Introduction
The human gastrointestinal tract consists of a diverse collection of microbes which are considered to be non-pathogenic and essential for intestinal homeostasis.1 Human intestinal microbiome is an anaerobic environment made up of trillions of microorganisms that have co-evolved with the host with unique functional characteristics.2 Reports suggest the presence of over 104 microorganisms in the gastro-intestinal tract, which is comparatively ten times higher than the cells that exist in the human body.3 Apart from beneficial microorganisms, anaerobic or facultative pathogens may also invade, colonize the gut and cause diseases.4 Colonized microbes may assist in the digestion of food through two main catabolic pathways viz., Saccharolytic and Proteolytic. In the saccharolytic pathway, gut microbiota breaks down sugars and produce vital short chain fatty acids (SCFAs). On the other hand, the proteolytic pathway is characterized by protein fermentation leading to the synthesis of substances such as ammonia, amines, thiols, phenols, and indoles apart from SCFAs. Accumulation of certain microbial metabolic by-products disrupts the intestinal tolerance, triggering severe health problems in the human system.5 Microbial metabolites regulate and activate host immunity to defend the body from pathogens. Antimicrobial peptides (AMPs) which are encoded from the host’s genes are a component of the host’s innate immune response against pathogenic infections. The expression of AMPs surges when bacteria colonize the gut in order to control the spreading of pathogenic bacteria. Maintaining a homeostatic state in the gut microbiome is a crucial factor in gut health as its imbalance may lead to many metabolic diseases such as metabolic endotoxemia, type 2 diabetes, inflammatory bowel disease or obesity.6 The equilibrium between the activation of the innate immune system and the microbial factors that regulate multifaceted functions is restored by gut microbes. Disappearance of microbial diversity results in the suppression of the innate immune system and higher incidence of non- communicable diseases.7,8,9 In a diseased condition, intestinal barrier turns leaky allowing the gut microbes to navigate into distant organs causing inflammation leading to conditions such as alcoholic fatty liver disease, non-alcoholic fatty liver disease, or cardiovascular diseases.10,11 Colonization of the gut starts when a fetus is in the lower uterus but an infant’s gut microbiota is established only after birth through two transitions in infancy. The first transition occurs after birth, during the period of lactation whereby dominance of the gut microbiota by Bifidobacterium is reported. The second transition occurs during the weaning period, where the introduction of solid foods contribute in the establishment of an adult-type complex microbiome dominated by the Bacteroidetes and Firmicutes phyla.12 Bacteroidetes and Firmicutes represent more than 90% of the adult gut microbiome with the ratio between the two phyla varying from person to person depending on the dietary, environmental and lifestyle factors.13,14 Early establishment of the gut microbiota is affected by various factors such as the mode of delivery (C-section or vaginal delivery), mode of milk intake (breastfeeding or formula feeding), and administration of antibiotics. Metagenomic analysis of fecal samples from a group of Swedish infants and their mothers reported that the mode of birth was one of the most important factors in shaping the gut microbiota of infants. Vaginally delivered newborns shared a 72% match in microbial diversity between the early colonizers of the newborn’s gut and the species found in the stool of their own mothers suggesting a possibility for an effective vertical mother-neonate microbial transfer. However, newborns delivered by C-section had only 41% of species match with their mother, indicating that mother-infant transmission was compromised during a C-section birth.15 The gut microbiota exhibit a vital role in maintaining the overall functioning of the newborn by contributing to digestion (metabolizing polysaccharides which are barely digestible), vitamin synthesis, barrier development against potentially pathogenic bacteria, detoxifying bacteria by aiding in the development of the host immune system, and the regulation of mood and behaviour by influencing the development of the nervous system.16,17 Maintenance of intestinal homeostasis by gut microbiome is of utmost importance in order to prevent gastro-intestinal diseases and in regulating disease conditions such as obesity and depression.178,181 Many approaches that include administration of probiotics, prebiotics, Fecal Microbial Transplant (FMT), antibiotic administration, supplementation of engineered bacteria capable of secreting metabolites have been studied for restoring the homeostatic condition. This review discusses the state of eubiosis, dysbiosis, their role in maintaining gut health and prevention of diseases.
Intestinal Epithelial Barrier
The intestinal barrier is made of a single layer of internal epithelial lining that maintains the integrity of the gut. The epithelial cells collectively form a polarized layer to establish a tight barrier with the help of intercellular tight junctions, adherens junctions and desmosomes effectively separating the luminal bacteria from the immune cells that are present in the intestinal mucosa. Lamina propria, a thin layer of connective tissue present below the epithelial layer contribute for a healthy communication between the gut microbiome and the internal mucosal immune cells (dendritic cells, T cells, B cells and macrophages) through the pattern recognition receptors (PRRs). Receptors present on the intestinal epithelial cells (IECs) are either Toll like receptors (TLRs) or nuclear oligomerization domain-like receptors (NLRs) that are responsible in recognizing bacterial components such as microbe associated molecular patterns (MAMPs) or pathogen associated molecular patterns (PAMPs) namely the microbial liposaccharides or peptidoglycans.18,19 Upon the interaction of TLRs with a PAMP, the innate immune system is activated thereby triggering signaling pathways leading to the production of inflammatory mediators such as interleukin-6 or tumor necrosis factor.20 Signaling of TLR also induce the synthesis of antimicrobial peptides and interferons that influence the responses of adaptive immunity. The interaction of TLRs with MAMP helps in promoting gut homeostasis by maintaining the integrity of the intestinal barrier in turn reducing the host’s inflammatory responses. The permeability of IECs ensures selective absorption of essential ions and allows nutrient passage through the barrier. Transportation of vital molecules occurs through three main pathways viz., trans-cellular pathway, carrier mediated pathway and paracellular pathway. The tight junction protein seals the gaps between the intestinal cells and selectively allows the transport of solute molecules and essential ions while restricting the movement of hydrophilic molecules and non-essential microbial-derived peptides. Alterations in the structure of the tight junctions lead to disruption of the epithelial integrity, allowing random to and fro movement of bacterial toxins affecting the gut permeability, epithelial physiology and immune homeostasis.21 IECs continuously interact with commensal bacteria in the GI tract and provide instruction to mucosal immune system to activate an immunological balance between active and quiescent state that ultimately contribute in intestinal homeostasis.179
Eubiosis and Dysbiosis
Establishment of the microbial pattern during host development is crucial as the gut microbiome plays a vital role in maintaining gut homeostasis. The gut microbiota established during infancy provides a qualitative and quantitative balance of various microorganisms that are required for metabolite synthesis and normal functioning of the host.24,25,26 Metabolic byproduct of gut bacteria regulate a diverse intestinal function such as digestion, permeability secretion, visceral sensing, motility, mucosal immunity, barrier effectiveness, etc.17 This state of a balanced gut microbiome is called as Eubiosis. Eubiotic state is generally characterized by the presence of beneficial species belonging mainly to Firmicutes, Bacteroidetes, Actinobacteria and Proteobacteria phyla. Firmicutes and Bacteroidetes are the main dominant phyla, while the others are sub dominant. Firmicutes include Lactobacillus, Mycoplasma, Streptococcus and Clostridium.22 Firmicutes are gram-positive bacteria composed mainly of Bacilli (facultative aerobes) and Clostridia (anaerobes) classes which are characterized with a low GC content and possess the ability to produce endospores that enable them to survive in adverse aerobic conditions.23 Bacteroidetes are anaerobic, gram negative bacteria belonging to about 20 genera and species that can tolerate the presence of oxygen but do not utilize it for its growth.22 The host and the microbiome share a mutually beneficial relationship wherein the host provides shelter and nutrients to the microbiota and in turn the microbes and their secretory metabolites protect the host from pathogens, minimise immune disorders by immunomodulation and improve various body functions. Association between the host and its microbiota is referred to as symbiosis. The microbiome and the host communicate through a crosstalk with the intestinal niche harboring its own microbial community.30 Gnotobiotic mice colonized with a single bacterial strain, Bacteroides thetaiotaomicron VPI-5482 exhibited relatively lesser weight gain and fat deposition compared to the germ free mice suggesting that even a single microbial species can influence the host metabolism.18,28 Upon addition of one more microbe (Methanobrevibacter smithii), the host showed an enhancement in the fat storage indicating the existence of microbial interaction between species in the gut. The metabolic products obtained from Bacteroides thetaiotaomicron’s fermentation were utilized by Methanobrevibacter smithii to produce methane that made the fermentation reaction thermodynamically favourable.29 Quantitative analysis of gene expression revealed the interactions between the host and the gut microbiome or between the various microbes colonising the gut. The complete genome analysis of Bacteroides thetaiotaomicron and Methanobrevibacter smithii afford evidences regarding the microbial adaptation in the human gut. However, the pathways of such interactions are not yet completely understood.18 Perturbations to the structure of the complex commensal microbial communities allow microbes to move to the peritoneal cavity and enter blood circulation causing few microbes to become pathogenic. Ali Metchnikoff suggested that most diseases begin in the digestive tract when the “good” bacteria are no longer able to control the “bad” ones. This condition where the bacteria no longer live together in mutual harmony is known as Dysbiosis, a widely used term that was originally defined as ‘difficult living’ or ‘life in distress’.17,31 The eubiotic and dysbiotic states are clearly depicted in Fig.1.
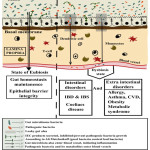 |
Figure 1: Differences in Eubiotic and Dysbiotic States of the Gut. |
Gut Microbiota and Eubiosis
Several factors viz., host-dependent factors (e.g., genetic background, age, sex, immune system and gut motility), treatment (e.g., antibiotics) and diet (e.g., non-digestible carbohydrates, fat) influence the microbial richness in the gut.32 Most of the dietary microbiota modulations are affected by the consumption of a wide range of fibres, administration of probiotics (live beneficial bacteria) and prebiotics (food which enhance the growth of probiotics).33 The microbes present in the gut are capable of digesting the indigestible dietary fibres like polysaccharides, oligosaccharides, proteins, peptides and glycoprotein by converting them into products such as short chain fatty acids (SCFAs) namely acetate, propionate, butyrate and other signaling molecules which can be easily absorbed by the host.34 The short chain fatty acids maintain the integrity of the intestinal epithelial barrier, reduce the translocation of bacteria, increase the expressions of hunger-suppressing hormones and also reduces inflammation. A decreased expression of the short chain fatty acids is associated with the alteration of the gut microbiota indicating an imbalanced state.35 Glycoside hydrolases coded by the gut microbiota genome are employed in breaking down plant polysaccharides rich in xylan, pectin and arabinose containing carbohydrate structures into an usable energy sources.36 Glycans present in the food cannot be degraded by the enzymes coded in the human genome. The gut microbiota ferment glycans into short chain fatty acids and obtain energy.18 Synthesised SCFAs serve as nutrients for colonocytes and the other gut epithelial cells. Bacteroides thetaiotaomicron, a common bacterium in the human gut flora, is known to produce high levels of digestive enzymes required for the breakdown of carbohydrates.24 The cross talk between the microbes or between the microbes and the host is maintained or can be enhanced by the various products secreted by the intestinal microbes.33 Indole is identified to be a major signaling molecule involved in the crosstalk between the microbiota and the gut wall which works by increasing epithelial cell tight junctions.22 Indole also helps in regulating the secretion of GLP-1(Glucagon like peptide) from the enteroendocrine cells (specialized cells found within the gastrointestinal tract, stomach and pancreas) that helps in maintaining the glucose levels.37 Genome of Proteobacteria, one of the few identified in the human gut contains high levels of antibiotic resistance genes against bacitracin, tetracycline and macrolides. Resistance against such antibiotics increases with age.38,39 Bifidobacterium, a gram positive bacterium is known to produce vitamin K, B12, Biotin, Folate, Thiamine, which help in preventing infections by pathogenic bacteria.40,41 Reduction of intestinal endotoxin levels and improved mucosal barrier in rodents are noted in groups supplemented with Bifidobacterium spp.42,43 A number of studies have shown that the intestinal bacteria and their metabolites play a key role in the activation of the immune system, proliferation and differentiation of the T cells. Anti-inflammatory compounds produced by the gut microbes also help in preventing inflammation due to the production of LPS, a component of cell walls of gram-negative bacteria.44
Factors Causing Dysbiosis
Lifestyle, antibiotic treatments, psychological and physical stress, pathogens, consumption of alcohol, and smoking are some of the factors that can disturb the balance of intestinal microbiota.45 Strong correlation between dysbiosis of the gut microbiota and disease etiology is observed in several metabolic diseases such as obesity where an increase in the number of Firmicutes and a decrease in the number of Bacteroidetes have been reported in human and animal models.46,47 Lifestyle factors that includes geographical location, sleep pattern, personal fitness, and hygiene practices influence the rate of eubiosis and dysbiosis.33,48 An alteration in the functioning of the gut barrier leads to a leaky gut that changes the gut microbiota composition reported to be associated with diet-induced obesity and genetic obesity.49 Antibiotic treatments disrupt the microbial assemblages and affect the functionality of gut microbes resulting in antibiotic associated diarrhea. Duration of the treatment, concentration of the antibiotic and the spectrum of the antibiotic are probable factors that can have an impact on the integrity of the intestinal barrier.50 Inflammatory response produced by the host cause a significant decrease in the beneficial bacteria population that leads to pathogen colonization in intestinal niche. Furthermore, substances such as nitrate, S-oxides, and N-oxides generated as by-products of inflammation, potentially colonize pathogenic Escherichia coli in mice model.45 Imbalances in the gut microbiome are known to influence the brain, including the mood of an individual.51 Studies show that individuals exposed to psychological stress have a significant decrease in the production of mucin and mucopolysaccharide layer lining in the mucosal surface.52 It is known that the mucopolysaccharide layer and mucous inhibits the growth of the pathogenic organisms thereby acting as a defensive barrier. Stress can also be one of the reasons for an individual to suffer from inflammatory bowel syndrome.33 Stress acts as a factor that causes gut impermeability allowing bacteria and bacterial antigens to cross the epithelial barrier thus altering the composition of the microbiome.182 Alcohol consumption is a another major factor that can have an impact on the gut microbiota. Ethanol can also affect intestinal cells by targeting many pre and post-transcriptional regulators, that mainly includes circadian clock genes and microRNA (short ribonucleic acid molecules of 22 nucleotides attached to complementary sequences of miRNA causing translational repression).53 Ethanol is metabolized by the microbes present in the intestine to produce acetaldehyde that causes disruption of the tight junction integrity resulting in significant decrease in the ‘good’ bacteria.53 According to a study by Chen (2015) in humans and mice, alcohol induce intestinal dysbiosis resulting in limiting the production of LCFA (long chain fatty acid). However, exogenous administration of fatty acids stabilized the intestinal barrier resulting in restoration of eubiosis condition.180 The changes induced due to smoking can lead to an increase in the count of Bacteroides-Prevotella in individuals that can lead to the increased risk of Crohn’s disease. Fermentation of high protein rich food by colonic microbes results in the synthesis of potentially harmful by-products that affect the morphology and functionality of the mucosal cells. Emergence of indole, ammonia, phenols, and amines in relation to high protein diet consumption also affects the intestinal homeostasis.50
Consequences of Dysbiosis
Dysbiosis is a major factor that is associated with the pathogenesis of many intestinal and extra-intestinal disorders. Intestinal disorders such as inflammatory bowel disease, irritable bowel syndrome (IBS), celiac disease, and extra-intestinal disorders such as allergy, asthma, metabolic syndrome, cardiovascular disease, and obesity are associated with dysbiosis. The malfunctioning of the microbiota turns the intestinal barrier leaky thereby allowing the gut microbes to progress into other organs. The bacterial composition varies from one location to another in the gastrointestinal tract with the higher microbial diversity reported in the distal large intestine and in fecal microbiota.54 Primarily, the changes caused as a result of dysbiosis cause an imbalance to the homeostatic state leading to an overall loss of the diverse microbial community present in the intestinal region. The alteration in the intestinal region allows translocation of pathogenic organisms to the gut. The decrease in the bacterial diversity favors the recognition of the pathogenic bacteria by the Pattern Recognition Receptor (PRR) system. Host response to such infectious agents might further affect the host metabolism and energy homeostasis. Re-localization of Nuclear Factor Kappa B (NF-kB) in the nucleus and increase in the cellular oxidative stress are responses related to disruption of host metabolism. Diseases including type 2 diabetes, atherosclerosis, Crohn’s disease are linked with low grade chronic inflammation.55 Loss of bacterial diversity causes a decrease in the load of beneficial bacteria and an increase in the pathogenic strains. Resulting imbalance in the microbial consortia has been linked with various diseases such as inflammatory bowel disease, crohn’s disease, obesity, metabolic syndrome, fatty liver disease, parkinson’s diseases etc. Fig. 2 summarizes the diseases associated with a leaky gut.
Inflammatory Bowel Disease
Inflammatory Bowel Disease (IBD) is a chronic condition wherein patients suffer from severe inflammation in the gastrointestinal tract.56 The disease has two clinical forms; Ulcerative colitis (UC) which is restricted to the rectum and the colon excluding the small intestine and Crohn’s disease (CD) wherein any part of the intestine can have inflammation including the small intestine and large intestine.21 Expression of abnormal Toll like receptors (TLR4) of the intestine is known to be associated with inflammatory bowel disease. Increased expression of TLRs, pro-inflammatory cytokines viz., tumor necrosis factor (TNF)-a, interleukin (IL)- 1b, IL-6, and IL-8 activates the intestinal inflammatory responses.56 Another cause of IBD is the loss of intestinal integrity caused due to the defect resulting from mucosal tolerance. Due to this imbalance, microbes and their metabolites continuously move in and out through the leaky gut causing uncontrollable inflammatory signal cascades.21 Extensive research is however required to prove that dysbiosis is one of the major causes of IBD. Pathogenesis is still not clear as to whether dysbiosis seen in IBD patients is an effect of the disease or a risk factor. It is observed that IBD patients have a decreased number of butyrate producing bacteria (Faecalibacterium prausnitzii) and also a decline in the diversity of Firmicutes.57 An increase in the number of sulphate reducing bacteria is also observed in patients with IBD. These bacteria metabolize sulphate to hydrogen sulphide which can block the utilization of butyrate by the intestinal epithelial cells, and decreases the expression of the tight junction proteins which in turn increases the permeability of the intestinal barrier resulting in the translocation of bacteria.57 The increase in sulphate reducing bacteria also inhibits phagocytosis and killing of bacteria. It has also been reported that healthy individuals have many obligate anaerobic populations but the IBD patients have a decreased population of obligate anaerobes and an increased population of facultative anaerobes. This change from obligate to facultative anaerobes in the intestine led to the hypothesis that oxygen may be a major factor that causes dysbiosis in IBD patients. An oxygen hypothesis was made which suggested that due to an increase in the reactive oxygen species, the anaerobic environment of the gut is disrupted which favors the facultative anaerobes causing dysbiosis.58 Metagenomic analysis of the gut microbiota in IBD patients exhibited a decreased expression of genes responsible for carbohydrate and amino acid metabolism and an increase in the oxidative stress pathway in IBD patients.59 Furthermore, a bulk of the studies focus on microbiome census and obliterate the role of viruses and fungi that is even less understood.185 There hasn’t been any specific pathogen fulfilling the Koch’s postulates identified in patients of IBD. Hence, dysbiosis leading to functional changes in the gut microbiota may be responsible for the pathophysiology of IBD. Research for improvement of epithelial integrity and correction of abnormal micro-host interaction are currently in progress to supplement anti-inflammatory and immune therapies for IBD patients. Moreover, interventions in bacterial engineering, next generation probiotics, microbe-specific bactericidal antibiotics and fecal microbiota transplantation therapy have promising results for IBD treatment.
Crohn’s Disease (CD)
Dysbiosis in patients of Crohn’s disease has been identified using both culture-dependent and culture-independent techniques. Studies have reported that the common sites of inflammation seen in CD patients are present in the human gut. The inflammation could be the result of a poor diet or long antibiotic courses in childhood.60 Biopsies of the colonic or small intestinal tissue of CD patients provide evidence that they have a reduced ability of killing pathogenic microbes compared to the control.61 The pathogenesis of bacteria in CD is well established in patients and dysbiosis is mainly associated with a lower number of Firmicutes and Clostridium cluster IV along with a higher amount of Bacteroidetes. Lower levels of Faecalibacterium prausnitzii has been commonly determined in fecal samples and biopsies in most of the CD patients. Mycobacterium avium subspecies paratuberculosis (MAP) and Escherichia coli strain LF82 are also reported to be more in number in CD patients.62,63,64 In the cohort studies, the predominant faecal microbiota which is believed to cause dysbiosis in CD was Dialister invisus, Faecalibacterium prausnitzii, Bifidobacterium adolescentis and Ruminococcus gnavus. A multifaceted interplay between host genetics and microbial assemblage is identified in the pathogenesis of CD. Genetic variants exhibit a varied inflammatory response related to membrane permeability, microbial sensing, and impediment in autophagy pathways causing increased inflammation in CD.65 Single nucleotide polymorphism in Mucin-19 has been associated with intestinal inflammation in CD, where intestinal mucus layer face structural anomalies. Typical dysbiosis signature of bacterial consortia viz., Faecalibacterium prausnitzii, Ruminococcus spp., Dialister invisus, Bifidobacterium spp., Clostridium spp. associated with CD were reported in fecal samples.66 Varied effects are observed due to altered luminal defensins and modified permeability in the brush border of the intestinal lining. Studies also show that even the unaffected relatives of patients had an entirely different microbiota compared to healthy controls. An analysis of biopsies in CD patients revealed that the alpha diversity of the tissue-associated intestinal microbiome remained lower which might be associated with continuous diarrhea that signifies marked alterations in intestinal microbiome of patients.186 A detailed study in CD-associated dysbiosis will help in understanding and untangling the role of bacteria involved in causing CD.
Obesity, Metabolic Endotoxemia and Type 2 Diabetes
Obesity is characterized by a cluster of many metabolic disorders that may be caused because of factors such as high fat diet, reduction in physical activities etc.183 Apart from these factors, a large shift in the abundance of certain microbes belonging to the intestine also play a very important role in the development of metabolic diseases associated with obesity.1 In a study on obese mice, a significant decrease of Bacteroidetes and an increase of Firmicutes were observed in the cecal microbiota.46 The development of obesity in mice affects the relative abundance of major gut bacteria derived from maternal inoculum. The mechanism responsible for the direct microbial changes in the gut is still undefined. It has also been reported that obesity due to the alteration of the diet leads to a decrease in Bifidobacterium spp. and a Bacteroidetes related bacteria, Eubacterium rectale-Blautia Coccoides.67 One of the key mechanisms known for the development of obesity is the inhibition of fasting-induced adipose factor (FIAF). FIAF inhibits lipoprotein lipase, promoting the release of triglycerides such as very low-density lipoprotein (VLDL) and chylomicrons. Limiting FIAF regulate obesity related disorders by controlling the release of triglycerides. Levels of short chain fatty acids in obese individuals are significantly lower than lean individuals. SCFAs inhibit the accumulation of fat in the adipose tissue by reducing energy intake and by increasing energy expenditure. Hence, decrease in SCFA leads to accumulation of fat. SCFAs, primarily propionate and butyrate reduce food intake by activating gut hormones through the free fatty acid receptor 2 and 3 (FFAR2; FFAR3). However, the same study reported the action of SCFAs independent of FFAR activation in food intake and body weight gain subjects.68 Propionate supplementation activated gut hormones, Peptide tyrosine kinase and glucagon like peptide (GLP-1) which in turn exhibited a profound effect in reducing food intake and reduced weight gain by limiting adipose tissue accumulation.69 Obesity and diabetes are associated with higher gut permeability leading to metabolic endotoxemia, initiation of low grade inflammation and insulin resistance in the liver, muscles and adipose tissue.49 Metabolic endotoxemia is characterized by an increase in the levels of LPS in the bloodstream. Lipopolysaccharide (LPS), a structural component of the cell wall of gram-negative bacteria contain Lipid A which causes an initiation of a signaling cascade to activate the pro-inflammatory pathways, increasing the oxidative stress as it binds to Toll Like Receptor 4.10,70 Many studies have been performed on CD14/TLR4 receptor knockout mice to observe the contribution of gut microbiota in regulating the pathophysiology of diseases such as obesity, type 2 diabetes, and metabolic endotoxemia as it is considered that high fat content in the diet causes an increase in the LPS levels. The LPS binds to the CD14/TLR4 receptors and cause secretion of pro-inflammatory cytokines which are closely associated with these diseases. Experimental mice that do not have CD14/TLR4 receptor were found to be resistant to high fat diet induced metabolic disorders. Simultaneously, studies carried out with various other combinations of high fat diet and LPS in CD14/TLR4 receptor knockout mice exhibited resistance to high fat diet induced metabolic disorders at lower LPS concentration. The conclusion drawn from the various studies reflected that high fat feeding induced low-grade inflammation, which was originated from intestinal absorption of LPS. 1,71,72 Various reports of higher levels of plasma LPS were found to be associated with metabolic endotoxemia and low-grade inflammation.73,74 Many studies have reported that an imbalance of specific microbes of the intestine is related to the increase in LPS levels.49 Mice that were fed with high fat diet had a significant decrease in the levels of Bifidobacterium spp. and Eubacterium rectale / Clostridium Coccoides compared to the ones that were fed with a standard high carbohydrate diet suggesting that high fat diet increased the proportion of LPS containing microbiota in the gut which leads to signaling of proinflammatory cytokines thereby leading to high fat diet-induced metabolic diseases. 67 Few experimental studies suggest that the development of low-grade inflammation is due to metabolic endotoxemia which is involved with insulin resistance and type 2 diabetes. Data also suggests that increased plasma LPS has a negative effect on glucose metabolism. Alteration of the microbiota can cause endotoxemia leading to insulin resistance. A decrease in the number of butyrate-producing bacteria and an increase in the number of Lactobacillus spp. is associated with type 2 diabetes.5 Butyrate is a short chain fatty acid which is an energy substrate for the epithelial cells of the gut.75,76 The epithelial cells will increase in number due to the increased production of butyrate thereby involved in the secretion of Glucagon-like peptide 1 (GLP-1). GLP-1 plays an important role in maintaining levels of glucose in the body by stimulating the secretion of insulin to convert glucose to glycogen. Impaired GLP-1 secretion can lead to an imbalance in the glucose levels causing diabetes. Metabolic syndrome is an amalgam of heart diseases, stroke and type 2 diabetes. Problems such as increased blood pressure, excess body fat around the waist, abnormal cholesterol or triglyceride levels and high blood sugar levels are associated with metabolic disorders.77 Increase in metabolic syndrome is attributed to increased food consumption and change in lifestyle activity.78 The gut microbiota regulates the host metabolism by regulating the composition of bile acid pools. The changes in the bile acid pool activates ligand binding to Farnesoid X Receptor leading to the transcription of genes that affects the glucose and lipid metabolism.79
Fatty Liver Disease (Non-Alcoholic and Alcoholic)
Non-Alcoholic Fatty Liver Disease (NAFLD) has become the most common chronic liver disease in the world184. Individuals who are usually obese and insulin resistant are in high-risk groups for NAFLD.16 Gut microbes connect to the liver through the portal vein and confer positive and negative effects. Pathways involving the gut–liver axis are influenced by the microbiome and their homeostatic state. In eubiotic state, butyrate secreting Faecalibacterium prausnitzii maintain the membrane integrity and tight junction formation. In dysbiotic state, damage causing bacteria such as Bilophila wadsworthia, forms a secondary layer above beneficial microbes and hinders metabolite synthesis. Dysbiosis related symptoms include systemic endotoxin levels, endogenous ethanol quantity and augmented intestinal permeability. In addition, the existence of harmful Klebsiella pneumonia in the gut synthesises endogenous alcohol which activates the reactive oxygen species chain in the liver, causing progression of NAFLD. Mechanisms that lead to endotoxemia and inflammation include diet induced intestinal bacterial overgrowth, leaky gut and the movement of endotoxins to distant parts that are similarly associated with NAFLD. Enhanced permeability and accumulation of bacterial metabolites in the liver is observed due to damages in the intestinal barrier by the triggering of TLR 4, TLR 5 and TLR 9 receptors causing abnormal retention of fats and hindering the metabolism of lipids.80 One of the mechanisms that relate the microbiota to NAFLD is the bacterial metabolism of choline. The deficiency of choline causes accumulation of triglycerides in the liver and further progressing to steatohepatitis, fibrosis, cirrhosis and liver cancer.81 Metabolism of choline also has an influence on obesity and insulin resistance.82 Mice susceptible to NAFLD, fed with a high fat diet had reduced expression of choline due its conversion into methylamines by microbiota, resulting in the inability to synthesize phosphatidylcholine causing subsequent accumulation of triglycerides in the liver.82 Accumulation of triglycerides further causes complications in the liver making individuals susceptible to NAFLD. Disruption of the gut barrier eventually leads to translocation of the gut microbes and their metabolites causing the activation of immune system leading to liver inflammation and injury.83 With respect to alcoholic fatty liver disease, consumption of alcohol leads to alterations of gut microbiota further causing a leaky gut and thereby allowing the microbes and their metabolites to translocate to the liver. Studies carried out in mice fed with ethanol showed a decrease in the population of phylum Firmicutes and genus Lactobacillus spp. Incidence of higher levels of few species belonging to Verrucomicrobia (genus Akkermansia muciniphila), Actinobacteria (genus Corynebacterium spp.) and Proteobacteria (genus Alcaligenes spp.) were observed.84 In the Tsukamoto-French model of alcoholic liver disease, a group of mice were given specific liquid diets and intra-gastric infusions of ethanol. On the other hand the control group was given the same liquid diet but was fed with dextrose instead of alcohol. Quantitative changes through real-time PCR and qualitative changes in the reduction of probiotic bacteria such as Lactobacillus, Pediococcus, Leuconostoc and Lactococcus in the gut of the experimental mice was observed.85 Recent lifestyle and dietary trends have added on the prevalence of non-alcoholic fatty liver disease / steatohepatitis (NAFLD/NASH) around the globe. Even-though non-invasive biomarker and imaging modalities are employed in the detection of individuals with high risk of rapid disease progression and low fibrosis stages, early detection will help to deliver efficient single or combination treatment that will have maximum benefits and fewer adverse events.187
Parkinson’s Disease
Parkinson’s disease (PD) is a multi-centric neurodegenerative disorder characterized by the accumulation and aggregation of alpha synuclein in the substantia nigra of the central nervous system and in other neural structures.86,87 Studies have found that gut microbiota can influence the central nervous system (CNS) and enteric nervous system and possess the potential to alter the CNS through the microbiota-gut-brain axis. Modulatory regulation involves immunological, neuro endocrine and direct neural mechanisms.88 Gastrointestinal (GI) dysfunction has been reported in PD patients which are considered to be the initial symptoms of PD, prior to the characteristic motor symptoms.89 Various dysbiosis symptoms have also been reported to be related to the PD associated GI dysfunction.90,91 Gut dysbiosis, small intestinal bacterial overgrowth or increased gut permeability will cause excessive stimulation of the innate immune system which may eventually produce systemic and/or CNS inflammation.92 Germ free animal with low or depleting microbiota showed compromise in microglia cell shape and maturation, leading to improper immune responses. Restoring the microbiota second time exhibited partial recovery of microglia cells.93 Deficiency of short chain fatty acids such as butyrate, propionate, acetate have also been directly linked to the deficiency in the inhibitory neurotransmitter GABA (gamma-aminobutyric acid).94 High numbers of Helicobacter pylori infections were reported in patients for a decade.95 Similarly a 50% increase of Helicobacter pylori related dysbiosis cases have been reported among Indian PD patients.96 Pathogenesis of PD related gut dysbiosis showed alterations in gut microbiome. Higher count of opportunistic pathogens such as Proteus and Enterobacter spp. and a lower count of Peptostreptococcus, Lactobacillus and Butyricicoccus spp. were noted.97 Manipulation of fecal microbial community, better and complete bowel movement was observed in PD subjects supplemented with fermented milk containing 250 * 109 of multiple probiotics comprising Streptococcus salivarius, Lactobacillus rhamnosus, Lactobacillus delbrueckii, Lactobacillus paracasei, Bifidobacterium etc., and prebiotic fibers.98 Commensal gut microbiota maintains stable neural and emotional indices exerting better cognitive and emotional improvement.99 Psychobiotic effects of Lactobacillus helveticus and Bifidobacterium longum conferring better psychological balance and mental health has been reported in rat and human volunteers.100 The imbalance of specific bacteria and its consequences pertaining to each of the diseases discussed in the previous section is depicted in Table 1.
Table 1: Microbial Imbalance in Gut Ecosystem and their Consequence.
| Sl. No | Disease | Lower Microbial Count | Higher Microbial Count | Consequence of Microbial imbalance |
| 1. | Inflammatory bowel disease | Butyrate producing bacteria (Faecalibacterium prausnitzii) | Sulphate producing bacteria | Increased expression of TLRs pro-inflammatory cytokine secretions and activation of acquired immune responses increases intestinal inflammatory responses.Hydrogen sulphide, toxic molecule produced when sulphate reducing bacteria metabolizes sulphate thereby causing decrease in the expression of tight junction proteins which eventually causes the intestine to become impermeable. |
| 2. | On Obesity | Bacteroidetes | Firmicutes | Inhibition of fasting-induced adipose factor and therefore it cannot inhibit lipoprotein lipase (LPL) protein leading to obesity.SCFAs inhibit accumulation of fat in the adipose tissue but a decrease in their levels causes obesity. |
| 3. | Type 2 diabetes | Butyrate producing bacteria | Lactobacillus spp. | Impaired GLP secretion causes imbalance in the glucose levels which leads to diabetes.Increased plasma LPS levels have a negative impact on glucose metabolism and causes alteration of the microbiota leading to insulin resistance. |
| 4. | Alcoholic fatty liver disease | Lactobacillus spp. | Enterococcus spp. | Excessive alcohol consumption causes alteration in the balance of the gut microbiota thereby causing a leaky gut and translocation of unwanted bacteria and metabolites to distant organs. |
| 5. | Crohn’s Disease | Firmicutes | Bacteroidetes | Colonic or small intestinal tissue has reduced ability of killing the microbes that are pathogenic compared to the normal tissues.MAP (chronic enteric pathogen) acts as a primary cause of inflammation of the intestine in a range of different species |
| 6. | Non- alcoholic Fatty Liver Disease | Erysipelotrichia | Gammaproteobacterial | Deficiency of choline causes accumulation of triglycerides in the liver which sometimes progresses from steatohepatitis to liver cancer.Impermeable gut leads to translocation of bacterial species and their metabolites thereby causing the activation of the immune system and leading to liver inflammation and injury. |
| 7. | Atheroscleros | Bacteroides | Ruminococcus | Increased levels of plasma LPS and its binding to TLR4 leads to the activation of pathways which causes an increased production of pro-inflammatory cytokines like IL-6, IL-1, IL-27 and tumor necrosis factor-alpha leading to increased risk of developing CVD. |
| 8. | Colorectal cancer | Butyrate producing Lachnospiraceae | Bacteroides fragilis, Enterococcus, Klebsiella, Streptococcus | Intestinal permeability induces the immune system to secrete cytokines which lead to a cascade of reactions ultimately causing inflammation. This local inflammation will lead to progression of tumor through pro-tumorigenic cytokines and chemokines that act as growth factors and promote angiogenesis. |
| 9. | Celiac disease | Bifidobacterium | Bacteroides,Prevotella,Clostridium histolyticum, Eubacterium rectale, Clostridium coccoides and Atopobium sp. |
Gluten containing food products are not completely digested. They are potential immunogenic gluten derived peptides which trigger immune response associated with celiac disease. |
| 10. | Ulcerative colitis | Firmicutes | Bacteroidetes and Facultative anaerobes | Dysbiosis causes a rapid increase in harmful bacteria in the intestine and aggravates intestinal inflammatory response.Enterotoxin increases the permeability of the intestinal mucosa and the production of immunosuppressive proteins resulting in immune dysfunction. Overgrowth of bacteria affects energy metabolism causing inflammation and damage to the intestinal mucosa. |
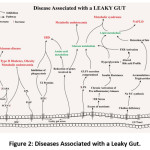 |
Figure 2: Diseases Associated with a Leaky Gut. |
Preventive and Therapeutic Approaches
Many approaches are proposed to restore the normal intestinal homeostasis in the gut. Assessment of gut microbial cascade is an essential factor towards designing a prophylactic treatment to treat dysbiosis.101 Employing probiotics, prebiotics, synbiotics, amino acids and carbohydrate as dietary interventions, fecal microbial transplantation, use of engineered bacteria, antibiotic administration, bacterial consortium transplantation and phage therapy are gaining importance to modulate the intestinal dysbiosis.101
Use of Probiotics, Prebiotics and Synbiotics
According to the World Health Organization, probiotics are “live microorganisms which when administered in adequate amounts confer a health benefit to the host”.49 The gastrointestinal tract is composed of beneficial microorganisms belonging to genus Lactobacilli and Bifidobacteria and other organisms such as Saccharomyces boulardii, Escherichia coli Nissle 1917, Streptococcus thermophilus, Faecalibacterium parausnitzi and Bacillus polyfermenticus.4 Lactobacillus reuteri is known to promote the release of incretin (GLP-1) which will in turn cause an increase in the secretion of insulin in obese glucose-tolerant subjects.102 Probiotics adhere to the intestinal walls and physically block the adhesion of pathogenic bacterial species onto the epithelial cells. The microorganisms also induce anti-inflammatory Tregs cells and lower the pH which helps in preventing the pathogenic bacteria from adhering to the inner lining of the gut. Probiotics help in increasing the mucus layer, enhancing the number of the epithelial cells and in the elongation of microvilli thereby increasing the surface area.103,104 Administration of certain Lactobacillus sp. has been observed to decrease the production of toxins such as dimethylamine and nitrosodimethylamine in patients with Chronic Kidney Disease (CKD). The gut microbes are also involved in the secretion of mucin and in the formation of muco-polysaccharides, which protects the intestinal cells. Administration of Bifidobacterium infantis in obese mice showed a decrease in the production of pro-inflammatory cytokines and white adipose tissue.67,68 Microbes such as Lactobacillus rhamnosus , Lactobacillus gasseri BNR17 and Bifidobacterium lactis isolated from human breast milk were reported to be beneficial in treating adiposity and obesity in which, mRNA levels of genes that are related to fatty acid oxidation, including Acyl-CoA oxidase (ACO), carnitine palmitoyle-transferase1 (CPT1) and peroxisome proliferator-activated receptor α and δ (PPAR-α and δ) were undoubtedly expressed higher. In addition, lower levels of fatty acid synthesis such as sterol regulatory element-binding protein-1c (SREBP-1c) and acetyl-CoA carboxylase (ACC) were noted. ACO and CPT1 are the enzymes that play key roles in energy homeostasis and adipogenesis, Therefore, increased expression of fatty acid metabolism gene and alternatively reduced fatty acid synthesis were responsible for antiobesity effect in the mice.105,188 Prebiotics are non-digestible substances that play an important role in maintaining the nutritional, physiological and immunological status of the host and are used as an alternative constituent to improve the activity of probiotics. Fruits, vegetables, and cereals are potential sources of prebiotics.106,107 Artificially synthesized prebiotics that include lactulose, galacto-oligosaccharides, malto-oligosaccharides, fructo-oligosaccharides, inulin and oligosaccharides have gained attention in intestinal homeostasis.108 Prebiotics are fermented by the gut bacteria thereby producing products such as SCFAs which play an essential role in preventing many ailments such as metabolic endotoxemia and type 2 diabetes. Inulin and fructo-oligosaccharides are prebiotic dietary fibres that are used to enhance the growth of bacterial species belonging to genera of Bifidobacterium and Lactobacillus.1 Prebiotic treatment also helps in increasing the number of enteroendocrine cells that produce GLP-1 and GLP-2 (L-cells) in the jejunum and colon.109 Changes in the gut microbiota of the mouse with the inclusion of prebiotics helped in increasing the plasma GLP-2 levels and hence can improve systemic and hepatic inflammation. It has also been reported that apart from maintaining the integrity of the gut barrier and enhancing the immune function, GLP-2 also enhances hepatic insulin sensitivity and is also known to play a key role in controlling glucose homeostasis.104 An approach was suggested to use prebiotic dietary fibres (oligofructose) to increase the number of Bifidobacterium spp.110 Hence, the dietary supplementation of prebiotics helps in improving glucose-tolerance, glucose-induced insulin-secretion and normalising low-grade inflammation (decreased endotoxemia, plasma and adipose tissue pro-inflammatory cytokines).111 Synbiotics, a combination of probiotics and prebiotics, is another therapeutic approach to restore intestinal homeostasis.112 Some examples of synbiotics include a combination of Lactobacillus rhamnosus GG, Bifidobacteria and Lactobacilli with fructo-oligosaccharides or inulin.113 Symbiotic effect of the prebiotics and probiotics contribute for a synergistic effect that complement higher tolerance to various environmental conditions of the host’s intestine such as oxygenation, pH and temperature.113 Synbiotics help in reducing the concentration of undesirable metabolites and also leads to an increase in the levels of short chain fatty acids, ketones, carbon disulphides and methyl acetates which have a positive effect on the host’s health.114 The use of a synbiotic product consisting of Lactobacillus rhamnosus and Bifidobacterium lactis as probiotics and inulin and oligofructose as prebiotics in the diet helped in increasing the levels of intestinal IgA. Synbiotics reduce the levels of blood cholesterol, blood pressure and also improve the absorption of calcium, magnesium, and phosphorus.115 Table 2 provides the list of probiotics, prebiotics and synbiotics with the respective function.
Table 2: Nutraceuticals and their Functions with Respect to Gastrointestinal Related Diseases.
| Sl. No. | Nutraceutical Agent | Type | Function |
| 1. | Bacillus subtilis | Probiotic | Secretes several antimicrobial agents such as Subtilin and Bacilysin that has broad-spectrum antimicrobial activity against pathogens.act as an immune-stimulant and helps to maintain intestinal homeostasis116. |
| 2. | Lactococcus lactis | Probiotic | Produces bacteriocins such as lactococcin A and nisin that have antimicrobial properties against pathogens117.Has a potent anti-inflammatory effect on intestinal epithelial cells helping with colitis118. |
| 3. | Akkermansia muciniphila | Probiotic | Employed in the prevention and treatment of metabolic disorders such as Obesity and Type 2 Diabetes.Better clinical trial results are obtained in patients having A. muciniphila during cancer immunotherapy119. |
| 4. | Faecalibacterium prausnitzii | Probiotic | Has anti-inflammatory properties with the ability to reduce IL-8 levels and induce IL-10 production120. |
| 5. | Bifidobacterium infantis | Probiotic | Considered as a very important coloniser of infant’s gut. Bacteria metabolizes human milk oligosaccharides (HMO) into SCFA such as lactate and acetate.The SCFA help other beneficial bacteria colonise the gut. Also known to help with maturation of innate immune response, decreases intestinal permeability and controls inflammation121. |
| 6. | Bifidobacterium animalis subsp. Lactis | Probiotic | It promotes a healthy bowel function by relieving symptoms of antibiotic induced diarrhoea and constipation.Improves immune function and lowers incidence of respiratory ailments122. |
| 7. | Lactobacillus reuteri | Probiotic | Secretes antimicrobial agents such as reuterin and lactic acid that are effective against many gastrointestinal infections.Prevents H. pylori attachment to its receptor. Reduces production of pro-inflammatory cytokines such as TNF-α, IL-1β and IL-6123. |
| 8. | Lactobacillus rhamnosus | Probiotic | Secretes Msp1 and Msp2 that has anti-apoptotic properties and increases the survival of intestinal epithelial cells.Promotes gastrointestinal health and inhibits gastrointestinal pathogens such as Salmonella or Shigella124. |
| 9. | Enterococcus faecium | Probiotic | Antimicrobials such as enterocins are effective against many food-borne pathogens.It has preventive and curative effect against diarrhoea and irritable bowel syndrome125. |
| 10. | Saccharomyces boulardii | Probiotic | Has antimicrobial activity that prevents pathogen colonisation and maintains homeostasis.Stabilizes the intestinal barrier by preventing apoptosis and by controlling pro-inflammatory cytokines.Effective against acute GI diseases such as diarrhoea and chronic GI diseases such as colitis and Irritable bowel syndrome126. |
| 11. | Inulin | Prebiotic | Promotes growth of desired Bifidobacterium sp. and Lactobacillus sp. in the gutHelps re-establish intestinal homeostasis in diseased conditions and reduces risk of GI diseasesIncreases calcium deposition and mineral contents of bone127. |
| 12. | Beta Glucans | Prebiotic | Promotes selective growth of Bifidobacterium spp. and Lactobacillus spp. in the gutKnown to have immune-modulatory and anti-tumour properties128. |
| 13. | Polydextrose | Prebiotic | Has a positive effect on the growth of beneficial bacteria in the gut.Protective effect against inflammatory GI diseases and colon cancer.The SCFAs produced after its metabolism improves lipid metabolism and lowers blood glucose levels129. |
| 14. | Lactulose | Prebiotic | Exhibits a dose-dependent growth promotion of beneficial gut microbiota.Re-establishment of intestinal health also improves bowel functions and defecation frequencies due to increase in Bifidobacterium spp.130,131 |
| 15. | Galacto oligosaccharides (GOS) | Prebiotic | GOS can be metabolized by many intestinal bacteria such as Bifidobacterium sp. and Lactobacillus sp. thereby promoting their growth.Decrease in cancer risk, blood pressure control, and reduction of serum cholesterol levels is also associated with GOS consumption132. |
| 16. | Bifidobacterium longum and inulin-oligofructose | Synbiotic | Reduced levels of TNF-ɑ and IL-1ɑ and other pro-inflammatory markers in active Ulcerative colitisReduction in colitis at the macroscopic and microscopic level133. |
| 17. | Bifidobacterium breve, Lactobacillus casei and galactooligosaccharides | Synbiotic | Used in the treatment of short bowel syndromeImproved nutritional state, suppressed pathogenic growth in gut and establishment of healthy gut microbiotaProbiotics improved intestinal motility and enhanced the intestinal immune system while the prebiotic promoted growth of Bifidobacterium spp. and Lactobacillus spp.134 |
| 18. | Lactobacillus plantarum and Inulin | Synbiotic | Reduced growth of pathogenic strains such as E. coli and Salmonella spp. in the gutThe prebiotic promoted growth of Lactobacillus spp. and Bifidobacteria spp. and the synbiotic feed reduced aberrant crypt foci formation (Marker of colon cancer)135. |
| 19. | Lactobacillus acidophilus, Bifidobacterium bifidum and oligofructose | Synbiotic | Reduction in serum cholesterol levels, increased HDL levels and showed a significant decrease in glycemia136. |
Dietary Induced Interventions
Change in dietary patterns is found to be the reason for dysbiosis in patients whose microbiota development was driven by diet. 137,138 Studies have found a correlation between improved health due to consumption of dietary fibers and lowering of IBD and colorectal cancer.139,140 Western diets with high amounts of protein and fat content and a very minimal fiber intake increases the incidence of colon related diseases. Consumption of less fiber content has been proven to hinder gut microbial establishment, while a diet with high fiber content mediates SCFA (Short-Chain fatty acids) synthesis by intestinal microbes. A severe reduction in faecal butyrate levels in obese individuals was observed, when the individuals were put on a low carbohydrate diet. Reduction in butyrate production may be due to lower number of butyrate producing Firmicutes sp. along with a sudden change in diet pattern.141
Fatty Acids
SCFAs like acetate, butyrate, pentanoate and propionate have two to five carbon atoms that are mainly produced through microbial fermentation of large complex polysaccharides inside the colon.76 SCFAs enter into the bloodstream of the host and transverse to the distal colon, from which they are transported to respective tissues.142 Microbes in the gut produce 99% of the blood SCFAs (acetate and PPA), that act on regulatory receptor Olfr78 and Gpr41 in physiologically opposite roles in response to the same SCFAs stimulus at different concentration levels to maintain appropriate blood pressure regulation. SCFAs produced by the bacteria in the gut interact with olfactory receptors, Olfr78 and Gpr41 in the kidney, that are key regulators of blood pressure. Activation of Olfr78 stimulate renin and prevent the blood pressure from lowering to dangerous levels.143 Maintaining the appropriate level of SCFAs (acetate and propionate) in the blood is important to prevent neuro degeneration. Mice treated with butyrate synthesized by Bifidobacterium helped in the restoration of microbial homeostasis inside the gut, thus improving gut integrity compared to control mice.144 Butyrate, a bacterial metabolic end product, constitutes 20% of SCFA in the human colon. Butyrate is the primary metabolite preferred by the intestinal epithelial cells (IECs) that enhance the IEC barrier by stabilising the hypoxia-inducible factor (HIF). Butyrate suppresses colonic inflammation and carcinogenesis by activating GPR109a receptors that promote anti-inflammatory properties in colonic macrophages and dendritic cells. Butyrate stimulates anti-inflammatory mechanisms that helps in mucosal barrier restoration by limiting inflammatory cytokine production.144 Similarly, another study reported that introduction of SCFAs such as Propionate (PA) into a mice model showed enhancement of CD25, CD4, Foxp3, Treg cells and ameliorated autoimmune encephalomyelitis. PA has the capacity to restore altered Treg cell: effector T cell balance in Multiple sclerosis (MS) patients by restraining CNS autoimmunity. 145
Amino Acids and Carbohydrate Supplements in Food
Introduction of amino acids and carbohydrates would help the depleting and dormant bacterial spores to get activated and transform into a vegetative state.Colonic microbiota to a large extent depends on the availability of microbiota-accessible carbohydrates (MACs) that is found in the dietary fibre. Diet with highly rich in resistance starch or non-starch polysaccharide fibre can enrich the gut with diverging bacterial growth. Microbial digestion releases the finest food particle in to blood stream and serve as an antioxidant material.188 Similarly, amino acid play a major role in supporting the growth and survival of gut bacteria that regulate energy and protein homeostasis in organism. Many elements of tryptophan catabolism would be extremely beneficial for maintaining gut homeostasis146. Tryptophan helps in the regulation of tight junction proteins in intestinal epithelial cells that aids in building a barrier inside the gut147. Studies suggest that N-acetylcysteine helps in increasing the barrier inside the gut and also stops endothelial cells from premature senescence.148,149 N-acetylcysteine is also known to increase the growth of fibroblast cells by reducing ROS levels and by increasing ATP production in fibroblast.150 However, in vitro studies show that over usage of amino acids like N-acetylcysteine and tryptophan could cause inflammation.151 The mTORC1 pathway activates pro-oxidant and pro-inflammatory proteins in the blood cells forming branched chain amino acids (BCAAs), Leucine, Isoleucine and Valine, thus could lead to daily elevation of blood levels from 3-6 m mol/l concentration.151
Engineered Bacteria and Antibiotic Administration
The eubiotic state can be restored by supplementing engineered bacteria that are capable of producing biomolecules possessing the potential to alter human metabolism.152 The engineered probiotic species include Escherichia coli Nissle, Lactococcus lactis, Bacteroides ovatus and Lactobacillus casei.153,154,155 Nanobodies specific to eukaryotic targets are infused in synthetic microbiota that strongly anchor the engineered bacteria in the challenging gastrointestinal epithelial cells.156 Engineered bacteria packed in the synthetic consortia are used for delivering therapeutic antimicrobial substances and immune-modulators to minimise inflammation. Appropriate modification of microbiome by incorporation of genetically engineered Escherichia coli Nissle 1917, capable of bio-synthesising N-acyl-phosphatidylethanolamines (NAPEs) in the intestinal tract reduces obesity related disorders. Daily administration of engineered Escherichia coli exhibited elevated levels of NAPEs that limits development of obesity.157Antibiotic administration is also another therapeutic approach used to manipulate the microbial community to restore eubiotic state. Though antibiotics are reported to decreases the species richness and the microbial mass in the human gut, they carry the advantage for eliminating gut pathogens and confer resistance to beneficial microbes as a collateral effect. Selective antibiotics such as rifaximin also retard the inflammatory response exerted by the gut microbes by inhibiting the expression of nuclear factor (NF)-κB. Response of gut microbes to antibiotics are species specific. Incidence of higher Lactobacilli count was observed in mouse model treated with rifaximin whereas similar effect was not noticed in neomycin supplemented groups.27 Antibiotics are also effective for primary treatment of Crohn’s disease at a dosage of 10 to 20 mg/kg/day.158 Tylosin is another antibiotic which is used to treat chronic enteropathies in dogs. A study showed that the effect of this antibiotic in healthy dogs helped in reducing the bacterial content in the jejunum and caused an increase in the number of beneficial Enterococcus species and the study did not report any short term clinical abnormality of intestinal diseases in the treated groups 159. Furthermore, treatment with polymyxin B helps in specifically eliminating Gram-negative bacteria and thereby further quenching LPS which results in diminishing hepatic steatosis160. In a cohort of dogs with inflammatory bowel disease, administration of rifaximin helped in significantly improving the clinical signs by either decreasing the pathogenic strains or by increasing the beneficial microbes.161,162
Fecal Microbial Treatment, Bacterial Consortium Treatment and Phage Therapy
Fecal microbial transplantation (FMT) is a process by which fecal bacteria is transplanted from a healthy donor to a diseased recipient’s gastrointestinal tract.3 Infusion of fecal microbiota exhibited clinical improvement in dysbiosis-associated diseases. Though colonization of microbial suspension varies from individuals, a stable intestinal ecosystem can be restored by employing a mixed bacterial consortium. A study using faecal microbiota transplantation (FMT) from lean donors to insulin-resistant patients with metabolic syndrome showed that faeces from lean subjects improved insulin sensitivity and was also enhanced the numbers of butyrate-producing bacteria.163 In a randomized control study that was carried out between a group of people who were given FMT treatment and another group wherein the people were given antibiotics to treat the patients from recurrent Clostridium difficile infection showed that Clostridium difficile associated diarrhea in the FMT group resolved 81% of patients whereas, only 31% of the antibiotic group recovered. Infusion of donor feces to host system improved the microbial diversity by increasing the load of Bacteroidetes species and clostridium cluster IV and XIVa (Firmicutes) along with the decrease in the count of proteobacteria species. 164 However, an infused bacterial consortium might not have similar clinical efficacy in restoring mucosal physiology. Contrasting results were observed in a randomized Ulcerative Colitis trial where patients did not show any significant changes in the gut microbiota after undergoing FMT therapy.165 Few other studies carried out on adult Ulcerative Colitis patients following this approach failed but a lot of structural changes in the gut microbiota was observed.166,167 It has also been reported that this approach helps to some extent in treating some of the gastro-intestine related disorders but it is unclear whether the benefits of this approach is derived from the transfer of viable microbes or through the delivery of a wide range of substances like proteins, SCFAs and many others which are present in the faeces. Possibly, this approach should be carried out periodically and repeatedly in order to maintain the eubiotic state in the donor to cure dysbiosis.168 Clinical application of FMT therapy is limited due to poor colonisation of bacteria in the intestinal tract and non-standardization of the treatment process. The other therapeutic approach used to restore eubiosis is Bacterial Consortium Transplantation (BCT). Intestinal dysbiosis and TNBS induced colitis gets ameliorated upon supplementation of a bacterial consortium containing ten bacterial strains. Microbial equilibrium is re-established in the transplanted mice samples offering a therapeutic option for colitis and IBD diseases.169 BCT is a more effective and safer approach as the bacterial consortium can be very specific based on the disease to be treated.170 Disruption of microbial homeostasis in the intestine due to ceftriaxone sodium induced dysbiosis in BALB/c mice was restored by complementary supplementation of 1 × 109.8 bacterial cells collected from fresh faeces. Commensal bacteria in faecal sample that include Bifidobacterium thermophilum, Enterococcus hirae, Lactobacillus reuteri, Bacteroides alanitronis, Streptococcus thermophilus, Veillonella parvula, Peptococcus niger, Eubacterium siraeum, Escherichia coli str. Staphylococcus aureus subsp etc enhanced the intestinal permeability and re-established the disrupted mucosal barrier.171
Surface imprinted nanobodies that anchor on the surface of bacteria are utilised for the synthesis of synthetic consortia. Surface display of nanobody helps the target microbe to bind strongly on the target entities that include eukaryotic cells, viruses and bacteria.172 Intestinal phages directly influence the co-existing bacteriome which directly contribute to intestinal homeostasis. Phage bacteriome interaction and Phage host interaction influence gut equilibrium. Activation of host immune response by a phenomenon called phage tropism stimulates the TLR pathway and stimulates adaptive immune response.173 The treatment of dysbiosis with respect to phage therapy is of great concern as many studies must be performed for the approval of phages as antibacterial drugs by Food and Drug Administration (FDA) and European Medicines Agency (EMA). It is known that phages are viruses that infect bacteria by recognizing the specific receptor proteins present on the bacterial surface and hence it is reported that these phages can be used for antimicrobial purposes. Phages can be genetically modified and can be used as “gene carriers” for the biosynthesis and degradation of nutrients and to modulate the intestinal microbiota.174 Studies have reported that the use of lytic phages have shown positive results of decrease in Pseudomonas aeruginosa PA14. Phages aid in the disaggregation of the biofilm thereby allowing the antibiotics to diffuse at a much higher rate resulting in the elimination of the pathogenic bacteria.175,176 Further studies need to be carried out to understand the mechanistic interaction of using phages as a therapeutic agent to manipulate the microbiota in order to attain eubiosis.177
Conclusion
The importance of the microbes inhabiting the intestinal region directly influences the metabolic functioning and contributes for disease management. Maintaining a eubiotic state will help in preventing the onset of gastro-intestinal diseases. Many therapeutic approaches are currently employed to restore the eubiotic state for a healthy gut homeostasis. Apart from the usage of prebiotics, probiotics and synbiotics, use of certain metabolites synthesized by the gut microbes aid in bringing back normalcy. The administration of gut peptides like GLP-1 and secretory products such as butyrate, short chain fatty acids help in maintaining homeostasis in the gut microbiome. Fasting Induced Adipocyte Factor (FIAF) activity can also be considered as a useful therapy to increase LPL-driven triglyceride clearance. Evidence based data will further help in considering the gut microbiota as a target to prevent intestinal dysbiosis and further robust investigation is required for it to be applicable in humans. This field involves great interest because there is no universal cure for an imbalance of the gut microbiota. Personalised medicine employing innovative approaches like bacteria therapy, phage therapy, next generation probiotics and bacterial consortium transplantation depends on the individual’s health condition, lifestyle, diet and other criteria.
Acknowledgement
The authors acknowledge the support received from the Department of Life Sciences and Centre for Research, CHRIST (Deemed to be University), India.
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding Sources
The project is funded under MRP-DSC-1936 by CHRIST (Deemed To Be University), Bangalore, India.
References
- Cani P., Delzenne N. The Role of the Gut Microbiota in Energy Metabolism and Metabolic Disease. Curr Pharm Des. 2009; 15(13): 1546-1558.
CrossRef - Carding S., Verbeke K., Vipond D. T., Corfe B. M., Owen L. J. Dysbiosis of the gut microbiota in disease. Microb Ecol Health Dis. 2015; 26(0).
CrossRef - LeBlanc A. Effect of probiotic administration on the intestinal microbiota, current knowledge and potential applications. World J Gastroenterol. 2014; 20(44): 16518.
CrossRef - Belizário J., Napolitano M. Human microbiomes and their roles in dysbiosis, common diseases, and novel therapeutic approaches. Front Microbiol. 2015; 6 :1050.
CrossRef - Tang W., Kitai T., Hazen S. Gut Microbiota in Cardiovascular Health and Disease. Circ Res. 2017; 120(7): 1183-1196.
CrossRef - Grenham S., Clarke G., Cryan J., Dinan G. T. Brain-Gut-Microbe Communication in Health and Disease. Front Physiol. 2011; 2:1-15.
CrossRef - Brown C. T., Sharon I., Thomas B. C., Castelle C. J., Morowitz M. J., Banfield J. F. Genome resolved analysis of a premature infant gut microbial community reveals a Varibaculum cambriense genome and a shift towards fermentation-based metabolism during the third week of life. Microbiome. 2013; 1(1): 30.
CrossRef - Sartor R. B., Mazmanian S. K. Intestinal Microbes in Inflammatory Bowel Diseases. Am J Gastroenterol Suppl. 2012; 1(1): 15-21.
CrossRef - Round J. L., Mazmanian S. K. The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol. 2009; 9(8): 313-323.
CrossRef - Pascale A., Marchesi N., Marelli C., Coppola A., Luzi L., Govoni S., Giustina A., Gazzaruso C. Microbiota and metabolic diseases. Endocrine. 2018; 61(3): 357-371.
CrossRef - Logan A. C., Jacka F. N., Prescott S. L. Immune-Microbiota Interactions: Dysbiosis as a Global Health Issue. Curr Allergy Asthma Rep. 2016; 16(2).
CrossRef - Tanaka M., Nakayama J. Development of the gut microbiota in infancy and its impact on health in later life. Allergol Int. 2017; 66(4): 515-522.
CrossRef - Tamburini S., Shen N., Wu H. C., Clemente J. C. The microbiome in early life: implications for health outcomes. Nat Med. 2016; 22(7): 713-722.
CrossRef - Carmody R. N., Gerber G. G., Luevano J. M., Gatti D. M., Somes L., Svenson K. L., Turnbaugh P. J. Diet Dominates Host Genotype in Shaping the Murine Gut Microbiota. Cell Host Microbe. 2015; 17(1): 72-84.
CrossRef - Bäckhed F., Roswall J., Peng Y., Feng Q., Jia H., Kovatcheva-Datchary P., Li Y., Xia Y., Xie H., Zhong H., Khan M. T., Zhang J., Li J., Xiao L., Al-Aama J., Zhang D., Lee Y. S., Kotowska D., Colding C., Tremaroli V., Yin Y., Bergman S., Xu X., Madsen L., Kristiansen K., Dahlgren J., Wang J. Dynamics and Stabilization of the Human Gut Microbiome during the First Year of Life. Cell Host Microbe. 2015; 17(6): 852.
CrossRef - Schnabl B., Brenner D. Interactions Between the Intestinal Microbiome and Liver Diseases. Gastroenterology. 2014; 146(6): 1513-1524.
CrossRef - Lebba V., Totino V., Gagliardi A., Santangelo F., Cacciotti F., Trancassini M., Mancini C., Cicerone C., Corazziari E., Pantanella F., Schippa S. Eubiosis and dysbiosis: the two sides of the microbiota. New Microbiol. 2016; 39(1): 1-12.
- Hsiao W., Metz C., Singh D., Roth J. The Microbes of the Intestine: An Introduction to Their Metabolic and Signaling Capabilities. Endocrinol Metab Clin North Am. 2008; 37(4): 857-871.
CrossRef - Sangiuliano B., Pérez N., Moreira D., Belizário J. Cell Death-Associated Molecular-Pattern Molecules: Inflammatory Signaling and Control. Mediators Inflamm. 2014; 1-14.
CrossRef - Abreu M., Fukata M., Arditi M. TLR Signaling in the Gut in Health and Disease. J Immunol. 2005; 174(8): 4453-4460.
CrossRef - Chelakkot C., Ghim J., Ryu S. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp Mol Med. 2018; 50(8):103.
CrossRef - Ryan P. M, Delzenne N. M. Gut Microbiota and Metabolism. In: Hyland N., Stanton C. The Gut-Brain Axis Dietary, Probiotic, and Prebiotic Interventions on the Microbiota. Academic press. Ireland. 2016, 391-401.
CrossRef - Paredes-Sabja D., Shen A., Sorg J. Clostridium difficile spore biology: sporulation, germination, and spore structural proteins. Trends Microbiol. 2014; 22(7): 406-416.
CrossRef - Xu J., Bjursell M. K., Himrod J., Deng S., Carmichael L. K., Chiang H. C., Hooper L. V., Gordon J. I. A Genomic View of the Human-Bacteroides thetaiotaomicron Symbiosis. Science. 2003; 299(5615): 2074-2076.
CrossRef - Hooper L. V., Wong M. H., Thelin A., Hansson L., Falk P. G., Gordon J. I. Molecular Analysis of Commensal Host-Microbial Relationships in the Intestine. Science. 2001; 291(5505): 881-884.
CrossRef - Hammer H. Gut Microbiota and Inflammatory Bowel Disease. Dig Dis. 2011; 29(6): 550-553.
CrossRef - Ponziani F. R., Zocco M. A., D’Aversa F., Pompili M., Gasbarrini A. Eubiotic properties of rifaximin: Disruption of the traditional concepts in gut microbiota modulation. World J Gastroenterol. 2017; 23(25): 4491–4499.
CrossRef - Backhed F., Ding H., Wang T., Lora V. H., Gou Y. K., Andras N., Clay F. S., Jeffrey I. G. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci. 2004; 101(44): 15718-15723.
CrossRef - Samuel B., Gordon J. A humanized gnotobiotic mouse model of host-archaeal-bacterial mutualism. Proc Natl Acad Sci. 2006; 103(26): 10011-10016.
CrossRef - Rawls J., Samuel B., Gordon J. From The Cover: Gnotobiotic zebrafish reveal evolutionarily conserved responses to the gut microbiota. Proc Natl Acad Sci. 2004; 101(13): 4596-4601.
CrossRef - Harris K., Kassis A., Major G., Chou J. , Is the Gut Microbiota a New Factor Contributing to Obesity and Its Metabolic Disorders. J Obes. 2012; 2012: 1-14.
CrossRef - Delzenne N., Neyrinck A., Bäckhed F., Cani P. D. Targeting gut microbiota in obesity: effects of prebiotics and probiotics. Nat Rev Endocrinol. 2011; 7(11), 639-646.
CrossRef - Conlon M., Bird A. The Impact of Diet and Lifestyle on Gut Microbiota and Human Health. Nutrients. 2014; 7(1): 17-44.
CrossRef - Shen J., Obin M., Zhao L. The gut microbiota, obesity and insulin resistance. Mol Aspects Med. 2013; 34: 39–58.
CrossRef - Amabebe E., Robert F., Agbalalah T., Orubu E. S. F. Microbial dysbiosis-induced obesity: role of gut microbiota in homoeostasis of energy metabolism. Br J Nutr. 2020; 123(10): 1127-1137.
CrossRef - Gill S. R., Pop M., Deboy R. T., Eckburg P. B., Turnbaugh P. J., Samuel B. S., Gordon J. I., Relman D. A., Fraser-Liggett C. M., Nelson K. E. Metagenomic Analysis of the Human Distal Gut Microbiome. Science. 2006; 312(5778): 1355-1359.
CrossRef - Chimerel C., Emery E., Summers D., Keyser U., Gribble F., Reimann F. Bacterial Metabolite Indole Modulates Incretin Secretion from Intestinal Enteroendocrine L Cells. Cell Rep. 2014; 9(4): 1202-1208.
CrossRef - Forslund K., Sunagawa S., Kultima J., Mende D. R., Arumugam M., Typas A., Bork P. Country-specific antibiotic use practices impact the human gut resistome. Genome Res. 2013; 23(7): 1163-1169.
CrossRef - Hu Y., Yang X., Qin J., Lu N., Cheng G., Wu N., Pan Y., Li J., Zhu L., Wang X., Meng Z., Zhao F., Liu D., Ma J., Qin N., Xiang C., Xiao Y., Li L., Yang H., Wang J., Yang R., Gao G. F., Wang J., Zhu B. Metagenome-wide analysis of antibiotic resistance genes in a large cohort of human gut microbiota. Nat Commun. 2013; 4(1):
CrossRef - Nicholson J., Holmes E., Kinross J., Burcelin R., Gibson G., Jia W., Pettersson S. Host-Gut Microbiota Metabolic Interactions. Science. 2012; 336(6086): 1262-1267.
CrossRef - Fukuda S., Toh H., Hase K., Oshima K., Nakanishi Y., Yoshimura K., Tobe T., Clarke J. M., Topping D. L., Suzuki T., Taylor T. D., Itoh K., Kikuchi J., Morita H., Hattori M., Ohno H. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature. 2011; 469(7331): 543-547.
CrossRef - Griffiths E., Duffy L., Schanbacher F., Qiao H., Dryja D., Leavens A., Rossman J., Rich G., Dirienzo D., Ogra P. L. In Vivo Effects of Bifidobacteria and Lactoferrin on Gut Endotoxin Concentration and Mucosal Immunity in Balb/c Mice. Dig Dis Sci. 2004; 49(4): 579-589.
CrossRef - Wang Z., Xiao G., Yao Y., Guo S., Lu K., Sheng Z. The Role of Bifidobacteria in Gut Barrier Function After Thermal Injury in Rats. J Trauma. 2006; 61(3): 650-657.
CrossRef - Sokol H., Pigneur B., Watterlot L., Lakhdari O., Bermúdez-Humarán L. G., Gratadoux J. J., Blugeon S., Bridonneau C., Furet J. P., Corthier G., Grangette C., Vasquez N., Pochart P., Trugnan G., Thomas G., Blottière H. M., Doré J., Marteau P., Seksik P., Langella P. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc Natl Acad Sci. 2008; 105(43): 16731-16736.
CrossRef - Winter S., Winter M., Xavier M. N., Thiennimitr P., Poon V., Keestra A. M., Laughlin R. C., Gomez G., Wu J., Lawhon S. D., Popova I. E., Parikh S. J., Adams L. G., Tsolis R. M., Stewart V. J., Bäumler A. J. Host-Derived Nitrate Boosts Growth of E. coli in the Inflamed Gut. Science. 2013; 339(6120): 708-711.
CrossRef - Ley R., Backhed F., Turnbaugh P., Lozupone C. A., Knight R. D., Gordon J. I. Obesity alters gut microbial ecology. Proc Natl Acad Sci. 2005; 102(31): 11070-11075.
CrossRef - Turnbaugh P., Ley R., Mahowald M. A., Magrini V., Mardis E. R., Gordon J. I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006; 444(7122): 1027-1031.
CrossRef - Voigt R., Forsyth C., Green S. J., Mutlu E., Engen P., Vitaterna M. H., Turek F. W., Keshavarzian A. Circadian Disorganization Alters Intestinal Microbiota. PLoS ONE. 2014; 9(5): e97500.
CrossRef - Cani P., Osto M., Geurts L., Everard A. Involvement of gut microbiota in the development of low-grade inflammation and type 2 diabetes associated with obesity. Gut Microbes. 2012; 3(4): 279-288.
CrossRef - Hawrelak J. A., Myers S. P. The causes of Intestinal Dysbiosis: A Review. Altern Med Rev. 2004; 9(2): 180-197.
- Clarke G., Grenham S., Scully P., Fitzgerald P., Moloney R. D., Shanahan F., Dinan T. G., Cryan J. F. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol Psychiatry. 2012; 18(6): 666-673.
CrossRef - Lizko N. N. Stress and intestinal microflora. Nahrung. 1987; 31(5-6): 443-447.
CrossRef - Elamin E., Masclee A., Dekker J., Jonkers D. M. Ethanol metabolism and its effects on the intestinal epithelial barrier. Nutr Rev. 2013; 71(7): 483-499.
CrossRef - Zhou Y., Mihindukulasuriya K., Gao H., La Rosa P. S., Wylie K. M., Martin J. C., Kota K., Shannon W.D., Mitreva M., Sodergren E., Weinstock G. M. Exploration of bacterial community classes in major human habitats. Genome Biol. 2014; 15(5): R66.
CrossRef - Paule A., Frezza D., Edeas M. Microbiota and Phage Therapy: Future Challenges in Medicine. Med Sci (Basel). 2018; 6(4): 86.
CrossRef - Hanauer S. Inflammatory bowel disease: Epidemiology, pathogenesis, and therapeutic opportunities. Inflamm Bowel Dis. 2006; 12: S3-S9.
CrossRef - Fava F., Danese S. Intestinal microbiota in inflammatory bowel disease: Friend of foe? World J Gastroenterol. 2011; 17(5): 557.
CrossRef - Knights D., Lassen K., Xavier R. Advances in inflammatory bowel disease pathogenesis: linking host genetics and the microbiome. 2013; 62(10): 1505-1510.
CrossRef - Morgan X., Tickle T., Sokol H., Gevers D., Devaney K. L., Ward D. V., Reyes J. A., Shah S. A., LeLeiko N., Snapper S. B., Bousvaros A., Korzenik J., Sands B. E., Xavier R. J., Huttenhower C. Dysfunction of the intestinal microbiome in inflammatory bowel disease and treatment. Genome Biol. 2012; 13(9): R79.
CrossRef - Kanauchi O., Mitsuyama K., Araki Y., Andoh A. Modification of Intestinal Flora in the Treatment of Inflammatory Bowel Disease. Curr Pharm Des. 2003; 9(4): 333-346.
CrossRef - Nuding S., Fellermann K., Wehkamp J., Stange E. F. Reduced mucosal antimicrobial activity in Crohn’s disease of the colon. Gut. 2007; 56(9): 1240-1247.
CrossRef - Packey C., Sartor R. Commensal bacteria, traditional and opportunistic pathogens, dysbiosis and bacterial killing in inflammatory bowel diseases. Curr Opin Infect Dis. 2009; 22(3): 292-301.
CrossRef - Bringer M.A., Glasser A.L., Tung C.H., Méresse S., Darfeuille-Michaud A. The Crohn’s disease-associated adherent-invasive Escherichia coli strain LF82 replicates in mature phagolysosomes within J774 macrophages. Cell Microbiol. 2006; 8(3): 471-484.
CrossRef - Sokol H., Seksik P., Furet J. P., Firmesse O., Nion-Larmurier I., Beaugerie L., Cosnes J., Corthier G., Marteau P., Doré J. Low counts of Faecalibacterium prausnitzii in colitis microbiota. Inflamm Bowel Dis. 2009; 15(8): 1183-1189.
CrossRef - Baker P., Love D., Ferguson L. Role of gut microbiota in Crohn’s disease. Expert Rev Gastroenterol Hepatol. 2009; 3(5): 535-546.
CrossRef - Joossens M., Huys G., Cnockaert M., De Preter V., Verbeke K., Rutgeerts P., Vandamme P., Vermeire S. Dysbiosis of the faecal microbiota in patients with Crohn’s disease and their unaffected relatives. Gut. 2011; 60(5): 631-637.
CrossRef - Cani P, Amar J., Iglesias M. , Poggi M., Knauf C., Bastelica D., Neyrinck A. M., Fava F., Tuohy K. M., Chabo C., Waget A., Delmée E., Cousin B., Sulpice T., Chamontin B., Ferrières J., Tanti J. F., Gibson G. R., Casteilla L., Delzenne N. M., Alessi M. C., Burcelin R. Metabolic Endotoxemia Initiates Obesity and Insulin Resistance. Diabetes. 2007; 56(7): 1761-1772.
CrossRef - Lin, H. V., Frassetto A., Kowalik E. J., Nawrocki, A. R., Lu, M. M., Kosinski, J. R., Hubert, J. A., Szeto, D., Yao X., Forrest G., Marsh D. J. Butyrate and Propionate Protect against Diet-Induced Obesity and Regulate Gut Hormones via Free Fatty Acid Receptor 3-Independent Mechanisms. PLoS ONE. 2012; 7(4): p.e35240.
CrossRef - Chambers E. S., Viardot A., Psichas A., Morrison D. J., Murphy K. G., Zac-Varghese S. E. K., MacDougall , Preston, T., Tedford C., Finlayson, G. S., Blundell J. E., Bell, J. D, Thomas, E. L, Mt-Isa, S., Ashby, D., Gibson, G. R., Kolida S., Dhillo W. S., Bloom, S. R., Morley W., Clegg S., Frost G. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut. 2014; 64(11): 1744-1754.
CrossRef - Ghanim H., Abuaysheh S., Sia C. L., Korzeniewski K., Chaudhuri A., Fernandez-Real J. M., Dandona P. Increase in Plasma Endotoxin Concentrations and the Expression of Toll-Like Receptors and Suppressor of Cytokine Signaling-3 in Mononuclear Cells After a High-Fat, High-Carbohydrate Meal: Implications for insulin resistance. Diabetes Care. 2009; 32(12): 2281-2287.
CrossRef - Shi H., Kokoeva M., Inouye K., Tzameli I., Yin H., Flier J. S. TLR4 links innate immunity and fatty acid–induced insulin resistance. J Clin Invest. 2006; 116(11): 3015-3025.
CrossRef - Song M., Kim K., Yoon J. M., Kim J. B. Activation of Toll-like receptor 4 is associated with insulin resistance in adipocytes. Biochem Biophys Res Commun. 2006; 346(3): 739-745.
CrossRef - Amar J., Burcelin R., Ruidavets J., Cani P. D., Fauvel J., Alessi M. C., Chamontin B., Ferriéres J. Energy intake is associated with endotoxemia in apparently healthy men. Am J Clin Nutr. 2008; 87(5): 1219-1223.
CrossRef - Pussinen P., Havulinna A., Lehto M., Sundvall J., Salomaa V. Endotoxemia Is Associated With an Increased Risk of Incident Diabetes. Diabetes Care. 2011; 34(2): 392-397.
CrossRef - Donohoe D., Garge N., Zhang X., Sun W., O’Connell T. M., Bunger M. K., Bultman S. J. The Microbiome and Butyrate Regulate Energy Metabolism and Autophagy in the Mammalian Colon. Cell Metab. 2011; 13(5): 517-526.
CrossRef - Donohoe D., Wali A., Brylawski B., Bultman S. J. Microbial Regulation of Glucose Metabolism and Cell-Cycle Progression in Mammalian Colonocytes. PLoS ONE. 2012; 7(9): e46589.
CrossRef - Alberti K., Zimmet P., Shaw J. The metabolic syndrome—a new worldwide definition. The Lancet. 2005; 366(9491): 1059-1062.
CrossRef - Grundy S. Metabolic Syndrome Pandemic. Arterioscler Thromb Vasc Biol. 2008; 28(4): 629-636.
CrossRef - Vijay-Kumar M., Aitken J., Carvalho F., Cullender T. C., Mwangi S., Srinivasan S., Sitaraman S. V., Knight R., Ley R. E., Gewirtz A. T. Metabolic Syndrome and Altered Gut Microbiota in Mice Lacking Toll-Like Receptor 5. Science. 2010; 328(5975): 228-231.
CrossRef - Miele L., Valenza V., La Torre G., Montalto M., Cammarota G., Ricci R., Mascianà R., Forgione A., Gabrieli M. L., Perotti G., Vecchio F. M., Rapaccini G., Gasbarrini G., Day C. P., Grieco A. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology. 2009; 49(6): 1877-1887.
CrossRef - Cheung O., Sanyal A. Recent advances in nonalcoholic fatty liver disease. Curr Opin Gastroenterol. 2009; 25(3): 230-237.
CrossRef - Corbin K., Zeisel S. Choline metabolism provides novel insights into nonalcoholic fatty liver disease and its progression. Curr Opin Gastroenterol. 2012; 28(2): 159-165.
CrossRef - Zhu L., Baker S., Gill C., Liu W., Alkhouri R., Baker R. D., Gill S. R. Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: A connection between endogenous alcohol and NASH. Hepatology. 2013; 57(2): 601-609.
CrossRef - Hartmann P., Seebauer C., Schnabl B. Alcoholic Liver Disease: The Gut Microbiome and Liver Cross Talk. Alcohol Clin Exp Res. 2015; 39(5): 763-775.
CrossRef - Yan A. W., Fouts D. E., Brandl J., Stärkel P., Torralba M., Schott E., Tsukamoto H., Nelson K. E., Brenner D. A., Schnabl B. Enteric dysbiosis associated with a mouse model of alcoholic liver disease. 2010; 53(1): 96-105.
CrossRef - Braak H., Tredici K., Rüb U., de Vos R. A., Jansen Steur E. N., Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging. 2003; 24(2): 197-211.
CrossRef - Dickson D. W., Fujishiro H., Orr C., DelleDonne A., Josephs K. A., Frigerio R., Burnett M., Parisi J. E., Klos K. J., Ahlskog J. E. Neuropathology of non-motor features of Parkinson disease. Parkinsonism Relat Disord. 2009; 15: S5.
CrossRef - Mayer E. A., Tillisch K., Gupta A. Gut/brain axis and the microbiota. J Clin Invest. 2015; 125(3): 926-938.
CrossRef - Martinez-Martin P., Rodriguez-Blazquez C., Kurtis M. M., Chaudhuri K. R. The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov Disord. 2011; 26(3): 399-406.
CrossRef - Cloud LJ., Greene JG. Gastrointestinal Features of Parkinson’s Disease. Curr Neurol Neurosci Rep. 2011; 11(4): 379-384.
CrossRef - Noh Y., Sung Y.H., Lee J, Kim E. Y. Nigrosome 1 Detection at 3T MRI for the Diagnosis of Early-Stage Idiopathic Parkinson Disease: Assessment of Diagnostic Accuracy and Agreement on Imaging Asymmetry and Clinical Laterality. AJNR Am J Neuroradiol. 2015; 36(11): 2010-2016.
CrossRef - Galland L. The Gut Microbiome and the Brain. J Med Food. 2014; 17(12): 1261-1272.
CrossRef - Erny D., Hrabě de Angelis A. L., Jaitin D., Wieghofer V., Staszewski O., David E., Keren-Shaul H., Mahlakoiv T., Jakobshagen K., Buch T., Schwierzeck V., Utermöhlen O., Chun E.,Garrett W. S., McCoy K. D., Diefenbach A., Staeheli P., Stecher P., Amit I., Prinz M. Host microbiota constantly control maturation and function of microglia in the CNS. Nat Neurosci. 2015; 18(7): 965-977.
CrossRef - Bienenstock J., Kunze W., Forsythe P. Microbiota and the gut–brain axis. Nutrition Reviews. 2015; 73(1): 28-31.
CrossRef - Charlett A., Dobbs R. J., Dobbs S. M., Weller C., Brady P., Peterson D. W. Parkinsonism: siblings share Helicobacter pylori seropositivity and facets of syndrome. Acta Neurol Scand. 2009; 99(1): 26-35.
CrossRef - Mridula K. R., Borgohain R., Chandrasekhar Reddy V., Bandaru V., Suryaprabha T. Association of Helicobacter pylori with Parkinson’s Disease. J Clin Neurol. 2017; 13(2): 181.
CrossRef - Sampson T. R., Debelius J. W., Thron T., Janssen S., Shastri G. G., Ilhan Z. E., Challis C., Schretter C. E., Rocha S., Gradinaru V., Chesselet M. F., Keshavarzian A., Shannon K. M., Krajmalnik-Brown R., Wittung-Stafshede P., Knight R., Mazmanian S. K. Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease. Cell. 2016; 167(6): 1469-1480.e12.
CrossRef - Barichella M., Pacchetti C., Bolliri C., Cassani E., Iorio L., Pusani C., Pinelli G., Privitera G., Cesari I., Faierman S. A., Caccialanza R., Pezzoli G., Cereda E. Probiotics and prebiotic fiber for constipation associated with Parkinson disease. Neurology. 2016; 87(12): 1274-1280.
CrossRef - Burokas A., Arboleya S., Moloney R. D., Peterson V. L., Murphy K., Clarke G., Stanton C., Dinan T. G., Cryan J. F. Targeting the Microbiota-Gut-Brain Axis: Prebiotics Have Anxiolytic and Antidepressant-like Effects and Reverse the Impact of Chronic Stress in Mice. Biol Psychiatry. 2017; 82(7): 472-487.
CrossRef - Messaoudi M., Lalonde R., Violle N., Javelot H., Desor D., Nejdi A., Bisson J. F., Rougeot C., Pichelin M., Cazaubiel M., Cazaubiel J. M. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticusR0052 and Bifidobacterium longumR0175) in rats and human subjects. Br J Nutr. 105(5): 755-764.
CrossRef - Gagliardi A., Totino V., Cacciotti F., Gagliardi A., Totino V., Cacciotti F., Iebba V., Neroni B., Bonfiglio G., Trancassini M., Passariello C., Pantanella F., Schippa S. Rebuilding the Gut Microbiota Ecosystem. Int J Environ Res Public Health. 2018; 15(8): 1679.
CrossRef - Simon M. C., Strassburger K., Nowotny B., Kolb H., Nowotny P., Burkart V., Zivehe F., Hwang J. H., Stehle P., Pacini G., Hartmann B., Holst J. J., MacKenzie C., Bindels L. B., Martinez I., Walter J., Henrich B., Schloot N. C., Roden M. Intake of Lactobacillus reuteri Improves Incretin and Insulin Secretion in Glucose-Tolerant Humans: A Proof of Concept. Diabetes Care. 2015; 38(10): 1827-1834.
CrossRef - Lennon G.., Balfe Á., Earley H., Devane L. A., Lavelle A., Winter D. C., Coffey J. C., O’Connell P. R. Influences of the colonic microbiome on the mucous gel layer in ulcerative colitis. Gut Microbes. 2014; 5(3): 277-476.
CrossRef - Mendis M., Leclerc E., Simsek S. Arabinoxylans, gut microbiota and immunity. Carbohydr Polym. 2016; 139: 159-166.
CrossRef - Mekkes M. C., Weenen T. C., Brummer R. J., Claassen E. The development of probiotic treatment in obesity: a review. Benef Microbes. 2014; 5(1): 19-28.
CrossRef - Macfarlane S., Macfarlane G., Cummings J. Review article: prebiotics in the gastrointestinal tract. Aliment Pharmacol Ther. 2006; 24(5): 701-714.
CrossRef - Flamm G., Glinsmann W., Kritchevsky D., Prosky L., Roberfroid M. Inulin and Oligofructose as Dietary Fiber: A Review of the Evidence. Crit Rev Food Sci Nutr. 2001; 41(5): 353-362.
CrossRef - Markowiak P., Sli´zewska K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. 2017; 9(9): 1021.
CrossRef - Everard A., Lazarevic V., Derrien M., Girard M., Muccioli G. G., Neyrinck A. M., Possemiers S., Van Holle A., François P., de Vos W. M., Delzenne N. M., Schrenzel J., Cani P. D. Responses of Gut Microbiota and Glucose and Lipid Metabolism to Prebiotics in Genetic Obese and Diet-Induced Leptin-Resistant Mice. 2011; 60(11): 2775-2786.
CrossRef - Tuohy K., Rouzaud G., Bruck W., Gibson G. R. Modulation of the Human Gut Microflora Towards Improved Health Using Prebiotics – Assessment of Efficacy. Curr Pharm Des. 2005; 11(1): 75-90.
CrossRef - Shi X., Zhou F., Li X., Chang B., Li D., Wang Y., Tong Q., Xu Y., Fukuda M., Zhao J. J., Li D., Burrin D. G., Chan L., Guan X. Central GLP-2 Enhances Hepatic Insulin Sensitivity via Activating PI3K Signaling in POMC Neurons. Cell Metab. 2013; 18(1): 86-98.
CrossRef - Ceapa C., Wopereis H., Rezaïki L., Kleerebezem M., Knol J., Oozeer R. Influence of fermented milk products, prebiotics and probiotics on microbiota composition and health. Best Pract Res Clin Gastroenterol. 2013; 27(1): 139-155.
CrossRef - Tsilingiri K., Rescigno M. Postbiotics: what else? Benef Microbes.2013; 4(1): 101-107.
CrossRef - Pandey K. R., Naik S. R., Vakil B. V. Probiotics, prebiotics and synbiotics- a review. Journal of Food Science and Technology. 2015; 52(12): 7577-7587.
CrossRef - Burcelin R. Gut microbiota and immune crosstalk in metabolic disease. Mol Metab. 2016; 5(9): 771-781.
CrossRef - Suva M. A., Sureja V. P., Kheni D. B. Novel insight on probiotic Bacillus subtilis: Mechanism of action and clinical applications. J Curr Res Sci Med. 2016; 2(2): 65.
CrossRef - Yerlikaya O. Probiotic potential and biochemical and technological properties of Lactococcus lactis ssp. lactis strains isolated from raw milk and kefir grains. J Dairy Sci. 2019; 102(1): 124-134.
CrossRef - Nishitani Y., Tanoue T., Yamada K., Ishida T., Yoshida M., Azuma T., Mizuno M. Lactococcus lactis cremoris FC alleviates symptoms of colitis induced by dextran sulfate sodium in mice. Int Immunopharmacol. 2009; 9(12): 1444-1451.
CrossRef - Zhang T., Li Q., Cheng L., Buch H., Zhang F. Akkermansia muciniphila is a promising probiotic. Microb Biotechnol. 2019; 12(6): 1109-1125.
CrossRef - Martín R., Miquel S., Benevides L., Bridonneau C., Robert V., Hudault S., Chain F., Berteau O., Azevedo V., Chatel J. M., Sokol H., Bermúdez-Humarán L. G., Thomas M., Langella P. Functional Characterization of Novel Faecalibacterium prausnitzii Strains Isolated from Healthy Volunteers: A Step Forward in the Use of prausnitzii as a Next-Generation Probiotic. Front Microbiol. 2017; 8:1226.
CrossRef - Chichlowski M., Shah N., Wampler J. L., Wu S. S., Vanderhoof J. A. Bifidobacterium longum Subspecies infantis (B. infantis) in Pediatric Nutrition: Current State of Knowledge. 2020; 12(6): 1581.
CrossRef - Jungersen M., Wind A., Johansen E., Christensen J. E., Stuer-Lauridsen B., Eskesen D. The Science behind the Probiotic Strain Bifidobacterium animalis subsp. lactis BB-12®. 2014; 2(2): 92-110.
CrossRef - Mu Q., Tavella V. J., Luo X. M. Role of Lactobacillus reuteri in Human Health and Diseases. Front Microbiol. 2018; 9:757.
CrossRef - Segers M. E., Lebeer S. Towards a better understanding of Lactobacillus rhamnosus GG host interactions. Microb Cell Fact. 2014; 13(Suppl 1): S7.
CrossRef - Hanchi H., Mottawea W., Sebei K., Hammami R. The Genus Enterococcus: Between Probiotic Potential and Safety Concerns—An Update. Front Microbiol. 2018; 9:1791.
CrossRef - Kelesidis T., Pothoulakis C. Efficacy and safety of the probiotic Saccharomyces boulardii for the prevention and therapy of gastrointestinal disorders. Therap Adv Gastroenterol. 2011; 5(2): 111-125.
CrossRef - Shoaib M., Shehzad A., Omar M., Rakha A., Raza H., Sharif H. R., Shakeel A., Ansari A., Niazi S. Inulin: Properties, health benefits and food applications. Carbohydr Polym. 2016; 20(147): 444-454.
CrossRef - Lam K. K., Chi-Keung Cheung P. Non-digestible long chain beta-glucans as novel prebiotics. Bioactive Carbohydrates and Dietary Fibre. 2013; 2(1): 45-64.
CrossRef - Do Carmo M. M. R., Walker J. C.L., Novello D, Caselato V. M., Sgarbieri V. C., Ouwehand A. C., Andreollo N. A., Hiane P. A., Dos Santos E. F. Polydextrose: Physiological Function, and Effects on Health. 2016; 8(9): 553.
CrossRef - Ruszkowski J., Witkowski J. M. Lactulose: Patient- and dose-dependent prebiotic properties in humans. 2019; 59: 100-106.
CrossRef - Sakai Y., Seki N., Hamano K., Ochi H., Abe F., Masuda K., Iino H. Prebiotic effect of two grams of lactulose in healthy Japanese women: a randomised, double-blind, placebo-controlled crossover trial. Benef Microbes. 2019; 10(6): 629-639.
CrossRef - Hong K. B., Kim J. H., and Kwon H. K., Han S. H., Park Y., Suh H. J. Evaluation of Prebiotic Effects of High-Purity Galactooligosaccharides in vitro and in vivo. Food Technol Biotechnol. 2016; 54(2): 156-163.
CrossRef - Furrie E., Macfarlane S., Kennedy A., Cummings J. H., Walsh S. V., O’neil D. A., Macfarlane G. T. Synbiotic therapy (Bifidobacterium longum/Synergy 1) initiates resolution of inflammation in patients with active ulcerative colitis: a randomised controlled pilot trial. 2005; 54(2): 242-249.
CrossRef - Kanamori Y., Hashizume K., Sugiyama M., Morotomi M., Yuki N. Combination Therapy with Bifidobacterium breve, Lactobacillus casei, and Galactooligosaccharides Dramatically Improved the Intestinal Function in a Girl with Short Bowel Syndrome. Dig Dis Sci. 2001; 46(9): 2010-2016.
CrossRef - Chaiyasut C., Pattanandecha T., Sirilin S., Suwannalert P., Peerajan S., Sivamaruthi B. S. Synbiotic preparation with Lactic acid bacteria and inulin as a functional food: In vivo evaluation of microbial activities, and pre-neoplastic aberrant crypt foci. Food Sci. Technol. 2017; 37(2): 328-336.
CrossRef - Moroti C., Souza Magri L. F., de Rezende Costa M., Cavallini D. C., Sivieri K. Effect of the consumption of a new symbiotic shake on glycemia and cholesterol levels in elderly people with type 2 diabetes mellitus. Lipids Health Dis. 2012; 11(1): 29.
CrossRef - Wu G. D., Chen J., Hoffmann C., Bittinger K., Chen Y. Y., Keilbaugh S. A., Bewtra M., Knights D., Walters W. A., Knight R., Sinha R., Gilroy E., Gupta K., Baldassano R., Nessel L., Li H., Bushman F. D., Lewis J. D. Linking Long-Term Dietary Patterns with Gut Microbial Enterotypes. 2011; 334(6052): 105-108.
CrossRef - Devkota S., Wang Y., Musch M. W., Leone V., Fehlner-Peach H., Nadimpalli A., Antonopoulos D. A., Jabri B., Chang E. B. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10−/− mice. Nature. 2012; 487(7405): 104-108.
CrossRef - Slavin J. Why whole grains are protective: biological mechanisms. Proc Nutr Soc. 2003; 62(1): 129-134.
CrossRef - McIntosh G. H., Noakes M., Royle P. J., Foster P. R. Whole-grain rye and wheat foods and markers of bowel health in overweight middle-aged men. Am J Clin Nutr. 2003; 77(4): 967-974.
CrossRef - Duncan S. H., Belenguer A., Holtrop G., Johnstone A. M., Flint H. J., Lobley G. E. Reduced Dietary Intake of Carbohydrates by Obese Subjects Results in Decreased Concentrations of Butyrate and Butyrate-Producing Bacteria in Feces. Appl Environ Microbiol. 2006; 73(4): 1073-1078.
CrossRef - Alenghat T., Artis D. Epigenomic regulation of host–microbiota interactions. Curr Opin Immunol. 2014; 35(11): 518-525.
CrossRef - Miyamoto J., Kasubuchi M., Nakajima A., Irie J., Itoh H., Kimura I. Microbial Short-Chain Fatty Acids and Blood Pressure Regulation. Curr Opin Nephrol Hypertens. 2017; 19(4):25.
CrossRef - Zheng L., Kelly C. J., Battista K. D., Schaefer R., Lanis J. M., Alexeev E. E., Wang R. X., Onyiah J. C., Kominsky D. J., Colgan S. P. Microbial-Derived Butyrate Promotes Epithelial Barrier Function through IL-10 Receptor–Dependent Repression of Claudin-2. J Immunol. 2017; 199(8): 2976-2984.
CrossRef - Haghikia A., Jörg S., Duscha A., Berg J., Manzel A., Waschbisch A., Hammer A., Lee D. H., May C., Wilck N., Balogh A., Ostermann A. I., Schebb N. H., Akkad D. A., Grohme D. A., Kleinewietfeld M., Kempa S., Thöne J., Demir S., Müller D. N., Gold R., Linker R. A. Dietary Fatty Acids Directly Impact Central Nervous System Autoimmunity via the Small Intestine. 2015; 43(4): 817-829.
CrossRef - Thorburn A. N., Macia L., Mackay C. R. Diet, Metabolites, and “Western-Lifestyle” Inflammatory Diseases. 2014; 40(6): 833-842.
CrossRef - Alvarez C. S., Badia J., Bosch M., Giménez R., Baldomà L. Outer Membrane Vesicles and Soluble Factors Released by Probiotic Escherichia coli Nissle 1917 and Commensal ECOR63 Enhance Barrier Function by Regulating Expression of Tight Junction Proteins in Intestinal Epithelial Cells. Front Microbiol. 2016; 7:1981.
CrossRef - Oz S., Okay E., Karadenizli A., Cekmen M. B., Ozdogan H. K. N-acetylcysteine improves intestinal barrier in partially hepatectomized rats. ANZ J Surg. 2007; 77(3): 173-176.
CrossRef - Xu R., Tao A., Bai Y., Deng Y., Chen G. Effectiveness of N ‐Acetylcysteine for the Prevention of Contrast‐Induced Nephropathy: A Systematic Review and Meta‐Analysis of Randomized Controlled Trials. J Am Heart Assoc. 2016; 5(9): E003968.
CrossRef - Douiev L., Soiferman D., Alban C., Saada A. The Effects of Ascorbate, N-Acetylcysteine, and Resveratrol on Fibroblasts from Patients with Mitochondrial Disorders. J Clin Med. 2016; 6(1): 1.
CrossRef - Zhenyukh O., Civantos E., Ruiz-Ortega M., Sánchez M. S., Vázquez C., Peiró C., Egido J., Mas S. High concentration of branched-chain amino acids promotes oxidative stress, inflammation and migration of human peripheral blood mononuclear cells via mTORC1 activation. Free Radic Biol Med. 2017; 104: 165-177.
CrossRef - Cohen L. J., Esterhazy D., Kim S. H., Lemetre C., R. Rhiannon, Aguilar, Gordon E. A., Pickard A. J., Cross J. R., Emiliano A. B., Han S. M., Chu J., Vila-Farres X., Kaplitt J., Rogoz A., Calle P. Y., Hunter C., Bitok J. K., Brady S. F. Commensal bacteria make GPCR ligands that mimic human signalling molecules. 2017; 549(7670): 48-53.
CrossRef - Hamady Z., Scott N., Farrar M. D., Wadhwa M., Dilger P., Whitehead T. R., Thorpe R., Holland K. T., Lodge J. P., Carding S. R. Treatment of colitis with a commensal gut bacterium engineered to secrete human tgf-β1 under the control of dietary xylan. Inflamm Bowel Dis. 2011; 17(9): 1925-1935.
CrossRef - Vandenbroucke K., de Haard H., Beirnaert E., Dreier T., Lauwereys M., Huyck L., Van Huysse J., Demetter P., Steidler L., Remaut E., Cuvelier C., Rottiers P. Orally administered L. lactis secreting an anti-TNF Nanobody demonstrates efficacy in chronic colitis. Mucosal Immunol. 2009; 3(1): 49-56.
CrossRef - Motta J., Bermudez-Humaran L. G., Deraison C., Martin L., Rolland C., Rousset P., Boue J., Dietrich G., Chapman K., Kharrat P., Vinel J. P., Alric L., Mas E., Sallenave J. M., Langella P., Vergnolle N. Food-Grade Bacteria Expressing Elafin Protect Against Inflammation and Restore Colon Homeostasis. Sci Transl Med. 2012; 4(158): 158ra144-158ra144.
CrossRef - Timmis K., Timmis J. K., Brüssow H., Fernández L. Á. Synthetic consortia of nanobody-coupled and formatted bacteria for prophylaxis and therapy interventions targeting microbiome dysbiosis-associated diseases and co-morbidities. Microb Biotechnol. 2018; 12(1): 58-65.
CrossRef - Chen Z., Guo L., Zhang Y., Walzem R. L., Pendergast J. S., Printz R. L., Morris L. C., Matafonova E., Stien X., Kang L., Coulon D., McGuinness O. P., Niswender K. D., Davies S. S. Incorporation of therapeutically modified bacteria into gut microbiota inhibits obesity. J Clin Invest. 2014; 124(8): 3391-3406.
CrossRef - Hammer H. F. Gut Microbiota and Inflammatory Bowel Disease. Dig Dis. 2011; 29(6): 550-553.
CrossRef - Suchodolski J. S., Dowd S. E., Westermarck E., Steiner J. M., Wolcott R. D., Spillmann T., Harmoinen J. A. The effect of the macrolide antibiotic tylosin on microbial diversity in the canine small intestine as demonstrated by massive parallel 16S rRNA gene sequencing. BMC Microbiol. 2009 9(1): 210.
CrossRef - Pappo I., Becovier H., Berry E. M., Freund H. R. Polymyxin B reduces cecal flora, TNF production and hepatic steatosis during total parenteral nutrition in the rat. J Surg Res. 1991; 51(2):106-112.
CrossRef - Menozzi A., Dall’Aglio M., Quintavalla F., Dallavalle L., Meucci V., Bertini S. Rifaximin is an effective alternative to metronidazole for the treatment of chronic enteropathy in dogs: a randomised trial. BMC Vet Res. 2016; 12(1):217.
CrossRef - Gerard L., Garey K. W., DuPont H.L. Rifaximin: a nonabsorbable rifamycin antibiotic for use in nonsystemic gastrointestinal infections. Expert Rev Anti Infect Ther. 2005; 3(2): 201-211.
CrossRef - Sharma M., Thandassery R. B., Bhargava N. Double Pylorus: An Optical Illusion or Reality? Gastroenterology. 2012; 143(2): e7-e8.
CrossRef - Van Nood E., Vrieze A., Nieuwdorp M., Fuentes, S., Zoetendal, E.G., de Vos, W.M., Visser, C.E., Kuijper, E.J., Bartelsman, J.F., Tijssen, J.G. and Speelman, P.,. Duodenal Infusion of Donor Feces for Recurrent Clostridium difficile. N Engl J Med. 2013; 368(5): 407-415.
CrossRef - Rossen N. G., Fuentes S., van der Spek M. J., Tijssen J. G., Hartman J. H., Duflou A., Löwenberg M., van den Brink G. R., Mathus-Vliegen E. M., de Vos W. M., Zoetendal E. G., D’Haens G. R., Ponsioen C. Y. Findings From a Randomized Controlled Trial of Fecal Transplantation for Patients With Ulcerative Colitis. Gastroenterology. 2015; 149(1): 110-118.e4.
CrossRef - Angelberger S., Reinisch W., Makristathis A., Lichtenberger C., Dejaco C., Papay P., Novacek G., Trauner M., Loy A., Berry D. Temporal Bacterial Community Dynamics Vary Among Ulcerative Colitis Patients After Fecal Microbiota Transplantation. Am J Gastroenterol. 2013; 108(10): 1620-1630.
CrossRef - Kump P. K., Gröchenig H.P., Lackner S., Trajanoski S., Reicht G., Hoffmann K. M., Deutschmann A., Wenzl H. H., Petritsch W., Krejs G. J., Gorkiewicz G., Högenauer C. Alteration of Intestinal Dysbiosis by Fecal Microbiota Transplantation Does not Induce Remission in Patients with Chronic Active Ulcerative Colitis. Inflamm Bowel Dis. 2013; 19(10): 2155-2165.
CrossRef - Khan I., Ullah N., Zha L., Bai Y., Khan A., Zhao T., Che T., Zhang C. Alteration of Gut Microbiota in Inflammatory Bowel Disease (IBD): Cause or Consequence? IBD Treatment Targeting the Gut Microbiome. Pathogens. 2019; 8(3): 126.
CrossRef - Li M., Li Z., Wen S., Liu Y., Wang Y., Tang L. Transplantation of a bacterial consortium ameliorates trinitrobenzenesulfonic acid-induced colitis and intestinal dysbiosis in rats. Future Microbiol. 2016; 11(7): 887-902.
CrossRef - Petrof E. O., Gloor G. B., Vanner S. J., Weese S. J., Carter D., Daigneault M. C., Brown E. M., Schroeter K., Allen-Vercoe E. Stool substitute transplant therapy for the eradication of Clostridium difficile infection: ‘RePOOPulating’ the gut. Microbiome. 2013; 1(1): 1-3.
CrossRef - Li M., Liang P., Li Z., Wang Y., Zhang G., Gao H., Wen S., Tang L. Fecal microbiota transplantation and bacterial consortium transplantation have comparable effects on the re-establishment of mucosal barrier function in mice with intestinal dysbiosis. Front Microbiol. 2015; 6:692.
CrossRef - Steeland S., Vandenbroucke R. E., Libert C. Nanobodies as therapeutics: big opportunities for small antibodies. Drug Discov Today. 2016; 21(7): 1076-1113.
CrossRef - Sausset R., Petit M. A., Gaboriau-Routhiau V., De Paepe M. New insights into intestinal phages. Mucosal Immunol. 2020; 13(2): 205-215.
CrossRef - Scarpellini E., Ianiro G., Attili F., Bassanelli C., De Santis A., Gasbarrini A. The human gut microbiota and virome: Potential therapeutic implications. Dig Liver Dis. 2015; 47(12): 1007-1012.
CrossRef - Chaudhry W. N., Concepción-Acevedo J., Park T., Andleeb S., Bull J. J., Levin B. R. Synergy and Order Effects of Antibiotics and Phages in Killing Pseudomonas aeruginosa Biofilms. PLOS ONE. 2017; 12(1): e0168615.
CrossRef - Yadav H., Lee J., Lloyd J., Walter P., Rane S. G. Beneficial Metabolic Effects of a Probiotic via Butyrate-induced GLP-1 Hormone Secretion. J Biol Chem. 2013; 288(35): 25088-25097.
CrossRef - Vrieze A., Van Nood E., Holleman F., Salojärvi J., Kootte R. S., Bartelsman J. F., Dallinga-Thie G. M., Ackermans M. T., Serlie M. J., Oozeer R., Derrien M., Druesne A., Van Hylckama Vlieg J. E., Bloks V. W., Groen A. K., Heilig H. G., Zoetendal E. G., Stroes E. S., de Vos W. M., Hoekstra J. B., Nieuwdorp M. Transfer of Intestinal Microbiota From Lean Donors Increases Insulin Sensitivity in Individuals With Metabolic Syndrome. Gastroenterology. 2012; 143(4): 913-916.e7.
CrossRef - Bloomfield, S. F., Rook, G. A., Scott, E. A., Shanahan, F., Stanwell-Smith, R., & Turner, P. Time to abandon the hygiene hypothesis: new perspectives on allergic disease, the human microbiome, infectious disease prevention and the role of targeted hygiene. Perspectives in Public Health. 2016; 136(4): 213-224.
CrossRef - Goto Y, Kiyono H. Epithelial barrier: an interface for the cross-communication between gut flora and immune system. Immunol Rev. 2012; 245(1):147-63.
CrossRef - Chen, P., Torralba, M., Tan, J., Embree, M., Zengler, K., Stärkel, P., Van Pijkeren, J.P., DePew, J., Loomba, R., Ho, S.B. and Bajaj, J.S. Supplementation of saturated long-chain fatty acids maintains intestinal eubiosis and reduces ethanol-induced liver injury in mice. Gastroenterology. 2015; 148(1): 203-214.
CrossRef - Anna, T., Alexey, K., Anna, B., Vyacheslav, K., Mikhail, T. and Ulia, M. Effect of in vitro gastrointestinal digestion on bioactivity of poultry protein hydrolysate. Current Research in Nutrition and Food Science Journal.2016;4:77-86.
CrossRef - Baghlaf M. A, Eid N. M. S. Prevalence, Risk Factors, Clinical Manifestation, Diagnosis Aspects and Nutrition Therapy in Relation to both IgE and IgG Cow’s Milk Protein Allergies among a Population of Saudi Arabia: A Literature Review. Curr Res Nutr Food Sci. 2021; 9(2).
CrossRef - Nuryani N, Muhdar I. N, Ramadhani F, Paramata Y, Adi D. I, Bohari B. Association of Physical Activity and Dietary Patterns with Adults Abdominal Obesity in Gorontalo Regency, Indonesia: A Cross-Sectional Study. Curr Res Nutr Food Sci. 2021; 9(1).
CrossRef - Shikha Srivastava S, Siddiqi Z, Singh T, Bala L. Beetroot Supplementation on Non-Alcoholic Fatty Liver Disease Patients. Curr Res Nutr Food Sci. 2019; 7(1).
CrossRef - Yu, L.CH. Microbiota dysbiosis and barrier dysfunction in inflammatory bowel disease and colorectal cancers: exploring a common ground hypothesis. J Biomed Sci. 2018; 25(1): 79.
CrossRef - Boland, K., Bedrani, L., Turpin, W., Kabakchiev, B., Stempak, J., Borowski, K., Nguyen, G., Steinhart, A.H., Smith, M.I., Croitoru, K. and Silverberg, M.S. Persistent diarrhea in patients with Crohn’s disease after mucosal healing is associated with lower diversity of the intestinal microbiome and increased dysbiosis.Clin Gastroenterol Hepatol. 2021; 19(2):296-304.
CrossRef - Tanaka, N., Kimura, T., Fujimori, N., Nagaya, T., Komatsu, M. and Tanaka, E. Current status, problems, and perspectives of non-alcoholic fatty liver disease research.World J Gastroenterol. 2019; 25(2): 163.
CrossRef - Kim, J., Yun, J.M., Kim, M.K., Kwon, O. and Cho, B. Lactobacillus gasseri BNR17 supplementation reduces the visceral fat accumulation and waist circumference in obese adults: a randomized, double-blind, placebo-controlled trial. J Med Food. 2018;21(5):454-461.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.






