Comparative Study on Chemical Composition and Antioxidant Activity of Annonamuricata Plant Parts Cultivated in Covenant University, Ota, Ogun State, Nigeria
Department of Biochemistry, College of Science and Technology, Covenant University, Km 10, Idiroko Road, Canaanland, P.M.B. 1023, Ota, Ogun State, Nigeria.
Corresponding Author Email: lola.omotosho@covenantuniversity.edu.ng
DOI : http://dx.doi.org/10.12944/CRNFSJ.6.3.23
ABSTRACT:
Annonamuricata plant parts possess a broad range of medicinal and biological properties. This research compared the chemical composition and antioxidant properties of Annonamuric at aparts. Proximate, mineral, total phenol and total flavonoid content as well as invitro antioxidant activity were examined. Results revealed the leaves contained significantly(p<0.05) higher composition of moisture (8.69±0.22%), ash (4.60±0.02%), protein (14.53±0.11%), crude fat (10.28±0.03%),chromium (0.38±0.05 mg/100g), nickel (1.75±0.04mg/100g), total phenol (1.01±0.03mgpyrocatechol/mL) and total flavonoid (1.12±0.03mgGAE/mL)compared to the respective values for root. Carbohydrate (9.29±0.24%), lead (0.13±0.02mg/100g) and cobalt (1.93±0.02mg/100g) composition was significantly lower(p<0.05) in the leaves compared to the respective compositions in the root. The leaf and root extract exhibited a concentration-dependent increase in hydroxyl radical scavenging activity with no observable(p<0.05)difference in their EC50 value. This study suggests the leaves of A. muricata found in Covenant University had better chemical composition when compared to the root. Nonetheless, these plant parts may be further exploited for not only their nutritive composition and mineral content but also a natural source of antioxidant agents.
KEYWORDS:Annonamuricata; Antioxidant; Favonoid ; Mineral content; Proximate analysis; Scavenging activity
Introduction
Plants are a rich bio resource of natural products and phyto constituents which make them potent as functional foods and for treatment and management of diseases.1 Despite the availability of various varieties of synthetic drugs which are highly effective in curing various diseases, there are people who still prefer using traditional folk medicines and therapy as a result of their less harmful effects. There is a wide diversity of compounds found in plants, especially secondary plant metabolites, identified and isolated from plants.2 Various studies have shown that these compounds exhibit various properties such as anticancer, antibacterial, analgesic, anti-inflammatory, antitumor and antiviral. Their ethnomedicinal use by the locals, suggests easy access which makes them an easily obtainable source of treatment.3 Annonamuricata L. commonly known as soursop, graviola, guanabana and sirsak which belongs to the Annonaceae family, is found in tropical and subtropical regions such as South and Central America, Asia and Africa including Nigeria.1 Soursop is the most versatile fruit from the Annonaceae family utilised in industrial processes due to its low oxidising rate as well as a huge retrieval of fruit pulp during processing. The fruit is 15-30 cm long with an oval irregular shape and sparse soft green curved spines. It has a white cotton like mesocarp which is fibrous, contains shiny black seeds, a sour taste, pleasing flavour and aroma.4 Extensive phytochemical evaluations carried out on Annona muricata L. different parts (leaves, roots, fruits, stem, bark and seeds) have shown the presence of several bioactive compounds such as alkaloids, megastigmanes, flavonoltriglycosides, phenolics, cyclopeptides, essential oils and annonaceous acetogenin compounds. Major minerals such as K, Ca, Na, Cu, Fe and Mg have been reported to be present,5 which makes them ideal dietary sources of electrolytes, essential nutrients and elements which are utilised in various biological processes.4 The fruit is used to produce juice, candy and sherbets.6 The leaves and seeds are known to possess anti-arthritic,7 anti-cancer,8 anticonvulsant,9 anti diabetic and hypolipidemic,10 anti-inflammatory and anti-nociceptive ,11 antioxidant ,12 antihypertensive13 and anti parasitic14 activities. The roots are known for their antihelmintic, antiphlogistic14, antiparasitic and pesticidal properties.6 Despite a broad study on these plant parts, there is a paucity of information on the comparative chemical composition of the plant parts. Hence, the current study was carried out to compare the proximate composition, mineral content and in vitro antioxidant activity of Annonamuricata leaves and roots found in Covenant University, Ota, Nigeria.
Materials and Methods
Sample Collection and Preparation
The leaves and roots of Annona muricata were obtained from Covenant University farm, Canaan land, Ota, Ogun State, Nigeria in December 2013 and identified. Samples were dried for two weeks at room temperature and pulverised. Dried leaf and root powder (50g) was packed into a Soxh let apparatus and macerated with 250mL methanol at 60–65°C for 3–4h. The extract was filtered through What man filter paper No. 1, and the filtrate was concentrated under reduced pressure at 40°C using a rotary evaporator.
Chemicals and Reagents
Sodium Carbonate (Na2CO3), Sodium Nitrate (NaNO3), Nitric Acid (HNO3), Aluminium Chloride (NaOH) and Hydrogen Peroxide (H2O2), were purchased from Merck, Germany. All other chemicals used were of analytical grade.
Proximate Analysis
Leaves and root of A. muricatawere pulverised to uniform size and analysed for moisture, crude protein, crude fat, ash, and crude fibre composition according to the standard method of AOAC 15.950.01, 15.976.05, 15.920.39, 15.955.03 and 15.962.09, respectively as described below.14
Determination of Moisture Content
Dry crucible in the oven and cool in the desiccator for 15 minutes. Weigh empty petri dish to get (W1), Weigh in 1.5g (liquid samples) or 2g (solid samples) into the petri dish and record as (W2) Put in oven for 6-12hrs at 105oC. Remove and cool in desiccators. Weigh after cooling to get (W3)
Calculation
![]()
W1 = weight of empty crucible.
W2 = weight of sample and crucible.
W3 = weight of after drying in the oven
Determination of Crude Protein
0.5-1g of dried samples was taken in digestion flask. 10-15mL of concentrated H2SO4 and 8g of digestion mixture i.e. K2SO4:CuSO4 (8:1) was added to the sample. The flask was swirled in order to mix the contents thoroughly then placed on heater to start digestion till the mixture became clear (blue green in colour). After 2 hrs the digest was cooled, transferred to 100mL volume tric flask and the volume was made up to mark by the addition of distilled water. 10mL of digest was introduced in the distillation tube then 10 mL of 0.5 N NaOH was gradually added. Distillation was continued for at least 10 min and NH3 produced was collected as NH4OH in a conical flask containing 20 mL of 4% boric acid solution with few drops of modified methyl red indicator. The distillate was then titrated against standard 0.1N HCl solution till the appearance of pink colour. A blank was also run through all steps as above.
Calculations
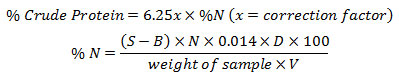
S = Sample titration reading
B = Blank titration reading
N = Normality of HCl
D = Dilution of sample after digestion
V = Volume taken for distillation
0.014 = Milli equivalent weight of Nitrogen
Determination of Crude Fat
1 g of moisture free sample was wrapped in filter paper, placed in fat free thimble and then introduced in the extraction tube. The receiving beaker was filled with petroleum ether and fitted into the apparatus. After 4-6 siphoning, ether was allowed to evaporate and beaker was removed before last siphoning. Extract was transferred into a clean glass dish with either washing and evaporated ether on water bath. Dish was placed in an oven at 105oC for 2 hrs and cooled in a desiccator.
Calculation
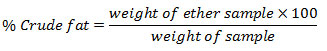
Determination of Ash
For the determination of ash, clean empty crucible was placed in a muffle furnace at 600oC for an hour, cooled in desiccator and then weight of empty crucible was noted (W1). One gram of each of sample was taken in crucible (W2). Then the crucible was placed in muffle furnace at 550oC for 2-4 h. The appearances of grey white ash indicated complete oxidation of all organic matter in the sample. After ashing furnace was switched off. The crucible was cooled and weighed (W3).
Calculation
![]()
W1 = weight of empty crucible.
W2 = weight of sample and crucible.
W3 = weight of after drying in the oven
Determination of Crude Fibre
0.153g of sample was weighed (W0) and transferred to porous crucible. 150 mL of preheated H2SO4 solution and some drops of foam-suppresser was added to each column. Sample was boiled and left for 30 mins. Valves were opened for drainage of acid and rinsed with distilled water thrice to completely ensure the removal of acid from sample. The same procedure was used for alkali digestion by using KOH instead of H2SO4. Sample was oven dried at 150oC for 1 hr, then cooled in a desiccator and weighed (W1). The sample crucibles were kept in muffle furnace at 55oC for 3-4 hrs and later cooled in a desiccator and weighed again (W2).
Calculations
![]()
Determination of Carbohydrate
Carbohydrate was calculated by difference after analysis of all the other items method in the proximate analysis, i.e.
Mineral Determination
The official method of AOAC15 was adopted for the mineral analysis of the samples: 1g of the sample was weighed in a Vycor dish, dried for 1hr at 150oC in air forced oven and then ashed overnight (16h) at 550oC before cooling in a desiccator. One mL of HNO3 was added to dissolve the ash. The sample is then transferred to a 250mL volumetric flask and made up to volume with H2O. Sodium (Na) and potassium (K) levels of the samples were ascertained using a flame emission photometer with NaCl and KCl as standards. All other metals were determined by atomic absorption spectrometry (AAS) method.16
Total Phenol Quantification
Total phenolic content of the extracts was analysed using the Folin-Ciocalteau reagent method described byLee et al.,17 with slight modification. 10μL of the extract was added to 600mL of distilled water followed by 50μL of 10% of Folin-Ciocalteau reagent. 150μL of 7% Na2CO3 was added and vor texed. The mixture was left at room temperature for 8 mins before 190μL of water was added and kept at room temperature for 2 hrs. Absorbance was read at 765nm. Total phenolic content was calculated from the calibration curve, and the results were expressed as mg of pyrocathecol equivalent per mL.
Total Flavonoid Quantification
The total flavonoid content of the crude extract was determined by the aluminium chloride colour imetric method described by Baba and Malik,18 with slight modification. Distilled water (490μL) was added to 10μL of extract, 30μL of 5% Sodium Nitrate, 30μL of 10% AlCl and incubated at room temperature for 5 mins. One M NaOH followed by 240μl of H2O was added and vortexed thoroughly. Absorbance was read at 510nm. The total flavonoid content was calculated from a calibration curve, and the result was expressed as mg gallic acid equivalent per mL.
Hydrogen Peroxide Scavenging Activity
The ability of extract to scavenge H2O2 was determined according to the method of Sharma et al.,19 with slight modification. Hundred μL of extract was incubated with 0.6mL of H2O2 (40mM in a phosphate buffer, 0.1M pH 7.4) in dark for 10mins. A negative control was set up in parallel with entire reagent except for either extract or standard. The absorbance of H2O2 at 230 nm was determined against a blank solution containing phosphate buffer.
Calculation
![]()
Where
A control is the absorbance of the control reaction
A test is the absorbance in the presence of the sample extract.
Statistical Analysis
Results were expressed as mean ± standard error of mean (SEM) of triplicate values. Statistical analysis was performed by one-way ANOVA followed by Dun can test as post hoc. IBM SPSS statistics 23 was used and a probability (p) value < 0.05 was considered to be statistically significant.
Result
Proximate Composition
The proximate composition as reported in table 1 on a dry weight basis showed there was a significantly (p<0.05) higher composition of moisture, ash, crude fat and protein in the leaves (8.69±0.22 %, 4.60±0.02 %, 10.28±0.03 % and 14.53±0.11 %) than that of the roots(2.40±0.03 %, 1.20±0.06 %, 6.46±0.04 % and 7.53±0.11 %). Carbohydrate was significantly(p<0.05) higher in the root(27.23±0.14 %) than the leaves (9.29±0.24 %).However, the crude fibre composition of leaves (52.63±0.36 %) was not significantly (p<0.05) different from that of the roots (55.19±0.02 %).
Table 1: Proximate Analysis of A. Muricata Leaves and Roots
| SAMPLE | LEAF | ROOT |
| Moisture (%) | 8.69±0.22a | 2.40±0.03b |
| Ash (%) | 4.60±0.02a | 1.20±0.06b |
| Crude Fats (%) | 10.28±0.03a | 6.46±0.04b |
| Crude Fibre (%) | 52.63±0.36a | 55.19±0.22a |
| Protein (%) | 14.53±0.11a | 7.53±0.11b |
| Carbohydrate (%) | 9.29±0.24a | 27.23±0.14b |
Values are expressed as mean ± SEM of 3 replicates. abValues with different superscript on a row are significantly different (p<0.05)
Mineral Composition
Mineral content as revealed in table 2 revealed chromium and nickel were significantly (p<0.05) higher in leaves (0.38±0.05, 1.75±0.04mg/100g) compared to the root (0.30±0.05, 1.25±0.03mg/100g) respectively. Lead and cobalt content was significantly (p<0.05) higher in roots (0.23±0.05, 2.80±0.04mg/100g) compared to the leaves (0.13±0.02, 1.93±0.02mg/100g) respectively. There was no significant (p<0.05) difference between the amount of zinc, cadmium, copper, magnesium, sodium, calcium, potassium and iron detected in the leaves and root.
Table 2: Mineral Content of A. Muricata Leaves and Roots
| SAMPLE | LEAF | ROOT |
| Zinc (mg/100g) | 8.70±0.29a | 8.40±0.18a |
| Cadmium (mg/100g) | 5.23±0.09a | 5.50±0.10a |
| Chromium (mg/100g) | 0.38±0.05a | 0.30±0.05b |
| Lead (mg/100g) | 0.13±0.02a | 0.23±0.05b |
| Copper (mg/100g) | 7.35±0.11a | 7.80±0.09a |
| Nickel (mg/100g) | 1.75±0.04a | 1.25±0.03b |
| Cobalt (mg/100g) | 1.93±0.02a | 2.80±0.04b |
| Magnesium (mg/100g) | 30.73±0.21a | 32.80±0.27a |
| Sodium (mg/100g) | 49.88±0.12a | 51.28±0.20a |
| Calcium (mg/100g) | 155.03±0.39a | 151.30±0.57a |
| Potassium (mg/100g) | 23.95±0.19a | 24.88±0.44a |
| Iron (mg/100g) | 37.55±0.23a | 38.55±0.88a |
Values are expressed as mean ± SEM of 3 replicates. abValues with different superscript on a row are significantly different (p<0.05)
Total Phenolic and Flavonoid Quantification
Results recorded in table 3 show total phenolic and flavonoid content was significantly (p<0.05) higher in the leaves (1.01±0.03 mg pyrocatechol/mL, 1.12±0.03 mgGAE/mL) compared with the root (0.74±0.02mg pyrocatechol/mL, 0.25±0.01mgGAE/mL). There was no significant (p<0.05) difference in the EC50 value for hydroxyl radical scavenging activity of the leaf (2.92±0.02) and root extract (2.65±0.03).
Table 3: Total Phenolic, Flavonoid Composition and EC50 Values for Hydroxyl Radical Scavenging Activity
| SAMPLE | Leaves | Roots |
| Total Phenol (mgpyrocatechol/mL) | 1.01±0.03a | 0.74±0.02b |
| Total Flavonoid (mgGAE/mL) | 1.12±0.03a | 0.25±0.01b |
| EC50 | 2.92±0.02a | 2.65±0.03a |
Values are expressed as mean ± SEM of 3 replicates. abValues with different superscript on a row are significantly different (p<0.05)
Hydroxyl Radical Scavenging Activity
A concentration-dependent increase in hydroxyl radical scavenging activity of A. muricata leaf and root methanolic extract was depicted in figure 1. There was, however, a slight scavenging activity decrease in the highest concentration of the leaf extract.
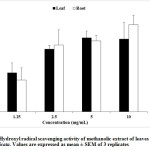 |
Figure 1: Hydroxyl radical scavenging activity of methanolic extract of leaves and roots of A. muricata. Values are expressed as mean ± SEM of 3 replicates |
Discussion
The results from the proximate composition of the leaves and root of A. muricata further show their nutritive and medicinal properties. Cellular metabolism thrives on and requires energy provided by carbohydrate to run continuously.20 Dietary fibre improves motility in the digestive system and plays a role in cardiovascular diseases and cancer prevention.21 The amount of crude fibre present in the leaf and root of A. muricata in this study, were very high compared to that of seed previously reported. However, the inverse was the case for the carbohydrate content as it was higher in the seed.22 The high carbohydrate and fibre content of the root shows that it can be used to aid digestion and as a source of energy.23 The moisture content of the leaves suggests it may be a better source of hydration, but none the less, it may be more prone to microbial attack during storage than the roots. This indicates that the leaf may have reduced shelf life which corresponds with a study on Costus afer24 as a high moisture content of any biological matter makes it highly susceptible to microbial attack during storage.25 The high crude fat, protein and ash contents of the leaf suggest that they could be used as a better source of plant fat, protein and minerals in feed supplementation. Fat is an essential macro molecule needed for energy and absorption of fat-soluble vitamins.26 Proteins are essential components of diets required for energy generation, maintenance of body tissues and synthesis of enzymes, hormones as well as other substances required for healthy functioning.27,28 The crude protein content of the leaves and roots shows A. muricata may be used as a natural protein supplement for animals. The protein and fat content of the leaves are higher than the 12.5% and 1.49% reported for Nauclea latifolia leaf but lower than the reported protein (27.74% and 20.72%) content for Vitex doniana and Moringa oleifera leaves respectively.29 The reported high fat content of the leaves and root were in line with the reported values (8.3 – 27.0%) of leafy vegetables.29 It is also corroborated by the findings of Agu and Okolie30 on the leaf and root extracts which may be as a result of the high presence of annonaceous acetogenins which are long chain fatty acids derivative.Minerals play a massive role in metabolic pathways, disease prevention and management.Their presence in A. muricata leaves and roots may be the reason for the folkloric use in treatment and management of diseases.24 High concentration of minerals was observed to be present in the leaves and root of A. muricata with calcium, sodium, iron, magnesium and potassium being the most abundant. Calcium is essential for healthy bones, teeth, blood, muscles,31 as well as absorption of dietary vitamin B, for the synthesis of the neurotransmitter acetylcho line and immune response.32 Magnesium, potassium as well as calcium are involved in enzyme synthesis, co factors for enzyme activation, biological structure promoter and optimal physiological function.33,34 Sodium is required for optimal acid-base balance, maintenance of osmotic pressure and cellular homeostasis.35 Iron is important for animal survival as it plays a role in respiration, DNA synthesis and blood functioning.36 Chromium prevents diabetes by being directly involved in insulin production and function.37 All other minerals present in the leaves and root play a part in the optimal function of various physiological and biochemical processes.38 Interestingly results of mineral content analysis of the leaves were in contrast with that of Usunomena and Paulinus39 except chromium which had almost similar value. This observation, may be as a result of the soil location, where this plant was cultivated, as well as the environmental practices of the surrounding populace. Phenolic substances are abundant low molecular weight bio active compounds in plants which have various health benefits.40 The dose-dependent increase in the scavenging capacity of the leaves and roots may be attributed to the presence of flavonoids and phenolics in the methanolic extracts, as they are known to be soluble in polar solvents.24 These plant secondary metabolites are known to possess potent antioxidant capacity which may be as a result of the presence of the hydroxyl groups present in their ring structures. These groups are effective in scavenging of reactive oxygen species by donating and accepting electrons with free radicals there by quenching them.36,41 The findings agree with the studies carried out by Agu and Okolie,30 Kalidindi et al.,42 and Mariod et al.,43 attributing the antioxidant properties of their plants to the phenol and flavonoid content.
Conclusion
This study suggests the leaves of A. muricata have more phenolic and flavonoid contents compared to the root. Nonetheless, these plant parts are a rich source of nutrients and can be capitalised for feed supplementation, the potential to supply sufficient amount of minerals for consumers and microbial media for micro organisms. Besides their nutritional and mineral value, they possess antioxidant activity, thus may be utilised as an alternative natural source of antioxidants.
Acknowledgement
The authors are grateful to Covenant University Centre for Research and Discovery, Ota, Ogun State for providing a platform for the publication of the research work.
Conflicts of Interest
None
References
- Moghadamtousi S., Fadaeinasab M., Nikzad S., Mohan G., Ali H., Kadir H. Annona muricata (Annonaceae): A review of its traditional uses, isolated acetogenins and biological activities. International Journal of Molecular Sciences. 16(7): 15625-15658.
CrossRef - Iqbal E., Salim K. A., Lim L. B. Phytochemical screening, total phenolics and antioxidant activities of bark and leaf extracts of Goniothalamus velutinus (Airy Shaw) from Brunei Darussalam. Journal of King Saud University-Science. 2015;27(3):224-232.
CrossRef - Somsak V., Polwiang N., Chachiyo S.In vivo anti malarial activity of Annona muricata leaf extract in mice infected with Plasmodium berghei.Journal of Pathogens;2016: Article ID. 2016;3264070.
- Iombor T.T., Olaitan I. N., Ede R. A. Proximate composition, antinutrient content and functional properties of soursop flour as influenced by oven and freeze drying methods. Current Research in Nutrition and Food Science. 2014;2(2):106-110.
CrossRef - Moghadamtousi S., Rouhollahi E., Hajrezaie M., Karimian H., Abdulla M., Kadir H.Annona muricata leaves accelerate wound healing in rats via involvement of Hsp70 and antioxidant defence. International Journal of Surgery. 2015;18: 110-117.
CrossRef - Adefegha S., Oyeleye S., Oboh G. Distribution of phenolic contents, antidiabetic potentials, antihypertensive properties and antioxidative effects of soursop (Annona muricata) fruit parts in vitro. Biochemistry Research International; 2015:Article ID 347673: 2015.
- Chan P., Ah R., Mh K. Anti-arthritic activities of Annona muricata leaves extract on complete Freund’s adjuvant (CFA)-induced arthritis in rats. Planta Medica. 2010;76(12):166.
CrossRef - Asare G., Afriyie D., Ngala R., Abutiate H., Doku D., Mahmood S., Rahman H. Antiproliferative activity of aqueous leaf extract of Annona muricata on the prostate, BPH-1 cells, and some target genes. Integrative Cancer Therapies. 2015;14(1):65–74.
CrossRef - N’gouemo P., Koudogbo B., Tchivounda H., Akono-Nguema C., Etoua M. Effects of ethanol extract of Annona muricata on pentylenetetrazol-induced convulsive seizures in mice. Phytotherapy Research. 1997;11(3):243–245.
CrossRef - Adeyemi D., Komolafe O., Adewole O., Obuotor E., Adenowo T. Anti-hyperglycemic activities of Annona muricata (Linn). African Journal of Traditional, Complementary and Alternative Medicine. 2009;6(1):62–69.
- Roslida A., Tay C., Zuraini A., Chan P. Anti-inflammatory and anti-nociceptive activities of the ethanolic extract of Annona muricata Journal of Natural Remedies. 2010;10(2);97–104.
- George V., Kumar D., Suresh P. Antioxidant, DNA protective efficacy and HPLC analysis of Annona muricata (soursop) extracts. Journal of Food Science and Technology. 2015;52(4);2328–2335.
CrossRef - Nwokocha C., Owu D., Gordon A., Thaxter K., McCalla G., Ozolua R., Young L. Possible mechanisms of action of the hypotensive effect of Annona muricata (soursop) in normotensive Sprague-Dawley rats. Pharmaceutical Biology. 2012; 50(11);1436–1441.
CrossRef - Osorio E., Arango G. J., Jiménez N., Alzate F., Ruiz G., Gutiérrez D., Paco M. A., Giménez A. Robledo S.Antiprotozoal and cytotoxic activities in vitro of Colombian Annonaceae. Journal of Ethnopharmacology. 111(3):630–635.
CrossRef - 1990. The Official Methods of Analysis of the Association of Official Analytical Chemists 15 ed. Arlington, Virginia.
- Pohl P., Dzimitrowicz A., Jedryczko D., Szymczycha-Madeja A., Welna M., Jamroz P.The determination of elements in herbal teas and medicinal plant formulations and their tisanes. Journal of Pharmaceutical and Biomedical Analysis. 2016;130:326-335.
CrossRef - Leng L. Y., Nadzri N, Shaari A. R., Norawanis A. R., Khor C. Y. Antioxidant capacity and total phenolic content of fresh, oven-dried and stir-fried tamarind leaves. Current Research in Nutrition and Food Science. 2017;5(3):282-287.
CrossRef - Baba S., Malik S. Determination of total phenolic and flavonoid content, antimicrobial and antioxidant activity of a root extract of Arisaema jacquemontii Journal of Taibah University for Science. 2015;9(4):449–454.
- Sharma A., Goyal R., Sharma L. Potential biological efficacy of Pinus plant species against oxidative, inflammatory and microbial disorders. BMC Complementary and Alternative Medicine. 2016;16(1):35-46.
CrossRef - Mensah J., Okoli R., Ohaju-Obodo J., Eifediyi K. Phytochemical, nutritional and medicinal properties of some leafy vegetables consumed by Edo people of Nigeria. African Journal of Biotechnology. 2008;7(14):2304-2309.
- Chinedu S., Oluwadamisi A., Popoola S., David B., Epelle T. Analyses of the leaf, fruit and seed of Thaumatococcus daniellii (Benth): Exploring potential uses. Pakistan Journal of Biological Sciences. 2014;17(6):849-854.
CrossRef - Onimawo I. A. Proximate composition and selected physicochemical properties of the seed, pulp and oil of sour sop (Annona muricata). Plant Foods for Human Nutrition. 2002;57:165-171.
CrossRef - Pazhanichamy K., Pavithra S., Rubini S., Lavanya B., Ramya I., Eevera T. Morphological, anatomical and proximate analysis of leaf, root, rhizome of Costus igneus. Journal of Pharmacy Research. 2010;3(4):747-752.
- Anyasor G., Onajobi F., Osilesi O., Adebawo O. Proximate composition, mineral content and in vitro antioxidant activity of leaf and stem of Costus afer (Ginger lily).Journal of Intercultural Ethnopharmacology. 2014;3(3):128-134.
- Hassan L., Muhammad M., Umar K., Sokoto A. Comparative study on the proximate and mineral contents of the seed and pulp of sugar apple (Annona squamosa). Nigerian Journal of Basic and Applied Science. 2008;16(2):179-182.
- Balogun I.O., Olatidoye O. P. Chemical composition and nutritional evaluation of velvet bean seeds (Mucuna utilis) for domestic consumption and industrial utilization in Nigeria. Pakistan Journal of Nutrition. 2012;11(2):116.
CrossRef - Ogunyinka B. I., Oyinloye B. E., Osunsanmi F. O., Kappo A. P., Opoku A.R. Comparative study on proximate, functional, mineral, and antinutrient composition of fermented, defatted, and protein isolate of Parkia biglobosa Food Science and Nutrition. 2017;5(1):139-147.
CrossRef - Francis E. U., Augustine A., Ozioma O. P. Comparative assessment of the nutritional and antioxidant status of Euphorbia heterophylla (Euphorbiaceae) and Morinda lucida (Rubiaceae) plants. Nigerian Journal of Pharmaceutical and Applied Science Research. 2016;5(2):49-57.
- Eze O. S., Ernest O. Phytochemical and nutrient evaluation of the leaves and fruits of Nauclea latifolia (Uvuru-ilu). Communications in Applied Sciences. 2014;2(1):8–24.
- Agu K. C., Okolie P. N. Proximate composition, phytochemical analysis, and in vitro antioxidant potentials of extracts of Annona muricata (Soursop). Food science & Nutrition. 2018;5(5):1029-1036.
CrossRef - Charles P. Calcium absorption and calcium bioavailability. Journal of Internal Medicine. 1992;231(2):161-165.
CrossRef - Pu F., Chen N., Xue S. Calcium intake, calcium homeostasis and health. Food Science and Human Wellness. 2016;5(1):8-16.
CrossRef - Alcázar A. Electrolyte and acid-base balance disorders in advanced chronic kidney disease. Nefrologia. 2008;28:87-93.
- Cowan J. Structural and catalytic chemistry of magnesium dependent enzymes. Biometals. 2002;15(3):225-235.
CrossRef - Farquhar W., Edwards D., Jurkovitz C., Weintraub W. Dietary sodium and health: more than just blood pressure. Journal of American College of Cardiology. 2015;65(10):1042–1050.
CrossRef - Kumari A., Parida A., Rangani J., Panda A. Antioxidant activities, metabolic profiling, proximate analysis, mineral nutrient composition of Salvadora persica fruit unravel a potential functional food and a natural source of pharmaceuticals. Frontiers in Pharmacology. 8: Article 61.
CrossRef - Sarfo D., Gyamfi K., Ampomah-Amoako E., Nyarko B., Serfor-Armah Y., Akaho E. Studies on the mineral composition of the leaves of graviola plant, Annona muricata, from some selected municipalities in Ghana by activation analysis. Elixir Food Science. 2011;41:5676-5680.
- Bamidele O., OjedokunO., Fasogbon B. Physico-chemical properties of instant ogbono (Irvingia gabonensis) mix powder. Food Science and Nutrition. 2015;3(4):313–318.
CrossRef - Usunomena U., Paulinus O. N. Phytochemical analysis and mineral composition of Annona muricata International Journal of Research and Current Development. 2016;1(1):7-10.
- Archana A., Babu K. Nutrient composition and antioxidant activity of gonads of sea urchin Stomopneustes variolaris. Food Chemistry. 2016;197:597–602.
CrossRef - Iheagwam F. N., Nsedu E. I., Kayode K. O., Emiloju O. C., Ogunlana O. O., Chinedu S. N. Bioactive screening and in vitro antioxidant assessment of Nauclea latifolia leaf decoction. AIP Conference Proceedings. 2018;1954(1):030015.
CrossRef - Kalidindi N., Thimmaiah N. V., Jagadeesh N. V., Nandeep R., Swetha S., Kalidindi B. Antifungal and antioxidant activities of organic and aqueous extracts of Annona squamosa leaves. Journal of Food and Drug Analysis. 2015;23(4):795-802.
CrossRef - Mariod A. A., Abdelwahab S. I., Elkheir S., Ahmed Y. M., Fauzi P. N. M., Chuen C. S. Antioxidant activity of different parts from Annona squamosa, and Catunaregam nilotica methanolic extract. Acta Scientiarum Polonorum Technologia Alimentaria. 2012;11(3):249-258.













