Microencapsulation of Peanut Worm (Siphonosoma australe) Extract from Indonesian Marine Waters as a Natural Umami Flavor Enhancer Obtained by Spray Drying
1Department of Food Technology, Muhammadiyah University Semarang, Semarang, Indonesia
2Department of Agribusiness, University of Muhammadiyah Semarang, Semarang, Indonesia
3Department of Maritime, University of Muhammadiyah Semarang, Semarang, Indonesia
4Department of Agricultural Product Technology, Sriwijaya University, Indonesia
Corresponding Author Email: yonata@unimus.ac.id
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.3.14
ABSTRACT:The peanut worm (Siphonosoma australe), a marine organism endemic to the coastal waters of Sulawesi, Indonesia, is rich in glutamic acid, making it a promising natural source of umami flavor. This study aimed to develop and characterize spray-dried microcapsules of peanut worm extract (MPWE) using maltodextrin (MD) and gum arabic (GA) as encapsulating agents, individually and in combination, with β-cyclodextrin (βCD) added to improve encapsulation efficiency. The spray-drying process yielded 53.26–70.98% of microcapsules retaining L-glutamic acid contents of 2594.33–3871.33 mg/100 g, with recovery rates of 31.26–61.81%. All formulations exhibited desirable physicochemical properties, including low moisture content (2.12–3.77%), water activity (0.31–0.35), hygroscopicity (8.27–11.20%), high solubility (88.78–98.84%), and good flowability. The microcapsules had particle sizes ranging from 5.71 to 8.47 μm with heterogeneous, wrinkled surfaces characteristic of spray-dried powders. Incorporation of βCD significantly enhanced the physicochemical stability of MPWE, though a slight reduction in solubility was observed. Among all formulations, the MD–GA–βCD (MGβ) combination produced the most favorable characteristics, suggesting its potential as an optimal encapsulation system for producing stable, umami-rich microcapsules from marine-derived ingredients. This work introduces a novel marine-derived umami microcapsule with strong potential for application in functional and savory food industries.
KEYWORDS:Glutamic acid; Marine-derived ingredients; Microcapsule; Molecular inclusion; Physicochemical properties; Umami flavor
Introduction
The peanut worm (Siphonosoma australe), a marine invertebrate belonging to the phylum Sipuncula, is characterized by its peanut-like shape and is predominantly distributed along the southern coast of Sulawesi, Indonesia.1 Traditionally, local communities have utilized peanut worms as snacks and traditional medicine.2 To enhance their value as a fishery resource, it is essential to develop innovative processing strategies that transform this underutilized species into high-value and sustainable products.3-5
Recent studies have revealed that peanut worms are rich in protein, with glutamic acid as the dominant amino acid.6 Given that glutamic acid imparts the umami taste, peanut worms have strong potential as a natural umami flavor enhancer. Extraction typically releases free glutamic acid, the key contributor to umami flavor.7,8 Acid-assisted extraction has been shown to improve the yield, purity, and stability of protein-derived bioactive compounds,9 with citric acid proving particularly effective for producing peanut worm extracts with high glutamic acid content.6 However, the extracted glutamic acid is unstable and prone to degradation during processing, limiting its practical application.10 This limitation can be effectively mitigated through microencapsulation technology.7,8
Microencapsulation enables controlled release and protection of active compounds, improving their stability and usability in food applications. Among various encapsulation techniques, spray drying is widely recognized for its efficiency, scalability, and cost-effectiveness in producing umami microcapsules.11 The selection of wall materials is a critical factor influencing encapsulation efficiency and stability.12 A combination of maltodextrin (MD) and gum arabic (GA) has been reported as an effective coating system for flavor compounds, offering good film-forming ability and emulsifying properties.13,14 However, MD–GA microcapsules often exhibit hygroscopicity and reduced shelf stability.8,15 Recent studies have demonstrated that incorporating β-cyclodextrin (βCD) into the encapsulation matrix can significantly enhance the retention of flavor and aroma compounds during processing and storage.16,17
βCD-based inclusion complexes have gained increasing attention for stabilizing and controlling the release of bioactive molecules.18 The unique structure of βCD, with a hydrophilic outer surface and a hydrophobic internal cavity,19,20 allows it to form stable complexes with various bioactive compounds.21,22 Despite its excellent thermal stability, βCD exhibits relatively low solubility in water, whereas MD and GA are more soluble but prone to moisture absorption. Therefore, combining MD and GA with βCD as composite wall materials may balance solubility, stability, and moisture resistance, thereby improving microcapsule performance.
To date, no study has reported the development of umami microcapsules from peanut worm extract using composite wall materials containing MD, GA, and βCD. Therefore, this study aims to prepare and characterize spray-dried peanut worm extract microcapsules using different combinations of these encapsulating agents. This research provides a novel approach to enhancing the functional and economic potential of Indonesian marine resources by producing stable, umami-rich microcapsules for use in the food industry.
Materials and Methods
Materials
This study sourced peanut worms as the primary raw material from fishermen at Toronipa Beach, Southeast Sulawesi. Extraction was performed using distilled water and commercial citric acid (Koepoe-Koepoe, Indonesia). Microencapsulation was carried out using three wall materials—maltodextrin (MD) (Lihua Starch, China), gum Arabic (GA) (Ingredion, Thailand), and beta-cyclodextrin (βCD) (Landor Trading Company, Thailand). All analytical procedures employed reagents of pro-analytical grade, which were sourced from Sigma (Sigma-Aldrich, USA).
Preparation of the microcapsules
The extraction process commenced with immersing 100 g of cleaned and dried peanut worms in 500 mL of 3% citric acid at ambient temperature for 36 h. The mixture was then passed through a Whatman No. 1 filter paper under vacuum. The filtrate obtained was adjusted to neutral pH using 1 N NaOH and subsequently concentrated by evaporation at 50 °C for 30 min. This extract served as the core material for spray-drying microencapsulation.4,5 For the preparation of each wall-material formulation (Table 1), 40 g of the designated coating agent was dissolved in distilled water (100 mL) and mixed using a homogenizer at 3000 rpm for 30 min. Afterward, 100 mL of the peanut worm extract was incorporated into the solution, yielding a feed mixture with approximately 45% total solids, and homogenization was continued for an additional 30 min at the same speed. The feed was spray-dried under operating conditions of 150 ± 1 °C inlet temperature, 80 ± 2 °C outlet temperature, a feed rate of 6.0 mL/min, and an atomization pressure of 5 bar. The microcapsule powders produced were immediately transferred into amber glass containers and stored at freezing temperatures until analysis. Formulations coated solely with MD served as control samples.
Table 1: Composition of coating materials
|
Code |
Coating Materials (%) | ||
| MD | GA |
βCD |
|
|
MD |
100 | 0 | 0 |
| MDβ | 98 | 0 |
2 |
|
GA |
0 | 100 | 0 |
| GAβ | 0 | 98 |
2 |
|
MG |
50 | 50 | 0 |
| MGβ | 49 | 49 |
2 |
MD: maltodextrin; MDβ: maltodextrin + β-cyclodextrin; GA: gum Arabic; GAβ: gum Arabic + β-cyclodextrin; MG: maltodextrin and gum Arabic; MGβ: maltodextrin and gum Arabic + β-cyclodextrin
Yield, glutamic acid content and glutamic acid recovery
The spray-drying yield was quantified by comparing the mass of the final powder with the total solids present in the initial raw material. The determination of glutamic acid concentration followed a previously described procedure.7 L-glutamate levels were assessed using an L-glutamic acid assay kit, in which the increase in absorbance at 492 nm was monitored using a UV–Vis spectrophotometer (Shimadzu UV-Vis 1601, Japan). For the analysis, 0.1 mL of the diluted sample (mixed with 2 mL distilled water) was combined with 0.5 mL of solution 1 (35 mL pH 8.6 buffer containing 0.02% w/v sodium azide), 0.2 mL of solution 2 (containing NAD⁺ and INT), and 0.05 mL of suspension 3 (diaforase). After a 2-minute incubation period, the initial absorbance (A₁) was measured at 492 nm. The reaction was then initiated by adding 0.05 mL of solution 4 (glutamate dehydrogenase, GIDH), and the mixture was incubated for approximately 8–10 minutes until completion, afterward the final absorbance (A₂) was recorded. The absorbance change (ΔA L-glutamate) was obtained by subtracting A₁ from A₂. The recovery of glutamic acid from the microencapsulated peanut worm extracts produced with different wall materials was then computed using the respective formula.
Color profile
Moisture content was quantified using a Shimadzu MOC63u moisture analyzer (Japan), while water activity was measured at 25 °C with a Rotronic Hygropalm-HP23 Aw-A instrument (Switzerland).
Hygroscopicity
The hygroscopic properties of the microcapsules were evaluated by placing 1 g of each sample in a sealed container equilibrated with saturated NaCl, which maintains approximately 75% relative humidity. The samples were stored at 25 °C for seven days. Hygroscopicity was expressed as the amount of water vapor absorbed per 100 g of dry sample.23
Solubility
Solubility was determined by weighing 1 g of the microcapsule powder and dispersing it in 100 mL of distilled water with magnetic stirring (10 minutes). The mixture was subsequently passed through a filter, and the obtained filtrate was dried at 105 °C until a stable weight was achieved. The solubility value was reported as the fraction of the initial solids that entered the aqueous phase.23
Dissolution time
The dissolution time was evaluated based on an established method.23 In this test, 1 g of the microcapsule powder was introduced into 100 mL of distilled water contained in a 250 mL beaker at room temperature. The time taken for the particles to completely submerge and no longer remain visible on the surface was recorded as the dissolution time.
Dispersibility
To assess dispersibility, 1 g of the microcapsule powder was combined with 10 mL of distilled water in a 50 mL beaker and stirred for 30 minutes to obtain a uniform dispersion free of sedimented clumps. The resulting mixture was filtered through a 200-mesh sieve, and the retained residue was subsequently dried to a constant mass.23
![]()
Within the formula, a refers to the sample mass applied in the analysis, b represents the powder’s moisture level, and TS corresponds to the quantified total solid fraction in the filtrate.
Flow properties
Bulk density (Bd) was measured by filling a 100 mL graduated cylinder with microcapsules up to the 25 mL mark and weighing the sample. The weight and volume data were used to calculate Bd in units of g/cm³. Tapped density (Td) was determined using the same procedure, with the addition of mechanical tapping of the cylinder until the volume stabilized. Td was calculated using the same formula as Bd [7,8]. Flowability of the microcapsules was evaluated using the compressibility index (CI) and Hausner ratio (HR), both derived from Bd and Td values according to equations previously reported by Areekal et al.24
Particle size distribution and morphology structure
Particle size analysis of the microcapsules was performed using a laser light–scattering technique with an LLPA-C10 particle size analyzer (Labron Equipment Ltd., UK). Surface morphology was observed using scanning electron microscopy (SEM) on a JSM-6510LA unit (Jeol Ltd., Japan). Before SEM observation, the samples were sputter-coated with a thin layer of gold and examined at an accelerating voltage of 12 kV.
Statistical Analysis
All measurements were performed out in five independent replicates, and data are reported as the mean with the standard deviation. Statistical processing was conducted using SPSS version 20.0. A one-way ANOVA was employed to assess differences among treatments at a 95% confidence level, followed by Duncan’s Multiple Range Test (DMRT) for post-analysis comparisons when significant effects were detected (p < 0.05).
Results
The results of this study are presented in Figures 1A–1C, which illustrate the yield, glutamic acid content, and glutamic acid recovery; Figures 2A–2B, which show the dissolution time and permeability; Figure 3, depicting the particle size distribution; and Figure 4, demonstrating the structural morphology. In addition, Table 2 presents the color profile, Table 3 summarizes the physical properties, and Table 4 reports the flow properties.
 |
Figure 1: Yield (A), glutamic acid content (B), and glutamic acid recovery (C) of microcapsule form peanut worm extract. |
Table 2: Color profile of microcapsule form peanut worm extract
|
Coating materials |
L* | a* | b* | WI |
|
MD |
94.65 ± 0.72d | 0.20 ± 0.04a | 2.37 ± 0.29a | 94.14 ± 0.69d |
| MDβ | 95.05 ± 0.87d | 0.17 ± 0.07a | 2.16 ± 0.22a |
94.59 ± 0.81d |
|
GA |
88.48 ± 0.85a | 0.21 ± 0.05a | 5.02 ± 0.22c | 87.43 ± 0.79a |
| GAβ | 90.88 ± 0.47b | 0.15 ± 0.06a | 2.82 ± 0.22b |
90.45 ± 0.44b |
|
MG |
91.38 ± 0.80bc | 0.17 ± 0.09a | 2.89 ± 0.20b | 90.90 ± 0.71b |
| MGβ | 92.32 ± 0.56c | 0.16 ± 0.05a | 2.75 ± 0.13b |
91.83 ± 0.55c |
Results are reported as the means ± standard deviations (n = 4).
Values within the same column sharing identical superscript letters do not differ significantly (p < 0.05).
MD: maltodextrin; MDβ: maltodextrin + β-cyclodextrin; GA: gum Arabic; GAβ: gum Arabic + β-cyclodextrin; MG: maltodextrin and gum Arabic; MGβ: maltodextrin and gum Arabic + β-cyclodextrin.
Table 3: Physical properties of microcapsule form peanut worm extract
|
Coating materials |
Moisture (%) | aw | Hygroscopicity (%) | Solubility (%) |
| MD | 2.12 ± 0.09a | 0.31 ± 0.02a | 11.29 ± 0.30f |
98.84 ± 0.43f |
|
MDβ |
2.43 ± 0.04b | 0.32 ± 0.02a | 9.90 ± 0.29e | 97.16 ± 0.78e |
| GA | 3.45 ± 0.06d | 0.35 ± 0.03a | 8.62 ± 0.30b |
90.57 ± 0.68b |
|
GAβ |
3.77 ± 0.07e | 0.33 ± 0.02a | 8.27 ± 0.21a | 88.78 ± 0.53a |
| MG | 2.38 ± 0.08b | 0.34 ± 0.02a | 9.50 ± 0.11d |
94.96 ± 0.54d |
|
MGβ |
2.69 ± 0.08c | 0.33 ± 0.02a | 9.14 ± 0.16c |
93.08 ± 0.92c |
Results are reported as the means ± standard deviations (n = 4).
Values within the same column sharing identical superscript letters do not differ significantly (p < 0.05).
MD: maltodextrin; MDβ: maltodextrin + β-cyclodextrin; GA: gum Arabic; GAβ: gum Arabic + β-cyclodextrin; MG: maltodextrin and gum Arabic; MGβ: maltodextrin and gum Arabic + β-cyclodextrin.
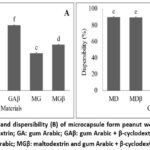 |
Figure 2: Dissolution time (A), and dispersibility (B) of microcapsule form peanut worm extract. |
Table 4: Flow properties of microcapsule form peanut worm extract
|
Coating materials |
Bd (g/cm3) | Td (g/cm3) | CI (%) | HR | Flowability |
| MD | 0.25 ± 0.01a | 0.28 ± 0.01a | 9.82 ± 0.82a | 1.11 ± 0.01a |
Excellent |
|
MDβ |
0.30 ± 0.01b | 0.35 ± 0.01b | 14.58 ± 0.54b | 1.17 ± 0.01b | Good |
| GA | 0.41 ± 0.01e | 0.53 ± 0.01e | 22.67 ± 1.36d | 1.29 ± 0.02d |
Passable |
|
GAβ |
0.44 ± 0.01f | 0.58 ± 0.01f | 24.78 ± 0.52e | 1.33 ± 0.01e | Passable |
| MG | 0.33 ± 0.01c | 0.40 ± 0.01c | 15.76 ± 1.26b | 1.19 ± 0.02b |
Fair |
|
MGβ |
0.39 ± 0.01d | 0.48 ± 0.01d | 19.73 ± 0.68c | 1.25 ± 0.01c |
Fair |
Results are reported as the means ± standard deviations (n = 4).
Values within the same column sharing identical superscript letters do not differ significantly (p < 0.05).
MD: maltodextrin; MDβ: maltodextrin + β-cyclodextrin; GA: gum Arabic; GAβ: gum Arabic + β-cyclodextrin; MG: maltodextrin and gum Arabic; MGβ: maltodextrin and gum Arabic + β-cyclodextrin.
 |
Figure 3: Particle size distribution of microcapsule form peanut worm extract. A: maltodextrin; B: maltodextrin + β-cyclodextrin; C: gum Arabic; |
 |
Figure 4: Morphology structure of microcapsule form peanut worm extract. A: maltodextrin; B: maltodextrin + β-cyclodextrin; C: gum Arabic; |
Discussion
Yield, glutamic acid content, and glutamic acid recovery
The product yield was determined by comparing the mass of the dried solids with the solid content present in the feed mixture. As illustrated in Figure 1A, the yield values varied between 53.26% to 70.98%. Overall, the MPWE samples produced using MD exhibited higher yields than those prepared with GA. The addition of βCD as part of the wall material markedly improved the yield, with the MGβ formulation achieving the highest recovery among all treatments. The lower yield observed for GA-based microcapsules is likely associated with the hydrophilic and branched molecular structure of GA, which can impede efficient atomization and increase wall deposition during spray drying, ultimately reducing product recovery.17 Incorporation of βCD into the encapsulating matrix enhanced the formation of molecular inclusion complexes during homogenization and elevated the glass transition temperature (Tg) of the resulting dried particles. When the Tg exceeds the drying temperature, undesirable physicochemical phenomena such as stickiness, caking, and thermal degradation are diminished, thereby improving coating performance and overall product yield.23,25 Collectively, these results demonstrate that βCD contributes to better stabilization of the microencapsulation system throughout the spray-drying process.
The L-glutamic acid levels in MPWE were found to range between 2584.33 and 3871.33 mg/100 g (Figure 1B), markedly exceeding those previously reported for umami microcapsules produced from seaweed extract (180–460 mg/100 g).7 The incorporation of MD as an encapsulating agent proved effective in safeguarding the umami constituents of PWE. This material exhibits high solubility and forms a porous matrix that can effectively entrap glutamic acid molecules.17 Notably, microcapsules formulated with GA exhibited even greater L-glutamic acid retention than those coated solely with MD, which may be attributed to GA’s enhanced film-forming properties that improve the preservation of core components.8 The addition of βCD to form composite wall systems (MDβ, GAβ, and MGβ) led to a further rise in L-glutamic acid content. This enhancement is likely associated with the ability of βCD to establish inclusion complexes within its hydrophobic cavity, supported by hydrogen bonding that facilitates the encapsulation of glutamic acid. The robust thermal stability of βCD also minimizes compound degradation during spray drying.17,26 Glutamic acid recovery rates, which varied from 41.24% to 61.81% (Figure 1C), demonstrate the protective efficiency of the different coating matrices. These recovery values closely correspond to the measured L-glutamic acid concentrations, reinforcing the role of βCD in mitigating oxidative and thermal losses throughout processing. The superior recovery observed in βCD-enriched formulations is likely driven by the concurrent effects of inclusion complex formation and the elevated glass transition temperature, both of which contribute to improved stability during the drying process.
Color profile
Color is one of the crucial attributes in food products, as consumers are highly attracted to products with visually appealing colors. Ideally, umami-enhancing ingredients should exhibit a high degree of brightness and remain visually neutral to avoid introducing color changes to the final product. According to Table 2, the microcapsules showed lightness (L*) values ranging from 88.58 to 95.05, indicating their relatively pale appearance. The luminance of the microcapsules was dictated mainly by the intrinsic color properties of the coating materials, which exerted a pronounced influence on the resulting L* values. Previous studies reported that the L* values of different coating materials varied considerably, with βCD exhibiting the highest L* value, followed by MD, while GA showed the lowest.17 No significant differences were observed in the a* values of the microcapsules (0.15–0.20), although a slight decreasing trend was noted with the presence of βCD. Meanwhile, higher b* values (2.16–5.02) were observed in microcapsules prepared with GA as the coating material, indicating a brighter yellowish hue characteristic of GA. This also contributed to the decrease in the whiteness index (WI) of the microcapsules prepared with GA. In general, MPWE exhibited a bright white color, superior to umami flavor enhancer microcapsules derived from seaweed extract and crab by-products.7,8 Microcapsules with bright color characteristics have broad applications in the food industry.27
Moisture content and water activity (aw)
Moisture content plays an important role in determining product quality, particularly in relation to shelf life, hygroscopicity, and texture. The moisture content of the produced MPWE ranged from 2.12% to 3.77% (Table 3). All microcapsules formulated with composite wall materials containing βCD exhibited relatively higher moisture content. Similar findings were reported in microcapsules of grape extract,28 seaweed extract,17 and turmeric extract.23 The hydrophobic cavity formed through the molecular inclusion of βCD allows more water to be trapped, most of which is difficult to evaporate during the drying of the feed solution.29 Microcapsules with moisture content below 5% are considered ideal and are known to have good long-term storage stability.30 Previous researchers suggested that the moisture content of spray-dried powders should be within the range of 4–5% to ensure storage stability.31 In contrast to the moisture content, the water activity (aw) of the microcapsules showed no statistically significant variation among formulations, with values ranging from 0.31 to 0.35. Despite the slight fluctuations, all treatments fell within the acceptable range for dry powdered products. Typically, aw in such products should remain below 0.4, as higher levels may facilitate the growth of spoilage microorganisms and promote aggregation during storage.32 Some studies have proposed a more lenient upper limit of 0.6 for dry materials.33 Nevertheless, maintaining aw below 0.4 is generally regarded as optimal to ensure long-term stability and minimize quality degradation.
Hygroscopicity and solubility
Hygroscopicity, a property associated with the uptake of moisture from the environment, directly impacts the stability, longevity, and flow performance of microcapsules. MPWE exhibited hygroscopicity levels ranging from 8.27% to 11.29%. MD-based microcapsules showed markedly higher hygroscopicity, whereas incorporating βCD into the coating composition significantly reduced this value (Table 3). GA has been reported to have a higher glass transition temperature compared to MD.34 The glass transition temperature of each coating material is a key factor determining the hygroscopicity of microcapsules. Furthermore, the decrease in hygroscopicity is also related to the limited presence of hydrogen and hydroxyl groups in the GA and βCD molecules.35 High hygroscopicity can cause aggregation in spray-dried powders, thereby affecting the stability of the encapsulated bioactive components and the flowability of the resulting microcapsules.23 Solubility represents a key quality parameter for powdered food products, indicating the extent to which the particles can completely dissolve during application.8 The solubility of MPWE ranged from 88.78% to 98.84% (Table 3), which is similar to that found in umami flavor enhancer microcapsules from seaweed extract, ranging from 93.18% to 94.49%.17 MD as a coating material exhibited better solubility compared to GA or βCD. This is related to the higher number of hydroxyl groups in MD molecules compared to GA and βCD,17,36 while βCD has a hydrophobic structure that leads to lower solubility, especially at room temperature.23 Previous studies have reported that spray-dried microcapsules prepared with βCD as the coating material showed solubility levels of no more than 60%.37
Dissolution time and dispersibility
The dissolution time of MPWE in this study ranged from 57.27 to 199.68 seconds (Figure 2A). An ideal flavor enhancer is expected to dissolve quickly, not float on the surface, and completely sink and dissolve in the solution. As shown in Figure 2A, the trend of MPWE dissolution time was similar to its solubility pattern (Table 3). Coating materials with abundant hydroxyl groups accelerated the dissolution time of the resulting microcapsules, whereas hydrophobic structures delayed the dissolution process.17 Furthermore, microcapsules with faster dissolution times exhibited better dispersibility. As shown in Figure 2B, MPWE prepared with MD as the coating material had the highest dispersibility (90.06%), while that prepared with GAβ had the lowest (77.70%). Microcapsules with high dispersibility are highly desirable in the food industry.23
Flow properties
The flowability characteristics of the powders were evaluated using bulk density (Bd), tapped density (Td), Carr index (CI), and Hausner ratio (HR), as summarized in Table 4. The GAβ-coated MPWE exhibited the highest Bd value (0.44 g/cm³), along with a correspondingly elevated Td value (0.58 g/cm³). These findings align with previous observations reported for umami microcapsules produced from seaweed extract.7 Powders with higher Bd and Td values generally contain less air, thereby reducing the likelihood of oxidation.38 The particle size distribution is a key factor influencing the Bd and Td values of microcapsules. Powders with smaller particle sizes generally exhibit higher Bd and Td values.8 Smaller particles tend to have fewer hollow structures and better cohesiveness. Good cohesiveness is indicated by low HR and CI values. In line with the Bd and Td results, MD-based MPWE showed the lowest CI (9.82–14.58%) and HR (1.11–1.17) values among all treatments, indicating the best flowability properties.17
Particle size distribution
Particle size distribution is a critical attribute of powdered products because it influences handling behavior during transportation and storage, as well as their physicochemical performance. The particle size profiles of MPWE are shown in Figures 4A–4F, exhibiting a unimodal pattern with mean particle diameters ranging from 5.71 to 8.47 μm. The microcapsules produced in this study were noticeably smaller than previously reported umami microcapsules derived from crab by-products (20.75–25.32 μm)8 and seaweed extract (16.01–34.10 μm).17 With respect to the wall materials, the MG formulation generated smaller particles than MD alone, whereas incorporating βCD as part of the composite coating resulted in larger microcapsules. This trend can be attributed to the higher viscosity of the feed mixture during atomization, as more viscous feeds typically form larger droplets, which in turn result in larger dried particles.35 MD contains more hydroxyl groups, which contribute to increased solution viscosity and may explain the observed differences. An enlargement in particle size with βCD incorporation has also been documented in the microencapsulation of curcumin.23 Although smaller microcapsules are often considered less favorable due to longer wetting times and reduced porosity, they are generally associated with improved technological and functional performance.7,39
Morphology structure
The SEM micrograph illustrating the morphological features of MPWE is shown in Figure 5. Beyond the differences observed in particle size, microcapsules produced with different wall materials displayed markedly distinct surface characteristics. Overall, the peanut worm extract microcapsules exhibited a broad variety of shapes. Notably, incorporating βCD as a coating agent generated particles with more rigid and uniform surfaces, reducing the presence of dents and wrinkles typically formed during heat exposure. This behavior may be related to the elevation of the particles’ glass transition temperature resulting from βCD addition.17 Flavor–βCD inclusion complexes are also known to form sizable aggregates in aqueous systems. These aggregates often possess regular geometric structures and are thought to contribute to improved thermal stability of the liquid phase during drying.8 Encapsulating agents are widely recognized as the main contributors to the morphological traits of microcapsules.40 The degree of smoothness or roughness of particle surfaces provides preliminary insight into the stability of the encapsulated products.18 Generally, smoother particles tend to exhibit lower interparticle interactions, which can be beneficial in specific applications. Conversely, previous studies have noted that microcapsules with rougher surfaces may demonstrate slower release behavior.23
Conclusion
Based on the analysis, each coating material significantly affected the yield, L-glutamic acid content, glutamic acid recovery, and the physicochemical properties of MPWE, except for the aw value. The combination of MGβ as the coating material produced MPWE with superior physicochemical characteristics compared to other coating materials. The yield of MPWE prepared with MGβ reached 70.98%, with a glutamic acid content of 3871.33 mg/100 g and a glutamic acid recovery of approximately 61.81%. Specifically, the presence of βCD in the coating formulation improved the color profile, hygroscopicity, flow properties, particle size, and morphological structure of MPWE compared to MD, GA, and MG. However, a decrease in quality was observed in the solubility and moisture content parameters of the resulting microcapsules. These findings provide valuable insights into the potential of PWE-based umami flavor enhancers in powder form, prepared by spray drying with MGβ as the coating material. The application of the resulting MPWE as a flavor enhancer or seasoning powder is a promising area for further exploration. Production on a small, medium, or large scale is quite promising for achieving high profit margins, given that peanut worms are available for free. However, additional research is needed to evaluate the product’s sensory properties and stability during storage before it can be commercialized.
Acknowledgement
The authors are thankful to the Ministry of Higher Education, Science and Technology, the Republic of Indonesia for financial support through the program of Fundamental Research Grant on behalf of Muhammad Yusuf, Ph.D.
Funding Sources
Fundamental Research Grant [grant numbers 127/C3/DT.04.00/PL/2025;023/LL6/PL/AL.4/
2025;012/061026/ PN/PRF/SP2H/2025] from Ministry of Higher Education, Science and Technology, the Republic of Indonesia.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Muhammad Yusuf: Conceptualization; Validation; Funding acquisition; Writing – Review and Editing.
- Nurul Puspita: Visualization; Project administration
- Aditano Yani Retawimbi: Supervision; Resources.
- Boby Pranata: Data curation; Writing – Review and Editing.
- Diode Yonata: Methodology; Data curation; Writing – Original draft preparation
References
- Bahtiar, Jiwani YE, Findra MN, et al. Temporal variation of peanot worm (Siphonosoma australe-australe) reproduction in Toronipa Beach of Southeast Sulawesi, Indonesia. Biodivers. 2024;25(8):2533-2540. doi:10.13057/biodiv/d250825
CrossRef - Rahayu R, Hudha AM, Sukarsono, et al. Analysis of nutritional content of fresh sea worm honingka (Siphonosoma australe-australe) as a potential food source for communities. In: IOP Conf Ser: Earth and Environ Sci. 2019:1-7. doi:10.1088/1755-1315/276/1/012026
CrossRef - Yusuf M, Hardiwinoto, Trondsen T, et al. Oppurtunities for frozen and processed shrimps to enter the Singaporean and Malaysian markets. Egypt Jour Aqua Bio Fish. 2024;28(3):1063-1072. doi:10.21608/EJABF.2024.361160
CrossRef - Yusuf M, Sukesti F, Puspita N, et al. Innovation to achieve sustainable competitive advantage of processed fishery products sector in Central Java. Egypt Jour Aqua Bio Fish. 2024;28(6):519-531. doi:10.21608/EJABF.2024.392256
CrossRef - Yusuf M, Sya’di YK, Yonata D, et al. Export performance and competitiveness analysis of Indonesian crab products in the ASEAN market. Egypt Jour Aqua Bio Fish. 2024;27(4):983-995. doi:10.21608/EJABF.2023.313718
CrossRef - Suwarjoyowirayatno, Hidayat C, Wahyuningsih TD, et al. Characterization of acid-soluble collagen from peanut worm (Siphonosoma australe) and its inhibitory activity of cyclooxygenase-2. Jour App Bio Biotech. 2024;12(6):123-129. doi:10.7324/JABB.2024.192617
CrossRef - Nurhidajah, Pranata B, Yusuf M, et al. Microencapsulation of umami flavor enhancer from Indonesian waters brown seaweed. Curr Res Nutr Food Scie. 2022;10(1):349-359. doi:10.12944/CRNFSJ.10.1.29
CrossRef - Yusuf M, Yonata D, Pranata P, et al. Utilization of swimming crab by- product as a seafood flavor microcapsule obtained by spray drying. AACL Biofl. 2022;15(2):716-724.
- Petreska-Stanoeva J, Balshikevska E, Stefova M, et al. Comparison of the effect of acids in solvent mixtures for extraction of phenolic compounds from Aronia melanocarpa. Nat Prod Comm. 2020;15(7):1-10. doi:10.1177/1934578X20934675
CrossRef - Zhou LY, Li W, Pan WJ, et al. Effects of thermal processing on nutritional characteristics and non-volatile flavor components from Tricholoma lobayense. Emi Jour Food Agr. 2017;29(4):285-292. doi:10.9755/ejfa.2016-12-1815
CrossRef - Harada-Padermo SdS, Dias-Faceto LS, Selani MM, et al. Umami Ingredient: flavor enhancer from shiitake (Lentinula edodes) byproducts. Food Res Intern. 2020;137:109540. doi:10.1016/j.foodres.2020.109540
CrossRef - Wang Y, Selomulya C. Spray drying strategy for encapsulation of bioactive peptide powder for food application. Adv Pow Techn. 2020;31(1):409-415. doi:10.1016/j.apt.2019.10.034
CrossRef - Bu Y, He W, Zhu L, et al. Effects of different wall materials on stability and umami release of microcapsules of Maillard reaction products derived from Aloididae aloidi. Inter Jour Food Scie Techn. 2021;56(12):6484-6496. doi:10.1111/ijfs.15341
CrossRef - Wu L, Zhang M, Liu Y, et al. Characteristics and release of monosodium glutamate microcapsules obtained by spray drying. Dry Techn. 2019;37(11):1340-1351. doi:10.1080/07373937.2018.1500483
CrossRef - Suparmi, Dewita, Desmelati, et al. Study of the making of hydrolizate protein powder of rebon shrimp as a food nutrition enhancement ingredient. Pharma Jour. 2021;13(5):1180-1185. doi:10.5530/pj.2021.13.151
CrossRef - Pellicer JA, Fortea MI, Trabal J, et al. Stability of microencapsulated strawberry flavour by spray drying, freeze drying and fluid bed. Powder Techn. 2019;347:179-185. doi:10.1016/j.powtec.2019.03.010
CrossRef - Aminah S, Nurrahman, Pranata B, et al. Microencapsulation of seafood flavor enhancers from Indonesian brown seaweed with maltodextrin, Arabic gum, and β-cyclodextrin. Egypt Jour Aqua Bio Fish. 2023;27(2):811-821. doi:10.21608/EJABF.2023.297775
CrossRef - Yonata D, Ulvie YNS, Soesanto E, et al. Stabilization and controlled release of curcumin from temulawak by spray- drying microencapsulation with composite wall materials. Trend in Scie. 2025;22(2):9125. doi:10.48048/tis.2025.9125
CrossRef - Guo X, Wei X, Chen Z, et al. Multifunctional nanoplatforms for subcellular delivery of drugs in cancer therapy. Prog Mat Scie. 2020;107: 100599. doi:10.1016/j.pmatsci.2019.100599
CrossRef - Chew SC, Tan CP, Nyam KL. Microencapsulation of refined kenaf (Hibiscus cannabinus) seed oil by spray drying using β-cyclodextrin/gum Arabic/sodium caseinate. Jour Food Eng. 2018;237:78-85. doi:10.1016/j.jfoodeng.2018.05.016
CrossRef - Saffarionpour S. Nanoencapsulation of hydrophobic food flavor ingredients and their cyclodextrin inclusion complexes. Food Bio Tech. 2019;12:1157-1173. doi:10.1007/s11947-019-02285-z
CrossRef - Benucci I, Mazzocchi C, Kombardelli C, et al. Inclusion of curcumin in b-cyclodextrin: a promising prospective as food ingredient. Food Additives & Contaminants. 2022;39(12):1942-1952. doi:10.1080/19440049.2022.2135764
CrossRef - Rosidi A, Soesanto E, Sulistyowati E, et al. A new approach in preparing curcumin microcapsules from temulawak (Curcuma xanthorrhiza) extract as a source of natural antioxidants for the pharmaceutical and food industries. Curr Res Nutr Food Scie. 2025;13:124-140. doi:10.12944/CRNFSJ.13.Special-Issue-July.08
CrossRef - Areekl NN, Chakkaravarthi A, Debnath S. Effect of microencapsulation on physical properties of powder developed from blended oils rich in PUFA. Journal of Food Science and Technology. 2024;61:2275-2286. doi: 10.1007/s13197-024-05992-4
CrossRef - Nguyen TK, Khalloufi S, Mondor M, et al. Moisture profile analysis of food models undergoing glass transition during air-drying. Journal of Food Engineering. 2020;281:109995. doi: 10.1016/j.jfoodeng.2020.109995
CrossRef - Deng C, Cao C, Zhang Y, et al. Formation and stabilization mechanism of β-cyclodextrin inclusion complex with C10 aroma molecules. Food Hydro. 2022;123:107013. doi:10.1016/j.foodhyd.2021.107013
CrossRef - Toledo NMV, Mondoni J, Harada-Padermo SS, et al. Characterization of apple, pineapple, and melon by-products and their application in cookie formulations as an alternative to enhance the antioxidant capacity. Jour Food Proces Preser. 2019;43(9):14100. doi:10.1111/jfpp.14100
CrossRef - Escobar-Avello D, Mardones C, Saéz V, et al., Pilot-plant scale extraction of phenolic compounds from grape canes: Comprehensive characterization by LC- ESI-LTQ-Orbitrap-MS. Food Res Inter. 2021;143:110265. doi:10.1016/j.foodres.2021.110265
CrossRef - Zhu G, Zhu G, Xiao Z. A review of the production of slow-release flavor by formation inclusion complex with cyclodextrins and their derivatives. Jour Inc Phen Macr Chem. 2019;95(1–2):17–33. doi:10.1007/s10847-019-00929-3
CrossRef - Michalska A, Lech K. The effect of carrier quantity and drying method on the physical properties of apple juice powders. Bever. 2018;4(1):1-15. doi:10.3390/beverages4010002
CrossRef - Ghandi A, Powel IB, Chen XD, et al. The effect of dryer inlet and outlet air temperature and protectant solids on the survival of Lactococcus lactics during spray drying. Drying Technology. 2012;30(4):1649-1657. doi:10.1080/07373937.2012.703743
CrossRef - Kowalska G, Rosicka-Kaczmarek J, Miśkiewicz K, et al. Influence of rye bran heteropolysaccharides on the physicochemical and antioxidant properties of honeydew honey microcapsules. Food Biop Proces. 2021;130:171-181. doi:10.1016/j.fbp.2021.09.014
CrossRef - Chew SC, Tan CP, Nyam KL. Microencapsulation of refined kenaf (Hibiscus cannabinus) seed oil by spray drying using β-cyclodextrin/gum arabic/sodium caseinate. Jour Food Eng. 2018;237:78-85. doi:10.1016/j.jfoodeng.2018.05.016
CrossRef - Laureanti EJG, Paiva TS, de-Matos JLM, et al. Microencapsulation of bioactive compound extracts using maltodextrin and gum arabic by spray and freeze-drying techniques. Inter Jour Bio Macro. 2023;253:126969. doi:10.1016/j.ijbiomac.2023.126969
CrossRef - Wu HT, Chuang YH, Lin HC, et al. Immediate release formulation of inhaled beclomethasone dipropionate-hydroxypropyl-beta-cyclodextrin composite particles produced using supercritical assisted atomization. Polymers. 2022;14(10):2114. doi: 10.3390/polym14102114
CrossRef - Avila EL, Rodríguez MC, Velásquez HJC. Influence of Maltodextrin and Spray Drying Process Conditions on Sugarcane Juice Powder Quality. Rev Fac Nac Agr Med. 2015;68(1):7509-7520. doi:10.15446/rfnam.v68n1.47839
CrossRef - Pudziuvelyte L, Marksa M, Jakstas V, et al. Microencapsulation of Elsholtzia ciliata herb ethanolic extract by spray-drying: impact of resistant- maltodextrin complemented with sodium caseinate, skim milk, and beta-cyclodextrin on the quality of spray-dried powders. Molec. 2019;24(8):1461. doi:10.3390/molecules24081461
CrossRef - Lourenço SC, Moldão-Martins M, Alves VD. Microencapsulation of pineapple peel extract by spray drying using maltodextrin, inulin, and arabic gum as wall matrices. Foods. 2020;9(6):1-17. doi:10.3390/foods9060718
CrossRef - Nurhidajah, Yonata D, Pranata B. Microencapsulation of anthocyanin-rich extract from Indonesian black rice using maltodextrin, arabic gum and skimmed milk powder as wall material by spray drying. Trends in Scie. 2024;21(8):7971. doi:10.48048/tis.2024.7971
CrossRef - Wu L, Zhang M, Liu Y, et al. Characteristics and release of monosodium glutamate microcapsules obtained by spray drying. Dry Tech. 2019;37(11):1340-1351. doi:10.1080/07373937.2018.1500483
CrossRef













