Comprehensive in vivo Antidiabetic Investigations and antioxidant potential of Caralluma edulis whole Plant
1Department of Pharmacy, Abdul Wali Khan University Mardan, Mardan, Pakistan.
2Department of Chemistry, College of Science, Princess Nourah Bint Abdulrahman University, Riyadh, 11671, Kingdom of Saudi Arabia.
3Department of Pharmacy, University of Naples Federico II, Naples, ITALY.
4International Research Center for Food Nutrition and Safety, Jiangsu University, Zhenjiang, China.
5Department of Pharmacy, Korea University, Sejong South Korea.
Corresponding Author E-mail: haroonkhan@awkum.edu.pk
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.Special-Issue-July.11
ABSTRACT:Diabetes is considered the most prevalent health disorders globally, with its incidence continuing to rise. The available therapeutic are unable to provide adequate hyperglycemic control. Caralluma edulis has been traditionally consumed as medicine worldwide for managing a range of ailments including diabetes mellitus. The present investigation aimed to analyze the antioxidant and antidiabetic properties of crude hydro-alcoholic extract/ fractions derived from C. edulis. Antioxidant property was determined using ABTS and DPPH methods. In vivo experiments included acute toxicity studies, oral glucose tolerance tests, and streptozotocin-induced antidiabetic activity. The effects on body weight, serum insulin levels, glycosylated hemoglobin, and liver glycogen were also evaluated. Histological analysis of pancreas was also performed. The highest level of antioxidant effect was shown by the ethyl acetate fraction. Oral glucose tolerance test results showed that after 60 and 120 min of glucose loading, there was notable drop in blood glucose levels for the C. edulis extract. The in-vivo antidiabetic investigation showed that crude hydroalcoholic extract at 200 mg/kg had the strongest antidiabetic potential when compared with glibenclamide. The tested medicinal plants led to significant improvement in the body weight, serum insulin, hepatic glycogen, and glycated hemoglobin concentrations. Histopathological analysis showed that the extract considerably reduced the pathological damages, including edema, fatty alterations, vascular congestion, inflammatory infiltrates, and necrosis, in a dose-dependent way. According to our findings, the whole plant of C. edulis demonstrates strong antioxidant activity and holds promising therapeutic potential in diabetes management, as supported by in vitro, in vivo, and histological analysis.
KEYWORDS:Antioxidant potential; Biochemical analysis; Caralluma edulis; Histological analysis; In vitro antidiabetic
Introduction
DM, or Diabetes mellitus is an insulin resistance (metabolic) syndrome that critically compromised the life style of millions of peoples around the globe. According to current projections, diabetes is predicted to affect more than 380 million persons worldwide by 2025, representing a significant increase from 194 million cases reported in 2003. Diabetes will mostly impact China, India, and the United States by 2025. 1,2 By 2021, 537 million adults globally are predicted to have diabetes, and by 2045, that figure is likely to rise to 783 million, according to the most recent prediction. The treatment of DM is a great challenge for the diabetologists. The conventional treatments strategies principally emphasis on managing blood glucose levels and mitigating symptoms, however they often fail to address the underlying causes and multiple pathways involved in the disease’s progression. 3 In this regard, new effective strategies and therapeutic agents are extremely important to cope with the management and completions of the syndrome for which, natural products could be better alternative.
The edible and nutritionally significant species C. edulis, sometimes referred to as chunga, chung, ‘Settu, or pippu, is a member of the Apocynaceae. Adapted to arid and semi-arid environments, the succulent xerophytic herb C. edulis exhibits a broad geographic distribution across several regions, including Saudi Arabia, Pakistan, Eritrea, Afghanistan, Somalia, Sudan, the United Arab Emirates, and India. Its occurrence has also been reported in Iran and Mauritania, indicating a wide ecological range and adaptability to diverse dryland habitats. 4,5 It is a plant found in arid, sandy areas 1,300 meters above sea level. 6 The C. edulis plant is an upright herb with substrate branches that ranges in height from 15 to 45 cm. If there are leaves, they are 6–13 mm long, 4 grooved, and may be sharp or linear. Pairs of flowers are seen in the axil of scale-like bracts. Pedicels are 2.5 mm long, oval, glabrous, or acute, and have five sepals. They range in length from 3.5 to 13 mm. The corolla campanulate has ovate-lanceolate lobes and is purple inside. This features five lobed outer corona, with two teeth on each lobe and 1mm long inner corona lobe. There is one pollinium in per flower anther cell. Follicles measure between 8.5 and 11 cm in length. 7
Traditionally, this plant has an exceptional therapeutic history. Numerous medical conditions, including hypertension, Alzheimer’s disease, rheumatism, stomach issues, and leprosy, have been suggested to be treated with it. In the past, diabetes was treated by boiling the stem in water and using the resulting extract. 7 The occurrence of several types of phytochemicals in C. edulis was investigated and the finding indicated the existence of alkaloids, proteins, carbohydrates, glycosides, flavonoids, terpenoids, saponins , tannins and phenol. The presence of several glycosides in C. edulis has been linked to its therapeutic benefits. 7 Previous studies have demonstrated distinct antioxidant and pharmacological activites of the plant, indicating the therapeutic significance of its bioactive phytoconstituents, which contribute to its anti-inflammatory, antioxidant, and antidiabetic effects. 8-10 Our study deals with the detail in vivo antidiabetic potential of crude extract of C. edulis via testing oral glucose tolerance, blood glucose level, body weight, various biomarkers as well as histological analysis. The study also evaluated the effects of the whole plant extract/fractions against various free radicals.
Material And Methods
Chemicals
Distilled water, analytical grade solvents including Methanol (SA/Sigma Aldrich), Ethyl acetate (SA), n-Hexane (SA), DPPH (SA), ABTS (SA), Ascorbic acid (SA), Potassium Phosphate Dibasic (SA), Potassium persulphate (SA), Potassium Phosphate Monobasic (SA), Hydrochloric acid (SA), P-nitrophenyl glucopyranoside (Pars Biochem), Sodium bicarbonate (SA), Streptozotocin (SA), Sodium citrate (SA), Citric acid (SA), Glucose, Dimethyl sulfoxide (DMSO) (SA), Hematoxylin and Eosin (H&E) dye.
Collection and Identification of Plant
The plant specimen was acquired from mountainous areas near Rustam (34°21’57.0″N 72°22’08.0″E), Mardan, KPK, Pakistan. The specimen of plant was taxonomically identified and authenticated by Dr. Mohib Shah, Department of Botany, AWKUM. Voucher Number allotted by Department of Botany, AWKUM is; C. edulis Awkum.Bot.150.1.1
Extraction and Maceration
The fresh plant material was washed to remove impurities and subsequently shade-dried under ambient conditions. The desiccated plant material was subsequently finely milled with an electric grinder and the resulting powder was subjected to maceration in a hydroalcoholic solvent (comprising 90% methanol and 10% distilled water) using an inert glass container for 14 days with intermittent stirring. Following the maceration period, the liquid extract (menstruum) was separated from the solid residue (marc) by passing the mixture through muslin cloth, followed by filtration with filter paper (Whatman No. 1). To ensure the maximum extraction yield, the process was repeated three times. The resulting filtrate was concentrated by evaporating the solvent using a rotary evaporator maintained at 30–40°C. The concentrated extract was subsequently dried in an oven at 40°C . The dry extracts were then stored in a refrigerator at 2-8°C until being utilized in the experiments.
Fractionation
The methanolic extract was treated with warm distilled water, subsequently subjected to liquid-liquid extraction with n-hexane using a separating funnel. Fractionation of the methanolic phase with ethyl acetate resulted in the separation of the n-hexane fraction; similarly, ethyl acetate and aqueous fractions were also obtained through this process. To ensure maximum yield, the fractionation procedure was repeated several times. The obtained fractions were concentrated using a rotary vacuum evaporator and then dried in an oven at approximately 40°C to remove residual solvents.
Antioxidant assay
DPPH (1,1-diphenyl-2-picrylhydrazyl) radical scavenging method
The DPPH radical scavenging activity was assessed in a 96-well microplate using the method described by Taha, M., et al. 11, with minor modifications. A reaction mixture containing 95 μL of DPPH (300 μM dissolved in methanol) and test sample (10 μL). Following this, the reaction mixture was incubated in the dark at 37 °C for 30 minutes. Absorbance was measured at 517 nm using a microplate reader. The percentage of radical scavenging activity was calculated by comparing the sample absorbance to that of methanol, which served as the negative control. Ascorbic acid was used as the positive control, and the percentage of antioxidant activity was determined using the following formula:
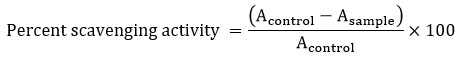
where A control represents the absorbance of the normal control reaction (comprising DPPH solution and methanol), and A sample shows the absorbance of test sample.
ABTS (2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) inhibitory assay
The ABTS radical scavenging property was assessed employing the previously reported method with minor adjustments. 12 At first, a 7 mM concentrated solution of ABTS was prepared by dispersing it in an aqueous medium. Moreover, ABTS•+ was produced by mixing the stock solution with 2.45 mM potassium persulfate and incubating the mixture in the dark at room temperature prior to use. After 12-16 hours, the ABTS•+ solution was diluted with PBS (pH 7.4) to achieve an absorbance of 0.7 ± 0.05 at 734 nm. A volume of 190 μL of the diluted ABTS•+ solution was combined with 10 μL of the standard, ascorbic acid, or sample in a microplate reader, and the absorbance was recorded at 734 nm against the appropriate solvent blank. The percentage of ABTS•+ radical scavenging was calculated for various concentrations of both the extract and the standard using the provided equation:
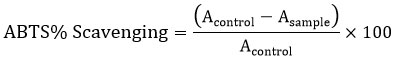
A control represent the absorbance of control reaction (comprising 7 mM ABTS, 2.45 mM K2S2O8), and A sample indicate of the test sample.
Experimental animals
Male and female Swiss albino mice, aged 6–9 weeks and weighing between 25–35 grams, were utilized in this study. Ethical approval for the experimental procedures was obtained from the Research Ethics Committee of Abdul Wali Khan University (Ethical Approval #: EC/AWKUM/2024/02). Experimental animals were procured from the National Institute of Health (NIH), Islamabad. All procedures involving animals adhered to the guidelines established by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), ensuring ethical handling and proper care. Prior to the commencement of experiments, the mice underwent a seven-day acclimatization period to reduce stress associated with transportation, which can impact physiological, neurological, immunological, endocrine, and cardiovascular functions. This acclimatization helped maintain stable environmental and behavioral conditions. Animals were housed under controlled conditions with a 12-hour light/dark cycle at ambient room temperature. Standard laboratory chow and tap water were provided ad libitum throughout the study duration.
Acute Toxicity
Acute oral toxicity of the samples was evaluated following the OECD (Organisation for Economic Co-operation and Development) Test Guideline 423. The study was conducted using Swiss albino mice, which were randomly divided into four groups, each comprising six animals. The test extract was administered orally at increasing dose levels of 250, 500, 1000, and 2000 mg/kg body weight. Throughout the experiment, animals were monitored closely for changes in body weight, food and water consumption, and physical appearance. Particular attention was paid to diabetes-related symptoms such as polyuria, polydipsia, polyphagia, and weight loss. Observations were made over a 24-hour period, and any signs of toxicity or mortality were meticulously recorded.
In Vivo Antidiabetic Activity
Induction of diabetes
Following a one-week acclimatization period, the experimental animals were subjected to a 6-hour fasting phase. Eight mice were randomly assigned to the Normal Control (NC) group and received an intraperitoneal injection of 0.06 mL distilled water. The remaining animals were administered streptozotocin (STZ) at a dose of 50 mg/kg body weight, freshly dissolved in cold citrate buffer (0.1 M, pH 4.5), via intraperitoneal injection. This treatment was repeated once daily for five consecutive days. On the sixth day, fasting blood glucose levels were measured, and mice exhibiting blood glucose concentrations greater than 11.1 mmol/L were classified as diabetic.
Oral glucose tolerance test (OGTT)
The Oral Glucose Tolerance Test (OGTT) was performed based on a previously reported protocol, with minor adjustments. 13 The animals were randomly assigned to six groups, each consisting of six mice. The first group received distilled water and served as the normal control (NC), while the second group, designated as the diabetic control, also received distilled water. The third group was administered glibenclamide, a standard antidiabetic agent, at a dose of 5 mg/kg. The remaining three groups were treated with the crude extract at oral doses of 50, 100, and 200 mg/kg, respectively, via gavage. Thirty minutes post-treatment, all animals received an oral glucose load of 2 g/kg. Blood samples were collected from the tail vein prior to drug administration and at 0, 30, 60, and 120 minutes following glucose administration. Blood glucose levels were measured immediately using a glucometer.
Experimental design
Following the confirmation of diabetes induction, the animals were placed randomly in six groups, with each group consisting of eight mice. Distilled water was administered to Group I, which act as the normal control. Group II (diabetic control) comprised diabetic mice that were administered distilled water. Group III (positive control) included diabetic mice treated with glibenclamide at a dose of 5 mg/kg body weight. Groups IV, V, and VI consisted of diabetic mice treated with the crude extract at doses of 50 mg/kg, 100 mg/kg, and 200 mg/kg body weight, respectively.
Glibenclamide (standard drug) and the crude extract were administered orally once daily for 21 consecutive days. Fasting blood glucose levels were monitored using an ACU-CHEK® glucometer, with blood samples collected from the tail vein on days 0, 7, 14, and 21 of the study. 14,15
Effect on body weight
Baseline body weights of all animals were recorded at the start of the experiment. During the treatment period, body weight was measured using an electronic balance on days 7, 14, and 21.
Estimation of glycosylated hemoglobin, serum insulin and liver glycogen
At the end of the 21-day experiment, mice were fasted for 12 hours and then anesthetized mildly. Blood samples were collected via cardiac puncture and transferred to EDTA tubes. The glycosylated hemoglobin (HbA1c) test was carried out following an established method. 16 Assessment of serum insulin concentration was done by using the standard protocols.17After 21 days of experimentation, the mice were fasted for 12 hours and sacrificed by cervical severance. Following rinsing with normal saline, approximately 1 g of liver tissue was excised and transferred into a centrifuge tube containing 2 ml of potassium hydroxide (KOH, 300 g/L). The mixture was heated for approximately 20 minutes with intermittent shaking. Subsequently, 0.2 ml of a saturated sodium sulfate solution was added and mixed thoroughly. Glycogen was precipitated by the addition of 5 ml of ethanol, and the resulting precipitate was dissolved in 10 ml of distilled water. A 1 ml aliquot of this solution was combined with 1 ml of hydrochloric acid (HCl) and the mixture was boiled for 2 hours. Upon completion of hydrolysis, the solution was neutralized with sodium hydroxide (NaOH) using phenol red as a pH indicator. The final volume was adjusted to 5 ml and transferred into a colorimetric cuvette. Absorbance was measured at 620 nm after appropriate blank correction. Hepatic glycogen content was expressed as mg per gram of liver tissue.18
Histopathological study
After the experiment, animals were dissected, pancreatic tissue samples were taken and preserved in 20% buffered formalin. The tissue samples were then cut into slices approximately 1 cm thick and placed in designated cassettes. These cassettes were transferred to a tissue processing machine, where they underwent automated stages, including alcohol dehydration, xylene and wax clearing, followed by impregnation with paraffin for approximately 14 hours. The tissue cassettes were subsequently embedded in molten paraffin, which solidified into blocks upon cooling. The paraffin blocks were trimmed and sectioned at a thickness of approximately 5 μm using a microtome. The sections were stained with Hematoxylin and Eosin (H&E), mounted with DPX, and examined microscopically.
Statistical analysis
All experiments were conducted in triplicate, and data are expressed as mean ± standard error of the mean (SEM). The half-maximal inhibitory concentration (IC₅₀) values were calculated using IBM SPSS Statistics version 21. Statistical analyses were performed using one-way ANOVA followed by Dunnett’s post hoc test for in vitro data, and two-way ANOVA followed by the Bonferroni post hoc test for in vivo data, using GraphPad Prism version 8.0.2. A p-value of less than 0.05 was considered indicative of statistical significance.
Results
Effect of antioxidant activity
Effect of DPPH inhibitory activity
The inhibitory effects of the crude hydro-methanolic extract of C. edulis and its fractions on DPPH were assessed through in vitro activity. Different concentrations, including 1000, 500, 250, 125, 62.5, 31.25, 15.63 and 7.81 µg/ml, were employed to evaluate the samples and determine their IC50 values. The ethyl acetate fraction demonstrated a significantly lower IC50 value of 20.324 µg/ml compared to the crude extract (27.291 µg/ml), aqueous (96.969 µg/ml), and n-hexane (205.474 µg/ml) fractions. In contrast, ascorbic acid, utilized as the standard, exhibited the maximum DPPH inhibition with an IC50 value of 10.159 µg/ml, as illustrated in Figure 1.
 |
Figure 1: Representing DPPH inhibitory activity of (a) Ethyl acetate fraction (b) Crude extract (c) Aqueous fraction (d) n-Hexane fraction and (e) standard drug, Ascorbic acid. |
Effect of ABTS activity
The crude hydro-alcoholic extract of C. edulis and its different fractions were examined for their capability to inhibit ABTS in vitro. To evaluate the samples and calculate their IC50 values, various concentrations of 1000, 500, 250, 125, 62.5, 31.25, 15.63 and 7.81 µg/ml were utilized. The ethyl acetate fraction demonstrated a notably lower IC50 value of 13.204 µg/ml when compared to the crude extract (16.856 µg/ml), aqueous (46.843 µg/ml), and n-hexane (120.383 µg/ml) fractions. In comparison, ascorbic acid, employed as the reference standard, exhibited the greatest inhibition of ABTS with an IC50 value of 12.342 µg/ml, as illustrated in Figure 2.
 |
Figure 2: Representing ABTS inhibitory activity of (a) Ethyl acetate fraction (b) Crude extract (c) Aqueous fraction (d) n-Hexane fraction and (e) standard drug, Ascorbic acid. |
In-vivo activities
Animals
Swiss albino mice, weighing between 25–35 grams, were obtained from the National Institute of Health (NIH), Islamabad, Pakistan. The animals were housed in sanitized cages under standard laboratory conditions, maintaining a temperature of 25 ± 2 °C, relative humidity of 50 ± 5%, and a 12-hour light/dark cycle. They were provided with a standard rodent diet and had unrestricted access to drinking water. The study protocol received approval from the Research Ethics Committee of the Department of Pharmacy, Abdul Wali Khan University, Mardan, Khyber Pakhtunkhwa, Pakistan, following thorough ethical review.
Acute toxicity
Crude hydro-methanolic extracts of whole plant C. edulis were orally administered in in vivo testing for LD50 at different doses, 250 mg/kg, 500 mg/kg, 1000 mg/kg and 2000 mg/kg of body weight. Each dose group included three replicates along with positive and negative controls. Animals were observed continuously for 4 hours for clinical signs of toxicity and further monitored for 24 hours for mortality. No treatment-related morbidity or mortality was recorded across all groups, as shown in the table 1. Based on these observations, the estimated median lethal dose (LD₅₀) for the crude hydro-methanolic extract of C. edulis was determined to be greater than 2000 mg/kg.
Table 1: Gross effects presented by animals in acute toxicity test of Caralluma edulis
| Sr.NO. | Doses mg/Kg | ||||
| Gross effects | 250 | 500 | 1000 | 2000 | |
| 1 | Abdominal contractions | – | – | – | – |
| 2 | Urination | – | ++ | + | +++ |
| 3 | Sedation | – | – | ++ | – |
| 4 | Tremors | – | – | – | – |
| 5 | Emesis | – | – | – | – |
| 6 | Lacrimation | – | – | ++ | – |
| 7 | Alertness | + | +++ | + | +++ |
| 8 | Death | – | – | – | – |
Key: + (mild), ++ (moderate), +++ (sever), + (present), – (absent)
Result of glucose tolerance test
The crude hydro-methanolic extract of C. edulis was evaluated for its effect on oral glucose tolerance (OGTT) at dose levels of 50, 100, and 200 mg/kg body weight. Blood glucose concentrations were measured at 0, 30, 60, and 120 minutes following glucose administration (2 g/kg). The diabetic control group exhibited significantly elevated glucose levels compared to the normal control. Both the experimental groups and the positive control (glibenclamide-treated) demonstrated a marked reduction in blood glucose levels relative to the diabetic control, with the exception of the 50 mg/kg group at 30 minutes post-glucose load. The crude extract elicited a statistically significant hypoglycemic effect, comparable to that of glibenclamide, as depicted in figure 3.
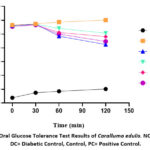 |
Figure 3: Oral Glucose Tolerance Test Results of Caralluma edulis. NC= Normal, DC= Diabetic Control, Control, PC= Positive Control. |
Results of in-vivo antidiabetic activity
Intraperitoneal administration of streptozotocin (STZ) at a dose of 50 mg/kg body weight resulted in a significant elevation of blood glucose levels in diabetic mice on day 0, compared to the normal control group. Subsequent oral administration of the crude hydroalcoholic extract of C. edulis at doses of 50, 100, and 200 mg/kg body weight for 21 consecutive days produced a significant reduction in blood glucose levels. This hypoglycemic effect was comparable to that of the standard drug, glibenclamide (5 mg/kg). The data are presented as mean ± SEM, as illustrated in the corresponding figure 4.
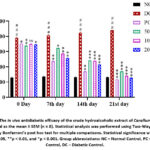 |
Figure 4: The in vivo antidiabetic efficacy of the crude hydroalcoholic extract of Caralluma edulis is illustrated as the mean ± SEM (n = 8). Statistical analysis was performed using Two-Way ANOVA, followed by Bonferroni’s post hoc test for multiple comparisons. |
Effect of crude hydroalcoholic extract on body weight of mice
The diabetic control group exhibited a significantly lower body weight (P < 0.01) compared to the normal control group on days 0, 7, 14, and 21. Treatment with the crude hydroalcoholic extract of C. edulis resulted in a significant improvement in body weight among diabetic mice, as presented in Table 2. Compared to the diabetic control group, mice in the extract-treated groups exhibited the significant increase in body weight on days 7, 14, and 21. Similarly, glibenclamide administration resulted in a marked improvement in body weight among diabetic mice at the same time points.
Table 2: Effect of crude hydro-methanolic extract of Caralluma edulis on body weight of mice
| Test Sample |
Dose (mg/kg) |
Body weight | |||
| Day 0 | 7th day | 14th day | 21st day | ||
| Normal control | 10ml | 28.2 ± 0.42 | 28.7 ± 0.263 | 29.5 ± 0.46 | 30.3 ± 0.36 |
| Diabetic control | No treatment | 33.7 ± 0.414 | 32.56 ± 0.59 | 30.85±0.47 | 28.78 ± 0.42 |
| Glibenclamide | 5mg | 29.1 ± 0.463 | 29.7 ± 0.410 | 30.4 ± 0.44 | 31.7 ± 0.26 |
| Caralluma edulis | 50mg | 28.6 ± 0.346 | 29 ± 0.542 | 29.4 ± 0.488 | 29.9 ± 0.553 |
| 100mg | 26.3 ± 0.336 | 26.9 ± 0.283 | 27.4 ± 0.542 | 28 ± 0.314 | |
| 200mg | 28.2 ± 0.596 | 29 ± 0.643 | 29.4 ± 0.584 | 30 ± 0.531 | |
Effect on glycosylated haemoglobin, serum insulin and liver glycogen
Diabetic control animals had significantly higher level of glycosylated haemoglobin (P<0.001) than normal control. The diabetic control group exhibited a significantly elevated level of glycosylated hemoglobin (HbA1c) compared to the normal control (P < 0.001). Treatment with the crude hydroalcoholic extract of C. edulis resulted in a dose-dependent and statistically significant reduction in HbA1c levels, as indicated in figure 5A, compared to the diabetic control group. A significant reduction in HbA1c levels was also observed with glibenclamide administration. Serum insulin levels in the diabetes control group were substantially lower than in the normal control group. Serum insulin levels in the experimental groups significantly improved as depicted in figure 5B, In comparison to the diabetic control group, the extract-treated groups exhibited a significant increase in serum insulin levels. The standard drug, glibenclamide, also produced a marked elevation in insulin concentration. Additionally, the diabetic control group showed a significant reduction in hepatic glycogen content relative to the normal control. Treatment with the extract led to a notable improvement in liver glycogen levels, as illustrated in the corresponding figure 5C. Glibenclamide also significantly increased the level of liver glycogen when compared to the diabetic control.
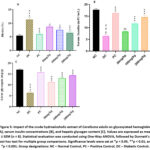 |
Figure 5: Impact of the crude hydroalcoholic extract of Caralluma edulis on glycosylated hemoglobin [A], serum insulin concentrations [B], and hepatic glycogen content [C]. Values are expressed as mean ± SEM (n = 8). |
Histopathological study
Normal diet-fed animals exhibited typical pancreatic tissue architecture without any pathological alterations. In contrast, STZ-induced diabetic mice displayed severe pathological changes, including edema, vascular congestion, fatty changes, inflammatory infiltrates, and tissue necrosis. Recovery from these pathological alterations occurred in a Dose-responsive manner following treatment with C. edulis extract. Pancreatic tissue samples were analysed and compared with the normal control. 50mg/kg extract resulted in the mild alterations, 100mg/kg showed mild to moderate and 200mg/kg displayed moderate improvements in the damage, as shown in the figure 6.
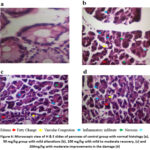 |
Figure 6: Microscopic view of H & E slides of pancreas of control group with normal histology (a), 50 mg/kg group with mild alterations (b), 100 mg/kg with mild to moderate recovery, (c) and 200mg/kg with moderate improvements in the damage (d) |
Discussions
This study assessed the antioxidant activity of the crude hydro-methanolic extract of the whole plant of C. edulis and its fractions, including ethyl acetate, aqueous, and n-hexane, was evaluated through DPPH and ABTS radical scavenging assays. Among the tested samples, the ethyl acetate fraction exhibited the highest inhibitory activity, likely due to its high phenolic compound content, which is well-documented for its antioxidant properties. 19,20 The crude extract demonstrated moderate inhibitory activity, whereas the aqueous and n-hexane fractions exhibited weak antioxidant potential compared to ascorbic acid, which was utilized as the standard reference compound.
In preclinical research, the Oral Glucose Tolerance Test (OGTT) is primarily employed to assess glucose homeostasis, rather than for diagnostic purposes as in clinical practice. Insulin resistance and sensitivity are evaluated through the glucose-insulin index derived from OGTT data in animal models. Impaired glucose tolerance (IGT) is typically indicated by an elevated incremental area under the plasma glucose curve (iAUC) during the OGTT. A significant elevation in AUC0-120 min observed during OGTT confirmed the effective establishment of a diabetic model in the experimental animals. 21,22 Subsequently, diabetic animal models are frequently utilized to assess the therapeutic potential of experimental agents, including nutrients and herbal extracts. A notable change in the glucose disposal phase slope, coupled with a reduced area under the curve (AUC) when compared to the vehicle-treated control, indicates that the evaluated compound may possess the ability to alleviate impaired glucose tolerance (IGT), potentially by improving glucose metabolism.23,24 In the present study, the crude hydro-methanolic extract of C. edulis effectively mitigated impaired glucose tolerance by significantly reducing blood glucose concentrations in a dose-dependent manner, with the most notable reduction observed at a dose of 200 mg/kg.
Diabetes was induced by administering streptozotocin (STZ) intraperitoneally at a dose of 50 mg/kg for a duration of five consecutive days. Streptozotocin, also referred to as Streptozocin, is a natural antibiotic derived from Streptomyces achromogenes, first identified in 1959. Its toxic effects on pancreatic β-cells were first described in 1963.25,26 It is one of the most widely used diabetogenic agents in experimental animal models to induce Type 1 and Type 2 diabetes mellitus (T1DM and T2DM). 27 The findings of our study indicate that the crude hydroalcoholic extract of C. edulis at a dose of 200 mg/kg demonstrated the most significant antidiabetic activity, surpassing the effects of the standard drug, glibenclamide. The observed reduction in blood glucose levels by C. edulis may be attributed to the presence of various bioactive phytochemicals, such as berberine, curcumin, rutin, anthocyanin, genistein, and quercetin, all of which are known to possess blood glucose-lowering properties.28
Elevated blood glucose levels and reduced insulin secretion induced by streptozotocin (STZ)-induced diabetes can lead to a decrease in body mass in experimental animals.18 Medicinal plants possess the capacity to counteract weight loss induced by diabetes. A prior study explored the antidiabetic properties of several traditional plant species. The results revealed that these plants significantly prevent the weight loss associated with STZ-induced diabetes in experimental rodents. 29 The findings of the present study also demonstrated that treatment with the crude hydro-methanolic extract of C. edulis significantly enhanced body weight in the treated mice, as compared to the diabetic control group.
The crude extract of C. edulis caused a significant reduction in glycosylated hemoglobin (HbA1c) levels, an effect that may be attributed to its high dietary fiber content, 30 this observation is consistent with previous reports indicating that diets high in dietary fiber are regarded as beneficial for individuals with diabetes, as they help to achieve a lower plasma glucose response. 31 Additionally, the plant contains ellagic acid and anthocyanins, which may contribute to the reduction in HbA1c levels. 28,32 A standard insulin assay quantifies both endogenous and exogenous insulin. In the absence of exogenous insulin administration, serum insulin levels serve as an indicator of pancreatic beta-cell function. Previous studies have shown that streptozotocin administration results in a significant reduction in serum insulin levels produced by the pancreas. 33 In the present study, C. edulis significantly enhanced serum insulin levels. This effect may be attributed to the presence of oleic acid, linoleic acid, and palmitic acid esters, which are recognized for their ability to stimulate insulin secretion under diabetic conditions. 34
Streptozotocin treatment has been reported to significantly reduce hepatic glycogen content in diabetic animal models. 35 In the current investigation, treatment with the crude extracts of C. edulis resulted in a significant increase in hepatic glycogen content compared to the diabetic control group. This effect is likely attributable to the presence of bioactive phytochemicals, including triterpenes and flavonoids, which have been associated with the modulation of hepatic glycogen metabolism. 36
Histological changes observed in diabetic animals including rats and mice closely resemble the human disease 37 Histopathological analyses of a pancreatic segment revealed damaged epithelial lining cells with bleeding and congested blood vessels, as well as inflammatory infiltration and necrosis in the interlobular septa. 38 In the present study, pancreatic tissues of mice from each tested and control group were analyzed. Cells of the normal control group revealed no damaging effect. Administration of the extract restored normal cellular architecture in a dose-dependent manner. At 50 mg/kg, the crude extract of C. edulis produced mild histological alterations, whereas a dose of 100 mg/kg resulted in mild to moderate restoration. The highest dose, 200 mg/kg, elicited marked improvements in cellular integrity when compared to the diabetic control group. Pathological damage, including edema, vascular congestion, fatty changes, inflammatory infiltrates, and necrosis, was significantly mitigated in a dose-dependent fashion by the investigated plant extract. Notably, the tissue architecture in all treated animals was preserved.
Conclusion
This study demonstrates the antioxidant property of the crude hydroalcoholic extract and fractions of C. edulis, with the ethyl acetate fraction showing the strongest antioxidant activity. Additionally, the crude extract exhibited antidiabetic effects in STZ-induced diabetic mice, improving blood glucose, serum insulin, body weight, and liver glycogen levels, while reducing glycosylated hemoglobin. Histopathological evaluations revealed dose-dependent improvements in edema, fatty changes, vascular congestion, inflammation, and necrosis. These findings suggest that C. edulis possesses significant antidiabetic potential, Moreover, further detailed studies at the molecular level are recommended for this plant.
Acknowledgement
The authors extend their appreciation to the Princess Nourah Bint Abdulrahman University Researchers Supporting Project number (PNURSP2025R13), Princess Nourah Bint Abdulrahman University, Saudi Arabia for funding.
Funding Sources
The authors gratefully acknowledge the financial support from the Higher Education Commission (HEC) of Pakistan, under project No: [Ref No. 20-16097/NRPU/R&D/HEC/2021 2021].
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The data that support the findings of the present study are available from the corresponding authors upon reasonable request
Ethics Statement
The ethical approval for the animal studies has already been included in the manuscript and is cited with the approval number: Ethical Approval #: EC/AWKUM/2024/02
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to Reproduce Material from Other Sources
Not Applicable
Clinical Trial Registration
This research does not involve any clinical trials.
Author Contributions
- Fazal Hadi: Experimental and writing the initial draft;
- Haseeba Sardar: Experimental and writing the initial draft;
- Jamelah Al-Otaibi: Experimental analysis and writing the initial draft;
- Abdul Saboor Pirzada Experimental and writing the initial draft;
- Waqas Alam: Histological analysis and writing the initial draft;
- Maria Daglia: Editing and final reversion of the article.
- Haroon Khan: supervised the overall project and refined the manuscript for publication. All authors approved the final version for submission.
References
- Tabish SA. Is Diabetes Becoming the Biggest Epidemic of the Twenty-first Century? Int J Health Sci (Qassim). Jul 2007;1(2):5-8.
- Aloke C, Egwu CO, Aja PM, et al. Current Advances in the Management of Diabetes Mellitus. Biomedicines. Sep 29 2022;10(10)doi:10.3390/biomedicines10102436
CrossRef - Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. Jun 2005;54(6):1615-1625. doi:10.2337/diabetes.54.6.1615
CrossRef - Tariq S. Biochemical, phytochemical, and bioogical evaluation of Caralluma edulis from Cholistan Desert. 2020.
- Aslam I, Iqbal J, Peerzada S, Afridi MS, Ishtiaq S. Microscopic investigations and pharmacognostic techniques for the standardization of Caralluma edulis (Edgew.) Benth. ex Hook. f. Microscopy research and technique. 2019;82(11):1891-1902.
CrossRef - Sharawy S, Kamel E, Karakish E, Loutfy M. A systematic revision on Caralluma species of Saudi Arabia based on karyological and molecular data. Pak J Bot. 2015;47(3):937-950.
- Ansari B, Behl T, Pirzada AS, Khan H. Caralluma edulis (Apocynaceae): A comprehensive review on its traditional uses, phytochemical profile and pharmacological effects. Current Topics in Medicinal Chemistry. 2022;22(18):1501-1514.
CrossRef - Ullah I, Gul S, Khan RU, et al. Antibacterial and antioxidant activity analysis of some wild medicinal plants. Journal of Entomology and Zoology Studies. 2017;5(6):1771-1775.
CrossRef - Khan M, Manzoor Z, Rafiq M, et al. Phytochemical screening, anti-inflammatory, and antidiabetic activities of different extracts from Caralluma edulis plant. Molecules. 2022;27(16):5346.
CrossRef - Minhas A, Khan A-u, Ansari M. Anti-inflammatory effect of Caralluma edulis against acute and chronic inflammation. JAPS: Journal of Animal & Plant Sciences. 2018;28(2).
- Taha M, Ismail NH, Jamil W, et al. Synthesis, evaluation of antioxidant activity and crystal structure of 2, 4-dimethylbenzoylhydrazones. Molecules. 2013;18(9):10912-10929.
CrossRef - Ahmed S. Antioxidant Assays in Phytonutrient Research: Translating Laboratory Innovations into Practical Applications: Antioxidant Assays in Phytonutrient Research. PHYTONutrients. 2025:08-27.
CrossRef - Pandhare RB, Sangameswaran B, Mohite PB, Khanage SG. Antidiabetic Activity of Aqueous Leaves Extract of Sesbania sesban (L) Merr. in Streptozotocin Induced Diabetic Rats. Avicenna J Med Biotechnol. Jan 2011;3(1):37-43.
- Sultan K, Zakir M, Khan H, et al. Antihyperglycemic effect of Persea duthieion blood glucose levels and body weight in alloxan induced diabetic rabbits pakistan Journal Pharmaceutical Sciences. 2016;29(3):837-842.
- Zakir M, Sultan K, Munir Y, et al. Biofunctional beverage: Antihyperglycemic effect of green tea in alloxan induced diabetic rabbits. Curr Bioact Comp. 2019;15:120-124.
CrossRef - Nayak SS, Pattabiraman T. A new colorimetric method for the estimation of glycosylated hemoglobin. Clinica Chimica Acta. 1981;109(3):267-274.
CrossRef - Soni LK, Dobhal MP, Arya D, Bhagour K, Parasher P, Gupta RS. In vitro and in vivo antidiabetic activity of isolated fraction of Prosopis cineraria against streptozotocin-induced experimental diabetes: A mechanistic study. Biomedicine & Pharmacotherapy. 2018/12/01/ 2018;108:1015-1021. doi:https://doi.org/10.1016/j.biopha.2018.09.099
CrossRef - Zafar M, Naqvi SN-u-H. Effects of STZ-Induced Diabetes on the Relative Weights of Kidney, Liver and Pancreas in Albino Rats: A Comparative Study. International Journal of Morphology. 2010;28(1)
CrossRef - Shalev Y, Hadaya O, Bransi-Nicola R, et al. Entourage effect for phenolic compounds on production and metabolism of mammary epithelial cells. Heliyon. Mar 2022;8(3):e09025. doi:10.1016/j.heliyon.2022.e09025
CrossRef - Sannigrahi S, Kanti Mazuder U, Kumar Pal D, Parida S, Jain S. Antioxidant Potential of Crude Extract and Different Fractions of Enhydra fluctuans Lour. Iran J Pharm Res. Winter 2010;9(1):75-82.
- Babbar R, Heni M, Peter A, et al. Prediction of glucose tolerance without an oral glucose tolerance test. Frontiers in endocrinology. 2018;9:82.
CrossRef - Abbas HT, Alic L, Erraguntla M, et al. Predicting long-term type 2 diabetes with support vector machine using oral glucose tolerance test. Plos one. 2019;14(12):e0219636.
CrossRef - Kuo SC, Li Y, Cheng JT. Glucose Tolerance Test Applied in Screening of Anti-Diabetic Agent (S). Current Research in Diabetes & Obesity Journal. 2018;7(4):69-71.
CrossRef - Kuo FY, Cheng KC, Li Y, Cheng JT. Oral glucose tolerance test in diabetes, the old method revisited. World J Diabetes. Jun 15 2021;12(6):786-793. doi:10.4239/wjd.v12.i6.786
CrossRef - Thurston DE, Pysz I. Chemistry and pharmacology of anticancer drugs. CRC press; 2021.
CrossRef - Capdevila J, Ducreux M, García Carbonero R, et al. Streptozotocin, 1982–2022: Forty years from the FDA’s approval to treat pancreatic neuroendocrine tumors. Neuroendocrinology. 2022;112(12):1155-1167.
CrossRef - Gauthier EL. Streptozotocin: uses, mechanism of action and side effects. Nova Science Publishers, Incorporated; 2014.
- Luo Y, Zeng Y, Peng J, et al. Phytochemicals for the treatment of metabolic diseases: Evidence from clinical studies. Biomedicine & Pharmacotherapy. 2023/09/01/ 2023;165:115274. doi:https://doi.org/10.1016/ j.biopha.2023. 115274
CrossRef - Swanston-Flatt SK, Day C, Bailey CJ, Flatt PR. Traditional plant treatments for diabetes. Studies in normal and streptozotocin diabetic mice. Diabetologia. 1990/08/01 1990;33(8):462-464. doi:10.1007/BF00405106
CrossRef - Enyiukwu D, Amadioha A, Ononuju C. Nutritional significance of cowpea leaves for human consumption. Greener Trends Food Sci Nutr. 2018;1:1-10.
CrossRef - Olopade OB, Odeniyi IA, Iwuala S, et al. Comparison of glycaemic indices of some local beans (Vigna unguiculata [Linn] Walp varieties) in Nigerians. Journal of Endocrinology, Metabolism and Diabetes of South Africa. 2017;22(3):51–55-51–55.
CrossRef - Uuh Narvaez JJ, Castellanos Ruelas AF, Olivera Castillo L, Puerto Castillo C, Segura Campos MR. Saponins from Vigna unguiculata husks obtained by microwave-assisted extraction: Identification and mechanism of inhibition on urease activity. South African Journal of Botany. 2023/03/01/ 2023;154:265-272. doi:https://doi.org/10.1016/ j.sajb.2023.01.038
CrossRef - Dulin W, Soret M. Chemically and hormonally induced diabetes. The diabetic pancreas. Springer; 1977:425-465.
CrossRef - Bhardwaj M, Yadav P, Yadav M, Chahal J, Dalal S, Kataria SK. Phytochemical Screening and Antidiabetic Efficacy of Balanites aegyptiaca Seed Extract and Their Silver Nanoparticles on Muscle and Pancreatic Cell Lines. ACS Omega. 2024/05/28 2024;9(21):22660-22676. doi:10.1021/acsomega.4c00327
CrossRef - Ros S, García-Rocha M, Calbó J, Guinovart JJ. Restoration of hepatic glycogen deposition reduces hyperglycaemia, hyperphagia and gluconeogenic enzymes in a streptozotocin-induced model of diabetes in rats. Diabetologia. 2011/10/01 2011;54(10):2639-2648. doi:10.1007/s00125-011-2238-x
CrossRef - Alam S, Sarker MMR, Sultana TN, et al. Antidiabetic Phytochemicals From Medicinal Plants: Prospective Candidates for New Drug Discovery and Development. Front Endocrinol (Lausanne). 2022;13:800714. doi:10.3389/fendo.2022.800714
CrossRef - Yamamoto T, Nakamura T, Noble NA, Ruoslahti E, Border WA. Expression of transforming growth factor beta is elevated in human and experimental diabetic nephropathy. Proceedings of the National Academy of Sciences. 1993;90(5):1814-1818.
CrossRef - AL-Aamery MSH, Alol LH, Ataee EHY. Study the Patho-physiological changes after induce diabetes in mouse. Global Journal of Bio-Sciences and Biotechnology. 2016;5:268-275.













