Introduction
Blueberry (Cyanococcus) belongs to the genus Vaccinum of the family Ericaceae and sub-family Vacciniaceae. Blueberry has a circumpolar distribution, mainly present in North America, Europe, and Asia. Commercial “blueberries” (including both wild and cultivated blueberries) are native of North America and were introduced in Europe in 1930. Blueberries are prostate shrubs that varied in size from 10 centimetres to 4 metres, and the fruit is a berry that varied from 5 – 16 mm in diameters with the flared crown at the end. They are dark purple in colour after maturing and are covered in a protective coating of powdery epicuticular wax known as the “bloom.” Blueberries have high antioxidant activity and phenolic content.1-4 These findings led to further investigation of the health benefits of blueberry on human health. Studies on blueberry supplementation in diets have been associated with metabolic syndrome,5-7 improving brain health and memory,8-10 reducing the risk of cancer,11-14 improving visual acuity,15 enhancing gut health16 and muscle repair17-20 and weight management.21
There is a significant demand for fresh fruits and their products. It is known that most fruits are seasonal and have a limited shelf life. They often required the application of heat treatments, via processing, in order to maintain and preserve the quality and provide various products such as pulp, jam, jellies, and juices throughout the year, mainly in the off-season. The fruits are generally consumed raw or in the form of ice creams, juices, jams, jellies, liquors, and various sweet formulations.22,23 These food products have pleasant flavours, high level of sugars, proteins, minerals, fatty acids, vitamin, and carotenoids.24
Probiotics are living microorganisms that provide beneficial effects on the host when taken in adequate amounts [25, 26]. The probiotic culture should be alive to confer the effects and must present in large quantities in the product, usually greater than 106-107 cells per millilitre or gram27 or 1×109 CFU per serving as probiotic.26
Fermentation can be regarded as a biological method of preservation. Fermented foods have a low risk of contamination when enriched in antimicrobial food products, such as bacteriocins, ethanol and organic acids. Fermented foods not only provide the new and desirable taste and texture but also promote the host health in ways not directly attributable to the starting food materials. Products of fermentation and the contribution of microbes provide the additional health benefits beyond essential nutrition. Strains of Lactobacilli and Bifidobacteria are available for human consumption to lower the risk of gastrointestinal infections.28 Consumption of probiotic provide some beneficial health effects such as improvement of intestinal health by the improvement of microbiota, synthesizing and enhancing the bioavailability of nutrients, reducing the risk of coronary heart disease, improving antibiotic-associated diarrhoea, helps in reducing the symptoms of lactose intolerance and food digestion, improving oral health, lowering the blood pressure, improving antioxidative activity, normalizing the serum cholesterol level, reducing constipation and ulcers, improving the parasitic infections and reducing the risk of urinary tract and upper respiratory tract infection.29-37
In Asia, any bacterial strain of a known species that are traditionally used can be added to the food for the preparation of probiotic food product. Nowadays, probiotic food products are promoted in the form of dairy products such as fermented milk and yoghurt. Lactose intolerance and cholesterol content are two significant disadvantages related to fermented dairy food products.38 People with special needs of vegetarians and people with allergic reactions to milk proteins have found a good substitute in the form of fruit and vegetable food products containing probiotics.39, 40 Fruit products have a high amount of antioxidants, dietary fibre, vitamins, minerals, and other useful nutritional substances and thus improve the health of the host.41, 42 Recently, various raw materials have been explored to ascertain the suitability of the substances to produce novel non-dairy probiotic products.43
According to,44 significant factors that could limit the viability of probiotic bacteria in fruit products are categorized as, (1) intrinsic food parameters such as pH, titrable acidity, microbial preservatives like bacteriocins and hydrogen peroxide; (2) processing parameters like incubation temperature, heat treatment, cooling rate, storage techniques;3 microbiological factors such as type of probiotic strains, intercompatibility of different strains with each other, proportion and rate of inoculum.
Food components can be modified in a controlled way by altering some structural characteristics of fruit and vegetable matrices with the advancement in technology.45 The interest in the development of fruit-based functional food products with probiotics is increasing among people of all age groups as they have a good taste profile and considered as healthy and refreshing products.43 In recent years, fruit and vegetable jams are considered an excellent basal medium for LAB fermentation. Advantage of these jams is accounted because of their low allergenicity and high health benefits.43
The present study demonstrates the fermentation of blueberry fruit with probiotic lactic acid bacteria to select an appropriate starter culture for developing lacto- fermented blueberry jam. Physico-chemical changes, microbiological analysis, antioxidant assay, and in vitro cholesterol reduction were carried out during fermentation.
Materials and Methods
Preparation of blueberry substrate
Fresh blueberry fruit was procured from fruit market, New Delhi, India. After arrival at the laboratory, blueberry fruits were appropriately washed. The blueberry jam (without supplementary water or nutrient) was prepared by putting them in a saucepan over heat. The jam was sterilized by autoclaving at 121℃ for 15 min.
Probiotic lactic acid bacteria
L. plantarum DB-2, L. fermentum J-1, P. acidilactici M-3, L. plantarum SK-3 and P. pentosaceus SM-2 were isolated from Dosa batter, Jalebi batter, Maida dough, Sauerkraut, and Soymilk, respectively. Probiotic attributes such as acid tolerance,46 bile tolerance,47 antibiotic susceptibility,48 hemolytic activity,49 gelatinase activity,50 autoaggregation,51 co-aggregation studies,51 hydrophobicity,52 bacteriocin production,53 lactic acid and hydrogen peroxide production.54 exopolysaccharide production55 were studied on all the five isolates. Genotypic characterization was done of all the five isolates.56 Analysis of the 16S rRNA sequences revealed that lactic acid bacteria isolated from Dosa batter, Jalebi batter, Maida dough, Sauerkraut, and Soymilk showed 99%, 99%, 97%, 100% and 99% homology with Lactobacillus plantarum, Lactobacillus fermentum, Pediococcus acidilactici, Lactobacillus plantarum and Pediococcus pentosaceus respectively. The 16S rRNA gene sequence was submitted to Genbank and assigned accession number MK246169, MK353735, MK461878, MK246167 and MK461882 for isolate DB-2, J-1, M-3, SK-3 and SM-2, respectively.57
Fermentation of probiotic blueberry jam
All the five isolates were sub-cultured in MRS broth at 35℃ till the colony count reaches up to 108 CFU/ml. 100 g of the blueberry jam was inoculated separately in triplicates. The probiotic enriched blueberry jam prepared in the present study was divided into three sets, i.e., Set A was inoculated with L. plantarum DB-2 (108 CFU/ml), Set B was inoculated with P. pentosaceus SM-2 and Set C was inoculated with consortia of probiotic strains viz. L. plantarum SK-3, L. fermentum J-1, P. acidilactici M-3 and P. pentosaceus SM-2 (1:1:1:1, @ 108 CFU/ml). Set D was control (without probiotic bacteria). The fermentation process was executed at 35℃ for 72 h. The fermentation was terminated by keeping these four sets at 4℃.
Physicochemical analysis
The blueberry jam was studied to determine the following parameters: pH, total soluble solids, titratable acidity, and ascorbic acid.
Preparation of the extracts
The blueberry jam extract was prepared as described by58 with slight modifications. The blueberry jam sample (10 g) was extracted with 50 ml of 80% methanol. Extraction was carried out under stirring using a magnetic blender for 40 min at room temperature. After agitation, the solution was centrifuged at 3000 g for 15 min at 4ºC. Then, the supernatant was filtered out using Whatman No. 3 filter paper and filled in a 100 mL volumetric flask and allowed to set in the dark for an hour and then stored at 4ºC until further analysis.
pH
The pH of the fermented blueberry jam was measured at 0, 7, 14, 21 and 28 days by using a digital pH meter (HI 8314, HANNA Instruments, Italy), calibrated with pH 4 and 7 buffers.
Total soluble solids (TSS)
TSS content of the blueberry jam was estimated using a portable refractometer (ARBO-95, Brix 0-95%).
Titratable acidity (in terms of lactic acid)
Titratable acidity was measured by the titrimetric method59 and expressed as % lactic acid. The fermented blueberry jam extract was titrated to pH 8.0 with 0.1 N NaOH. 2-3 drops of 1% phenolphthalein was used as an indicator.
![]()
Ascorbic acid
Ascorbic acid content of blueberry jam was evaluated by visual titration method given by [59]. 1 ml of the jam extract was diluted with 10 ml of 3% metaphosphoric acid. After centrifugation, titration was done with the standard dye solution (sodium salt of 2, 6-dichlorophenolindophenol) to a pink colour end point.
![]()
Antioxidative properties of lacto-fermented blueberry jam
The fermented blueberry jam extract was analyzed for anti-oxidative properties viz. total antioxidant property, reducing power, and anti-scavenging property. Samples were taken at 0, 7, 14, 21, and 28 days for analysis.
The antioxidant activity
The ABTS antioxidant activity of lacto-fermented blueberry jam was determined using the method of [60]. The antioxidant activity was measured by adding 1 ml of each peroxidase (Sigma-Aldrich), Hydrogen peroxide (Merck), 100 M ABTS [2,2-azino-bis (3-ethylbenz-thiazoline-6-sulphonic acid)] (Sigma-Aldrich) and distilled water. After proper mixing, the reaction mixture was incubated in the dark at 25℃ for 1 h. 1 ml of blueberry jam extract was added. Finally, the absorbance was measured at 734 nm. The antioxidant activity was calculated by the formula mentioned below:
Total antioxidant activity (%) = [1- (A734 nm sample/A734 nm control)] x 100
Reducing power
The reducing power of lacto-fermented blueberry jam was determined following the method of.61 It was determined by adding 1 ml of blueberry jam extract, 0.5 ml of 0.5 M phosphate buffer (Merck, pH 6.6) and 2.5 ml of potassium hexacyanoferrate solution (Merck, 1% w/v). The reaction mixture was heated at 50℃ for 20 min. After the mixture was cooled to room temperature, the reaction was terminated by adding 0.5 ml of 10% trichloroacetic acid (Merck). After centrifugation at 3000 g for 10 min, 1 ml of supernatant was mixed with 1 ml of distilled water and 0.1 ml of 0.1% ferric chloride (Merck). The reaction mixture was incubated for 10 min at room temperature. The absorbance was measured at 700 nm using a spectrophotometer.
Free radical scavenging activity (FRSA)
FRSA was performed according to the method of.62 It was measured by adding 1 ml of blueberry jam extract with 5 ml of 0.1 mM DPPH-methanolic solution (Sigma-Aldrich). The mixture was kept in the dark for 1 h, and absorbance was measured at 517 nm by using a spectrophotometer. FRSA was calculated by the following formula:
Scavenging activity (%) = [1 – (A517 nm sample/ A517 nm blank)] x 100
Microbiological analysis
The viability of probiotic cultures in the lacto-fermented blueberry jam was determined by standard plate count method (CFU/ml) given by63 on MRS agar medium after serial dilution ranging from 10-1 to 10-9. The agar plates were incubated at 35℃ for 24 h. The samples were taken at 0, 7, 14, 21, and 28 days.
Health promoting effect of probiotic juice
In vitro cholesterol lowering property
In vitro cholesterol lowering property of the lacto-fermented blueberry jam was performed by following the method of [46]. 0.3% oxgall (Himedia) was added to MRS broth (Himedia) and autoclaved at 121℃ for 15 minutes. Water-soluble cholesterol (Sigma-Aldrich) (after filter sterilization) was added to autoclaved MRS broth (Himedia). 10% of the lacto-fermented blueberry jam extract was added to it and incubated at 35℃ for 24 h. Centrifugation was performed at 50000 g for 10 min at 4℃. 1 ml of supernatant was taken into a sterilized test tube and added with 1 ml and 2 ml of KOH (33% w/v) and absolute ethanol, respectively. The mixture was incubated at 35℃ for 15 min after vortexing for 1 min. The reaction mixture was cooled to room temperature. Then, it was mixed with 2 ml of distilled water and 3 ml of hexane. Vortexing was done for 1 min. 1 ml of hexane layer was separated and evaporated under nitrogen gas. The residue was dissolved in 2 ml of o-phthalaldehyde solution. 0.5 ml of conc. Sulphuric acid was added after proper mixing and vortexed for 1 min. The mixture was incubated for 10 min at room temperature, and absorbance was measured at 550 nm. Cholesterol-lowering capacity was measured by the following formula:
Cholesterol reduction (%) = 1 – (absorbance of culture supernatant550/ absorbance of control550) x 100
Sensory Evaluation
The lacto-fermented blueberry jam was subjected to sensory evaluation by 22 untrained panellists. A total of 13 untrained female panellists and 9 untrained male panellists between the ages of 18-53 years old completed the sensory evaluation. Sensory evaluation was carried out at room temperature in the Department of Microbiology, Kurukshetra University, Haryana, India. Tastings occurred between the hours of 11:00 am to 2:00 pm. Sensory evaluation performa, along with the consent form, was prepared and distributed to the panellists. Each panellist was given a pen for recording on his/ her evaluation sheet and a glass of water to cleanse the mouth between tastings. The evaluation of the product was carried out on appearance, colour, taste, texture, flavour, and overall acceptability. A nine-point hedonic scale in which 1= ‘liked extremely,’ 5= ‘neither liked nor disliked’ and 9= ‘disliked extremely’ was used by each participant for sample evaluation. Approximately 20 mg of each sample was presented in a glass container. A control sample of the non-fermented blueberry jam was first tasted, and then the lacto-fermented blueberry jam was served. The plain blueberry jam was used as a control product to eliminate first-order bias in which consumers may unintentionally rate the first sample more or less favourable than the lacto-fermented blueberry jam, regardless of the product under evaluation [64]. Results from the hedonic scale record sheets were collected and input a Microsoft Excel 2010 database, then imported into the IBM SPSS Statistic 23 for analysis.
Statistical Analysis
All the experimental results were recorded as mean ± SD (Standard Deviation). For every observation, 3 determinations were used. The data were statistically analyzed. The significant differences between means were calculated by a one-way analysis of variance (ANOVA) using Duncan’s multiple range test at P < 0.05.
Results and Discussion
Physicochemical Analysis
All the strains (L. plantarum DB-2, L. fermentum J-1, P. acidilactici M-3, L. plantarum SK-3, and P. pentosaceus SM-2) grew well on sterilized blueberry jam without any nutrient supplementation. Table 1 (a – d) expressed the physicochemical analysis of blueberry jam fermented with L.plantarum DB-2, L. fermentum J-1, P. acidilactici M-3, L.plantarum SK-3, and P. pentosaceus SM-2. During fermentation up to 28 days, the pH of blueberry jam showed a reduction from 3.31 to 2.94 in Set A, 3.30 to 2.97 in Set B, 3.20 to 2.92 in Set C. The corresponding decrease in pH was observed in Set D (Control), i.e., 3.33 to 3.20. The pH of Marola and strawberry guava jam showed a slight decrease from 3.31 to 3.27 during the first 6 months of storage and increased up to 3.33 after 12 months of storage.65 The decrease in pH (3.9 to 3.7) was observed in sugar-free guava jam during storage.66 The cherry jam was evaluated between 0 and 15 days and found a decrease in pH values from 3.66 to 3.29.67 The decrease in pH values is associated with dissociation of organic acids68 and the utilization of carbohydrates and sugars by lactic acid bacteria to produce lactic acid. The changes in brix in the blueberry jam was evaluated over 28-day of fermentation and observed the decrease in all the Set (A to C) along with the control. Highest titratable acidity was observed in Set C which produced about 6.67% lactic acid whereas Set A and Set B produced 5.88% and 5.96% titratable acidity in terms of lactic acid, respectively after 28-day of fermentation at 35℃. Control did not exhibit the titratable acidity in terms of lactic acid. The production of acids resulted in a decrease in pH. The capability of a microorganism to survive and grow in fruit jam is more dependent on pH than on titratable acidity.69 The ability of probiotic lactic acid bacteria to grow at such low pH may be due to the heterogeneous characteristics that allow them to survive in various ecological niches. The gradual decrease in ascorbic acid content was observed during 28 days of storage from an initial value of 39.65 to 36.36 mg/100 g, 39.57 to 37.13 mg/100 g, 39.67 to 38.16 mg/100 g in Set A, Set B, and Set C, respectively. Set D (Control) also showed the same pattern of reduction of ascorbic acid from an initial value of 7.94 to 6.54 mg/100 g over 28-days of fermentation.
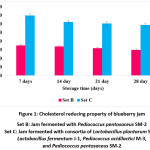 |
Figure 1: Cholesterol reducing property of blueberry jam Click here to View figure |
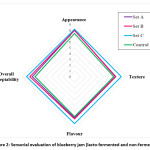 |
Figure 2: Sensorial evaluation of blueberry jam (lacto-fermented and non-fermented) Click here to View figure |
Table 1: Physicochemical analysis of blueberry jam fermented with different lactic acid bacteria
- pH
| Storage time Time (days ) | Ph | |||
| Set A | Set B | Set C | Set D (Control) | |
| 0 | 3.31 ± 0.11A | 3.30 ± 0.05A | 3.20 ± 0.03A | 3.33 ± 0.02A |
| 7 | 3.22 ± 0.03A | 3.21 ± 0.03A | 3.11 ± 0.01A | 3.31 ± 0.05A |
| 14 | 3.10 ± 0.04B | 3.12 ± 0.06 A | 3.03 ± 0.01B | 3.28 ± 0.01A |
| 21 | 3.00 ± 0.01B | 3.05 ± 0.03B | 2.98 ± 0.04B | 3.24 ± 0.06A |
| 28 | 2.94 ± 0.02C | 2.97 ± 0.02B | 2.92 ± 0.05B | 3.20 ± 0.03A |
Each value represents the mean ± SD (n=3). Data bearing different uppercase superscript letters in the same column are significantly different (P<0.05).
- TSS
| Storage time Time (days ) | TSS (ºB) | |||
| Set A | Set B | Set C | Set D (Control) | |
| 0 | 74.38 ± 0.08A | 74.72 ± 0.04A | 74.63 ± 0.07A | 68.05 ± 0.03A |
| 7 | 73.84 ± 0.03A | 73.49 ± 0.05A | 73.43 ± 0.16A | 67.67 ± 0.05A |
| 14 | 72.10 ± 0.17A | 72.72 ± 0.02B | 72.27 ± 0.11B | 65.33 ± 0.37B |
| 21 | 71.88 ± 0.06B | 71.93 ± 0.06C | 71.18 ± 0.03B | 63.98 ± 0.11C |
| 28 | 69.48 ± 0.05B | 69.57 ± 0.12D | 70.83 ± 0.08C | 63.24 ± 0.06C |
Each value represents the mean ± SD (n=3). Data bearing different uppercase superscript letters in the same column are significantly different (P<0.05).
- Titratable acidity
| Storage time Time (days ) | Titratable acidity (in terms of lactic acid) | |||
| Set A | Set B | Set C | Set D (Control) | |
| 0 | 0.07 ± 0.06D | 0.08 ± 0.22D | 0.08 ± 0.05D | – |
| 7 | 1.94 ± 0.14D | 1.96 ± 0.11C | 1.97 ± 0.05 C | – |
| 14 | 2.57 ± 0.08C | 2.78 ± 0.04C | 2.99 ± 0.14B | – |
| 21 | 3.78 ± 0.07B | 3.94 ± 0.06B | 4.12 ± 0.22A | – |
| 28 | 5.88 ± 0.12A | 5.96 ± 0.03A | 6.67 ± 0.11A | – |
Each value represents the mean ± SD (n=3). Data bearing different uppercase superscript letters in the same column are significantly different (P<0.05).
- Ascorbic acid
| Storage time Time (days ) | Ascorbic acid (mg/ 100 g) | |||
| Set A | Set B | Set C | Set D (Control) | |
| 0 | 39.65 ± 0.16A | 39.57 ± 0.37A | 39.67 ± 0.12A | 7.94 ± 0.35A |
| 7 | 39.27 ± 0.25A | 39.11 ± 0.46A | 39.42 ± 0.23A | 7.10 ± 0.12A |
| 14 | 38.78 ± 0.21A | 38.77 ± 0.19A | 39.01 ± 0.10A | 7.00 ± 0.04B |
| 21 | 37.34 ± 0.05B | 37.22 ± 0.02B | 38.28 ± 0.07B | 6.73 ± 0.36C |
| 28 | 36.36 ± 0.17C | 37.13 ± 0.28B | 38.16 ± 0.14B | 6.54 ± 0.11C |
Each value represents the mean ± SD (n=3). Data bearing different uppercase superscript letters in the same column are significantly different (P<0.05).
Microbiological Analysis
The survival of probiotic bacteria during storage evaluated in all the sets of blueberry jam, which is mentioned in Table 2 and expressed as mean count ± SD. All the strains were capable of growing during the storage period of 28 days at 4℃. All the five strains grew rapidly in the blueberry jam and reached 7.98 ± 0.35 (log CFU/ml ± SD), 8.01 ± 0.25 (log CFU/ml ± SD) and 8.21 ± 0.27 (log CFU/ml ± SD) in Set A, Set B and Set C, respectively whereas Set D (Control) showed no viable cells of lactic acid bacteria after 30 days of storage. A probiotic food product must contain 106 CFU/ml viable cells to behave as a health-promoter product.70 During 4 weeks of storing at 4℃, the number of probiotic bacteria is reduced due to sugar consumption and nutrients in fruit jam. The survival ability of the lactic acid bacteria in a probiotic food product is the essential factor during storage under refrigeration for producing health benefits to the host. Results of the present study demonstrate that all the probiotic strains were able to survive in a blueberry jam during storage, establishing that such a medium could be a good candidate as a vehicle of probiotics. In the present study, the formulation of blueberry jam seemed to support the probiotic viability better. This is in accordance with previous reports which concluded that solid matrices might protect bacteria during the storage of food.71, 72 Lactobacilli have been considered as demanding microorganisms due to their requirement for various essential amino acids and vitamins.73 Some Lactobacilli have been found to survive in fruit matrices at refrigerated conditions.43, 74, 75 Many researchers have shown the survivability of probiotic bacteria in matrices with low pH and high acidity during storage under refrigeration (4-5ºC).38, 76 L. plantarum SK-3 and P. acidilactici M-3 showed viable counts of more than 106 in probiotic noni and mulberry juice.77
Table 2: Viability of lactic acid bacteria in lacto- fermented blueberry jam
| Storage interval (Days) | Viability of lactic acid bacteria (log CFU/ml ± SD) | |||
| Set A | Set B | Set C | Set D (Control) | |
| 0 | 9.88 ± 0.35 | 10.07 ± 0.58 | 10.56 ± 0.16 | – |
| 7 | 9.12 ± 0.29 | 9.76 ± 0.72 | 10.02 ± 0.22 | – |
| 14 | 8.79 ± 0.17 | 9.18 ± 0.21 | 9.39 ± 0.39 | – |
| 21 | 8.13 ± 0.52 | 8.49 ± 0.16 | 8.84 ± 0.41 | – |
| 28 | 7.98 ± 0.24 | 8.01 ± 0.25 | 8.21 ± 0.27 | – |
Table 3: Antioxidant activity of lacto-fermented blueberry jam
- Total antioxidant activity
| Storage time (days) | Total antioxidant activity | |||
| Set A | Set B | Set C | Control (Set D) | |
| 0 | 73.29 ± 2.57A | 74.68 ± 3.27A | 75.29 ± 2.84A | 69.81 ± 3.58A |
| 7 | 73.79 ± 1.14A | 73.27 ± 1.19A | 74.78 ± 1.32A | 68.46 ± 2.73A |
| 14 | 72.85 ± 3.91A | 72.22 ± 2.12A | 73.32 ± 3.28A | 67.27 ± 3.17A |
| 21 | 71.34 ± 2.80B | 71.48 ± 4.29A | 72.57 ± 2.65A | 65.46 ± 2.52A |
| 28 | 70.52 ± 3.52B | 70.25 ± 3.18B | 71.47 ± 3.57A | 64.12 ± 2.47B |
Each value represents the mean ± SD (n=3). Data bearing uppercase superscript letters in the same column (different sampling time) are significantly different (P<0.05).
- Reducing power
| Storage time (days) | Reducing Power | |||
| Set A | Set B | Set C | Control (Set D) | |
| 0 | 0.33 ± 0.02A | 0.35 ± 0.04A | 0.39 ± 0.01A | 0.29 ± 0.01A |
| 7 | 0.32 ± 0.02A | 0.34 ± 0.01A | 0.38 ± 0.03A | 0.28 ± 0.03A |
| 14 | 0.31 ± 0.01A | 0.34 ± 0.01A | 0.37 ± 0.02A | 0.27 ± 0.02A |
| 21 | 0.30 ± 0.03B | 0.33 ± 0.02A | 0.35 ± 0.03B | 0.27 ± 0.02A |
| 28 | 0.30 ± 0.01B | 0.32 ± 0.02B | 0.36 ± 0.02C | 0.25 ± 0.03B |
Each value represents the mean ± SD (n=3). Data bearing uppercase superscript letters in the same column (different sampling time) are significantly different (P<0.05).
- Free radical scavenging activity
| Storage time (days) | Free radical scavenging activity | |||
| Set A | Set B | Set C | Control (Set D) | |
| 0 | 73.79 ± 3.29A | 74.28 ± 3.84A | 74.67 ± 3.79A | 65.48 ± 2.79A |
| 7 | 72.61 ± 2.52A | 73.78 ± 1.12A | 73.49 ±1.43A | 64.89 ± 1.25A |
| 14 | 71.59 ± 4.61A | 72.48 ± 2.68A | 72.79 ± 2.25A | 63.61 ± 3.83A |
| 21 | 70.37 ± 2.55B | 71.83 ± 4.68A | 72.12 ± 1.46A | 62.82 ± 2.52B |
| 28 | 69.87 ± 2.13B | 70.47 ± 3.29B | 71.62 ± 1.69B | 61.42 ± 3.45B |
Each value represents the mean ± SD (n=3). Data bearing uppercase superscript letters in the same column (different sampling time) are significantly different (P<0.05).
Antioxidative properties of lacto-fermented blueberry jam
In this study, the antioxidative activity of cell-free extract of lactic acid bacteria was determined using various antioxidant assays. Table 3 (a to c) showing the antioxidant activity of blueberry jam using different starter cultures of L. plantarum DB-2, L. fermentum J-1, P. acidilactici M-3, L. plantarum SK-3, and P. pentosaceus SM-2. Fermented blueberry jam exhibited high antioxidative activity in terms of total antioxidant activity, reducing power and DPPH radical scavenging activity irrespective of the starter culture used in them. Antioxidant activity of blueberry jam (fermented with all the five strains) reduced with the storage time. Blueberry jam showed a reduction of 2.77%, 4.43% and 3.82% in Set A, Set B, and Set C, respectively whereas Set D (Control) showed a decrease of approximately 5.69% over 28 days of storage time at 4℃. The reducing power of the blueberry jam showed a reduction during storage at 4℃ for 28 days. Radical scavenging activity of blueberry jam was varied with that of control because of the starters used. Set C showed good DPPH scavenging activity in comparison with Set A and Set B. Four weeks of storage at 4℃ reduced the DPPH scavenging activity of blueberry jam.
Free radicals play an essential role in numerous chronic pathologies as they are involved in the process of lipid peroxidation. A compound with radical scavenging property serves as a potential antioxidant.67,78
Cholesterol-lowering property
The lowering of serum cholesterol level could be an important health benefit of lactic acid bacteria, demonstrated in humans as well as in animal studies.79 In the present study, blueberry jam fermented with probiotic lactic acid starters showed to reduce the in vitro cholesterol level (Fig. 1).80 observed the same results in cocktail fermented with Lactobacillus plantarum and noted the reduction of 13.6% after weeks of consumption.81 reported the significant level of reduction in the total cholesterol and low-density lipoprotein-c level after the consumption of kimchi by the young, healthy volunteers. No literature was found for in vitro reduction of cholesterol by fruit jam. The cholesterol level and its fraction levels in serum were reduced after the treatment with the probiotic bacteria B. animalis VKL, B. animalis VKB and L. casei IMV B-7280 in 6-8 weeks old female BALB /c mice but the effects were strain-specific on serum lipid profiles.82 L. plantarum SK-3 and P. acidilactici M-3 showed to reduce the in vitro cholesterol level in both probiotic noni as well as probiotic mulberry juice.77
Sensory evaluation
Appearance serves as a preliminary parameter for the acceptance of food and indicates the strength of fruit products for consumption. The lacto-fermented blueberry jam was rated by the panellists, which is quite reasonable for a product. The difference in appearance among different sets of the lacto-fermented blueberry jam was negligible. Appearance was rated 8.01 ± 0.23, 8.00 ± 0.28, 8.20 ± 0.14 and 7.45 ± 0.19 for Set A, Set B, Set C and Control, respectively. Flavour means an overall integrated perception of taste and aroma associated with the product.82 The flavour for the lacto-fermented blueberry jam was rated as 7.05 ± 0.08 for Set A, 7.13 ± 0.19 for Set B, 8.08 ± 0.12 for Set C, and 6.75 ± 0.11 for Control. The texture is related to the mouth-feel of a food product. It was rated highest for Set C as 8.10 ± 0.23, 7.25 ± 0.21 for Set B, 7.10 ± 0.29 for Set A and lowest for Control as 6.85 ± 0.17. Overall acceptability is based on multiple organoleptic quality parameters, i.e., colour, flavour, texture, etc. and reveals the accumulative perception and acceptance by the panellists. Overall acceptability was observed as 7.30 ± 0.12 in Set A, 7.70 ± 0.16 in Set B, 8.28 ± 0.20 in Set C, and 6.80 ± 0.05 in Control, as shown in Fig. 2.
Conclusion
In the present study, the survival of L. plantarum, L. fermentum, P. acidilactici, and P. pentosaceus probiotic strains in the blueberry jam was reported for the first time. The study assessed the physicochemical changes during fermentation and storage period. The viability of different probiotic strains was also examined in a blueberry jam during fermentation and under refrigerated storage. The probiotic cultures added to the jam did not remarkably modify the colour and appearance of the product; however, the metabolism of lactobacilli changes the pH which, in turn, accelerates the rate of hydrolysis of sugar into simpler ones. Fruit jam represents a suitable carrier for the delivery of probiotics. Blueberries are a good candidate for producing a novel and healthy non-dairy probiotic food which could effectively deliver probiotic L. plantarum, L. fermentum, P. acidilactici and P. pentosaceus strains under refrigerated conditions. Incorporation of probiotics into fruit jam makes the jam healthier as fruits are naturally rich in essential macro- and micro-elements. Concerning the performance, L. plantarum DB-2, L. fermentum J-1, P. acidilactici M-3, L. plantarum SK-3, and P. pentosaceus SM-2 were observed to be suitable for fermentation of blueberry jam and may have the ability for a possible industrial application in the production of lacto-fermented blueberry jam. Further work on optimizing the fermentation conditions and in vitro and in vivo functionality of the fermented blueberry jam is highly recommended. Lacto-fermented blueberry jam can increase consumer acceptability.
Compliance with Ethics Requirements
Funding
No scholarship/ fellowship/ funding received for this study.
Conflict of interest
The authors declare no commercial or financial conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
References
- Brambilla A, Lo Scalzo R, Bertolo G, et al. Steam-blanched High-bush blueberry (Vaccinium corymbosum L.) juice: phenolic profile and antioxidant capacity in relation to cultivar selection. J Agric Food Chem. 2008; 56(8): 2643- 2648.
- Eichholz I, Huyskens-Keil S, Kroh LW, et al. Phenolic compounds, pectin and antioxidant activity in blueberries (Vaccinium corymbosum L.) influenced by boron and mulch cover. Journal of Applied Botany and Food Quality. 2011; 84(1): 26-32.
- Howard LR, Castrodale C, Brownmiller C, et al. Jam processing and storage effects on blueberry polyphenolics and antioxidant capacity. Journal of Agricultural and Food Chemistry. 2010; 58(7):4022-4029.
- Koca I, Karadeniz B. Antioxidant properties of blackberry and blueberry fruits grown in Black sea Region of Turkey. Scientia Horiculturae. 2009; 121(4): 447- 450.
- Bolling S. Blueberries make their mark on cardiovascular and diabetes risks. Retrieved from http://www.med.umich.edu/prmc/media/newsroom/details.cfm?ID=1113; 2009.
- Basu B, Hazra B. Evaluation of nitric oxide scavenging activity, in vitro and ex vivo, of selected medicinal plants traditionally used in inflammatory diseases. Phytother Res. 2006; 20:896-900.
- Pranprawit A. Effect of Highbush blueberry consumption on markers of metabolic syndrome. PhD Thesis, Massey University, Palmerston North, New Zealand; 2014.
- Andres-Lacueva C, Shukitt-Hale B, Galli RL, et al. Anthocyanins in aged blueberry-fed rats are found centrally and may enhance memory. Nutritional Neuroscience. 2005; 8:111-120.
- Krikorian R, Shidler MD, Nash TA, et al. Blueberry supplementation improves memory in older adults. Journal of Agricultural and Food Chemistry. 2010; 58(7):3996-4000.
- Willis L, Bickford P, Zaman V, et al. Blueberry extract enhances survival of intraocular hippocampal transplants. Cell Transplant 2005; 14(4):213-223.
- Bomser J, Madhavi DL, Singletary K, et al. In vitro anticancer activity of fruit extracts from Vaccinium species. Planta Medica. 1996; 62(3):212-216.
- Vinson JA, Su X, Zubik L, et al. Phenol antioxidant quantity and quality in foods and fruits. Journal of Agriculture and Food Chemistry. 2001; 49:5315-5321.
- Schmidt M, Erdman J, Ann Lila M. Effects on food processing on blueberry antiproliferation and antioxidant activity. Journal of Food Science. 2005; 70:389-394.
- Yi W, Akoh CC, Fischer J, Krewer G. Effects of phenolic compounds in blueberries and muscadine grapes on HepG2 cell viability and apoptosis. Food Research International. 2006; 39(5): 628-638.
- Kajimoto O. Blueberries and eyesight. Food Style. 1999; 21(3):3.
- Molan A, Lila M, Mawson J, et al. In vitro and in vivo evaluation of prebiotic activity of water-soluble blueberry extracts. World Journal of Microbiology and Biotechnology. 2009; 25(7):1243-1249.
- Connolly DAJ, McHugh MP, Padilla-Zakour OI. Efficacy of a tart cherry juice blend in preventing the symptoms of muscle damage. British Journal of Sports Medicine. 2006; 40:679-683.
- Galvano F, Fauci LL, Lazzarino G, Fogliano V. Cyanidins: metabolism and biological properties. Journal of Nutrition and Biochemistry. 2004; 15:2-11.
- Hurst RD, Wells RW, Hurst SM, et al. Blueberry fruit polyphenolics suppress oxidative stress-induced skeletal muscle cell damage in vitro. Molecular Nutrition and Food Research. 2010; 54(3):353-363.
- McLeay Y, Barnes M, Mundel T, et al. Effect of New Zealand blueberry consumption on recovery from eccentric exercise-induced muscle damage. Journal of the International Society of Sports Nutrition. 2012; 9(1):19.
- Molan A, Lila M, Mawson J. Satiety in rats following blueberry extract consumption induced by appetite-suppressing mechanisms unrelated to in vitro and in vivo antioxidant capacity. Food Chem. 2008; 107(3):1039-1044.
- Almeida SP. “Frutas nativas do cerrado,” in Cerrado: Ambiente e Flora. Embrapa-CPAC, Planaltina. 1998; 244-285.
- Silva DB, Silva JA, Junqueira NTV, and Andrade LRM. “Frutas do cerrado,” Embrapa Bras´ılia, 2001.
- Agostini-costa T, Vieira RF. Frutas Nativas do Cerrado: Qualidade Nutricional e Sabor Peculiar. Embrapa Recursos Gen´eticos e Biotecnologia, Brasilia; 2000.
- FAO/ WHO. (2002). Guidelines for the evaluation of probiotics in food. Report of a Joint Food and Agriculture Organization of the United Nations, World Health Organization Working Group of Drafting Guidelines for the Evaluation of Probiotic in food, Ontario, Canadá, 2002.
- Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014; 8:506–14.
- Donkor ON, Nilmini SLI, Stolic P. Survival and activity of selected probiotic organisms in set-type yoghurt during cold storage. International Dairy Journal. 2007; 17:657-65.
- Salminen SJ, Gueimonde M & Isolauri E. Probiotics that modify disease risk. J Nutr. 2005; 135: 1294–1298.
- Kumar M, Behare PV, Mohania D, Arora S, Kaur A & Nagpal R. Health-promoting probiotic functional foods: potential and prospects. Agro Food Ind Hi Tech. 2009(a); 20: 29–33.
- Kumar M, Mohania D, Poddar D, Behare PV, Nagpal R, Kumar A & Aggarwal PK. A probiotic fermented milk prepared by mixed culture combination reduces pathogen shedding and alleviates disease signs in rats challenged with pathogens. Int J Probiotics Prebiotics. 2009(b); 4: 211–218.
- Kumar M, Kumar A, Nagpal R. Cancer-preventing attributes of probiotics: an update. Int J Food Sci Nutr. 2010; 61:473–496.
- Nagpal R and Kaur A. Synbiotic effect of various prebiotics on in-vitro activities of probiotic lactobacilli. Ecol Food Nutr. 2011; 50: 63–68.
- Nagpal R, Yadav H, Puniya AK, Singh K, Jain S and Marotta F. Potential of probiotics and prebiotics for synbiotic functional dairy foods. Int J Probiotics Prebiotics. 2007; 2: 75–84.
- Nagpal R, Kumar A and Arora S. In-vitro probiotic potential of lactobacilli from indigenous milk products. Int J Probiotics Prebiotics. 2010; 5: 103–110.
- Yadav H, Jain S and Sinha PR. Formation of oligosaccharides in skim milk fermented with mixed dahi cultures, Lactococcus lactis ssp. diacetylactis and probiotic strains of Lactobacilli. J Dairy Res. 2007(a); 74: 154–159.
- Yadav H, Jain S and Sinha PR. Anti-diabetic effect of probiotic dahi containing Lactobacillus acidophilus, Lactobacillus casei and Lactococcus lactis bacteria in high fructose diet fed rats. Nutrition. 2007(b); 72: 62–68.
- Yadav H, Jain S and Sinha PR. Oral administration of dahi containing probiotic Lactobacillus acidophilus and Lactobacillus casei ameliorated the Streptozotocin-induced oxidative stress and dyslipidemia in rats. J Dairy Res. 2008; 75: 189–195.
- Yoon KY, Woodams EE, Hang YD. Probiotication of tomato juice by lactic acid bacteria. Journal of Microbiological Methods. 2004; 42:315-318.
- Marhamatizadeh MH, Rezazadeh S, Kazemeini F, et al. The study of probiotic juice product conditions supplemented by the culture of Lactobacillus acidophilus and Bifidobacterium bifidum. Middle-East J Sci Res. 2012; 11(3):287-295.
- Rößle C, Auty MAE, Brunton N, Gormley RT, Butler F. Evaluation of fresh-cut apple slices enriched with probiotic bacteria. Innova Food Sci Emerg Technol. 2010; 11: 203–209.
- Pereira ALF, Maciel TC, Rodrigues S. Probiotic beverage from cashew apple juice fermented with Lactobacillus casei. Food Res Int. 2011; 44: 1276–1283.
- Nagpal R, Ashwani K and Kumar M. Fortification and fermentation of fruit juices with probiotic lactobacilli, Annals of Microbiology. 2012; 62(4):1573-1578.
- Sheehan VM, Ross P, Fitzgerald GF. Assessing the acid tolerance and the technological robustness of probiotic cultures for fortification in fruit juices. Innovative Food Science and Emerging Technologies. 2007; 8:279–284.
- Tripathi, MK and Giri, SK. Probiotic functional foods: Survival of probiotics during processing and storage. Journal of Functional Foods. 2014; 9: 225–241.
- Luckow T, Delahunty C. Which juice is ‘healthier’? A consumer study of probiotic non-dairy juice drinks. Food Quality and Preference. 2004; 15:751-759.
- Liong MT and Shah NP. Acid and bile tolerance and cholesterol removal ability of Lactobacilli strains. Journal of Dairy Science. 2005; 88:55-56.
- Walker D K and Gilliland S E. Relationships among bile tolerance, bile salts deconjugation and assimilation of cholesterol by Lactobacillus acidophilus. Journal of Dairy Science. 1993; 76:956-961.
- Thirabunyanon M, Boonprasom P and Niamsup P. Probiotic potential of lactic acid bacteria isolated from fermented dairy milks on antiproliferation of colon cancer cells. Biotechnology Letters. 2009; 31:571–576.
- Harrigan W F. Laboratory methods in food microbiology. Academic Press: New York, 1998.
- Harrigan W F and McCance M E. Laboratory Methods in Food and Dairy Microbiology. Academic Press, London, 1990.
- Del Re B, Sgorbati B, Miglioli M and Palenzona D. Adhesion, autoaggregation and hydrophobicity of 13 strains of Bifidobacterium longum. Letters in Applied Microbiology. 2000; 31:438-442.
- Rosenberg M, Gutnick D and Rosenberg E. Adherence of bacteria to hydrocarbons: a simple method for measuring cell surface hydrophobicity. FEMS Microbiology Letters. 1980; 9:29-33.
- Papagianni M and Anastasiadou S. Pediocins: the bacteriocins of Pediococci: sources, production, properties and applications. Microbial Cell Factories. 2009; 8:3.
- AOAC. Official methods of analysis of association of official analytical chemists, 16th edn. Vol I and II. Association of Official Analytical Chemists. Arlington, Virginia, USA, 1995.
- Mora D, Fortina MG, Parini C, Ricci G, Gatti M, Giraffa G and Manachini PL. Genetic diversity and technological properties of Streptococcus thermophilus strains isolated from dairy products. Journal of Applied Microbiology. 2002; 93:278-287.
- Hashem S, Sabit HH, Amin M., Tawakkot W and Shamseldin AF. Molecular characterization of Egyptian isolates of Lactobacillus and Bifidobacterium. Journal of American Science, 2010; 6(11):959-964.
- Aditya Chaudhary and Baljeet Singh Saharan. Probiotic properties of Lactobacillus plantarum. Journal of Pure and Applied Microbiology. 2019; 13(2): 933-948.
- Benmeziane F, Djamai R, Cadot Y, et al. Optimization of extraction parameters of phenolic compounds from Algerian fresh table grapes, (Vitis Vinifera). International Food Research Journal. 2014; 21(3): 1061-1065.
- AOAC. Official methods of analysis of the association of official analytical chemist, Hortwits W (ed). Association of official analytical chemists, Washington, D. C. USA, 1984.
- Miller NJ, Rice-Evans CA. The relative contributions of ascorbic acid and phenolic antioxidants to the total antioxidant activity of orange and apple fruit juices and blackcurrant drink. Food Chem. 1997; 60:331-337.
- Duh PD, Yen GC. Antioxidative activity of three herbal water extracts. Food Chem. 1997; 60:639-645.
- Shimada K, Fujikawa K, Yahara K, et al. Antioxidative properties of xanthan on the autooxidation of soybean oil in cyclodextrin. J Agric Food Chem. 1992; 40:945-948.
- David B Fankhauser. Pour plate technique for bacterial enumeration. Uni. Of Cincinnati Clermont College. Batavia OH; 2005, 45103:1-3.
- Meilgaard CC. Sensory Evaluation Techniques 3rd Edition,” CRC Press, Boca Raton, 1999.
- Damiani C, Asquieri ER, Lage ME, et al., “Study of the shelf-life of amixed arac¸a (Psidium guineensis Sw.) and marolo (Annona crassiflora Mart.) jam,” Food Science and Technology. 2012; 32(2):334–343.
- Mesquita KS, Borges SV, Carneiro JDDS, et al. Quality alterations during storage of sugarfree guava jam with added prebiotics,” Journal of Food Processing and Preservation. 2013; 37(5):806–813.
- Rababah TM, Al-U’Datt M, Al-Mahasneh M. Effect of Jam processing and storage on phytochemicals and physiochemical properties of cherry at different temperatures. Journal of Food Processing and Preservation. 2014; 38(1):247–254.
- Khouryieh HA, Aramouni FM, Herald TJ. Physical, chemical and sensory properties of sugar-free jelly. Journal of Food Quality. 2005; 28(2):179–190.
- Sadler GD, Murphy PA. pH and titrable acidity. In: Nielsen, S. (Ed.): Food analysis. 4th edition. New York, Springer US, 2010; 219–238.
- Angelov A, Gotcheva V, Hristozova T, Gargova S. Application of pure and mixed probiotic lactic acid bacteria and yeast cultures for oat fermentation. J Sci Food Agric. 2005; 85:2134-2141.
- Ong L, Henriksson A, Shah NP. Development of probiotic cheddar cheese containing Lactobacillus acidophilus, Lb. casei, Lb. paracasei and Bifidobacterium spp. and the influence of these bacteria on proteolytic patterns and production of organic acid. International Dairy Journal. 2006; 16: 446-456.
- Vinderola CG, Mocchiutti P, Reinhermer JA. Interactions among lactic acid starter and probiotic bacteria used for fermented dairy products. Journal of Dairy Science. 2002; 85:721- 729.
- Salminen S, Von Wright A. Lactic Acid Bacteria. New York: Marcel Dekker Inc., 1993.
- Saarela M, Virkajärvi I, Alakomi HL, et al. Stability and functionality of freeze-dried probiotic Bifidobacterium cells during storage in juice and milk. International Dairy Journal. 2006; 16(12),1477- 1482.
- Champagne CP, Roy D, Gardner NJ. Challenges in addition of probiotic cultures to foods. Critical Reviews in Food Science and Nutrition. 2005; 4:61-84.
- Kyung YY. Woodams EE, Hang YD. Fermentation of beet juice by beneficial lactic acid bacteria. Lebensmittel Wissenschaft Und Technologie. 2005; 38:73-75.
- Aditya Chaudhary, Vishnu Sharma and Baljeet Singh Saharan. Probiotic potential of Noni and Mulberry Juice fermented with lactic acid bacteria. Asian J Dairy Food Res. 2019; 38(2):114-120.
- Khan T, Ahmad M, Khan R, et al. Evaluation of phytomedicinal potentials of selected plants of Pakistan. Am. Lab. 2006; 38(9): 20-22.
- Wang CH, Lai P, Chen ME, et al. Antioxidative capacity produced by Bifidobacterium- and Lactobacillis acidophilus-mediated fermentations of konjac glucomannan and glucomannan oligosaccharides. J Sci Food Agric. 2008; 88:1294-1300.
- Natural news. Lowering cholesterol with fermented foods. http://www.nyrnaturalnews.com/diet-2/2012/11/lowering-cholesterol-with-fermented-foods, 2012.
- Choi IH, Noh JS, Han JS, et al. Kimchi, a fermented vegetable, improves serum lipid profiles in healthy young adults: Randomized clinical trial. J Med Food. 2013; 6(3):223-229.
- Bubnov RV, Babenko LP, Lazarenko LM, Mokrozub VV, Demchenko OA, Nechypurenko OV, Spivak MY. Comparative study of probiotic effects of Lactobacillus and Bifidobacteria strains on cholesterol levels, liver morphology and the gut microbiota in obese mice. EPMA J. 2017;8(4):357-376.
- Meilgard M, Caviled GV, Carr BT. Sensory evaluation techniques 4th edition. CRC Press Inc., 2007.

This work is licensed under a Creative Commons Attribution 4.0 International License.







