Effectiveness of Selenium Supplement Combined with Exercise on Ankle Brachial Index and Bone Mineral Density
1Faculty of Allied Health Sciences, Department of Nutrition and Dietetics, Burapha University, Chonburi, Thailand.
2Trace Element Speciation Laboratory (TESLA), Institute of Chemistry, University of Graz, Graz, Austria.
3Department of Chemical Metrology and Biometry, National Institute of Metrology, Pathum Thani, Thailand.
Corresponding Author Email: alongkote@go.buu.ac.th
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.3.33
ABSTRACT:Selenium (Se) is an essential trace element involved in fatty acid metabolism and bone formation. Arterial disease resulting from excessive fat accumulation and low bone mineral density (BMD) are prevalent health concerns in the general population. This study aimed to investigate the effectiveness of Se supplementation combined with exercise on the Ankle-Brachial Index (ABI) and BMD. Sixty participants were randomly assigned to either a control group (n = 30) or an intervention group (n = 30). Both groups followed a structured exercise program for 12 weeks. In addition to the exercise regimen, participants in the intervention group received Se supplementation in the form of selenomethionine (200 µg per capsule daily) for the same duration, while the control group received no supplementation. Data on ABI, BMD, and body composition were collected from all participants at baseline and at the end of the study. At the study endpoint, participants in the intervention group showed significantly higher right and left ABI scores compared to the control group (p < 0.05). Similarly, BMD measurements at the wrist and ankle were significantly higher in the intervention group than in the control group (p < 0.05). Furthermore, significant positive correlations were observed between Se intake and both ABI (left ABI: r = 0.30) and BMD improvements (wrist: r = 0.47; ankle: r = 0.31; p < 0.05). In conclusion, the findings suggest that Se supplementation combined with exercise is more effective in improving ABI and BMD than exercise alone.
KEYWORDS:Ankle Brachial Index; Body Composition; Bone Mineral Density; Exercise; Selenium
Introduction
Selenium (Se) is a vital trace element essential for the physiological functioning of the human body. It plays important roles in various biological systems, including the thyroid gland, reproductive system, and immune system, among others.1 A common method for assessing Se status is through blood Se concentration, which should not fall below 120 µg/L.2 Previous studies have reported the prevalence of Se deficiency across populations in various countries. For instance, a study conducted in Spain among children found that 13.93% had blood Se levels below 60 µg/L.3 Similarly, research in Saudi Arabia revealed that 56% of the adult population had Se levels below 56 µg/L.4 In Thailand, data on Se intake among the general population are limited. However, one study on HIV-infected children in Thailand reported that 56% of participants were so deficient.5 In addition, previous research has highlighted the benefits of Se in relation to vascular health. Se, particularly in the form of selenoproteins, has been shown to stimulate intracellular fat metabolism, potentially aiding in the regulation of blood lipid levels and supporting normal blood circulation.6 For example, a longitudinal study conducted in China among adults monitored blood Se levels over time and found that individuals with normal Se levels exhibited lower triglyceride and cholesterol levels compared to those with Se deficiency, who were more likely to experience lipid abnormalities.7 These findings suggest that adequate Se intake plays a key role in lipid regulation and contributes to the maintenance of healthy vascular function.
Peripheral Arterial Disease (PAD) is caused by the accumulation of fatty deposits on the inner walls of arteries, leading to arterial hardening and the narrowing or blockage of peripheral arteries in the arms and legs. The severity of PAD varies depending on the extent of arterial obstruction. Common symptoms include leg and calf pain, particularly during walking, which may progress to chronic wounds on the feet or toes due to reduced blood flow resulting from narrowed arteries.8 The etiology of PAD involves several risk factors, including diabetes, kidney disease, and elevated blood lipid levels, all of which contribute to the deposition and accumulation of lipids on the inner vascular walls.9 PAD significantly impairs patients’ quality of life, with global estimates indicating that approximately 236 million individuals are affected worldwide.10 In Thailand, one study reported a PAD prevalence rate of 7.9% in non-urban areas of the lower northern region.11 Screening for PAD can be conducted by assessing peripheral arterial circulation using the Ankle-Brachial Index (ABI), a non-invasive method that evaluates arterial patency in the arms and legs. This test is painless, safe, provides immediate results, and does not require fasting beforehand.12 According to diagnostic criteria, a normal ABI ranges from 0.9 to 1.4. An ABI value below 0.9 indicates peripheral arterial stenosis and is considered abnormal. This measurement technique has been widely accepted in previous research as a reliable and accurate indicator of PAD risk.13,14 Furthermore, previous studies have suggested that Se plays a role in stimulating fatty acid metabolism in the human body.15 Therefore, adequate Se intake may help reduce fat accumulation in the bloodstream. In addition to regulating lipid levels, regular physical exercise is also recognized as an effective strategy for improving peripheral arterial circulation.16
Low bone mineral density (BMD) and osteoporosis are significant global health concerns. These conditions are primarily associated with increased bone turnover, often resulting from physiological changes—particularly hormonal fluctuations—alongside insufficient physical activity and inadequate intake of nutrients essential for bone health, especially calcium, during early life.17,18 The consequences of low BMD substantially affect individuals’ quality of life, leading to pain, increased risk of fractures, and diminished capacity for self-care.19 Current data indicate that, in the United States, approximately 10 million individuals aged 50 and older suffer from osteoporosis.20 In Japan, the number of osteoporosis cases is estimated at 15 million.21 In Thailand, the prevalence of bone fragility and osteoporosis is increasing, largely due to the country’s transition to an aging society. For example, a previous study reported that 21.3% of early postmenopausal women attending a menopausal clinic at a tertiary care hospital in Thailand were diagnosed with osteoporosis.22 In addition to inadequate intake of bone-supporting nutrients such as calcium and vitamin D, and lack of exercise, Se has been identified in previous studies as a mineral associated with BMD. Se contributes to antioxidant defense and inhibits the activity of inflammatory mediators, including cytokines released during systemic inflammation.23 These inflammatory substances have been linked to accelerated bone turnover.24 Se is believed to mitigate internal inflammation by suppressing the release of these cytokines, thereby potentially slowing bone loss.25 Thus, prior research suggests that adequate Se intake may be associated with reduced bone resorption and improved bone health.26 Given the relevance of both peripheral arterial circulation and BMD in public health, and considering the possible beneficial role of Se, this study aims to investigate the effectiveness of Se supplementation combined with exercise in improving ABI and BMD values. Despite emerging evidence, studies specifically exploring the combined effects of Se and exercise on these outcomes remain limited.
Materials and Methods
Study tools
Basic information questionnaire for participants
A closed-ended questionnaire was designed and developed to collect basic demographic information from participants, such as gender, age, body mass index (BMI), education level, and so on. The participants completed this questionnaire by themselves.
Three-days food record interview form
A closed-ended questionnaire was developed for participants to record their food intake over three days following their participation in the study and after receiving education on food portion calculations. Participants recorded their food intake for two weekdays and one weekend day during the final week of the study.27 The recorded data were then submitted for analysis to calculate energy intake and nutrient consumption, providing information on total energy intake, energy distribution, and daily Se intake. This nutritional data, particularly Se levels, was used to support the interpretation of ABI measurements in the final week of the study. The dietary energy and Se intake data from this questionnaire were analyzed using the INMUCAL-Nutrient Version 4.0 food analysis program, developed by the Institute of Nutrition, Mahidol University, Thailand, in conjunction with the United States Department of Agriculture (USDA) database.
Se Dietary Supplement
A dietary supplement containing Se in capsule form was obtained from general pharmacies. The Se in this product is in the organic form of selenomethionine (SeMet), which has the highest absorption and utilization rate in the human body compared to other forms of Se.28 Each capsule contains 200 μg of Se as SeMet. Participants in the intervention group took one capsule daily after lunch. This dosage of Se has been shown to be effective in various clinical contexts, as demonstrated by multiple previous studies.29,30
ABI testing device
This device (HBP-8000, OMRON, Kyoto, Japan) is used to measure peripheral arterial function. It undergoes regular calibration and maintenance to ensure accurate and reliable results. The device measures the ABI of peripheral arteries in various areas, including both the left and right arms and legs, and prints the results along with their interpretation. A normal ABI value ranges from 0.9 to 1.4, while a value below 0.9 indicates a risk of arterial disease. Additionally, the device measures the Arterial Stiffness Index (ASI), which reflects the pressure range within the arterial volume. An optimal ASI value is less than 70; values between 70 and 180 indicate a moderate risk of arterial stiffness, while values above 180 indicate a high risk.31
Bone mineral density measurement device
A DEXA-based bone density measurement device (Osteo Checker, Ampall Co. Ltd., Seoul, South Korea) was used to measure bone density at the wrist and ankle. The device reports results as T-scores and bone mineral density (BMD) values. The instrument was calibrated before each use.
Body composition analysis devices
All participants underwent body composition analysis using the BOD POD device (COSMED Inc., Rome, Italy), which was calibrated before each use, and the InBody270 device (InBody Co., Seoul, South Korea) to measure parameters such as body fat percentage, muscle mass, and body fluid percentage, both before and after the study.
The study instruments, including questionnaires, were validated for accuracy and appropriateness by three qualified experts in nutrition and dietetics. After incorporating their suggested revisions, the questionnaires and detailed study procedures—conducted in accordance with the Declaration of Helsinki—were submitted for ethical approval to the Human Research Ethics Committee of Burapha University, Thailand (approval number IRB1-099/2567).
Participants
After enrollment, a total of 60 healthy participants residing in Saensuk Sub-District Municipality, Mueang District, Chonburi Province, were recruited for this study. The sample size was determined based on a power analysis using G*Power software (version 3.1), which indicated that a minimum of 52 participants was required to detect a medium effect size (d = 0.5) with 80% power at a significance level of 0.05. To account for potential dropouts and incomplete data, the target sample size was increased by approximately 15%, resulting in the recruitment of 60 participants. The inclusion criteria were: Thai ethnicity and nationality, good health with no chronic diseases, legal adults aged 18 to 60 years, and the ability to read and write Thai. The exclusion criteria were: a history of allergies to food or dietary supplements; muscle or bone injuries that impede exercise; serious communicable diseases; oral problems affecting chewing or swallowing; current use of other dietary supplements or herbal products; pregnancy or breastfeeding; intellectual disabilities affecting communication; withdrawal from the study before completion; and incomplete questionnaire responses or data. All participants signed informed consent forms prior to participation.
Study procedures
At the first appointment, all 60 participants were invited to a designated room at the Nutrition and Dietetics Division, Faculty of Allied Health Sciences, Burapha University, where they were provided with study details and each participant signed the informed consent form. Subsequently, participants were divided into two equal groups using quota sampling: an intervention group (n=30) and a control group (n=30). After group allocation, baseline data were collected using the aforementioned instruments. These measurements included weight and height, peripheral arterial function assessed by an ABI device (measuring ASI and ABI), body composition parameters (% body fat, % fat-free mass, muscle mass, % body fluid, thoracic gas volume) measured using BOD POD and InBody270, and wrist and ankle bone density (T-score and BMD) assessed by the Osteo Checker. Participants then completed a basic information questionnaire and received education on food exchange lists using food models. They were provided with a 3-day food record questionnaire to document their intake over two weekdays and one weekend day during the final week of the study.
Both groups were assigned to exercise according to WHO guidelines,32 which included 30–45 minutes of aerobic exercise 4–5 days per week and strength training 1–2 days per week for 12 weeks. The control group was asked to exercise only, while the intervention group additionally received Se supplements (200 μg SeMet per capsule) to be taken once daily after any meal for 12 weeks.33 In the final week (week 12), participants submitted their 3-day food records for calculation of energy intake, energy distribution, and Se intake. Final measurements were taken, including peripheral arterial function (ABI), body composition (BOD POD and InBody), and bone mineral density (Osteo Checker), for subsequent statistical analysis.
Statistical analyses
Participants’ data on sex and education were reported as percentages, and differences were assessed using Fisher’s Exact Test. Duration of weekly exercise, daily sun exposure, and other ABI and BMD scores were reported as mean ± standard deviation (SD). Skewness and kurtosis were used to assess data normality, and the Mann-Whitney U test was applied to determine significant differences between groups. Pearson correlation coefficients were used to examine the relationships between Se intake and ABI and BMD scores at the study endpoint. Statistical analyses were performed using SPSS software (version 26.0). A p-value of less than 0.05 was considered statistically significant.
Results
At the end of the final week of the study (week 12), all participants completed the study with no dropouts. The background data collected during the first week indicated no significant differences between the two groups in terms of general characteristics. The average age of participants in the control group was 26.36 years, while that of the intervention group was 25.20 years. The majority were female in both the control group (70%) and the intervention group (86.67%). Twenty-six participants (86.67%) in the control group and 28 participants (93.33%) in the intervention group held a bachelor’s degree. The average weekly exercise duration was 60.66 minutes in the control group and 68.50 minutes in the intervention group. Daily sun exposure averaged 22.50 minutes in the control group and 30.16 minutes in the intervention group (Table 1).
Table 1: Background characteristics of participants
|
Characteristics |
Control group
(n=30) |
Intervention group
(n=30) |
p value |
| Age (year) 1, mean (SD) | 26.36 (5.15) | 25.20 (5.39) |
0.15 |
|
Sex2 Male, n (%) Female, n (%) |
9 (30.00) 21 (70.00) |
4 (13.33) 26 (86.67) |
0.20 |
| Education2
Bachelor degree, n (%) Graduate degree, n (%) |
26 (86.67) 4 (13.33) |
28 (93.33) 2 (6.67) |
0.67 |
|
Length of weekly exercise (minute) 1, mean (SD) |
60.66 (56.19) | 68.50 (79.71) | 0.58 |
| Length of daily sun exposure (minute) 1, mean (SD) | 22.50 (19.06) | 30.16 (30.89) |
0.33 |
At the end of the study, the right and left ABI values at week 12 (final week) for participants in the intervention group were 1.33 and 1.30, respectively, which were significantly higher than those in the control group (1.19 and 1.21, respectively; p < 0.05). For the ASI, the right and left ASI values of participants in the intervention group were 70.53 and 68.30, respectively, which were significantly lower than the corresponding values in the control group (80.50 and 80.03; p < 0.05). Regarding BMD, the wrist and ankle BMD values at the endpoint for participants in the intervention group were 0.55 and 0.55 g/cm², respectively, significantly higher than those in the control group (0.46 and 0.50 g/cm², respectively; p < 0.05). Moreover, the body mineral weight of participants in the intervention group was 2.88 kg, significantly greater than that of the control group (2.44 kg; p < 0.05). Additionally, when examining changes within each group at the endpoint, several parameters showed significant improvement compared to baseline in both groups, including right and left ABI, right ankle ASI, ankle BMD values, and thoracic gas volume (p < 0.05) (Table 2).
Table 2: ABI scores, BMD scores, and body composition of participants
|
Parameters |
Baseline | p value | Endpoint | p value | ||
| Control
(n=30) |
Intervention
(n=30) |
Control
(n=30) |
Intervention (n=30) |
|||
|
Right ABI, mean (SD) |
1.08 (0.14) | 1.04 (0.12) | 0.24 | 1.19 (0.13)^ | 1.33 (0.08)^ | <0.05* |
| Left ABI, mean (SD) | 1.09 (0.15) | 1.02 (0.13) | <0.05* | 1.21 (0.13)^ | 1.30 (0.10)^ |
<0.05* |
|
Right brachial ASI, mean (SD) |
34.60 (8.88) | 36.43 (10.83) | 0.67 | 32.13 (6.04) | 31.46 (8.03)^ | 0.18 |
| Left brachial ASI, mean (SD) | 36.76 (7.59) | 38.13 (8.89) | 0. 29 | 34.23 (8.95) | 32.56 (11.79)^ |
0.12 |
|
Right ankle ASI, mean (SD) |
89.40 (26.01) | 85.90 (21.78) | 0.66 | 80.50 (21.51)^ | 70.53 (21.51)^ | <0.05* |
| Left ankle ASI, mean (SD) | 83.83 (18.11) | 82.83 (16.30) | 0.98 | 80.03 (14.96) | 68.30 (14.42)^ |
<0.05* |
|
T-score of wrist bone, mean (SD) |
-1.18 (0.80) | -1.24 (0.67) | 0.85 | -1.42 (0.66)^ | -1.20 (0.69) | 0.19 |
| BMD value of wrist (g/cm2), mean (SD) | 0.47 (0.07) | 0.51 (0.09) | 0.06 | 0.46 (0.06) | 0.55 (0.09)^ |
<0.05* |
|
T-score of ankle bone, mean (SD) |
-1.36 (0.82) | -1.25 (0.77) | 0.59 | -1.42 (0.84) | -1.23 (0.84) | 0.22 |
| BMD value of ankle (g/cm2), mean (SD) | 0.47 (0.04) | 0.50 (0.06) | 0.08 | 0.50 (0.05)^ | 0.55 (0.07)^ |
<0.05* |
|
BMI, mean (SD) |
21.86 (2.93) | 22.10 (2.63) | 0.28 | 22.33 (2.59) | 22.40 (2.42) | 0.74 |
| Body fluid (L), mean (SD) | 28.95 (3.00) | 29.59 (2.67) | 0.34 | 28.77 (3.04) | 29.66 (2.46) |
0.29 |
|
Muscle mass (kg), mean (SD) |
19.55 (2.32) | 19.83 (2.04) | 0.59 | 19.57 (2.06) | 20.16 (1.87) | 0.41 |
| Body fat mass (kg), mean (SD) | 20.45 (3.35) | 20.55 (2.49) | 0.66 | 21.04 (3.34) | 20.07 (2.12) |
0.37 |
|
Body weight by mineral (kg), mean (SD) |
2.52 (0.18) | 2.55 (0.24) | 0.92 | 2.44 (0.22) | 2.88 (0.16)^ | <0.05* |
| Thoracic gas volume (L), mean (SD) | 2.93 (0.32) | 2.94 (0.31) | 0.80 | 3.12 (0.31)^ | 3.11 (0.23)^ |
0.72 |
|
Estimated daily total kcal requirement (kcal), mean (SD) |
1,810.66 (143.00) | 1,832.36 (156.44) | 0.34 | 2,282.36^ (156.44) | 2,313.50^ (167.74) |
0.63 |
* Significant difference between groups using Mann-Whitney U test.
^ Significant difference within group when compared with baseline using Mann-Whitney U test.
The 3-day food records completed by the participants were used to assess their dietary habits during the study. Results showed that participants in the intervention group derived 59.83% of their total calories from carbohydrates, which was significantly higher than the control group (55.66%; p < 0.05). The intervention group obtained 27.23% of their total calories from fat, significantly lower than the control group (31.26%; p < 0.05). Additionally, with Se supplementation, the average daily Se intake of participants in the intervention group was 268.80 µg, significantly higher than that of the control group (63.50 µg; p < 0.05) (Table 3).
Table 3: Dietary habits of participants
|
Nutrients |
Control group
(n=30) |
Intervention group
(n=30) |
p value |
| Total kcal consumed (kcal), mean (SD) | 2,234.53 (248.42) | 2,269.33 (189.84) |
0.48 |
|
%kcal from carbohydrate, mean (SD) |
55.66 (6.12) | 59.83 (7.34) | <0.05* |
| %kcal from protein, mean (SD) | 13.06 (5.16) | 12.93 (5.57) |
0.69 |
|
%kcal from fat, mean (SD) |
31.26 (3.90) | 27.23 (4.42) | <0.05* |
| Amount of Se intake (µg), mean (SD) | 63.50 (10.97) | 268.80 (13.97) |
<0.05* |
* Significant difference between groups using Mann-Whitney U test.
When examining the correlations between Se intake and arterial health outcomes among all 60 participants with similar exercise habits, results showed a significant positive correlation between Se intake and left ABI (r = 0.30; p < 0.05), as shown in Figure 1B, and a significant negative correlation between Se intake and left ASI (r = -0.35; p < 0.05), as shown in Figure 1F.
 |
Figure 1: Correlation of the amount of Se intake and ankle brachial indexes (A) Right ABI and amount of Se intake; (B) Left ABI and amount of Se intake; (C) Right brachial ASI and amount of Se intake; |
For BMD, the results indicated significant positive correlations between Se intake and wrist BMD (r = 0.47; p<0.05) and ankle BMD (r = 0.31; p<0.05) among all 60 participants with similar exercise habits (Fig. 2B and 2D, respectively).
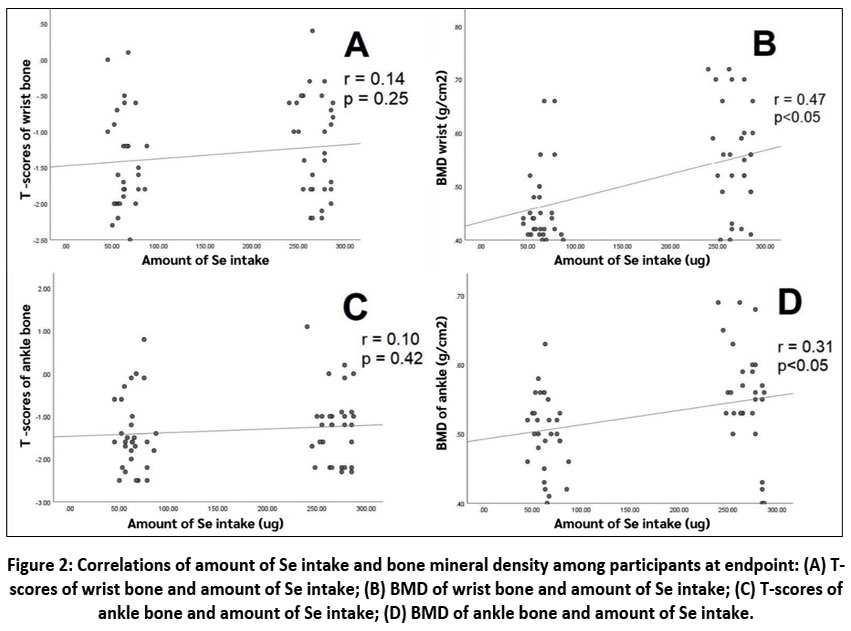 |
Figure 2: Correlations of amount of Se intake and bone mineral density among participants at endpoint: (A) T-scores of wrist bone and amount of Se intake; (B) BMD of wrist bone and amount of Se intake; |
Discussion
In this study, participants were assigned to follow the same exercise regimen according to WHO guidelines throughout the study to minimize confounding factors arising from differing physical activity levels. This was reflected in the lack of significant difference in total energy expenditure between groups. Se supplementation combined with exercise in the intervention group was observed to be more effective in improving arterial function than exercise alone. Evidence suggests that Se plays a crucial role in metabolic processes within several tissues, including the liver, white adipocytes, and muscles. It is particularly important in the thermogenesis of adipocytes, encompassing both brown and white adipose tissue, making Se a potential dietary intervention for treating obesity and metabolic disorders. Specifically, in the thermogenesis of brown adipose tissue (BAT), the Se-containing enzyme type 2 iodothyronine deiodinase (DIO2) is critical for initiating adaptive thermogenesis.34,35 Se may influence thermogenesis in BAT through multiple mechanisms. One key pathway involves selenoproteins, particularly DIO2, which has long been recognized as essential for thermogenic adaptation. Additionally, Se intake can enhance the activity of another selenoprotein, glutathione peroxidase 1 (GPX1), leading to reduced glutathione levels and increased reactive oxygen species (ROS) production, ultimately triggering thermogenesis.36,37 For these reasons, the improved arterial function observed in the intervention group receiving Se supplementation may be attributable to enhanced blood lipid metabolism. However, participants in the control group also showed improved ABI values at the endpoint compared to baseline, supporting previous studies that reported the benefits of exercise on vascular and cardiac function.38,39 Previous studies have demonstrated the anti-inflammatory benefits of Se and exercise, particularly through the suppression of pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), and interleukin-6 (IL-6), which are known to adversely affect arterial wall tissues.40,41 These mechanisms may underlie the findings of the present study, wherein participants in the intervention group—who received both Se supplementation and engaged in exercise—exhibited superior arterial function compared to the control group, who participated in exercise alone. Regarding bone mineral density (BMD), although the control group showed some improvement in bone health at the study endpoint compared to their baseline values, the intervention group demonstrated a greater enhancement. This improvement in the control group may be attributed to the role of exercise in promoting bone formation through osteoblast stimulation.42 However, the more pronounced improvement in the intervention group may be explained by prior research suggesting that Se can suppress inflammatory cytokines such as TNF-α and IL-6, both of which are implicated in bone resorption.43,44 Therefore, the administration of Se in the intervention group may have contributed to a downregulation of these cytokines, thereby reducing bone resorption and promoting osteoblast activity.45,46 This dual action of Se and exercise likely accounts for the more significant improvement in BMD observed in the intervention group compared to the control group, who received exercise only.
At the endpoint, participants showed no significant changes in body composition, suggesting that Se may not play a role in muscle building when combined with exercise. Previous studies have demonstrated that combining adequate intake of high-biological-value protein with weight training is most effective in inducing skeletal muscle hypertrophy.47 While the control group consumed a higher percentage of calories from fat than the intervention group, and the intervention group consumed a higher percentage of calories from carbohydrates than the control group, their regular exercise routine was likely the primary factor in maintaining their body fat mass.48 Results from the final week showed that both groups increased their estimated energy expenditure compared to baseline due to regular exercise, but their BMI remained unchanged. The balance of calories consumed may be key to maintaining body weight.49
Interestingly, participants in the control group who did not receive Se supplementation had a daily Se intake of 63.50 µg, which slightly exceeds the recommended daily allowance of 55 µg/day for adults.50 This may be attributed to the study location—an urban seaside area where participants have easy access to a variety of foods. Moreover, Se-rich foods such as fish and other seafood are readily available at reasonable prices in this coastal setting.51-53 Additionally, fish commonly consumed in Thailand from both freshwater and marine sources exhibit high Se bioavailability and lower levels of toxic heavy metal contamination.54,55 Limitations of this study include the lack of biochemical marker data related to Se status, such as GPX1, selenoprotein P, and superoxide dismutase, as well as missing information on blood lipid profiles (which are involved in arterial function), serum vitamin D, and parathyroid hormone (which are involved in bone health). Future studies are recommended to investigate these biochemical markers and different types of exercise to explore the best practical approaches for improving ABI and BMD values.
Conclusion
The findings of this study showed that Se supplementation at 200 µg per day for 12 weeks combined with exercise tends to be effective in improving arterial function (as measured by ABI) and promoting BMD (as measured by DEXA) compared with exercise alone. The supplemented amount exceeds the recommended daily intake of 55 µg for adults but remains well below the established upper intake limit of 400 µg. These results support the importance of regularly adequate Se intake (at least 12 weeks) at an optimal level to enhance arterial function and BMD. Foods rich in Se, such as seafood, marine fish, and certain vegetables, should be promoted in diverse portions to help individuals achieve sufficient Se intake. Importantly, regular exercise, in combination with adequate Se intake, is a key factor in improving these health parameters.
Acknowledgement
The author would like to thank Thai Dietetics Association for granting the research work. The author is also profoundly grateful to the Faculty of Allied Health Sciences, Burapha University for their facilities support to conduct the study.
Funding Sources
This study was received funding by the Thai Dietetics Association (TDA Junior Research Award 2024).
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This study obtained ethical approval by the Human Research Ethics Committee of Burapha University, Thailand (approval number IRB1-099/2567).
Informed Consent Statement
All participants were signed the informed consent before participated in this study.
Clinical Trial Registration
This trial is registered at Thai Clinical Trials Registry with the registration number TCTR20250214001
Permission to Reproduce Material from Other Sources
Not applicable
Author Contributions
- Arluk Udomwongyon – Conceptualized and collected the data
- Busakorn Yindee – Conceptualized and collected the data
- Chanidapa Yathuam – Conceptualized and collected the data
- Chonnikarn Limpaninchart – Conceptualized and collected the data
- Juthathip Nakieam – Conceptualized and collected the data
- Oradee Saikiseng – Conceptualized and collected the data
- Surangkhana Phalaket – Conceptualized and collected the data
- Thanaphorn Pumlamjeak – Conceptualized and collected the data
- Jörg Feldmann – Gave advice about the study protocol and proofread the manuscript draft
- Nunnapus Laitip – Gave advice about the study protocol and proofread the manuscript draft
- Alongkote Singhato – Conceptualization of methodology, statistical analyses, and manuscript writing
Reference
- Barchielli G, Capperucci A, Tanini D. The Role of Selenium in Pathologies: An Updated Review. Antioxidants (Basel). 2022;11(2):251. doi: 10.3390/antiox11020251.
CrossRef - Bizerea-Moga TO, Pitulice L, Bizerea-Spiridon O, et al. Evaluation of Serum Selenium Status by Age and Gender: A Retrospective Observational Cohort Study in Western Romania. Nutrients. 2021;13(5):1497. doi: 10.3390/nu13051497.
CrossRef - Navia B, Ortega RM, Perea JM, et al. Selenium status in a group of schoolchildren from the region of Madrid, Spain. J Hum Nutr Diet. 2014;27(2):239-46. doi: 10.1111/jhn.12126.
CrossRef - Al-Saleh I, Billedo G. Determination of selenium concentration in serum and toenail as an indicator of selenium status. Bull Environ Contam Toxicol. 2006;77(2):155-63. doi: 10.1007/s00128-006-1045-4.
CrossRef - Bunupuradah T, Pinyakorn S, Puthanakit T, on behalf of the Psg. Prevalence of selenium deficiency in Thai HIV-infected children without severe HIV symptoms. Eur J Clin Nutr. 2012; 66(11):1278-78. doi: https://doi.org/10.1038/ejcn.2012.116
CrossRef - Brown KM, Arthur JR. Selenium, selenoproteins and human health: a review. Public Health Nutr. 2001;4(2b):593-9. doi: 10.1079/phn2001143.
CrossRef - Chen C, Jin Y, Unverzagt FW, et al. The association between selenium and lipid levels: a longitudinal study in rural elderly Chinese. Arch Gerontol Geriatr. 2015;60(1):147-52. doi: 10.1016/j.archger.2014.09.005.
CrossRef - Olin JW, Sealove BA. Peripheral artery disease: current insight into the disease and its diagnosis and management. Mayo Clin Proc. 2010;85(7):678-92. doi: Peripheral artery disease: current insight into the disease and its diagnosis and management
CrossRef - Aday AW, Everett BM. Dyslipidemia Profiles in Patients with Peripheral Artery Disease. Curr Cardiol Rep. 2019;21(6):42. doi: 10.1007/s11886-019-1129-5.
CrossRef - Horváth L, Németh N, Fehér G, et al. Epidemiology of Peripheral Artery Disease: Narrative Review. Life (Basel). 2022;12(7). doi: 10.3390/life12071041.
CrossRef - Suwannasrisuk P, Sattanon S, Taburee W, et al. Prevalence and predictors of peripheral arterial disease determined by ankle brachial index in diabetes population treated within primary care services in a non-urban area of lower northern Thailand. Diab Vasc Dis Res. 2020;17(6):1479164120966997. doi: 10.1177/1479164120966997.
CrossRef - Casey S, Lanting S, Oldmeadow C, et al. The reliability of the ankle brachial index: a systematic review. J Foot Ankle Res. 2019;12:39. doi: 10.1186/s13047-019-0350-1.
CrossRef - Rac-Albu M, Iliuta L, Guberna SM, et al. The role of ankle-brachial index for predicting peripheral arterial disease. Maedica (Bucur). 2014;9(3):295-302.
- Cáceres-Farfán L, Moreno-Loaiza M, Cubas WS. Ankle-brachial index: more than a diagnostic test? Arch Peru Cardiol Cir Cardiovasc. 2021;2(4):254-62. doi: 10.47487/apcyccv.v2i4.168
CrossRef - Tinkov AA, Ajsuvakova OP, Filippini T, et al. Selenium and Selenoproteins in Adipose Tissue Physiology and Obesity. Biomolecules. 2020;10(4):658. doi: 10.3390/biom10040658.
CrossRef - Aday AW, Kinlay S, Gerhard-Herman MD. Comparison of different exercise ankle pressure indices in the diagnosis of peripheral artery disease. Vasc Med. 2018;23(6):541-48. doi: 10.1177/1358863X18781723.
CrossRef - Akkawi I, Zmerly H. Osteoporosis: Current Concepts. Joints. 2018;6(2):122-27. doi: 10.1055/s-0038-1660790.
CrossRef - Pouresmaeili F, Kamalidehghan B, Kamarehei M, et al. A comprehensive overview on osteoporosis and its risk factors. Ther Clin Risk Manag. 2018;14:2029-49. doi: 10.2147/TCRM.S138000
CrossRef - Gold T, Williams SA, Weiss RJ, et al. Impact of fractures on quality of life in patients with osteoporosis: a US cross-sectional survey. J Drug Assess. 2019;8(1):175-83. doi: 10.1080/21556660.2019.1677674.
CrossRef - Clynes MA, Harvey NC, Curtis EM, et al. The epidemiology of osteoporosis. Br Med Bull. 2020;133(1):105-17. doi: 10.1093/bmb/ldaa005
CrossRef - Iki M. [Epidemiology of osteoporosis in Japan]. Clin Calcium. 2012;22(6):797-803.
- Charoenngam N, Pongchaiyakul C. Current issues in evaluation and management of osteoporosis in Thailand. Osteoporos Sarcopenia. 2023;9(2):53-59. doi: 10.1016/j.afos.2023.05.002.
CrossRef - Xie H, Wang N, He H, et al. The association between selenium and bone health: a meta-analysis. Bone Joint Res. 2023;12(7):423-32. doi: 10.1302/2046-3758.127.BJR-2022-0420.R1.
CrossRef - Terkawi MA, Matsumae G, Shimizu T, et al. Interplay between Inflammation and Pathological Bone Resorption: Insights into Recent Mechanisms and Pathways in Related Diseases for Future Perspectives. Int J Mol Sci. 2022;23(3). doi: 10.3390/ijms23031786.
CrossRef - Huang Z, Rose AH, Hoffmann PR. The role of selenium in inflammation and immunity: from molecular mechanisms to therapeutic opportunities. Antioxid Redox Signal. 2012;16(7):705-43. doi: 10.1089/ars.2011.4145.
CrossRef - Yang T, Lee SY, Park KC, et al. The Effects of Selenium on Bone Health: From Element to Therapeutics. Molecules. 2022;27(2). doi: 10.3390/molecules27020392.
CrossRef - Singhato A, Booranasuksakul U, Khongkhon S, et al. Effectiveness of the Therapeutic Lifestyle Change Diet Intervention to Improve Biochemical Markers of Cardiovascular Diseases in HIV-Infected Patients with Dyslipidemia. Ann Nutr Metab. 2024;80(4):202-10. doi: 10.1159/000538578.
CrossRef - Wang N, Tan HY, Li S, et al. Supplementation of Micronutrient Selenium in Metabolic Diseases: Its Role as an Antioxidant. Oxid Med Cell Longev. 2017;2017:7478523. doi: 10.1155/2017/7478523.
CrossRef - Abdulah R, Koyama H, Miyazaki K, et al. Selenium supplementation and blood rheological improvement in Japanese adults. Biol Trace Elem Res. 2006;112(1):87-96. doi: 10.1385/BTER:112:1:87.
CrossRef - Combs GF, Jr., Midthune DN, Patterson KY, et al. Effects of selenomethionine supplementation on selenium status and thyroid hormone concentrations in healthy adults. Am J Clin Nutr. 2009;89(6):1808-14. doi: 10.3945/ajcn.2008.27356.
CrossRef - Woo JS. Ankle brachial index: a simple path to the future. Korean J Intern Med. 2023;38(3):277-79. doi: 10.3904/kjim.2023.141
CrossRef - Bull FC, Al-Ansari SS, Biddle S, et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br J Sports Med. 2020;54(24):1451-62. doi: 10.1136/bjsports-2020-102955.
CrossRef - Bahmani F, Kia M, Soleimani A, et al. Effect of Selenium Supplementation on Glycemic Control and Lipid Profiles in Patients with Diabetic Nephropathy. Biol Trace Elem Res. 2016;172(2):282-89. doi: 10.1007/s12011-015-0600-4.
CrossRef - Marsili A, Aguayo-Mazzucato C, Chen T, et al. Mice with a targeted deletion of the type 2 deiodinase are insulin resistant and susceptible to diet induced obesity. PLoS One. 2011;6(6):e20832. doi: 10.1371/journal.pone.0020832.
CrossRef - Shimada BK, Watanabe LM, Swanson S, et al. Selenium and selenoproteins in thermogenic adipocytes. Arch Biochem Biophys. 2022;731:109445. doi: 10.1016/j.abb.2022.109445.
CrossRef - Lettieri Barbato D, Tatulli G, Maria Cannata S, et al. Glutathione Decrement Drives Thermogenic Program In Adipose Cells. Sci Rep. 2015;5:13091. doi: 10.1038/srep13091.
CrossRef - Song Y, Zhu M, Islam MA, et al. Glutathione peroxidase 3 is essential for countering senescence in adipose remodelling by maintaining mitochondrial homeostasis. Redox Biology. 2024;77:103365. doi: 10.1016/j.redox.2024.103365.
CrossRef - Kobayashi R, Asaki K, Hashiguchi T, et al. Effect of aerobic exercise training frequency on arterial stiffness in middle-aged and elderly females. J Phys Ther Sci. 2022;34(5):347-52. doi: 10.1589/jpts.34.347.
CrossRef - Kresnajati S, Lin YY, Mündel T, et al. Changes in Arterial Stiffness in Response to Various Types of Exercise Modalities: A Narrative Review on Physiological and Endothelial Senescence Perspectives. Cells. 2022;11(22). doi: 10.3390/cells11223544.
CrossRef - hang F, Li X, Wei Y. Selenium and Selenoproteins in Health. Biomolecules. 2023;13(5):799. doi: 10.3390/biom13050799.
CrossRef - Luo B, Xiang D, Ji X, et al. The anti-inflammatory effects of exercise on autoimmune diseases: A 20-year systematic review. J Sport Health Sci. 2024;13(3):353-367. doi: 10.1016/j.jshs.2024.02.002.
CrossRef - Chang X, Xu S, Zhang H. Regulation of bone health through physical exercise: Mechanisms and types. Front Endocrinol (Lausanne). 2022;13:1029475. doi: 10.3389/fendo.2022.1029475.
CrossRef - Ansar S. Effect of Selenium on the Levels of Cytokines and Trace Elements in Toxin-Mediated Oxidative Stress in Male Rats. Biol Trace Elem Res. 2016;169(1):129-33. doi: 10.1007/s12011-015-0403-7.
CrossRef - Xu J, Yu L, Liu F, et al. The effect of cytokines on osteoblasts and osteoclasts in bone remodeling in osteoporosis: a review. Front Immunol. 2023;14:1222129. doi: 10.3389/fimmu.2023.1222129.
CrossRef - Lee JG, Jang JY, Baik SM. Selenium as an Antioxidant: Roles and Clinical Applications in Critically Ill and Trauma Patients: A Narrative Review. Antioxidants (Basel). 2025;14(3):294. doi: 10.3390/antiox14030294.
CrossRef - Poleboina S, Sheth VG, Sharma N, et al. Selenium nanoparticles stimulate osteoblast differentiation via BMP-2/MAPKs/β-catenin pathway in diabetic osteoporosis. Nanomedicine (Lond). 2022;17(9):607-625. doi: 10.2217/nnm-2021-0401.
CrossRef - Deldicque L. Protein Intake and Exercise-Induced Skeletal Muscle Hypertrophy: An Update. Nutrients. 2020;12(7). doi: 10.3390/nu12072023.
CrossRef - Kolnes KJ, Petersen MH, Lien-Iversen T, et al. Effect of Exercise Training on Fat Loss-Energetic Perspectives and the Role of Improved Adipose Tissue Function and Body Fat Distribution. Front Physiol. 2021;12:737709. doi: 10.3389/fphys.2021.737709.
CrossRef - Hall KD, Farooqi IS, Friedman JM, et al. The energy balance model of obesity: beyond calories in, calories out. Am J Clin Nutr. 2022;115(5):1243-54. doi: 10.1093/ajcn/nqac031.
CrossRef - Institute of Medicine Panel on Dietary A, Related C. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington (DC): National Academies Press (US).
- Yamashita Y, Yamashita M, Iida H. Selenium content in seafood in Japan. Nutrients. 2013;5(2):388-95. doi: 0.3390/nu5020388
CrossRef - Singhato A, Judprasong K, Sridonpai P, et al. Effect of Different Cooking Methods on Selenium Content of Fish Commonly Consumed in Thailand. Foods. 2022;11(12):1808. doi: 10.3390/foods11121808.
CrossRef - Singhato A, Judprasong K, Sridonpai P, et al. In Vitro Bioaccessibility of Selenium from Commonly Consumed Fish in Thailand. Foods. 2022;11(21):3312. doi: 10.3390/foods11213312.
CrossRef - Rueangsri N, Judprasong K, Sridonpai P, et al. Influence of Various Cooking Methods on Selenium Concentrations in Commonly Consumed Seafood Species in Thailand. Foods. 2024; 14(15):2700. doi: https://doi.org/10.3390/foods14152700
CrossRef - Sirisangarunroj P, Monboonpitak N, Karnpanit W, et al. Toxic Heavy Metals and Their Risk Assessment of Exposure in Selected Freshwater and Marine Fish in Thailand. Foods. 2023;12(21):3967. doi: https://doi.org/10.3390/foods12213967
CrossRef













