | -0.086 | 0.517 | 0.311 | 0.085 | 0.001 | 0.502 | 0.607 | 0.494 | -0.087 |
| Nutty | 0.312 | 0.264 | 0.339 | 0.236 | -0.603 | 0.257 | 0.215 | 0.248 | -0.277 |
| Smokey | 0.259 | 0.422 | 0.376 | 0.284 | 0.179 | 0.458 | 0.535 | 0.459 | 0.535 |
| Rancid | -0.836 | -0.359 | -0.192 | -0.018 | 0.163 | -0.303 | -0.293 | -0.337 | -0.293 |
| Acid | 0.356 | 0.473 | 0.375 | 0.447 | 0.266 | 0.482 | 0.459 | 0.510 | 0.459 |
| Cereal | 0.197 | 0.497 | 0.627 | 0.760 | -0.114 | 0.560 | 0.509 | 0.557 | 0.218 |
| Pungent | -0.197 | -0.292 | -0.462 | -0.446 | 0.543 | -0.313 | -0.238 | -0.305 | 0.143 |
| Earthy | 0.094 | -0.598 | -0.633 | -0.648 | 0.661 | -0.588 | -0.488 | -0.582 | 0.683 |
| Fermented | 0.183 | 0.294 | 0.586 | 0.735 | -0.259 | 0.382 | 0.314 | 0.371 | 0.314 |
Conclusion
Volatile compounds found in RB consist of ester, hydrocarbon, aldehyde, benzene, alcohol, ketone, acid, phenol, furan, lactone, monoterpene, thiazole, sesquiterpene, pyridine and pyrazine. A total 114 volatile compounds were found, out of which 106 were contained in fermentation RB and 94 in non-fermentation RB. Fermentation on RB formed some new volatile compounds such as oct-2-enal; (3Z)-pent-3-en-2-one; 6-methyl-3,5-heptadiene-2-one; 3-methylbutan-1-ol; butane-2,3-diol; 3-pyridinemethanol (nicotinyl alcohol); methyl hexanoate; methyl (E)-2-hexenoate; methyl octanoate; ethyl dodecanoate; methyl pentadecanoate; methyl (9Z)-hexadec-9-enoate; ethyl (E)-hexadec-9-enoate; butyl hexadecanoate; ethyl octadecanoate; methyl (9Z,12Z,15Z)-octadeca-9,12,15-trienoate (methyl linolenate); dihydro-3-hydroxy-4,4-dimethyl-2(3H)-furanone; 2,3-dimethylpyridine and 2-methyl-5-[(E)-prop-1-enyl]pyrazine. The result between GC-MS identification has some positive correlation with QDA; hexanal was correlated with grass; oct-2-enal, ethyl octadecanoate and ethyl hexadecanoate were correlated with fatty; 2-methylpropan-1-ol and 3-methylbutan-1-ol were correlated with fermented aroma; acetic acid was correlated with pungent; ethyl (9Z)-octadec-9-enoate was correlated with acid and 2-methyl-5-[(E)-prop-1-enyl] pyrazine was correlated with smokey.
Acknowledgments
The authors gratefully thank you to Anang Juni Yastanto, Desi Arofah, Yudha Restu Ginanjar Windi and all sensory panelists for technical assistance and helpful suggestions during these studies.
Funding Source
This work was supported by grants from the Ministry of Education, Culture, Research and Technology, Republic of Indonesia (contract No. 163/E4.1/AK.04.PT/2021).
Conflict of Interest
All authors declare no conflict of interest.
References
- Goufo, P., Trindade, H. Factors Influencing Antioxidant Compounds in rice. Critical Reviews In Food Science and Nutrition. 2017; 57(5):893–922.
CrossRef - Sasmita, P., Suprihanto, Nugraha, Y., Hasmi, I., Satoto, Rumanti, I. A., Susanti, Z., Kusbiantoro, B., Rahmini, Hairmansis, A., Sitaresmi, T., Suharna, Norvyani, M., Arismiati, D.. Deskripsi Varietas Unggul Padi. Balitbangtan; 2020.
- Stabilitas dan Adaptabilitas Varietas Padi Merah Lokal Daerah Istimewa Yogyakarta. Buletin Plasma Nutfah. 2016; 16(2):103.
CrossRef - Kristamtini, K., Taryono, T., Basunanda, P., Murti, R. H. Keragaman Genetik Kultivar Padi Beras Hitam Lokal Berdasarkan Penanda Mikrosatelit. Jurnal AgroBiogen. 2016; 10(2): 69.
CrossRef - Chumpolsri, W., Wijit, N., Boontakham, P., Nimmanpipug, P., Sookwong, P., Luangkamin, S., Wongpornchai, S. Variation of Terpenoid Flavor Odorants in Bran of Some Black and White Rice Varieties Analyzed by GC×GC-MS. Journal of Food and Nutrition Research. 2015; 3(2):114–120.
CrossRef - Zeng, M., Zhang, L., He, Z., Qin, F., Tang, H., Huang, X. Determination of Flavor Components of RB by GC-MS and Chemometrics. Analytical Methods Journal.2012; 4(2):539-45.
CrossRef - Arsa, S., Theerakulkait, C. Preparation, Aroma characteristics and Volatile Compounds of Flavorings From Enzymatic Hydrolyzed RB Protein Concentrate. Science Food and Agricultural. 2018; 98(12):4479-4487.
CrossRef - Ardiansyah, Nada, A., Rahmawati, N. T. I., Oktriani, A., David, W., Astuti, R. M., Handoko, D. D., Kusbiantoro, B., Budijanto, S., Shirakawa, H. Volatile Compounds, Sensory Profile and Phenolic Compounds in Fermented Rice Bran. Plants. 2021; 10(6):1073.
CrossRef - Schmidt, C.G., Goncalves, L.M., Prietto,L. Antioxidant Activity and Enzyme Inhibition of Phenolic Acids from Fermented RB with Fungus Rhyzopusoryzae. Food Chemistry. 2014; 146 (4):371–377.
CrossRef - Martins, S., Mussatto, SI., Martínez, G.A., Montañez JS.,Cristobal, N. Bioactive Phenolic Compounds: Production and Extraction by Solid-state Fermentation. A review. Biotechnology Advances.2011; 29(11):365–373.
CrossRef - Oliveira, M. D. S., Feddern, V., Kupski, L., Cipolatti, E. P., Badiale-Furlong, E., de S.S.A. Changes in Lipid, Fatty Acids and Phospholipids Composition of Whole Rice Bran After Solid-State Fungal Fermentation. Bioresource Technology. 2011; 102(17):8335-8338.
CrossRef - Janarny, G., and Gunathilake K.D.P.P. Changes in Ricebran Bioactives, Their Bioactivity, Bioaccessibility and Bioavailability with Solid-State Fermentation by Rhizopus Oryzae. Biocatalysis and Agricultural Biotechnology. 2020; 23(2):1878-8181.
CrossRef - Ardiansyah, David, W., Handoko, D.D., Kusbiantoro, B., Budijanto, S., Shirakawa, H. Fermented Rice Bran Extract Improves Blood Pressure and Glucose in Stroke-prone Spontaneously Hypertensive Rats. Nutrition & Food Science. 2019; 49(5): 844-853.
CrossRef - Choi, S., and Lee, J. Volatile and Sensory Profiles of Different Black Rice (Oryza sativa ) Cultivars Varying in Milling Degree. Food Research International. 2021; 141:110150.
CrossRef - Meilgaard, M.C., Civille, G.V.,and Carr, Sensory Evaluation Techniques. Ed.Washington. CRC Press. CLL; 1999.
CrossRef - Kondjoyan, N., Berdagué, J.-L., & Berdague, J. L. A Compilation of Relative Retention Indices for the Analysis of Aromatic Compounds. INRA de Theix: Saint Genes Champanelle; http://books.google.co.uk/books?id=CYDKAAAACAAJ.
- Goodner, K. L. Ã. Practical Retention Index Models of OV-101, DB-1, DB-5, and DB-Wax for Flavor and Fragrance Compounds. LWT – Food Science and Technology. 2008; 41(6):951–958.
CrossRef - Pino, J. A., Marbot, R., Fuentes, V. Characterization of Volatiles in Bullock’s Heart (Annona reticulata L.) Fruit Cultivars from Cuba. Journal of Agricultural and Food Chemistry. 2003; 51(13):3836–3839.
CrossRef - Gao, C., Li, Y., Pan, Q., Fan, M., Wang, L., Qian, H. Analysis of the Key Aroma Volatile Compounds in Rice Bran during Storage and Processing via HS-SPME GC/MS. Journal of Cereal Science. 2021; 99:103178.
CrossRef - Njoroge, S. M., Ukeda, H., Sawamura, M. Changes of the Volatile Profile and Artifact Formation in Daidai (Citrus aurantium) Cold-Pressed Peel Oil on Storage. Journal of Agricultural and Food Chemistry. 2003; 51(14):4029–4035.
CrossRef - Cullere, L., Escudero, A., Cacho, J., Ferreira, V. Gas Chromatography − Olfactometry and Chemical Quantitative Study of the Aroma of Six Premium Quality Spanish Aged Red. Journal of Agricultural and Food Chemistry. 2004; 52:1653–1660.
CrossRef - Ouzouni, P. K., Koller, W. D., Badeka, A. V., Riganakos, K. A. Volatile compounds from the fruiting bodies of three Hygrophorus mushroom species from Northern Greece. International Journal of Food Science and Technology. 2009; 44(4):854–859.
CrossRef - Cho, I. H., Choi, H.-K. K., Kim, Y.-S. S., In, H. C., Choi, H.-K. K., Kim, Y.-S. S. Difference in the Volatile Composition of Pine-Mushrooms (Tricholoma matsutake Sing.) According to Their Grades. Journal of Agricultural and Food Chemistry. 2006; 54(13):4820–4825.
CrossRef - Umano, K., Hagi, Y., Nakahara, K., Shyoji, A., & Shibamoto, T. Volatile Chemicals Formed in the Headspace of a Heated D-Glucose/L-Cysteine Maillard Model System. Journal of Agricultural and Food Chemistry. 1995; 43(8):2212–2218.
CrossRef - Wedge, D. E., Klun, J. A., Tabanca, N., Demirci, B., Ozek, T., Baser, K. H. C., Liu, Z., Zhang, S., Cantrell, C. L., Zhang, J., Husnu. Bioactivity-Guided Fractionation and GC/MS Fingerprinting of Angelica Sinensis and Angelica Archangelica Root Components for Antifungal and Mosquito Deterrent Activity. Journal of Agricultural and Food Chemistry. 2009; 57(2):464–470.
CrossRef - Sanz, M. L., Sanz, J., Martı́nez-Castro, I. Presence of Some Cyclitols in Honey. Food Chemistry. 2004; 84(1):133–135.
CrossRef - Dregus, M., Engel, K. H. Volatile Constituents of Uncooked Rhubarb (Rheum rhabarbarum L.) Stalks. Journal of Agricultural and Food Chemistry. 2003; 51(22):6530–6536.
CrossRef - Wu, S., Zorn, H., Krings, U., Berger, R. G. Characteristic Volatiles from Young and Aged Fruiting Bodies of Wild Polyporus sulfureus (Bull.:Fr.) Fr. Journal of Agricultural and Food Chemistry. 2005; 53(11):4524–4528.
CrossRef - Wu, Y., Pan, Q., Qu, W., Duan, C. Comparison of Volatile Profiles of Nine Litchi (Litchi chinensis Sonn. Cultivars from Southern China. Journal of Agricultural and Food Chemistry. 2009; 57(20):9676–9681.
CrossRef - Cheng, Y., Huynk-Ba, T., Blank, I., Robert, F., Heng, Y. O. N. G. C., A, T. U. H. U., Lank, I. M. R. E. B., Obert, F. A. R. Temporal Changes in Aroma Release of Longjing Tea Infusion : Interaction of Volatile and Nonvolatile Tea Components and Formation of 2-Butyl-2-octenal upon Aging. Journal of Agricultural and Food Chemistry. 2008; 56:2160–2169.
CrossRef - Bobo-Garcia, G.; Daidov-Pardo, G.; Arroqui, C.; Virseda, P.; Marin-Arroyo, M.R.; Navarro, M. Intra-laboratory Validation of Microplate Methods for Total Phenolic Content and Antioxidant Activity on Polyphenolic Extracts, and Comparison with Conventional Spectrophotometric Methods. Journal of the Science of Food and Agriculture. 2014; 95:204–209.
CrossRef - Selli, Serkan, Rannou, C., Prost, C., Robin, J., Serot, T. Characterization of Aroma-Active Compounds in Rainbow Trout (Oncorhynchus mykiss) Eliciting an Off-Odor. Journal of Agricultural and Food Chemistry. 2006; 54(25):9496–9502.
CrossRef - Mahajan, S. S. S., Goddik, L., Qian, M. C. C. Aroma Compounds in Sweet Whey Powder. Journal of Dairy Science. 2004; 87(12):4057–4063.
CrossRef - Du, X., Finn, C. E., Qian, M. C. Bound Volatile Precursors in Genotypes in the Pedigree of ‘Marion’ Blackberry (Rubus Sp.). Journal of Agricultural and Food Chemistry. 2010; 58(6): 3694–3699.
CrossRef - org: From Flavornet. Available from: http://flavornet.org/flavornet.html. Accessed: May 15, 2021.
- Lee, S.-J., Ahn, B. Comparison of Volatile Components in Fermented Soybean Pastes using Simultaneous Distillation and Extraction (SDE) with Sensory Characterization. Food Chemistry. 2009; 114(2):600–609.
CrossRef - Chung, H. Y., Fung, P. K., Kim, J.-S. Aroma Impact Components in Commercial Plain Sufu. Journal of Agricultural and Food Chemistry. 2005; 53(5):1684–1691.
CrossRef - Lopes-Lutz, D., Alviano, D. S., Alviano, C. S., Kolodziejczyk, P. P. Screening of Chemical Composition, Antimicrobial and Antioxidant Activities of Artemisia Essential Oils. Phytochemistry. 2008; 69(8):1732–1738.
CrossRef - Beck, J. J., Merrill, G. B., Higbee, B. S., Light, D. M., Gee, W. S. In Situ Seasonal Study of the Volatile Production of Almonds (Prunus dulcis) Var. ‘Nonpareil’ and Relationship to Navel Orangeworm. Journal of Agricultural and Food Chemistry. 2009; 57(9):3749–3753.
CrossRef - Lee, K. G., Lee, S. E., Takeoka, G. R., Kim, J. H., Park, B.-S. Antioxidant Activity and Characterization of Volatile Constituents of Beechwood Creosote. Journal of the Science of Food and Agriculture. 2005; 85(9):1580–1586.
CrossRef - Xu, Y., Fan, W., Qian, M. C., Ian, M. I. C. Q. Characterization of Aroma Compounds in Apple Cider Using Solvent-Assisted Flavor Evaporation and Headspace Solid-Phase Microextraction. Journal of Agricultural and Food Chemistry. 2007; 55(8):3051–3057.
CrossRef - Chung, H. Y., Ma, W. C. J., Kim, J.-S. S., Chen, F., Hau, Y. C., Ma, W. C. J., Kim, J.-S. S., Chen, F. Odor-Active Headspace Components in Fermented Red Rice in the Presence of a Monascus Species. Journal of Agricultural and Food Chemistry. 2004; 52(21):6557–6563.
CrossRef - Chinnici, F., Guerrero, E. D., Sonni, F., Natali, N., Marin, R. N., Riponi, C. Gas Chromatography-Mass Spectrometry (GC-MS) Characterization of Volatile Compounds in Quality Vinegars with Protected European Geographical Indication. Journal of Agricultural and Food Chemistry. 2009; 57(11):4784-4792.
CrossRef - The good scents company information system: From The Good Scents Company. Available from: http://www.thegoodscentscompany.com. Accessed: June 5, 2021.
- Miller, G.H. Whisky Science: Treshold Data. Springer, Cham; 2019: p.421-530.
CrossRef - Yi, C., Zhu,H., Yang, R., Bao, J., Hailun, H., Niu, M. Links Between Microbial Compositions and Volatile Profiles of Rice Noodle Fermentation Liquid Evaluated by 16S rRNA Sequencing and GC-MS. Food Science and Technology. 2020; 118:108774.
CrossRef - Verma, D. K., Mahato, D. K., Billoria, S., Srivastav, P. P. Solid Phase Micro Extraction (SPME): A Modern Extraction Method for Rice Aroma Chemicals. in D. K. Verma, P. P. Srivastav (Eds.). Science and Technology of Aroma, Flavour and Fragrance in Rice. USA: Apple Academic Press; 2018: p.93–140.
- Park, H. J., Lee, S. M., Song, S. H., Kim, Y. S. Characterization of Volatile Components in Makgeolli, a Traditional Korean Rice Wine, with or without Pasteurization, During Storage. 2013; 18:5317-5325.
CrossRef - Jasti, T., Chintala, R. K., & Kolluru, V. C. Biological functions of volatile compounds extracted from the rice bran- A review. Biomedical and Pharmacology Journal. 2020; 13(1):213–231.
CrossRef - Sukhonthara, S., Theerakulkait, C., Miyazawa, M. Characterization of Volatile Aroma Compounds from Red and Black Rice Bran. Journal of Oleo Science. 2009; 58(3):155–161.
CrossRef - Yu A-N, Tan Z-W, Shi B-A. Influence of the pH on the Formation of Pyrazine Compounds by the Maillard Reaction of L-ascorbic Acid with Acidic, Basic and Neutral Amino Acids. Asia Pacific Journal of Chemical Engineering. 2012; 7(3):455–462.
CrossRef - Arsa, S., Puechkamutr, Y. Pyrazine Yield and Functional Properties of Rice Bran Protein Hydrolysate Formed by the Maillard Reaction at Varying pH. Journal of Food Science and Technology. 2021.
CrossRef - Buttery, R. G., Orts, W. J., Takeoka, G. R., & Nam, Y. Volatile Flavor Components of Rice Cakes. Journal of Agricultural and Food Chemistry. 1999; 47(10):4353–4356.
CrossRef - Arsa, S., Theerakulkait, C., Cadwallader, K.R. Quantitation of Three Strecker Aldehydes from Enzymatic Hydrolyzed Rice Bran Protein Concentrates as Prepared by Various Conditions. Journal of Agriculture and Food Chemistry. 2019; 67:8205-8211.
CrossRef - Lee, S.M., Hwang, Y.R., Kim, M.S., Chung, M.S., Kim, Y. S. Comparison of Volatile and Nonvolatile Compounds in Rice Fermented by Different Lactic Acid Bacteria. 2019; 24:1183.
CrossRef - Culleré, L., Escudero, A., Cacho, J., Ferreira, V. Gas Chromatography-Olfactometry and Chemical Quantitative Study of the Aroma of Six Premium Quality Spanish Aged Red Wines. Journal of Agricultural and Food Chemistry. 2004; 52(6):1653-1660.
CrossRef - Shetty, K., Paliyath, G., Pometto, A., Levin, R. E. Food Biotechnology. New York. Taylor and Francis Group; 2006.
- Bocharova, O., Reshta, S., Eshtokin, V. Toluene and Benzyl Alcohol Formation in Fruit Juice Containing Benzoates. Journal of Food Processing and Preservation. 2016; 41(4):1-8.
CrossRef - Tang, H., Ma, J.-K., Chen, L., Jiang, L.-W., Xie, J., Li, P. GC-MS Characterization of Volatile Flavor Compounds in Stinky Tofu Brine by Optimization of Headspace Solid-Phase Microextraction Condition. Molecules. 2018; 23(12):3155.
CrossRef - Lombion, S., Comte, A., Tatu, L., Brand, G., Moulin, T., Millot, d. J.-L. Pattern of Cerebral Activation during Olfactory and Trigeminal Stimulations. Human Brain Mapping. 2009; 30(3):821-828.
CrossRef - Jayasena, D.D., Ahn, D.U., Nam, K.C., Jo, C. Flavour Chemistry of Chicken Meat: a Review. Asian-Australasian Journal of Animal Science.2013; 26(5):732-742.
CrossRef - Pruckler, M., Lorenz, C., Endo, A., Kraler, M., Dürrschmid, K., Hendriks, K. Comparison of Homo-and Heterofermentative Lactic Acid Bacteria for Implementation of Fermented Wheat Bran in Bread. Food Microbiol. 2015; 49:211–219.
CrossRef - Lee, J., Xiao, L., Zhang, G., Ebeler, S.E., Mitchell, A.E. Influence of Storage on Volatile Profiles in Roasted Almonds (Prunus dulcis). Journal of Agricultural and Food Chemistry. 2014; 62:11236-11245.
CrossRef - Neugebauer, A., Granvogl, M., Schieberle, P., Characterization of the Key Odorants in High-Quality Extra Virgin Olive Oils and Certified Off-Flavor Oils to Elucidate Aroma Compounds Causing a Rancid Off-Flavor. Journal of Agricultural and Food Chemistry. 2020; 68:5927-5937.
CrossRef - Liu, S., Yang, L., Zhou, Y., He, S., Li, J., Sun, H., Yao, S., Xu, S. Effect of Mixed Moulds Starters on Volatile Flavor Compounds in Rice Wine. Food Science and Technology. 2019; 112:108215.
CrossRef - Rong, J. H., Xiong, S., Zhang, L. Z., Xie, S. L., Xiong, S. B. Analysis of Volatile
Flavor Components in Crisp Grass Carp Muscle by Electronic Nose and SPME-GC-MS.
Food Science. 2015; 36(10):124–128.
CrossRef - Miyazawa, N., Nakanishi, A., Tomita, N., Ohkubo, Y., Maeda, T., Fujita, A. Novel Key Aroma Components of Galbanum Oil. Journal of Agricultural and Food Chemistry. 2009; 57(4):1433–1439.
CrossRef - Tamura, H., Boonbumrung, S., Yoshizawa, T., Varanyanond, W. Volatile Components of the Essential Oils in the Pulp of Four Yellow Mangoes (Mangifera indica ) in Thailand. Food Science and Technology Research. 2000; 6(1):68–73.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.






