Introduction
Indonesia, especially in the Barrang Lompo Island, South Sulawesi Province, is a famous area for the diversity of species of invertebrate animals such as starfish (Asterias vulgaris), snake star (Ophioderma superba), sea urchin (Diadema setosum), sea urchin (Echinus esculentus) and many more. Sea urchins are small and spiny, has a high selling value and mostly consumed by Japanese people (sushi),1 South America and France as well as in the United States (Boston, California, New York, British Columbia).2 The shell that is known to contain various pigments is polyhydroxylated naphthoquinone (PHNQ) spinochromes3 of antibacterial effect. Sea urchin part gonads have potential as antibacterial because has a compound polyhydroxylated naphthoquinone,4 according to research,5 extraction polyhydroxylated naphthoquinone from the spines and shell sea urchin evechinus chloroticus from New Zealand with six separate macroporous resins as an alternative to organic solvent extraction. The instrument HPLC and GC-MS have been found to be susceptible to degradation in light exposure, with PHNQ aminated being the least stable. Research,6 extracting sea urchin Echinometra mathaei shell and spine parts by using a solvent diethyl ether. Screening uses HPLC instrument and radical scavenging assay (antioxidant) 1-diphenyl-2 picrylhydrazyl. It is confirmed with the use of photodioid array detector and LC–ESI – MS PHNQ acquired (Spinochrome B and C, Echinochrome A and Spinochrome A). Results show that marine urchin shells and spines, which are mainly disposed of as waste, can act as a new biologically active resource.
The compounds’ chemical nature influences the extraction of phenolic and bacterial compounds from sea urchin materials, sample size, method of extraction, time and storage and the occurrence of agents such as proteins and carbohydrates.7–9 Has been reported that the use of aquades solutions of ethanol, methanol, and acetone dramatically improves the extraction of polyphenols compared to a single-compound solvent system.10 Extraction methods that most reported is maceration with solvents, hot-water extraction, alkaline extraction, resin-based extraction, enzyme-assisted extraction, extractions based on gamma and electron-beam irradiation and extraction using supercritical fluids. Some of these methods can cause a loss of bioactive compounds due to the use of high temperatures and long extraction times, or, in the case of irradiation, it can represent a health risk if the proper care does not take.11 Sustainable innovative green techniques have been used to reduce time, to consume energy, to increase the efficiency of the extraction and to help preserve the natural environment, by reducing the use of solvents and water in Ultrasound-Assisted Extraction (UAE) and Ultrasound Microwave-Assisted Extraction (UMAE),12 generation of hazardous substances and fossil energy.13–15 Ultrasonic radiation use power 20-100 kHz to extract natural compounds provides high reproducibility, secure handling, low solvent, and energy consumption, low-temperature processing and a smaller loss of bioactive compounds.16 The extraction techniques can be compared with the microwave-assisted extraction, the ultrasound equipment is cheaper and the operation is easier. In addition, ultrosonic extraction, such as maceration, can be used for the extraction of a wide range of natural compounds with any solvent.17
Ultrasound can hydration and facilitate swelling of vegetal tissue. According to research,10 ultrasound can allow high rates of diffusion across the cell and increase the transfer of mass. On the other hand, cavitation produced by ultrasonic waves can also disrupt the cell, and then release the contents. In research,18 the reported use frequency of 25 kHz from orange peel using an ultrasonic processor operated can produce higher extraction yields of polyphenols. Sonication is a faster, simpler, and more effective technique than maceration to extract organic compounds.19 Extraction of a bioactive compound from sea urchin using Ultrasonic Assisted Extraction (UAE). Therefore, the aim of this study was to assess the effect of ultrasonic treatment on the total bioactive compound content of the sea urchin and shell gonad.18
Material and Methods
Sea Urchin
Sea urchin (Diadema setosum) was obtained by trawling off the coast Barrang Lompo Island in Makassar, South Sulawesi, Indonesia. The gonads separated from the sea urchin shell (fig.1), then washed to remove other components and taken to the laboratory by carrying in the coolbox, and stored in the freezer (-20oC) until the gonads and shell sea urchin processed in Food Science and Instrumental Analysis Laboratory, Chemical Engineering Department, Politeknik Negeri Ujung Pandang, Indonesia.
Chemicals and Tools
All chemicals subtances were of analytical grade, methanol (CAS: 67-56-1), aquadestilate (CAS: 7732-18-5), and ethyl acetate (CAS: 141-78-6), supplied by Merck Millipore (Burlington, Massachusetts, United States). The tools used are water bath (Memmert WNB 7 Basic control) Hettich Zentrifugen EBA-20, and Hitachi centrifuge brands, Ultrasonic Assisted Extraction instrument (Elmasonic P30), Shimadzu GC-MS 2010 brand Gas Chromatography-Mass Spectrometry plus.
Preparation of Extract
Sea urchin was bled to death; tissues and various organs dissected out carefully and collected. Sea urchin divided into intestinal organs, gills, eggs, and body walls (including plates, feet, and spines).20 After removal of the internal organs (shell and gonad) a stream of cold water washed and cut into small pieces.
 |
Figure 1: Sea urchin (Diadema setosum): a) Sea urchin harvest in the sea by fishers; b) Sea urchin collected; c) Sea urchin shell, and 4) Sea urchin gonad Click here to View figure |
Solvent Extraction
The extraction process began with the weighing stage of the sample, two hundred grams of sea
urchin (gonad and shells) and 300 ml of methanol, added in a 500 ml glass flask. The mixture put in a water bath (Elmasonic P30 instrument) with a power of 40 kHz at 35°C for 30 min and 60 min to remove bioactive compounds from the sea urchin materials.17 Same treatment for ethyl acetate solvents. The extract was purified and evaporated at 39oC by a rotary evaporator, working principle of the rotary evaporator not only lies in heating but by lowering the pressure and regulating the velocity at a certain point so that the solvent methanol and ethyl acetate will evaporate and the soluble compounds in the solvent do not follow Evaporate but settlers. The boiling point of methanol and ethyl acetate solvent ranges from 64.7oC and 77.1oC, by heating below the solvent boiling point, so that the compounds in the solvent are not affected at high temperatures.17 The high polarity and volatility of methanol and ethyl acetate was used as solvent to enhance yield and concentrate the desired compounds.21 Methanol and ethyl acetate were dissolved into the sea urchin.22 The solvent will evaporate perfectly when the evaporation process on the rotary evaporator until obtained the solvent that has not dripped again on the round base flask and can also be seen with the more potent substances present in the sample round base flask So that the bubbles are formed on the surface of substances.23 The yield of sea urchin (%) is the ratio of the resulting sea urchin (g) compared to the sample weight used in the process (g). The formula calculates the calculation of the yield:

The Steps of Ultrasonic-Assisted Extraction (UAE)
Elmasonic Instrument P30 is paired with a cooling coil operated heating system (Fisher Scientific Inc St Louis, USA); and a cooling system for chillers; and water pumps (HJ-111 versions, 250 L/h submersible pumps, Sunshine Inc., Zhejiang, China). Ultrasonic waves are produced locally from micro cavitations around the material to be extracted so that the material is heated and the extract compounds are released. Double effects are generated, i.e. the cell wall screed to release the compound content in and the local heating in liquids and to increase the diffusion of the extracts. Variables selected temperatures of 30°C, 40°C, and 50°C; power settings of 30%, 50%, and 70%; and high durations of 10 min, 20 min, and 30 min. Cinetic energy is transferred to all parts of the fluid, followed by the formation of cavitation bubbles on the wall or surface, thereby increasing the mass transfer between the solid-liquid surfaces. The mechanical effect is to increase the penetration of the fluid into the cell membrane wall, support the release of the cell components and increase the transfer of mass.24 Research25 states that ultrasonic cavitation generates broken power that breaks the cell wall mechanically and improves material transfer. The variety of solvents used for extraction in Table 1.
Table 1: Ultrasonic Assisted Extraction with Variations of Solvents and Time Extraction.
| Ingredients | Sea urchin (Shell) | Sea urchin (gonad) | ||
| Methanol (min) | Ethyl acetate (min) | Methanol (min) | Ethyl acetate (min) | |
| Sea urchin (Diadema setosum) | 30 | 30 | 30 | 30 |
| 60 | 60 | 60 | 60 | |
Analysis of Bioactive Compounds (Gas Chromatography-Mass Spectrometry)
In a Shimadzu GCMS-QP 2010 Plus SHIMADZU instrument under computer control at 70 eV, the bioactive compound was determined using GC-MS analysis.26 About 1 μl of the methanol extract injected into the GC-MS using a microsyringe, and complete scanning in 45 min. Starting from the phase selection of the motion or commonly called the gas carrier specified according to the analysis of the sample to be performed. In this research, gas carrier He (Helium) with Principal level is 99.99%, and temperature maintained at 70-280°C or UHP (Ultra High Purity) type. Then the gas carrier is channelled through Stainless Steel tubing, and the pressure value of the gas carrier entered in Gas Chromatography is controlled by the AFC (Advanced Flow Controller) through the analysis software used by GC-Solution. The column used Elite 1 ( 100% dimethyl polysiloxane) to isolate components.27,28 The Advanced Flow Controller (AFC) controls the flow going into the column. Compounds were then identified by comparing their spectra of the spectral mass libraries of the Wiley and NIST/EPA/NIH.
Results
Extraction Yield of Sea Urchin
The yield of each sample was recorded on the dry basis of sample weight from 0.18 to 4.61% using ultrasonic-assisted extraction method and methanol or ethyl acetate solvents, as shown in Fig. 2. Maximum extraction yields of 1.61% and 4.61% were obtained from shell and gonad sea urchin, respectively, with methanol solvents at 60 min.
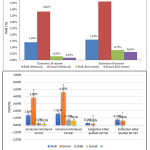 |
Figure 2: The yield of sea urchin: a) Total yield per solvent; b) Values were presented as mean + SD (n=12) Click here to View figure |
Unlike the above, the minimum yield is achieved by using ethyl acetate solvent at 30 min was 0.29% and 0.18% for shell and gonad sea urchin (Diadema setosum), respectively. Methanol solvents are capable of extracting components derived from alkaloids, phenolic, rubberonoid, tannin, amino acids, sugars, and glycosides. Besides, that methanol can destroy cell walls, so that components to break down and dissolve in methanol solvents.29,30 Standard deviation (SD) test results indicate that SD value is smaller than the mean value, so data is not significant (P < 0.05), mean the yield amount does not affect the number of bioactive compounds produced by each solvent in a sample of a sea urchin gonads and shell.
The bioactive compound of sea urchin shell with methanol solvent extraction
GC-MS test has performed showing the presence of 50 idealized compound components in methanol extract of sea urchin shell (Fig. 3), and 20 compounds identified in methanol extract of sea urchin gonads (Fig. 4). Some of the compounds detected at methanol extract of sea urchin shell, and gonad contains antibacterial substances. These antibacterial compounds include pentadecanoat acid, palmitic acid, stearic acid, ethyl cyclohexane, Tetradecanoic acid (Stearic acid), Phthalic Acid, 1-Eicosanol, and Ergosterol. Another study done by31 informed that the white Oyster mushroom alcohol extract contains palmitic acid, 9.12-octadecadienoic acid, methyl ester (E, E)-18.55, 14.17-acid Oktadekadenoat, cholestatic-3, 7, 1, 25-tetraacetic extracts, methyl ester (E, E)-5.59, ethyl ester-3.84, (3a, 5a, 7a, 12a)-55.20 is potentially an antibacterial agent. The analysis of the chemical components of the sea urchin shell and gonad methanol extract using GC-MS showed several active compounds that serve as antioxidant, antitumor, anticancer, analgesic agent and anti-inflammatory drugs. These active compounds include arachidonic acid, batilol, methyl ester, methyl ester, 9-octadecenoic acid (Z)-, tetradecanoic acid, and oleic acid. Analysis results also show that antibacterial components and anti-inflammatory drugs have the highest content, among other compounds. The antibacterial part detected is Cholest-5-EN-3-OL (3. Beta.) containing 39.51%.
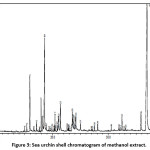 |
Figure 3: Sea urchin shell chromatogram of methanol extract. Click here to View figure |
 |
Table 1: Major phytochemicals identified by a sea urchin shell methanol extract. Click here to View Table |
A Bioactive compound of sea urchin gonad with methanol solvent extraction
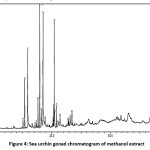 |
Figure 4: Sea urchin gonad chromatogram of methanol extract. Click here to View figure |
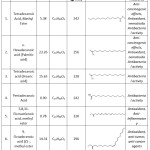 |
Table 2: Major phytochemicals identified by a sea urchin gonad methanol extract. Click here to View Table |
The bioactive compound of sea urchin shell with ethyl acetate solvent extraction
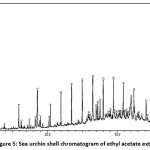 |
Figure 5: Sea urchin shell chromatogram of ethyl acetate extract. Click here to View figure |
The GC-MS test showed the presence of 60 idealized compound components in the sea urchin extract of ethyl acetate (Fig. 5), and 60 compounds identified in the extract of ethyl acetate from sea urchin gonades (Fig. 6). Some compounds found in ethyl acetate extract of sea urchin shell and gonad contain antibacterial and anti-fungal substances. These antibacterial compounds include pentadecanoat acid, Cholest-5-EN-3-OL (3. BETA.), Stigmast-8 (14) -EN-3.BETA.-OL, palmitic acid, stearic acid, ethyl cyclohexane, tetradecanoic acid (stearic acid), phthalic acid, pentacosane, and ergosterol. The analysis of the chemical components of the sea urchin shell and gonad ethyl acetate extract using GC-MS showed several active compounds that serve as an antioxidant, antitumour, anticancer, analgesic agent and anti-inflammatory drugs. These active compounds include 9-octadecenoic acid (Z) -, methyl ester, pentacosane, 2,2-Dimethylpropanoic acid, p-Anisic acid, benzenepropanoic acid, 3,5-bi’s (1,1-dimethyl ethyl) -4-hydroxy-, pentatriacontane, n-hexadecanoic acid or palmitic acid, Cholest-5-EN-3-YL acetate, and octadecyl ester. Some of the octacosanol compounds amounting to 0.81% (total amount) function as Anti-fatigue and Anti-Parkinson’s effects. As well as a small fraction of 2.81% of the gamma.-sitosterol A compound can function as an antihyperglycemic. Analysis results also show that antibacterial components and anti-inflammatory drugs have the highest content, among other compounds. The most significant content is CholesT-5-EN-3-OL (3. beta.) or 46.24% steroid as an antibacterial agent.
 |
Table 3: Major phytochemicals identified by a sea urchin shell ethyl acetate extract. Click here to View Table |
A Bioactive compound of sea urchin gonad with ethyl acetate solvent extraction
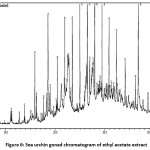 |
Figure 6: Sea urchin gonad chromatogram of ethyl acetate extract. Click here to View figure |
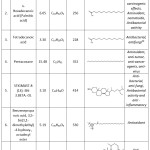 |
Table 4: Major phytochemicals identified by a sea urchin gonad methanol extract. Click here to View Table |
Conclusion
This study resulted in data types of bioactive compounds, quantities, benefits and phytochemical characteristics of sea urchin gonads and shells. Sea urchin (Diadema setosum) was obtained by trawling off the coast Barrang Lompo Island in Makassar, South Sulawesi, Indonesia. Contains chemical constitutions that are useful for different herbal formulations as anti-tumour, anti-cancer, anti-inflammatory, analgesic, anti-fatigue, anti-Parkinson effect, and anti-hypoglycemic. And potentially as a preservative in food and beverages because it is as antibacterial and antifungal.
Suggestions
Need to do further research to uses ultrasonic microwave-assisted extraction in extracting the sea urchin
Acknowledgement
The authors are thankful to the Research Institute of Politeknik Negeri Ujung Pandang, which facilitated and funded this research.
References
- Kuwahara R, Hatate H, Chikami A, Murata H, Kijidani Y. Quantitative separation of antioxidant pigments in purple sea urchin shells using a reversed-phase high performance liquid chromatography. LWT – Food Sci Technol. 2010. doi:10.1016/j.lwt.2010.03.005
- Amarowicz R, Synowiecki J, Shahidi F. Chemical composition of shells from red (Strongylocentrotus franciscanus) and green (Strongylocentrotus droebachiensis) sea urchin. Food Chem. 2012;133(3):822-826. doi:10.1016/j.foodchem.2012.01.099
- Anderson HA, Mathieson JW, Thomson RH. Distribution of spinochrome pigments in echinoids. Comp Biochem Physiol. 1969. doi:10.1016/0010-406X(69)91347-4
- Lebedev A V., Levitskaya EL, Tikhonova E V., Ivanova M V. Antioxidant properties, autooxidation, and mutagenic activity of echinochrome as compared with its etherified derivative. Biokhimiya. 2001.
- Hou Y, Vasileva EA, Mishchenko NP, Carne A, McConnell M, Bekhit AEDA. Extraction, structural characterization and stability of polyhydroxylated naphthoquinones from shell and spine of New Zealand sea urchin (Evechinus chloroticus). Food Chem. 2019. doi:10.1016/j.foodchem.2018.08.046
- Soleimani S, Yousefzadi M, moein S, Rezadoost H, Bioki NA. Identification and antioxidant of polyhydroxylated naphthoquinone pigments from sea urchin pigments of Echinometra mathaei. Med Chem Res. 2016. doi:10.1007/s00044-016-1586-y
- Koffi, Dodehe T, 4 SS. Effect of solvent type on extraction of polyphenols from twenty three Ivorian plants. J Anim Plant Sci. 2010.
- García-Márquez E, Román-Guerrero A, Cruz-Sosa F, Pérez-Alonso C, Jiménez-Alvarado R, Vernon-Carter EJ. Effect of solvent-temperature extraction conditions on the initial antioxidant activity and total phenolic content of muitle extracts and their decay upon storage at different pH. Rev Mex Ing Qum. 2012.
- Guneidy RA, Shahein YE, Abouelella AMK, Zaki ER, Hamed RR. Inhibition of the recombinant cattle tick Rhipicephalus (Boophilus) annulatus glutathione S-transferase. Ticks Tick Borne Dis. 2014. doi:10.1016/j.ttbdis.2014.03.009
- Akhbari M, Hamedi S, Aghamiri Z sadat. Optimization of total phenol and anthocyanin extraction from the peels of eggplant (Solanum melongena L.) and biological activity of the extracts. J Food Meas Charact. 2019. doi:10.1007/s11694-019-00241-1
- Liu CT, Wu CY, Weng YM, Tseng CY. Ultrasound-assisted extraction methodology as a tool to improve the antioxidant properties of herbal drug Xiao-chia-hu-tang. J Ethnopharmacol. 2005. doi:10.1016/j.jep.2005.02.018
- Piccolella S, Crescente G, Nocera P, Pacifico F, Manti L, Pacifico S. Ultrasound-assisted aqueous extraction, LC-MS/MS analysis and radiomodulating capability of autochthonous Italian sweet cherry fruits. Food Funct. 2018. doi:10.1039/c7fo01977g
- Chemat F, Rombaut N, Sicaire AG, Meullemiestre A, Fabiano-Tixier AS, Abert-Vian M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason Sonochem. 2017. doi:10.1016/j.ultsonch.2016.06.035
- Zhou P, Wang X, Liu P, et al. Enhanced phenolic compounds extraction from Morus alba L. leaves by deep eutectic solvents combined with ultrasonic-assisted extraction. Ind Crops Prod. 2018. doi:10.1016/j.indcrop.2018.04.071
- Adeel S, Rehman F ur, Iqbal MU, et al. Ultrasonic assisted sustainable dyeing of mordanted silk fabric using arjun (Terminalia arjuna) bark extracts. Environ Prog Sustain Energy. 2019. doi:10.1002/ep.13048
- Pan Z, Qu W, Ma H, Atungulu GG, McHugh TH. Continuous and pulsed ultrasound-assisted extractions of antioxidants from pomegranate peel. Ultrason Sonochem. 2012. doi:10.1016/j.ultsonch.2011.05.015
- Wang L, Weller CL. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci Technol. 2006. doi:10.1016/j.tifs.2005.12.004
- Khan MK, Abert-Vian M, Fabiano-Tixier AS, Dangles O, Chemat F. Ultrasound-assisted extraction of polyphenols (flavanone glycosides) from orange (Citrus sinensis L.) peel. Food Chem. 2010. doi:10.1016/j.foodchem.2009.08.046
- Jacques RA, dos Santos Freitas L, Pérez VF, et al., The use of ultrasound in the extraction of Ilex paraguariensis leaves: A comparison with maceration. Ultrason Sonochem. 2007. doi:10.1016/j.ultsonch.2005.11.007
- Haug T, Kjuul AK, Styrvold OB, Sandsdalen E, Olsen ØM, Stensvåg K. Antibacterial activity in Strongylocentrotus droebachiensis (Echinoidea), Cucumaria frondosa (Holothuroidea), and Asterias rubens (Asteroidea). J Invertebr Pathol. 2002;81(2):94-102. doi:10.1016/S0022-2011(02)00153-2
- Yusuf M, Indriati S, Attahmid NFU. KARAKTERISASI ANTOSIANIN KUBIS MERAH SEBAGAI INDIKATOR PADA KEMASAN CERDAS Characterization Antosianin of Red Cabbage as a Indicator in Smart Packaging. J Galung Trop. 2018;7(April):46-55.
- Altemimi A, Watson DG, Choudhary R, Dasari MR, Lightfoot DA. Ultrasound assisted extraction of phenolic compounds from peaches and pumpkins. PLoS One. 2016. doi:10.1371/journal.pone.0148758
- Teng H, Choi YH. Optimization of ultrasonic-assisted extraction of bioactive alkaloid compounds from rhizoma coptidis (coptis chinensis franch.) using response surface methodology. Food Chem. 2014. doi:10.1016/j.foodchem.2013.06.136
- Keil FJ. Modeling of Process Intensification.; 2007. doi:10.1002/9783527610600
- Liu QM, Yang XM, Zhang L, Majetich G. Optimization of ultrasonic-assisted extraction of chlorogenic acid from Folium eucommiae and evaluation of its antioxidant activity. J Med Plants Res. 2010.
- Mohammad A, Imad H. Autosomal STR: From locus information to next generation sequencing technology. Res J Biotechnol. 2013.
- Hameed IH, Jebor MA, Ommer AJ, Abdulzahra AI, Yoke C. Haplotype data of mitochondrial DNA coding region encompassing nucleotide positions 11,719–12,184 and evaluate the importance of these positions for forensic genetic purposes in Iraq. Mitochondrial DNA. 2016. doi:10.3109/19401736.2014.945576
- Kareem MA, Abdulzahra AI, Hameed IH, Jebor MA. A new polymorphic positions discovered in mitochondrial DNA hypervariable region HVIII from central and north-central of Iraq. Mitochondrial DNA. 2016. doi:10.3109/19401736.2015.1007369
- Anwar F, Przybylski R. Effect of solvents extraction on total phenolics and antioxidant activity of extracts from flaxseed (Linum usitatissimum L.). Acta Sci Pol Technol Aliment. 2012.
- Anwar F, Kalsoom U, Sultana B, Mushtaq M, Mehmood T, Arshad HA. Effect of drying method and extraction solvent on the total phenolics and antioxidant activity of cauliflower (Brassica oleracea L.) Extracts. Int Food Res J. 2013.
- Jananie R, Priya V, Vijayalakshmi K. Determination of bioactive components of Cynodon dactylon by GC-MS analysis. New York Sci J. 2011.
- Vijisaral Elezabeth D, Arumugam S. GC-MS analysis of bioactive constituents of indigofera suffruticosa leaves. J Chem Pharm Res. 2014.
- Graikou K, Kapeta S, Aligiannis N, et al. Chemical analysis of Greek pollen – Antioxidant, antimicrobial and proteasome activation properties. Chem Cent J. 2011. doi:10.1186/1752-153X-5-33
- Cantrell CL, Rajab MS, Franzblau SG, Fronczek FR, Fischer NH. Antimycobacterial ergosterol-5,8-endoperoxide from Ajuga remota. Planta Med. 1999. doi:10.1055/s-1999-14053
- Zhao J, Mou Y, Shan T, et al. Antimicrobial metabolites from the endophytic fungus pichia guilliermondii Isolated from Paris polyphylla var. yunnanensis. Molecules. 2010. doi:10.3390/molecules15117961
- Mujeeb F, Bajpai P, Pathak N. Phytochemical evaluation, antimicrobial activity, and determination of bioactive components from leaves of aegle marmelos. Biomed Res Int. 2014. doi:10.1155/2014/497606
- Park N, Kwon B, Kim SD, Cho J. Characterizations of the colloidal and microbial organic matters with respect to membrane foulants. J Memb Sci. 2006. doi:10.1016/j.memsci.2005.08.020
- Rajeswari G, Murugan M, Mohan VR. GC-MS analysis of bioactive components of Hugonia mystax L. (Linaceae). Res J Pharm Biol Chem Sci. 2012.
- Nedeltcheva-Antonova D, Ivanova D, Antonov L, Abe I. Insight into the aroma profile of Bulgarian tobacco absolute oil. Ind Crops Prod. 2016. doi:10.1016/j.indcrop.2016.08.047
- Agoramoorthy G, Chandrasekaran M, Venkatesalu V, Hsu MJ. Antibacterial and antifungal activities of fatty acid methyl esters of the blind-your-eye mangrove from India. Brazilian J Microbiol. 2007. doi:10.1590/S1517-83822007000400028

This work is licensed under a Creative Commons Attribution 4.0 International License.







