Effect of Sago Analogue Rice and Red Bean Diet to the Improvement of β-cell Pancreas in Streptozotocin-Nicotinamide (STZ-NA) Induced Diabetic Rats
1Department of Agricultural Product Processing, Faculty of Agricultural Technology, Universitas Semarang, Semarang, Indonesia
2Center of Technology Assessment and Application Board, Ministry of Research, Technology, and Higher Education of Republic Indonesia
Corresponding Author Email: sribudiwahjuningsih5678@gmail.com
DOI : http://dx.doi.org/10.12944/CRNFSJ.8.2.32
ABSTRACT:The study aims to determine the effect of sago analogue rice and red beans in diabetic rats to repair pancreatic β-cells. Thirty-five males Wistar rats were divided into 5 groups: normal group diet (STD), the diabetic group (STDD) with a standard feed diet, the diabetic group with mentik wangi rice (MWRD), the diabetic group with sago analogue rice (SARD) and the diabetic group with sago analogue rice with the addition of 10% red bean flour (SARKBD). All groups were analysed for dietary interventions, blood glucose level, insulin level for HOMA-β and HOMA S indices and measurement of insulin level by using IHC analysis. In addition, short-chain fatty acids (SCFA) analysis was performed in the caecum. This study showed that decreasing blood glucose level shown in SARD and RASKBD groups. The pancreatic β-cell number indicated an increase in the SARD group compares to the STDD group. The pool total of SCFA in SARD group was the highest among of all groups, as well as the acetate, propionate and butyrate pools. These results indicate that the sago analogue rice diet could repair and increase the expression of pancreatic β-cell through absorption inhibition mechanisms and by increasing insulin sensitivity and the SCFA level.
KEYWORDS:β-cell pancreas; Diabetic Mellitus; Insulin Sensitivity; Red Bean; Sago Analogue Rice
Introduction
Diabetes mellitus (DM) is a metabolic disease characterized by increasing blood sugar (hyperglycemia) caused by a lack of insulin sensitivity.1 More than 90% of Diabetes Mellitus Type 2 (DMT2) cases, which is diabetes caused by low insulin sensitivity.2 According to the International Diabetic Federation,3 around 415 million people have DM and this may increase to 642 million in 2040. Basic Health Research also states that DM proportion at the age of> 15 years was increasing to 5,7%.4 Furthermore, 90-95% of cases of DM are type 2 DM and caused by the lack of insulin sensitivity and resistance of peripheral insulin.5
Type 2 diabetes mellitus (DMT2) was caused by the imbalance between free radicals and antioxidants that can reduce sensitivity to insulin (Evans et al., 2002). Insulin resistance is indicated by the malfunction production of insulin by pancreatic β-cell, in other words, it shows the decrease of insulin sensitivity. The diabetes conditions are usually followed by hyperlipidemia cases.6 The (DMT2) case also could be controlled with diet management properly. Diet management involving resistant starch is one of the alternatives to improve glucose response in diabetics.7–10 Resistant starch (RS) is a fraction of the edible part of plants or their extracts, or synthetic analogue that are resistant to the hydrolysis of amylase digestive enzymes in the small intestine.10 Both of analogue sago rice and sago rice with the addition of red beans can be used as a new ingredient in DM diet therapy because they are rich in resistant starch. Sago analogue rice and red beans were expected to reduce insulin resistance and decrease blood glucose level as well as improve lipid profiles in diabetic rats.6 The purpose of this study is to determine the effects of sago analogue rice and kidney beans on the improvements of pancreatic β-cell in the diabetic rats induced by Streptozotocin-Nicotinamide (STZ-NA).
Materials and Methods
Materials
The sago starch (Metroxylon spp.) Meranti variety was obtained from Selat Panjang, Riau and local varieties of red beans (Phaseolus vulgaris) were taken from farmers in Parakan Subdistrict, Temanggung Regency. The Mentik wangi rice was obtained from a supermarket in Yogyakarta.
Animal Model
In this study, 2-3 months male Wistar rats (200-250g) were obtained from the Experimental Animal Laboratory, UPT LPPT, Universitas Gadjah Mada, Yogyakarta. The rats were conditioned with a 12-hour dark/light cycle, an adequate air ventilation in the cage at the temperature of 25oC. They were then divided into five groups and each group was treated with different diet formulation. The groups consisted of: one normal control group (STD) which follow standard AIN93M feed diet 11 and four diabetic induced groups which were treated with different kind of diets: standard diet (STDD), mentik wangi rice (MWRD), 100% sago rice (SARD) and 10% red bean sago rice (SARKBD). The food and water were given in ad libitum. Diabetes induction was carried out with nicotinamide (NA) in 0.9% buffer saline (NaCl) at a dose of 230 mg/kg intraperitoneally, fifteen minutes later the rat was induced with Streptozotocin 60 mg/kg (Gupta and Sharma, 2012). After induction, the rat was given a 5% glucose solution in water for 24 hours to reduce the possibility of death due to hypoglycemic effects (Saeed et al., 2012). This conditions and handling were approved by the Ethics Commission for Preclinical Research, Gadjah Mada University No. 00070/04/LPPT/X/2016.
Analogue Rice Production
Analogue rice was used in 2 different formulas; analogue rice from sago starch (SARD) and sago analogue rice with 10% red bean flour (SARKBD).12
Food Formulation
Food formulation refers to formula of AINM 1993. The details of the formula can be found in the previous study.6
In Vivo Analysis
Five days after STZ-NA induction, the blood glucose and fasting insulin level were measured using glucose oxidase phenol aminophenazone or GOD-PAP methods 13. The blood was aspirated from vena retroorbital by microcapillery technique. The criterion of rat diabetic is fasting blood glucose level more than 200 mg/dL. The intervention period of the treatment was 4 weeks and blood glucose level were analyzed each week.
HOMA Analysis
The Homeostatic model assessment and insulin resistance (HOMA-IR) index were analyzed according to Matthews et al.,14 while Homeostasis Model of Assessment-ß (HOMA-ß) was analyzed according to Hirata et al.15 methods.
Immunohistochemistry Analysis
Immunohistochemistry assays were performed on formalin-fixed paraffin in pancreatic tissue. The organ was deparaffinized in xylol and rehydrated with graded alcohol and then washed in phosphate buffer solution (PBS) pH 7.4. The endogenous peroxidase blocking was performed with 3% H2O2 solution in distilled water for 30 minutes, it was then washed with PBS solution. The blocking was performed for 5 minutes with goat serum and dripped anti-insulin primary antibody (1: 300) for overnight in the 4℃, after that, it was washed in PBS. The secondary antibodies used were biotinylated goat anti-polyvalent and was performed for 10 minutes and the conjugate of the streptavidin peroxidase enzyme was added. The substrate and chromogen 3,3 diaminobenzidine (DAB) were added, afterwards, the mixture was incubated for 10 minutes at room temperature in the dark. The slide was then washed with distilled water solution and was treated with counterstain with hematoxylin for 10 seconds. The result of immunohistochemistry staining was visualized and analyzed using BX53 (Olympus) with 200X Magnifications.
SCFA Analysis
The short-chain fatty acid (SCFA) level were analyzed by the gas chromatography method in the caecum. The caecum was weighed then centrifuged at 10,000 rpm for 15 minutes. The supernatant was separated and injected into the GCMS column (Shimadzu GC 8A brand, with FID detector). The caecal SCFA pool was concentrated in the caecum (μmol/g) by the mass of the caecal contents (g) and was expressed in micromoles per 100 g of body weight.16
Statistical Analysis
Statistical analysis was conducted using variance (ANOVA) with IBM SPSS Statistics version 20 and DMRT test with 5% confidence level.
Results and Discussion
The level of Blood Glucose
The blood glucose level of rat induced by STZ-NA were not significantly different in all groups, the changes ranging from around 150-160% in first week period with STDD, MWRD, SARD and SARKBD were155.09%, 155.66%, 156.22%, and 161.12%, respectively. (Table 1).
Table 1: Glucose level changes (%) in every week treatment of STD, STDD, MWRD, SARD and SARKBD rat groups.
| Treatment | STD | STDD | MWRD | SARD | SARKBD |
| Glucose changes 1 | 1.01a ± 0.35 | 155.09b ± 3.64 | 155.66b ± 5.09 | 156.22b ± 6.69 | 161.12b ± 10.83 |
| Glucose changes 2 | 5.77c ± 1.92 | 4.21c ± 7.79 | -22.79b ± 7.49 | -57.27a ± 5.19 | -55.69a ± 10.26 |
| Glucose changes 3 | 1.29d ± 0.57 | 1.57d ± 3.97 | -12.78c ± 1.11 | -20.29a ± 0.48 | -16.01b ± 4.25 |
| Glucose changes 4 | 0.93d ± 0.23 | 1.27d ± 0.49 | -2.93c ± 0.42 | -14.21a ± 0.81 | -11.22b ± 1.65 |
| Glucose changes 5 | 0.50d ± 0.28 | 1.55e ± 0.51 | -1.82c ± 0.64 | -28.99a ± 0.99 | -13.45b ± 1.31 |
Note : Data were presented as mean ± STDEV of three repetitions samples (n=3). Different superscripts in the same column showed significant different (p>0.05).
The STDD group showed positive changes in every week of treatment, while the MWRD, SARD and SARKBD showed the negative changes. The positive changes indicate that the group still have diabetes condition. Negative blood glucose level was demonstrated by the MWRD, SARD and SARKBD with an average percentage of 10.08%, 30.19%, and 24.09%, respectively. The highest alteration of blood glucose was shown by the SARD group. Feed intervention on all group of diabetes-induced rats demonstrated a decrease in blood glucose levels at the end intervention period.
STZ-NA is one of the diabetic agents that has a toxic ability to destroy pancreatic β-cell. It contains Nicotinamide (NA) which is a derivative of vitamin B3 (niacin) that has an antioxidant capacity to reduce toxic effects from STZ.9,17 In β cells, STZ attenuates the glucose oxidation and insulin synthesis, also decrease the glucose transport and glucokinase activity. SARD group showed the highest changes in blood glucose levels in every week of treatment, and this is followed by SARKBD and MWRD group. This research showed that dietary intervention groups can lower blood glucose due to the presence of resistant starch. RS in sago rice (SARD and SARKBD) was 12.25% and 11.80%, respectively.9 Meanwhile, the content of RS in fragrance rice was only 10.72%. According to Marsono et al.18, RS could reduce blood glucose level because it cannot be digested in the intestine which impacts to the decrease of the amount of glucose availability. Besides, the viscous characteristic of RS able to inhibit small intestine absorption. In addition, the Glycemic index of sago and red bean-based analogue rice was 40.7 and 50.49 while, rice was 80.18,19 Similarly, this was also supported by the research of Okoniewska and Witwer20 which states that RS in the small intestine can reduce the glycemic and insulin response in diabetics and hyperinsulinemic patients.
HOMA β and HOMA S Level
The index data of HOMA β and HOMA S are shown in Table 2 in which it shows that SARD group has the highest index among other groups at the end of intervention.
Table 2: HOMA β and HOMA S level of STD, STDD, MWRD, SARD and SARKBD rat groups.
| Treatment | STD | STDD | MWRD | SARD | SARKBD |
| Homa β | 2.84b ± 0.94 | 0.40a ± 0.15 | 0.38a ± 0.05 | 0.83a ± 0.07 | 0.46a ± 0.15 |
| Homa S | 1.07c ± 0.40 | 0.11a ± 0.03 | 0.19a ± 0.03 | 0.54b ± 0.06 | 0.54b ± 0.20 |
Note : Data were presented as mean ± STDEV of three repetitions samples (n=3). Different superscripts in the same column showed significant different (p>0.05).
The high value of HOMA β and HOMA S in the treated group showed that the β-cell function and insulin sensitivity were still high. In this study, treatment with Sago and read bean analogue rice showed a decrease in blood glucose levels in every week of treatment and this occurs through the increase of insulin sensitivity. Some previous studies reported that RS could improve insulin sensitivity by increasing the glucose uptake from the blood to muscle.21–25
Immunohistochemistry Analysis
The expression profile of insulin in the islets of Langerhans is shown in Figure 1. The insulin expression profile was higher in SARD and SARKBD group compare to STDD group. While, the number of pancreatic β-cell for SARD, SARKBD, and STDD, were 92.42%, 85.62% and 21.97% respectively.
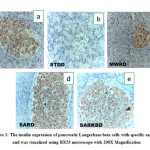 |
Figure 1: The insulin expression of pancreatic Langerhans beta cells with specific antibody and was visualized using BX53 microscope with 200X Magnification. |
The high expression of pancreatic β-cell could affect the alteration of insulin level, increase insulin sensitivity and improve the β-cell function.26,27 This result showed that SARD and SARKBD interventions have high expression profile values compared to other diet groups. This indicates that the sago rice diet can enhance insulin production and sensitivity.
On the other hand, STTD group showed the lowest expression of insulin in pancreatic Langerhans beta cells. In this case, it is argued that STZ-NA induction can damage the pancreatic Langerhans beta cells and standard food is not capable enough to repair the defect.
The sago analogue rice can be a promising diet treatment for improving insulin sensitivity by increasing insulin expression and regeneration in pancreatic Langerhans β-cell.27 We expected that this is due to the effect of resistant starch characteristic on Sago analogue rice that impact insulin sensitivity.
SCFA Analysis
The pool of short chain fatty acids (SCFA) level of all group are shown in Table 3. The SARD group showed the highest pool SCFA level of acetate, propionate, butyrate and in total. Meanwhile, the STDD group was the lowest concentration among other groups. Furthermore, the STD group showed the lower amount of SCFA than SARKBD and MWRD group. After all, the STDD group was significantly different (p <0.05) with SARKBD, MWRD, STDD and STD.
Table 3: The Pool SCFA of STD, STDD, MWRD, SARD and SARKBD rat groups.
| Pool SCFA | STD | STDD | MWRD | SARD | SARKBD |
| Acetate | 47.27b ± 4.49 | 20.44a ± 1.21 | 51.93c ± 1.81 | 148.12e ± 3.13 | 84.79d ± 0.53 |
| Propionate | 21.89b ± 2.08 | 10.26a ± 0.61 | 28.28c ± 0.99 | 58.88e ± 1.24 | 38.56d ± 0.24 |
| Butyrate | 7.85b ± 0.74 | 3.04a ± 0.18 | 9.38c ± 0.33 | 30.90e ± 0.65 | 15.56d ± 0.09 |
| Total | 77.01b ± 7.31 | 33.74a ± 2.00 | 89.58c ± 3.13 | 237.89e ± 5.02 | 138.92d ± 0.87 |
Note : Data were presented as mean ± STDEV of three repetitions samples (n=3). Different superscripts in the same column showed significant different (p>0.05).
The enhancement of insulin sensitivity can be caused by the production of SCFA through RS fermentation. Canfora et al.28 explained that acetate and propionate from RS fermentation products can increase buffering capacity which decreases the fatty acids in the muscles.28 This can cause a decrease of lipid storage and increase insulin sensitivity.
The mechanism of blood glucose reduction was due to the ability of SCFA to reduce blood glucose through several mechanisms. Gao et al.29 stated that the products of RS fermentation bring a hipolipidemic effect on STZ-NA induced-rats. This is also supported by Luo et al.30 which mention that high fatty acids can inhibit the process of glucose utilization in muscle tissue and insulin resistance disorder. Other researchers reported that the increase in SCFA concentration in humans suppress the formation of free fatty acids and thus, improves insulin sensitivity.31,32
According to Aluko33, the high level of free fatty acid can cause disruption of GLUT4 activity and impact on the increase of blood glucose level. Whereas, the SCFA can increase fatty acid β oxidation to avoid high free fatty acids level and induce the activity of GLUT4 to increase.30 Furthermore, in this study, butyrate was found in the high level in sago analogue rice and red bean group treatment. The butyrate and acetate are fatty acid that can increase AMPK (AMP-activated protein kinase) for stimulating glucose absorption. According to Wronkowska et al.16 the resistant starch diet produced the highest SCFA concentrations including butyrate compared to the standard diet. The butyrate can increase energy production and improve the mitochondrial function to increase insulin sensitivity and it also improves glucose metabolism.29,34 Another study, Gao et al.29 reported that butyrate also induces PGC-1α (PPAR Gamma Coactivator -1α) in the muscle so that improve the mitochondrial function, increase fatty acid oxidation and reduce the risk of decreased insulin sensitivity.
Conclusion
This study concludes that sago analogue rice and sago analogue rice with 10 % red bean flour in diabetic rats have an effect to improve and increase pancreatic Langerhans β-cell expression. The mechanism involves the role of resistant starch in the Sago analogue rice that reduce absorption, increase insulin sensitivity and improve the better result for total pool of SCFA alteration.
Funding Sources
The authors acknowledge The Ministry of Research, Technology, & Higher Education of Republic Indonesia for supporting and funding this research.
Conflict of Interest
There are no conflict interests
References
- Eisenbarth GS, Buse JB. Type 1 diabetes mellitus. In: In: Melmed S, Polonsky KS, Larsen PR, Kronenberg HM Eds. Williams Textbook of Endocrinology 12th Ed. Philadelphia. PA: Saunders Elsevier. Chap.32. ; 2008.
- Naik SR, Niture NT, Ansari AA, Shah PD. Anti-diabetic activity of embelin: Involvement of cellular inflammatory mediators, oxidative stress and other biomarkers. Phytomedicine. 2013.
CrossRef - DIABETES ATLAS 8th Edition 2017 [International Diabetes Federation].; 2017.
- Riset Kesehatan Dasar (Riskesdas) 2013.
- ADA (American Diabetes Association). Standards of Medical Care in Diabetes.; 2017.
- Wahjuningsih SB, Haslina, Marsono Y. Hypolipidaemic Effects of High Resistant Starch Sago and Red Bean Flour- based Analogue Rice on Diabetic Rats. Mater Socio Medica. 2018.
CrossRef - Behall KM, Hallfrisch J. Plasma glucose and insulin reduction after consumption of breads varying in amylose content. Eur J Clin Nutr. 2002;56(9):913-920.
CrossRef - Wolever TMS, Mehling C. High-carbohydrate–low-glycaemic index dietary advice improves glucose disposition index in subjects with impaired glucose tolerance. Br J Nutr. 2002;87(5):477-487.
CrossRef - Wahjuningsih SB, Marsono Y, Praseptiangga D, Haryanto B. Resistant starch content and glycaemic index of Sago (Metroxylon spp.) starch and red bean (Phaseolus Vulgaris) based analogue rice. Pakistan J Nutr. 2016;15(7):667-672.
CrossRef - Englyst HN, Kingman SM, Cummings J. Classification and measurement of nutritionally important starch fractions. Eur J Clin Nutr. 1992;46(2):S33-S50.
- Reeves PG, Nielsen FH, Fahey GC. AIN-93 Purified Diets for Laboratory Rodents: Final Report of the American Institute of Nutrition Ad Hoc Writing Committee on the Reformulation of the AIN-76A Rodent Diet. J Nutr. 1993;123(11):1939-1951.
CrossRef - Wahjuningsih SB, Praseptiangga D, Haryanto B. A study of sago starch and red bean flour-based analogue rice development as functional food. 2016;(August):23-25.
- Barham D, Trinder P. An improved colour reagent for the determination of blood glucose by the oxidase system. Analyst. 1972;97(1151):142-145.
CrossRef - Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412-419.
CrossRef - Hirata A, Maeda N, Hiuge A, et al. Blockade of mineralocorticoid receptor reverses adipocyte dysfunction and insulin resistance in obese mice. Cardiovasc Res. 2009;84(1):164-172.
CrossRef - Wronkowska M, Juśkiewicz J, Zduńczyk Z, Soral-Śmietana M, Krupa-Kozak U. Influence of chemically-modified potato starch (RS type 4) on the nutritional and physiological indices of rats. Polish J Food Nutr Sci. 2011;61(2):143-151.
CrossRef - Ghasemi A, Khalifi S, Jedi S. Streptozotocin-nicotinamide-induced rat model of type 2 diabetes (review). Acta Physiol Hung. 2014;101(4):408-420.
CrossRef - Marsono Y. Resistant starch: pembentukan, metabolisme dan aspek gizi-nya. Agritech. 1998;18(4):29-35.
- Thompson S V., Winham DM, Hutchins AM. Bean and rice meals reduce postprandial glycemic response in adults with type 2 diabetes: A cross-over study. Nutr J. 2012;11(1):1-7.
CrossRef - Okoniewska M, Witwer RS. Natural resistant starch: an overview of health properties as useful replacement for flour, resistant starch may also as boost insulin sensitivity and satiety. 2007;New York.
- Robertson MD, Bickerton AS, Dennis AL, Vidal H, Frayn KN. Insulin-sensitizing effects of dietary resistant starch and effects on skeletal muscle and adipose tissue metabolism. Am J Clin Nutr. 2005;82(3):559-567.
CrossRef - Johnston KL, Thomas EL, Bell JD, Frost GS, Robertson MD. Resistant starch improves insulin sensitivity in metabolic syndrome. Diabet Med. 2010;27(4):391-397.
CrossRef - Shen L, Keenan MJ, Raggio A, Williams C, Martin RJ. Dietary-resistant starch improves maternal glycemic control in Goto-Kakizaki rat. Mol Nutr Food Res. 2011;55(10):1499-1508.
CrossRef - Mattace Raso G, Simeoli R, Russo R, et al. Effects of Sodium Butyrate and Its Synthetic Amide Derivative on Liver Inflammation and Glucose Tolerance in an Animal Model of Steatosis Induced by High Fat Diet. PLoS One. 2013;8(7):1-13.
CrossRef - Wijanarka A. Tepung Gayam (Inocarfus Fagifer Forst.): Indeks Glikemik, Sifat Fungsional, Modifikasi Siklus Autoclaving Cooling Serta Sifat Gizi In Vitro dan In Vivo. Disertasi, Univ Gadjah Mada. 2013.
- Kaku K. Pathophysiology of Type 2 Diabetes and Its Treatment Policy. J Japan Med Assoc. 2009;138(1):28-32.
- Erwin E, Muttaqien, Pangestiningsih TW, Widyarini S. Ekspresi Insulin pada Pankreas Mencit (Mus musculus) yang Diinduksi Dengan Streptozotocin Berulang. J Kedokt Hewan. 2013;7(2):97-100.
CrossRef - Canfora EE, Jocken JW, Blaak EE. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat Rev Endocrinol. 2015;11(10):577-591.
CrossRef - Gao Z, Yin J, Zhang J, et al. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes. 2009;58(7):1509-1517.
CrossRef - Luo J, Yperselle M Van, Rizkalla SW, Rossi F, Bornet FRJ. Human Nutrition and Metabolism Chronic Consumption of Short-Chain Fructooligosaccharides Does Not in Type 2 Diabetics 1. J Nutr. 2000;130(January):1572-1577.
CrossRef - Higgins JA, Higbee DR, Donahoo WT, Brown IL, Bell ML, Bessesen DH. Resistant starch consumption promotes lipid oxidation. Nutr Metab. 2004;1:1-11.
CrossRef - Murphy KM, Travers P, Walport M. Janeway’s Immunobiology (Immunobiology: The Immune System (Janeway)) 7th Edition. Science. New York: Garland; 2008.
CrossRef - Aluko R. Bioactive Lipids 2 2.1. Funct Foods Nutraceuticals. 2012:23-35.
CrossRef - Den Besten G, Van Eunen K, Groen AK, Venema K, Reijngoud DJ, Bakker BM. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J Lipid Res. 2013;54(9):2325-2340.
CrossRef













