Valorization of Fruit and Vegetable Processing Wastes as Prebiotics: A Review
1Computational Modeling and Nanoscale Processing Unit, Department of Food Process Engineering, National Institute of Food Technology Entrepreneurship and Management - Thanjavur (NIFTEM-T), Thanjavur, India
2Medicaps University, Indore, Madhya Pradesh, India
Corresponding Author Email: moses.ja@iifpt.edu.in
Download this article as:
ABSTRACT:Although fruit and vegetable processing wastes are a great source of several macro/micronutrients and nutraceutical ingredients, in most cases, they are discarded or underutilized. Given the advancements in technology and the increasing awareness about the prospects of resource recovery, it is indeed a sustainable approach to effectively valorize such biomasses for both food and non-food applications. One promising concept is their prebiotic potential. Particularly, these agro-processing wastes are rich in dietary fiber and an array of bioactive compounds, making them an excellent substrate for probiotics, which are valued for their numerous health benefits. The effective extraction and inclusion of prebiotic fibers, oligosaccharides and bioactives from processing residues into a variety of functional foods, beverages, and supplements has been made possible by advancements in food technology. Considering the increasing research interest in this area, this review presents the fundamental concepts, expands on compositional merits, provides commodity-specific advantages, and concludes with the relevance and rising thrust of sustainability and the circular food economy. The sector benefits from the synergy between the circular economy policies and increased industry focus on waste valorization strategies, which further improve commercial viability. With consumers placing a greater emphasis on sustainability, clean-label products, and vegan-friendly options, plant-based prebiotics, including those made from fruit and vegetable byproducts, dominate market shares. Emphasis is placed on recent insights and innovations. With promising prospects and a keen focus on a circular food economy, the approach must consider scalability and commercialization, as many waste streams require pretreatment, the heterogeneous nature of waste makes process optimisation difficult and can lead to scalability issues
KEYWORDS:Circular food economy; Food processing waste; Prebiotic; Probiotic; Resource recovery; Waste valorization
Introduction
Globally, every year, over 0.9 billion tonnes of fruits and 1.2 billion tonnes of vegetables are produced.1 Although such figures are enormous, a sizable portion gets wasted or lost, with levels varying from country to country. Over time, agro-food processing solutions have been continuously explored to address this issue. However, even during the processing and value addition of these commodities, significant levels, ranging from 40 to 50%, are collected as ‘wastes’ and ‘discards’.2 This waste can be cumulative of peels, seeds, pomaces, shells, rags, stones, pods, vines, skins, etc. Unfortunately, current practices fall far short of the concept of ‘zero discards’; massive volumes of organic materials are often regarded as garbage and are burned, broken down anaerobically, or composted to provide energy, heat, and fertilizers, or sent as animal feed. Although not well reported, a sizable fraction ends up in landfills or is discarded in water bodies, making rotting biomass a pressing environmental concern. Furthermore, these processing waste streams are highly perishable, making them susceptible to microbial contamination, including mold growth and the introduction of aflatoxins into the food system. Over time, this can be utilized as a fertilizer in agriculture, as live feed in aquaculture, and as a biogas production source in renewable energy. However, these are considered waste by the food industry. Currently, the world is moving towards sustainability and the circular economy, so there is an urgent need to develop solutions that maximize the value of these waste materials, thereby achieving social, environmental, and financial benefits.
Probiotic bacteria are live microorganisms that, when administered in sufficient amounts, confer health benefits to the host.3 Probiotics such as Bifidobacteria spp. and Lactobacilli spp. support human health in many ways.4 The beneficial substances produced by probiotics during their growth that impart gut health are named postbiotics. Although they may be given as supplements, several indigenous fermented foods are also known to be rich in probiotics. As a practical approach, promoting the growth of these existing microbes through increased dietary intake of prebiotics can also improve the health-promoting activities of these microorganisms.5 The nondigestible food materials linked by glucosidic bonds that have a β-configuration are selectively metabolized by these beneficial organisms and are termed prebiotics. These act as a substrate for probiotics to stimulate their growth. Prebiotics possess certain characteristic features6 and these are summarized in Figure 1.
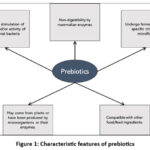 |
Figure 1: Characteristic features of prebiotics |
Apart from their significance to probiotics, which is most widely recognized, prebiotics render diverse health benefits. Therefore, there has been tremendous focus on identifying prebiotic substances that can bring about the most effective solutions for commercial-scale applications. Asparagus, sugar beet, garlic, chicory, onion, Jerusalem artichoke, wheat, honey, banana, barley, tomato, rye, soybean, human’s and cow’s milk, peas, and beans are the natural sources of prebiotics and are low in concentration; hence, conventionally, these prebiotics are sourced from lactose, sucrose, and starch as raw material.7 However, considering the requirements listed in Figure 1 and the wide availability of fruit and vegetable wastes, trends indicate that these agro biomasses can be exploited as prebiotic materials (Figure 2).
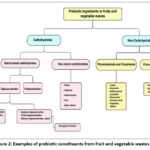 |
Figure 2: Examples of prebiotic constituents from fruit and vegetable wastes |
Overall, this review covers scientific evaluations of the utilisation of waste generated from fruit and vegetable fractions as prebiotics, which are crucial for probiotics. This approach serves two purposes: to address the rising concerns in waste management within agro-processing industries more effectively and to effectively utilise a widely available resource that is often discarded as ‘zero-value waste.’
Prebiotics and global markets
Until recently, all recognized prebiotics were carbohydrates, mainly oligosaccharides (OS). Nondigestible oligosaccharides (NDOs) belong to carbohydrate-type prebiotics and reach the colon intact by escaping the upper gut due to their nondigestible nature, and are fermented by intestinal microorganisms and produce volatile fatty acids. These, in turn, express several health benefits.8 A variety of non-carbohydrate materials, such as polyunsaturated fatty acids and polyphenols, are emerging prebiotics.9 There are more than 8000 recognized polyphenols in fruits, vegetables, and plants. Many make it to the colon unharmed and intact, where they are used by the microbes that live there.10 A few polyphenols have demonstrated prebiotic efficacy, such as extracts high in cranberries that stimulate Akkermansia muciniphila or offer pathogen-fighting antimicrobial activity.11
The market for prebiotics is not limited to humans but extends to animal health applications. With increasing awareness, several industrial sectors are finding increasingly expanding avenues for prebiotic substances. The main sectors involved in prebiotic synthesis are the food and beverage, pharmaceutical and healthcare, biotech, and animal feed industries.12 Prebiotics, as a functional ingredient, are used in many food industries; dairy products, confectionery, infant formulas, whole wheat bread, cereal bars, chocolate, and meat products are a few among them.13 Trends predict the prebiotic market to boom and fetch a profit of approximately $20.89 billion by 2030; the market was $6.71 billion in 2022 and will grow at a CAGR of 15.24% over the forecast period 2023-2030.14 Similarly, it is estimated that the market will grow at a CAGR of 12.30% from 2024 to 2034, from a market size of USD 8.97 billion in 2024 and USD 10.08 billion in 2025, to reach approximately USD 28.62 billion by 2034.15
Plant polysaccharides, bioactives, and their prebiotic potential
The most researched prebiotic component found in waste and byproducts from agro-food production operations is nondigestible carbohydrates. These are made of plant polysaccharides, which can be used as water-retaining substances (alginates and pectins), structural components (cellulose, hemicellulose, and pectin), or calorie reserves (starch or inulin). Further, fruit and vegetable waste and byproducts are rich in complex, nondigestible carbohydrates and polyphenols, which are considered prebiotic substrates. Flavonoids are one of the main classes of polyphenols, which influence the growth of the gut microbiota composition and benefit the host.16 Naturally occurring components in fruit and vegetable wastes can enhance intestinal microbial diversity and encourage the growth of probiotics. Intestinal flora breaks prebiotics down in the large intestine to create secondary metabolites, which are then absorbed by the intestinal epithelium or transported to the liver via the portal vein. These metabolites have many positive health effects on the host, such as enhancing intestinal barrier function, lowering blood lipid levels, regulating immunity, and preventing infections (Figure 3).
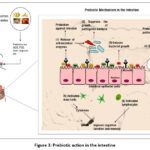 |
Figure 3: Prebiotic action in the intestine |
Oxygen and reactive oxygen species (ROS) generated by the electron transport system and TCA cycle during the oxidative phosphorylation of oxygen in mitochondria surround these microbes. Overexposure to ROS may be harmful to the probiotic population.17 As prebiotics, fructans (fructo-oligosaccharides and inulin) have a protective impact. In addition to activating antioxidant enzymes like glutathione S-transferases (GSTs), the short-chain fatty acids (SCFAs) generated as a result of their intestinal fermentation can indirectly scavenge ROS (Figure 4). An ex vivo experimental model reported the contrasting effect of inulin on the oxidative stress induced by lipo-polysaccharide in human colonic mucosa.18
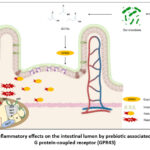 |
Figure 4: Anti-inflammatory effects on the intestinal lumen by prebiotic associated SCFA activated G protein-coupled receptor (GPR43) |
The generated galacto-oligosaccharides from the β-galactosidase transferase enzyme acting upon lactose favor Bifidobacteria proliferation. This was attributed to the galactose polymer-associated β-linked glucose monomer. The prebiotic effectiveness of lactulose was reported to depend on the dose given and the recipient’s physiology.19 Studies show that lactulose given at a dose of 5 g per day helps maintain an optimal balance of the members of the microbial community, comprising Bifidobacteria, Lactobacilli, and Anaerostipes. Such an intake also supports the generation of SCFAs. On the other hand, a high dose of 10 g per day was seen to suppress butyrate production while increasing the output of acetate, a byproduct of Bifidobacteria metabolism.20 Also, xylose-oligosaccharides exhibited a more significant bifidogenic effect on Bifidobacterium lactis compared to fructose-oligosaccharides in an artificial colon model. Isomaltooligosaccharides (IMO) acted in synergism with green tea extract in an in vivo model. Studies also demonstrated beneficial impacts on visceral adipose tissue, the synthesis of pro-inflammatory cytokines, and the regulation of lipid and glycemic levels, leading to enhancements in insulin, glucagon, and leptin concentrations.21
Carbohydrate components in fruit and vegetable wastes with prebiotic properties
Cellulose
Cellulose makes up most of the cell walls of all plants and is a polysaccharide consisting of anhydro-β-D-glucopyranose units that are favored by β-1,4-glycosyl linkages. Cellobiose, cellotriose, and cellotetraose are the names of the different oligosaccharides of cellulose and are sometimes referred to as cellodextrins. These include dimers, trimers, and tetramers. Because each bundle of fibers contains hydrogen bonds, cellulose has the strength and resistance to chemical and biological hydrolysis.22 It can absorb large volumes of water molecules, tends to swell, and is known to increase satiety and control body weight.23
Hemicellulose
After cellulose, hemicelluloses are the second most common biopolymer in the kingdom of plants. They form a primary component of the cell wall and are a relatively homogeneous 1-4-β-linked polyglucan, which sets them apart from cellulose. Hemicelluloses are a class of heteropolysaccharides that have complex structures and, depending on the source, vary in proportions of glucose, xylose, mannose, galactose, arabinose, fucose, glucuronic acid, and galacturonic acid.24
Lignin
Lignin is a complex natural aromatic polymer made up primarily of monolignols of substituted phenylpropanoid alcohols with methoxy (-OCH3) and hydroxy (-OH) groups. Its molecular weight ranges from 0.6 to 1.5 x 106 kg/kmol. Lignin originates from the enzymatic dehydrogenative polymerization of coniferyl alcohol, sinapyl alcohol, and p-coumaryl alcohol, the three main precursors of monolignols. The polymeric structure of lignin shows several connections between the monolignols, with the majority being alkyl- and aryl-ether linkages (β-O-4, α-O-4, 4-O-5, etc.) and less carbon-carbon and ester linkages.25
Pectin
Pectin is found in the cell walls of all higher plants. It is a complex polysaccharide rich in galacturonic acid (GalA). Three main pectin domains can be identified based on their structural characteristics: homogalacturonan (HG), which is the linear region of the polysaccharide, and rhamnogalacturonan-I (RG-I) and rhamnogalacturonan-II (RG-II), which are the branched domains of the polymer. HG is a homopolymer comprising a-D-GalA residues with some methyl-esterified C-6 carboxyl groups.26 However, the RG-I family of highly branched pectic polymers has a repeating disaccharide backbone (→4)-a-D-GalA-(1→2)-aL-Rha-(1→), in which side chains primarily composed of Rhamnose residues can be replaced with galactosyl and/or arabinosyl residues. The RG-II has about nine GalA residue backbones and four hetero-oligomeric side chains with a known and constant composition and length.27
Resistant starch (RS)
RS is a general term for a wide range of starch classes that can arise from different sources and are all resistant to upper gastrointestinal tract digestion. RS is primarily made up of the linear portion with α-(1-4) D-glucan units, which are primarily derived from the retrograded amylose fraction of starch and have a relatively low molecular weight (1.2×105 Da).28 Additionally, the authors reported that this type of starch is one of the most prevalent dietary sources of nondigestible carbohydrates and can be found in large quantities in whole grains, seeds, legumes, cooked and chilled pasta, potatoes, rice, unripe bananas, and cooled heat-treated starchy foods. From a physiological viewpoint, RS behaves generally in a manner akin to that of soluble, fermentable fiber.
Oligosaccharides
Galacto-oligosaccharides (GOS)
GOS are lactose-derived substances that have galactose or glucose at the reducing end and β-linked galactose moieties. Leguminous plant seeds are a natural source of GOS. The GOS comprises several β-connected galactose moieties, ranging in degree of polymerization (DP) from 2 to 8, such as β-(1 → 2), β-(1 → 3), β-(1 → 4), and β-(1 → 6).29 The most promising nondigestible oligosaccharides, GOS, have gained interest in the functional food industry because of their established health advantages and capacity to enhance the nutritional value of a wide range of meals. Because GOS are not digestible in the upper digestive system but are still fermentable by gut microorganisms, they are an excellent prebiotic. As a result, these are sometimes referred to as “nondigestible fibers” or “befidiogenic prebiotics” since they support the growth of bacterial species related to Bifidobacteria.30,31 Currently, GOS is also utilized as a low-calorie sweetener in a variety of food items, including yogurt, beverages, confections, and bread.32
Fructo-oligosaccharides (FOS)
Inulin and FOS are collectively termed ‘fructans.’ FOS are a type of oligosaccharides that have prebiotic potential as a functional food ingredient. FOS are composed of linear chains of fructose connected by β-2,1-links; typically, with glucose positioned at the end. Hydrolytic enzymes are unable to hydrolyse the linkage in inulin or FOSs. These substances have the distinct quality of being prebiotics.33 They are broken down by intestinal bacteria into L-lactate, CO2, H2, and SCFA, among other metabolites. To improve host health, the goal of FOS is to specifically encourage the growth and/or activity of one or a few beneficial bacterial species in the colon. One glucose (G) moiety and two to sixty fructose (F) moieties, such as those found in 1-kestose (1G, 2F), nystose (1G, 3F), and fructofuranosyl nystose (1G, 4F), are connected by (2–1)/ (2–6) glycosidic linkages.34 Typically, the sweetness of FOS decreases when DP rises. FOS lowers cholesterol, phospholipids, and fatty acid levels and improves the absorption of minerals like Ca+2, K+ and Mg+2.5 It has been discovered that two cultivated plants that are good providers of FOS are yacon and jerusalem artichokes.35 It is reported to have at least 4 g/day but preferably 8 g/day of FOS to show significant elevation in Bifidobacteria spp. in the human gut.36
Xylose-oligosaccharides (XOS)
Nondigestible oligosaccharides, or XOS, are produced from xylan-rich compounds and are short (DP 2–10) oligosaccharides linked together by β 1,4-linkages.37 Additionally, some researchers recommend considering compounds of xylose DP up to 20 as XOS.29 XOS are oligomers with an arabinose unit, uronic acid, or acetyl group as a replacement for the 1,4-linked xylose backbone. The degree of polymerization, the degree of substitutions (ratio of arabinose to xylose), and the links of XOS vary greatly. The kind of substitutions, how often they occur, and the molecular weight distribution on the xylan backbone determine the physical and biological properties of XOS.38 XOS is generated by the physical or chemical hydrolysis of lignocellulosic biomass and the enzymatic hydrolysis of plant biomass, and is used as a sweetener in food additives.39 Because XOS resists hydrolytic enzymes and promotes the growth and stimulation of gut bacteria, primarily Bifidobacterium spp., it demonstrates prebiotic qualities.40 Lignocellulosic biomass, such as agro-industrial wastes, is a sustainable source for XOS manufacture for industrial applications.39
Arabinoxylo-oligosaccharides (AXOS)
The AXOS are a class of oligosaccharides that can be derived from agro-processing wastes is characterized by a β-(1 → 4)- xylose backbone with arabinose residues attached to O-2 and/or O-3 positions and some arabinose esterified at O-5 with ferulic acid.41 Arabinoxylans, upon enzymatic treatment, will yield AXOS with different average degrees of polymerization and substitution, which may exhibit different prebiotic properties.42 AXOS has a DP of up to 20, with DP 2-6 potentially having the most prebiotic potential.43,44
Mannan-oligosaccharide (MOS)
The MOS class is derived from hemicellulosic polysaccharides found in plant cell walls and seeds.45 Linear mannans and glucomannan both have manno- and glucopyranose moieties linked together with similar linkages; however, the side chains in each of these two MOS are α-1,6-linked galactopyranosyl units, and the MOS are referred to as “galactomannans” and “galactoglucomannans,” respectively.29
Cello-oligosaccharides (COS)
Recently, a relatively new class of nondigestible oligomers called COS has been investigated. These oligomers have D-glucose units with β-1,4 connections.46 COS fall under the NDO category of oligosaccharides and are water-soluble and nondigestible to the human gut due to their linear chain of D-glucose coupled with a β-1,4-glycosidic bond present with a low DP (~6).46 While the longer COS produce insoluble cellulose material, the shorter COS, or disaccharides, are water soluble and include three (cellotriose) to six (cellohexose) glucose monomer units. COS enhances the growth and development of gut microorganisms, particularly Lactobacillus and Bifidobacterium species.47 Plant biomass contains cellulose, the most prevalent polysaccharide in nature, which serves as a natural source for the production of COS.29 Probiotic strain growth promotion was investigated using COS, and results indicated up to 4.1-fold increase in cell density for L. rhamnosus, L. lactis, L. paracasei, and Clostridium butyricum compared to inulin, trans-galacto-oligosaccharides, and cellobiose, indicating that COS works as a helpful functional prebiotic carbohydrate. These results point to new understandings of COS commercial use and industrial production.6
Malto- and iso-maltooligosaccharides
The α-1,4-bonds, which hold glucose moieties together in maltooligomers, are produced by amylolytic and pullulanase enzymes from polymeric carbohydrates like starch and glycogen. Isomaltooligosaccharides (IMO) are made up of isomaltose/triose/tetraose/pentaose, panose, nigerose, and kojibiose oligomers that contain α-1,6- and α-1,2-/1,3/1,4 bonded glucose. They are produced by starch-hydrolyzing enzymes and then transglycosylated by α-transglucosidase enzymes.34 As a naturally occurring component of many fermented foods and sweets, including sake, soybean sauce, and honey, IMO is made up of glucose monomers connected by α-1,6 (and rarely α-1,4) glucosidic linkages.48
Isomaltulose is a natural component of honey and sugar cane. Isomaltulose (6-O-α-D-Glucopyranosyl-D-fructose) is a disaccharide that is especially useful as a non-cariogenic substitute for sucrose.49 Bananas, rye, dragon fruit, onions, chicory root, garlic, asparagus, barley, wheat, tomatoes, leeks, and Stevia rebaudiana are among the fruits and vegetables from which this chemical is isolated.50,51 Isomaltotriose or α-D-glucopyranosyl (1→6) (1-6)-α-D-glucopyranosyl-D-glucose encourages the development of gut-dwelling good bacteria. The glucosyltransferase enzyme displaces the D-glucosyl groups during the production of D-glucosyl oligosaccharides.35
Specific fruits and vegetable wastes and their prebiotic constituents
Potential prebiotics can be found in many fruits and vegetables. This section presents commodity-specific examples, focusing mainly on fractions that are often discarded as waste. To alter functionality, future prebiotic molecules may potentially undergo structural or chemical modification using oxidation, high-pressure, acid, enzyme, and sonication treatments. Furthermore, novel benefit profiles may be created by combining distinct prebiotic combinations in optimized mixtures.52 Figure 2 illustrates the major bioactive molecules sources of fruit and vegetable waste.
Fruits
Fruit wastes contain proteins, fibers, phytochemicals, flavor compounds, polysaccharides, etc., which are utilized in the making of high-value products such as medicines, food items, and cosmetics.
Fruit wastes and their prebiotic benefits
Citrus fruits
Oranges, grapefruit, lemons, and mandarins constitute the majority of industrialized crops. Albedo, or the interior of the mesocarp, and flavedo make up the shell and refer to the white, spongy, cellulose tissue that is thought to be the primary source of dietary fiber in shells. Using HPLC-DAD, several phenolic and flavonoid chemicals, including coumaric acid, tangerine, and nobiletin, have been screened in orange peels.53 Further, Czech et al.54 found that all citrus fruits’ peels contained much more phenolic components than their pulp.
Apple
Apple peel, pulp, and seeds make up 25–30% of the total fruit. These are rich sources of fiber (4.4-47.3 g/ 100 g pomace), and the ratio of soluble to insoluble fibers varies depending on the types of apples used to obtain pomace. Along with fermentable carbohydrates, flavonoids and other antioxidants present in these discards can influence prebiotic action. A study on three commercial probiotic strains (L. rhamnosus, L. casei and B. lactis) showed that a small percentage (2%) of powdered apple, banana, and mango peels could be effectively used as prebiotics to improve lactic acid bacteria.55
Berries
Blackberries, blueberries, strawberries, raspberries, red, white, and black currants, sea buckthorn, and other berries are a few of the popular berries and the berry pomace or press cake typically includes seed, stem, and skin pieces, significantly rich in dietary fiber. Dietary fiber comes in the forms of inulin, cellulose, hemicellulose, pectin, and lignin.56 Importantly, polyphenols attached to the polysaccharides showed prebiotic activity. For example, cranberries are rich in anthocyanins, p-coumaric acid and quercetin, which may play a vital role in prebiotic mechanisms.57
Pomegranate
Chemical analysis of dried pomegranate peels showed about 11% crude fiber, 29% neutral detergent fiber, 19% acid detergent fiber, 15% cellulose and 10% hemicellulose.58 Bioactive phenolic acids like punicalagin, ellagic acid glucoside and flavonoids like anthocyanins are found in pomegranate peel. Punicalagin can be metabolized by lactic acid bacteria and transformed into ellagic acid and further to urolithins, and easily uptaken by the intestine.59 Studies have also shown that pomegranate peel extracts outperform other wastes in terms of their soluble phenolic content and antioxidant activity.60
Watermelon
Roughly 40% of the watermelon’s mass is its rind. Insoluble fiber is the main kind of fiber present in watermelon rinds. Since watermelon has a high pectin concentration, it has a strong potential to function as a prebiotic.61 The impact of freeze-dried watermelon peel powders (WPP) and banana peel powders (BPP) on the survival, growth kinetics, and bile salt tolerance of L. acidophilus and Lactiplantibacillus plantarum was studied, concluding with beneficial effects.62 Similarly, the polysaccharide extract of yellow watermelon peel was able to significantly raise L. rhamnosus and B. bifidum counts.63
Kiwi
Bagasse, skin, and seeds are essentially obtained as waste from kiwi processing. The dietary fiber from the pomace leftovers of kiwis was found to be around 26%.64 The fruit peel contains more polyphenols than its flesh: catechin, quercetin, epicatechin, epigallocatechin and p-coumaric acid are a few of them, which produce better-absorbing metabolites like hydroxyphenyl acetic acid and hydroxyphenyl propionic acids when metabolized by gut microbiota.57,65
Pear
Pear pomace contains 44 to 79% fiber on a dry weight basis, and is rich in polyphenols apart from organic acids and triterpenes.66 Arbutin, chlorogenic acid, catechin, quercetin, kaempferol, various hydroxycinnamoylmalic acids and their ethyl esters, hydroxycinnamoyl malates, procyanidins and triterpenes are the few reported compounds found in the peel of the pear at levels higher than in the pulp.67
Grape
The total amount of dietary fiber found in the grape pomace was 78% (d.w.), of which 9.5% was soluble and the remaining 68% was insoluble. Grape pomace is a rich source of phenolic compounds, which are available for the gut microbiome and produce 3,5-dihydroxybenzoic acid and phenylacetic acid, which can be absorbed into the body.68 LC-MS/MS analysis showed that grape pomace is rich in phenolic compounds like resveratrol, anthocyanins (cyanidin, delphinidin, malvidin, and petunidin derivatives), flavonols (quercetin, laricitrin, syringetin, and myricetin glycosides), and flavan-3-ols (catechin/epicatechin and their procyanidin oligomers).69
Mango
Processing mangos to remove pulp for juice and other value-added products often results in 35–60% of waste, which contains dietary fiber.70 Roughly 50% of the total dietary fiber content is found in mango peels and pulp. Hydrolysis with pectinase on these substances produces reducing sugars and releases phenolic compounds, which provide prebiotic properties as lactic acid bacteria are capable of conducting polyphenol hydrolysis to produce smaller molecules with antibacterial properties such as cinnamic, vanillin, ferulic, and p-coumaric acids, luteolin, and kaemferol.71 Importantly, for every combination of probiotic and enteric strains examined, the prebiotic activity score of mango peel powder was higher than that of commercial prebiotic maltodextrin.72
Dragon fruit
The unusable portion from cacti makes up 25–30% of the overall fruit mass and is rich in natural colors, phytochemicals, and dietary fiber. Pectin, cellulose, hemicellulose, and simple sugars are the main types of fiber present in the leftovers.73 Phenolic compounds (betacyanins, flavonoids, and phenolic acids) present in dragon fruits are known to stimulate the growth of probiotic bacteria, and they may act as prebiotics.74
Jackfruit
The fruit of the jackfruit is made up of 25.35% rags, 18-25% seeds, 30% pulp, and 31.0% skins.75 Jackfruit rags contain a substantial quantity of cellulose (20.5%), protein (0.3-0.6%), reducing sugar (1.7-4.50%), total sugar (1-6.8%), and pectin (1.10-1.63%), while the skin contains 8.94%-15.14% of pectin.76 The unused parts of jackfruits are also known to be a source of β-carotene, polyphenols and chlorogenic acid, which have proven prebiotic activity.77
Passion fruit
In passion fruits, the peel and seeds account for 50% of the weight of the fruit and peels from passion fruit have a good amount of cellulose, hemicellulose and pectic substancesAccording to Bussolo de Souza et al.,78 orange bagasse and passion fruit peels have demonstrated excellent fermentation profiles that enhance bacterial diversity and SCFA production. Further, neochlorogenic acid is a phenolic acid found abundantly in passionfruit, which has a stimulatory role in the growth of gut bacteria.79
Date palm
The byproducts of the date processing industries include date seed, date pomace, and discarded dates. Date seeds are a rich source of cellulose, hemicelluloses, lignin, protein and fatty acids such as oleic acid, date processing wastes contain fermentable fibers and sugars, The dietary fiber content of date seed is estimated to be 39.89% and exhibits prebiotic activity.80 Using isolated polysaccharides (date flesh polysaccharide and date seed polysaccharide), the chemical structure, functional characteristics, prebiotic potential, and metabolite production of L. plantarum and B. animalis subsp. lactis were examined. On B. animalis, date seed polysaccharide had a significant effect on proliferation (9.32 ± 0.05 log CFU/g), and so in the case of L. plantarum (8.66 ± 0.06 log CFU/g); additionally, B. animalis generated increased levels of SCFA in date seed polysaccharide cultured cells.81
Others
Banana peel is a good source for polysaccharides (pectin, cellulose and hemicellulose), and the prebiotic activity of pectin and cellulose oligosaccharides from banana peel is well-known.82 Similarly, guava wastes include peel, seed, and pulp leftovers and are rich in cellulose with proven prebiotic activity.83 Also, in pineapples, the peel makes up around 29–40% of all waste, and is rich in ferulic acid, gallic acid, epicatechins, and catechins, all of which can be utilized as potent antioxidant components.84 Many phenolic chemicals, including gallic acid, catechin, epicatechin, anthocyanins, kaempferol, and isoquercitrin, are present in pineapple and banana peels.84,85 The prebiotic action of peels from bananas, oranges, and watermelons has been demonstrated, and banana peels had the highest growth boost of L. casei.86
Vegetables
Vegetable wastes include peels, pomace, rinds, cores, leaves, and scraped portions. These are rich in protein, polyphenols, polyunsaturated fatty acids, enzymes, essential oils, and other phytochemicals, which are used in biocomposites, packaging, food, and the pharmaceutical sector.
Vegetable waste and its prebiotic benefits
Cabbage
Around 30% of cabbages are wasted, but this fraction is rich in carbohydrates and dietary fibre, which constitute a significant component of the waste. Further, prebiotic sugars, phenolic compounds like kaempferol, quercetin, and anthocyanin (red cabbage), are present in cabbages; these compounds have proven prebiotic activity.87
Carrot
Carrot pomace is high in fiber, with cellulose accounting for 51.6% of the total fiber content, followed by lignin (32.2%), hemicellulose (12.3%), and pectin (3.88%).88 Current studies have focused on the polyphenols associated with dietary fiber, exhibiting prebiotic and antioxidant activities. Interestingly, the dietary fiber was found to include 42 different polyphenolic components, including p-coumaric acids, caffeic acids, and p-hydroxybenzoic acids.89
Cauliflower
The amount of dietary fiber in each portion of the cauliflower varies: the florets have 40% dietary fiber, the upper stem has 48%, and the lower stem has 65% (all in d.w.). Cauliflower’s upper stems and florets possess 50–60% pectic components, but the lower stem portion contains 51% cellulose and 21% hemicellulose.90
Onion
There are plenty of nutritional fibers in varied proportions throughout the onion’s layers, and compositions are known to vary significantly with variety. It is also reported that polyphenols like rutin and quercetin present in onions may contribute to prebiotic activity.91
Potato
Potato wastes, primarily peel wastes, contain significant levels of bioactive substances and dietary fiber. Further, potato peels contain about 50% of the phenolic compounds in potatoes, and rutin, quercetin, and anthocyanins are of prebiotic importance.92
Tomato
Roughly 50% (d.w.) of dietary fibers are found in tomato pomace.93 Dietary fiber extracted from dried tomato peels had 83% of total dietary fiber and a 10:1 ratio of soluble to insoluble dietary fibers.94 In addition, phenolic compounds, lycopene, and carotenoids in tomatoes support prebiotic activity.
Artichokes
Around 80-85% of the material is discarded.95 The byproducts of artichokes are stems, bracts, and leaves, and these byproducts are high in inulin, a polysaccharide that can be broken down by digestive enzymes.96 The essential oils, sesquiterpenes, and caffeoylquinic acids present in artichokes exert a positive effect on the gut microbiota.97
Broccoli
The stems and leaves of broccoli are underutilized and often discarded without valuing the potential of functional ingredients present in it. Stems are rich in carbohydrates compared to leaves; more of these carbohydrates are insoluble dietary fiber. On storage, insoluble dietary fiber composition increased by 13% but soluble dietary fiber remained unaffected (Schafer J). More SCFAs (acetic acid, propionic acid, butyric acid, isovaleric acid, isobutyric acid, isocaproic acid, caproic acid, and valeric acid) were discovered to be formed by broccoli leaf fiber than by stems and inflorescences, showing its superior prebiotic potential.98 Glucosinolates are a group of specialised metabolites present in vegetables of the Brassicaceae family, which have no biological activity, but upon fermentation by gut bacteria, bioactive isothiocyanates, nitriles, thiocyanates, epithionitriles, and oxazolidinethiones etc. are formed, which have enhanced bioactivities.99
Kale
The prebiotic data on leafy vegetables are scarce. The identified prebiotic carbohydrates in kale are sugar alcohol (sorbitol, mannitol), simple sugars (glucose, fructose, sucrose) and hemicellulose (arabinose, mannose, xylose).100
Others
Asparagus byproducts mainly contain dietary fibers (62-77%)101 oligosaccharides can be extracted from sugar beet molasses, soybean whey, and saccharified vegetable starch derived from peas, beans, and lentils can also be utilized as prebiotics.102 Arugula, Brussels sprouts etc., also account for fermentable fibre content.103
Residue of fruit and vegetable wastes with rich nutritional profiles can be incorporated into food formulations at different levels (Tables 1 and 2). Additionally, the rich content of prebiotic ingredients in fruits and vegetable wastes can be used in developing novel functional food supplements by incorporating probiotics in novel food compositions (Tables 3 and 4).
Table 1: Recent studies highlighting the potential of utilization of fruit waste fractions in different food formulations
| Plant Source | Prebiotic significance | Pre-processing method | Food product |
Level of incorporation (% weight basis) | Bioactivity | Key observations |
|
Apple pomace (Malus domestica) |
Improves polyphenolic content, viable cell count, and antioxidant activity. | Lyophilization | Fermented soy milk |
Antioxidant activity | 24% pectin yield with a degree of esterification of 38%
Enhances the functional properties of fermented soy milk.104 |
|
| Apple peel
(Malus domestica) |
Enhances the growth of lactic acid bacteria | Homogenization | Functional ingredients | Antioxidant activity | Used as a thickening agent due to its higher fiber content, good water-holding capacity, and as a source of natural fiber105 | |
|
Apple peel (Malus domestica)
|
Natural source of high-quality antioxidant and bioactive compounds
Increases the viable count of probiotics. |
Lyophilization | Yogurt | Antioxidant activity |
No modification in texture and flavor Good functional and physical characteristics of yogurt106 |
|
|
Apple peel (Malus domestica) |
Apple peel polyphenol extract (APPE) increases the acidity
Decreases the melting rate and enhances the overrun of ice cream |
Lyophilization | Yoghurt ice cream | 5% | Antioxidant activity | The fortification with APPE increases the yogurt ice cream’s hardness, rheological, and sensory attributes107 |
|
Apple pomace (Malus domestica) |
Dietary fiber, pectin
higher phytochemicals and dietary fiber delivery (6%) |
Lyophilization | Agitated type yoghurt and yoghurt drink | 1 to 3% (w/w) | Antioxidant activity |
Acts as a stabilizer and reduces sedimentation of protein aggregates pH of yoghurt decreases (2 and 3%) Lightness value decreases with increasing dosage (from 1 to 6%)108 |
| Red pitaya peel (Hylocereus polyrhizus) | Dietary fiber: pectin, lignin, cellulose, hemicellulose
Replacer of fat in ice cream |
Grinding | Ice cream | 1% | Antioxidant activity | Addition of fiber improves the overrun, rheological properties, increases nutritional properties, and higher overall acceptability of the sample109 |
| Grapefruit peel
(Cabernet sauvignon) |
Nanofibrillated cellulose reduces the digestibility of fat in the human intestine (0.30 % (w/w)) | Drying, milling, lyophilization | Ice cream | 0.2, 0.3, 0.4% | – |
Great shape retention achieved (0.2%) Increases the sensory properties (smoothness, chewiness, and mushiness) by 0.4 %110 |
| Grape pomace (Cabernet sauvignon) | Dietary fiber and prebiotic compounds: lignin, cellulose, oligosaccharide
Digestibility of chocolate spread is 25% to 84% |
Drying, grinding | Chocolate spread | 3–5%
|
Antioxidant activity | Bitter taste due to phenols (5%)
Higher impact on the product particle size111 |
| Pomegrana seeds (Punica granatum) | Dietary fiber: lignin, cellulose
Pomegranate seed flour (PSF) increases the intake of protein and dietary fibers (5%) Maintains sensory acceptability |
Drying, milling | Bread | 5, 7.5 & 10% | Antioxidant activity |
Wheat flour PSF bread shows good sensory and technological quality (5%) There are no changes observed in the water activity of bread (5% PSF) The firmness of bread increases with increasing PSF (5%, 7.5%, 10%)112 |
| Pomegrana seeds (Punica granatum) | Improve metabolite levels in intestinal bacteria | Boiling | Value added polysaccharide | – | – | Jack fruit peel powder does not alter the molecular weight during upper gastrointestinal digestion113 |
| Banana peel | Probiotic substance: oligosaccharides – Enhanced growth of lactic acid bacteria | Fermentation | Value added polysaccharides | – | Antioxidant activity |
Pectinase is used for the preparation of pectin oligosaccharides high enzyme cost114 |
|
Banana peel (Musa paradisiaca) |
Prebiotics: Pectin | Drying | Salad cream | 2% | Antioxidant activity |
Alternative sources of pectin and fat replacers in food products Decreases in viscosity and lightness value due to the incorporation of banana peel powder into salad cream (30 %)115 |
|
Mango peel powder (Mangifera indica) |
Dietary fiber: Better prebiotic score than
commercial prebiotic maltodextrin |
Drying, grinding | Synbiotic products | – | Antioxidant and antiproliferative activity | Increased total phenolic flavonoid content, antioxidant capacity, and water and oil holding capacity.116 |
|
Mango peel powder (Mangifera indica) |
Dietary fiber: Cellulose, hemicellulose | Bleaching, drying | Instant drink | 2% (w/v) | Antioxidant and antiproliferative activity | Sensory characteristic decreases during storage117 |
| Pineapple pomace
(Ananas comosus) |
Dietary fiber: lignin, cellulose, hemicellulose | Steaming, lyophilization, milling | Vienna sausages | – | Antioxidant and antidiabetic activity |
Decrease in nitrites, moisture, shear strength, and shrinkage Increase in carotenoids, antioxidants, polyphenols118 |
|
Feijoa peel (Acca sellowiana) |
Dietary fiber: lignin, cellulose, hemicellulose- alternative source of bioactive ingredients. | Steaming, drying | Flour | – | – | Flour-containing mesocarp shows a higher water-holding capacity than the flour-containing mesocarp and epicarp119 |
| Melon peel, seed
(Cucumis metuliferus) |
Dietary fibre | Drying, grinding | Fermented milk | Whole fruit powder – 0.5%,
Peel powder – 0.7%, Seed powder – 1% w/v |
Antioxidant activity |
Low pH (1%) Changes in sensory properties due to natural oxidation, dehydration, microbial activity, and enzymatic browning during storage120 |
|
Melon peel (Cucumis melo L. inodorus) |
Dietary fiber and phenolic compound – positively impact the gut microbiota diversity
|
Milling, drying | Melon juice powder | 2% (w/v) | Antioxidant activity | Good accessibility index of sugars (85–90%), organic acids (90–93%), phenolics (80%), and antioxidant activity (54–76%)121 |
| Papaya peel powder | Dietary fibre | Drying | Yogurt | 10% | Antioxidant and antidiabetic activity | Papaya peel powder acts as a functional ingredient and yields bioactive components in the Greek yogurt matrix122 |
|
Rambutan peel (Nephelium lappaceum) |
Dietary fiber – breakdown leads to the production of an enzyme that metabolizes fiber sources and produces lactic acid, which maintains the intestinal health | Drying, grinding | Plant-base rice milk | – | Antioxidant activity |
Enhance food waste utilization Enhanced shelf-life of up to 20 days and flavor, oil-holding capacity, antioxidant activity, and the color pigments of flavonoids and carotenoids123 |
|
Mangosteen peel (Nephelium lappaceum) |
Increased dietary fibre (58%)
|
Drying, grinding, blending | Rice sausage | 17% – outer pericarp,
48% – inner pericarp |
– | The cardamon oil-treated mangosteen peel was added as a supplement in fermented rice sausage to extend shelf life up to 21 days124 |
| Grapes, pomegranate seeds (Punica granatum L), rosehips | Dietary fiber – Increases antioxidant activity | Grinding, sifting | Eriste (Turkish noodle) | 10% | Antioxidant activity | Sample with pomegranate seed powder obtained the highest appreciation from a sensory point of view125 |
Table 2: Recent studies highlighting the potential of utilization of vegetable waste fractions in different food formulations
| Plant Source | Prebiotic
significance |
Pre-processing method | Food product | Level of incorporation (% weight basis) | Bioactivity | Key observations |
|
Carrot baggase (Daucus carota) |
Fructo-oligosaccharide | Fermentation | Natural functional beverages | – | – | High nutritional value contributes to improving human health126 |
| Carrot pomace
(Daucus carota) |
Dietary fiber: pectin, lignin, cellulose, hemicellulose | Lyophilization | Agitated yogurt | 1 & 2% | Antioxidant activity |
Strawberry flavor was added to improve the color and smell of the sample Increases the water-holding capacity of yogurt and has high consumer acceptance (1%) Decreases in apparent viscosity (2%)127 |
| Carrot pomace
(Daucus carota) |
Dietary fiber: pectin, lignin, cellulose, hemicellulose | Drying, grinding, sifting | Donut | 6.45% | Antioxidant activity | A low-fat donut with acceptable physical, image, and sensorial properties (6.45 %)128 |
| Sweet potato peel
(Ipomoea batatas) |
Dietary fiber – Promotes bowel movement, prevention of constipation | Milling, sieving | Bread | 10% | – |
Orange-flushed sweet potato peel powder of 10% and haricot bean flour of 20% show better consumer acceptability The protein content of composite flour increased (11.25 to 17.4 g/100g)129 |
|
Sweet potato root powder (Ipomoea batatas) |
Resistance starch, nystose, ketose
Sweet potato flour (SPRF) of 20 g/l had an average positive prebiotic activity score (0.11–0.55)
|
Blanching, drying, grinding | Ingredients for functional foods or dietary supplements | – | – | The factors affecting the outcome of host health are diet composition, types of microorganisms forming the gut microbiota, and individual host response130 |
|
Potato peel (Solanum tuberosum) |
Dietary fiber, inulin or fructooligosaccharides
20- 30% prebiotics resulted in a considerable increase in the colon content of Lactobacillus sp and Bifidobacteria sp. |
Drying, grinding, sieving | Cupcake | 5, 10, 15, 20% | Antioxidant activity |
Increase in the hardness of cupcakes Better sensory attributes (15 %)131 |
Table 3: Recent studies on utilization of fruit waste fractions in different food formulations along with the addition of probiotics
| Source and part used | Food product into which it was incorporated | Level of incorporation of prebiotic source (% weight basis) | Probiotic culture added and level | Key observations | Probiotic viability and functionality |
| Rosehip seed powder (Rosa canina L.) | Probiotic yoghurt | 1%, 2%, and 3% RSP | Yoghurt starters (2.5 mL/kg milk)- Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus
Probiotic bacteria (0.1% w/w) –L. acidophilus LA-5 and B. animalis subsp. lactis BB-12 |
Improved syneresis, firmness and adhesiveness and reduced parameters like aftertaste, taste, consistency and appearance
Improved antioxidant activity and total phenolic content |
Bacterial counts > 6 log CFU/mL were present in all yogurt samples132
|
| Red dragon fruit peel (Hylocereus polyrhizus) | Yogurt | 0%, 2%, 5%, and 7% (w/v) | Yogurt starter (0.02% w/v) – Lactobacillus bulgaricus and Streptococcus thermophilus | Incorporation of 7% RD peel powder gave the most noticeable antioxidant activity in yogurt
The highest sensory scores were obtained with 2% RD peel powder
|
Total viable counts in all treatments ranged from 56.64×104 to 64.23×104 (CFU/mL). The yogurts’ bacteriological status fell within the permissible range (<106 CFU/mL) for goods made from fermented milk 133 |
|
Sour orange peel (Citrus aurantium) |
Yogurt | 0.5%, 1.0%, and 2.0% | L. acidophilus, S. thermophilus, and Bifidobacterial sp. (1.5 × 108 CFU/mL) |
Viable counts (log CFU/g) of L. acidophilus, S. thermophilus, and Bifidobacteria sp. decreased from 8.25 to 6.10, 8.35 to 6.70 and 8.20 to 6.80 on 28th day 134 |
|
| Pineapple crown powder (Ananas comusus) | White finger millet vegan beverage | 1–3% w/v | Lactobacillus rhamnosus GG (LGG) NCDC 347 (6.5 log CFU/mL) | Increase in viscosity of sample beverages on increasing PCP incorporation due to the presence of 8.71% soluble dietary fibers in the powder | After 6 h of incubation, the viable cell count increased from 6.5 to 9 log CFU/ml in pineapple crown powder-added beverages 135 |
|
Pink Guava seed and pulp (Psidium guajava L) Variety- Sungkai and Semenyih |
Yogurt | 1:100
(w/v) of UHT-treated milk |
Probiotic strains (1g of starter culture) Lactobacillus rhamnosus ATCC 9595, Lactobacillus plantarum ATCC 14917 Lactobacillus brevis KCTC 3102 Enteric strain– Escherichia coli ATCC 25922
|
Cellulase and xylanase-treated seed and pulp increased the yogurt’s texture by 22.28–28.05% for hardness and 67.6–68.6% for stickiness
Adhesiveness ranged from 84.11 to 86.44%. Enzymatically treated byproducts of processed guava caused the growth of Lactobacillus species |
Enzymatic treatment on yogurt increased the probiotic activity of Lactobacillus spp. up to 8.05 log CFU/mL 136
|
| Mango peel powder (Mangifera indica) | Freeze-dried yogurt | 2% (w/v) | Mixed starter culture (2%)- Lacticaseibacillus casei (431®), Lacticaseibacillus rhamnosus (LGG®) and Bifidobacterium subsp. Lactis (Bb-12®) | Increased fat, ash, and protein contents | The net count of probiotics in yogurt fortified
with MPP increased by at least 1 log CFU/g after 4 weeks of refrigerated storage 137
|
| Cavendish banana peel powder (Musa acuminta) | Freeze-dried yogurt | 2% (w/v) | Mixed starter culture (2%) – Lacticaseibacillus casei (431®), Lacticaseibacillus rhamnosus (LGG®) and Bifidobacterium subsp. Lactis (Bb-12®)
|
Increased fat, ash, and protein contents in inoculated samples |
The net count of probiotics in yogurt fortified with BPP increased by at least 1 log CFU/g after 4 weeks of refrigerated storage 137 |
|
Cranberry pomace powder (Vaccinium oxycoccus) |
Yogurt | 2%, 3%, and 4.5% (w/v) | Starter culture (5 mL/100 mL)-Streptococcus thermophilus and Lactobacillus bulgaricus | Higher total solids content (13.84% to 17.92%) with the increase in CPP
Protein content increased from 5.83% to 6.01% The dietary fiber level rose by around 1.45% with 2% CPP incorporation High dietary fiber content and better water holding capacity at 4.5% CPP incorporation |
Viable counts of probiotic Lactobacillus spp. in yogurt was >9 log CFU/g 138
|
| Cornelian Cherry peel powder (Cornus mas L.) | Probiotic Icecream | 3, 6, and 9% | Bifidobacterium lactis (Bl-04) 1% v/v of 109 CFU/mL of bacteria cells | The highest viscosity of ice-cream samples was obtained at 9% incorporation of peel powder
Water absorption ability in peel powder enhanced the melting resistance and viscosity of ice cream |
Increment of 6% and 9% peel powder in ice cream increased the viability of B. lactis during 120-day storage and after gastrointestinal simulation
Polyphenol compounds increase the survival of B. lactis 139 |
| Yellow watermelon peel (Citrullus lanatus) | Peel extract | 1% w/v | Lactobacillus rhamnosus and Bifidobacterium bifidum. (1ml of starter culture)
|
Higher amount of indigestible non-starch polysaccharides with WPP incorporation
WPP can withstand the artificial acidic conditions of gastric juice in the stomach and digestion by α-amylase in the small intestine |
WPP stimulated the growth of L. rhamnosus from 8.66 to 9.74 (log CFU/g dry extract) within 48 h
B. bifidum growth increased from 9.00 to 9.90 (log CFU/g dry extract) within 48 h 140 |
| Honeydew peel (Cucumis melo) | Peel extract | 1% w/v | Lactobacillus rhamnosus and Bifidobacterium bifidum. (1ml of starter culture) | Honeydew peel extract showed the least resistance to artificial human gastric juice and α-amylase digestion at 13.80% and 13.68% of the polysaccharides are being hydrolyzed, |
Peel extract stimulated the growth of L. rhamnosus from 8.65 to 9.82 (log CFU/g dry extract) within 48 h 140
|
| Guava seed powder (Psidium guajava L.) | Yogurt | 1, 2 and 3% (w/v) | ABT-5 culture (0.025%) Streptococcus thermophilus, Lactobacillus acidophilus, and Bifidobacterium BB-12 | Increase in GSP showed an increase in crude fiber, acidity, viscosity, total phenolic content, and antioxidant activity
A decrease in yogurt syneresis was also noted |
Viable counts of probiotic Streptococcus thermophilus, Lactobacillus acidophilus and Bifidobacterium BB-12 reduced from 8.92 to 8.13, 8.87 to 7.99, 8.76 to 8.00 (log of CFU/g) 141
|
|
Apple peel (Malus domestica) Variety- Red Delicious, Golden Delicious, Royal Gala, Amri, and Gacha |
Yogurt | 1%, 2%, 3%, 4% and 5% (w/v) | Starter culture-Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus salivarius subsp. thermophilus and Probiotics strains- Bifidobacterium lactis BB-12 and Lactobacillus acidophilus La-5
|
Loss of viability in yogurts incorporated with 1%, 2%, 3%, 4% and 5% (w/v) of apple peel powder were 1.67, 1.48, 1,24, 0.79 and 0.47 log cycles, respectively, after 21 days 142
|
|
| Pomegranate peel (Punica granatum) | Yogurt | 0.5% w/v | L.acidophilus and Bifidobacterium bifidum |
Tannin-free PPP enhanced the viability of probiotic culture under gastrointestinal conditions All probiotic cultures were maintained at counts around 8 log CFU /g in stirred yogurt supplemented with pomegranate peels after 21 days 143 |
|
| Pineapple peel and pomace (Ananas comusus) | Yogurt | 1% w/v | Streptococcus thermophilus ASCC 1275 and L. delbrueckii spp. bulgaricus Lb1466 1 % (v/v) L.acidophilus, L. casei and L. paracasei monocultures at 1 % (v/v) |
Enhanced growth of L.acidophilus (8.13–8.71 log CFU/g) and L. casei, L. paracasei (8.12–8.72 log CFU/g) Probiotic and proteolytic activity of starter cultures increased on the incorporation of pineapple waste powder 144 |
Table 4: Recent studies on utilization of vegetable waste fractions in different food formulations, along with the addition of probiotics
| Source and part used | Prebiotic significance | Food product into which it was incorporated | Level of incorporation of prebiotic source (% weight basis) | Probiotic culture added and level | Key observations | Probiotic viability and functionality |
| Pumpkin peel powder (Cucurbita maxima) | Peel powder | Yogurt | 2%, 4% (w/v) | Lactobacillus delbruckii subsp. Bulgaricus and Streptococcus thermophilus, (50 U culture per 250 L of milk)
|
The springiness and adhesiveness of the yogurt decreased with the increase in the pumpkin peel powder levels
Increased antioxidant activity was observed in supplemented samples 145 |
– |
| Broccoli leaves(Brassica oleracea L. var Italica, cv. Parthenon) | Leaves | Antidiabetic beverage | 1% w/v | Lactiplantibacillus plantarum (CECT 749) (6 log CFU/mL) | HHP (200 MPa, 10 min) preserved the highest concentration of sulforaphane (4.38–8.82 mg/L)
High stability of antidiabetic potential after refrigeration |
Levels of LAB in the broccoli leaves increased to 8.37 log CFU/g at the end of fermentation146
|
| White potato powder (Solanum tuberosum) | Peel powder | Yogurt | 2% potato peel flour in 1 L of rehydrated skim milk (10% w/v). | Streptococcus thermophilus and Lactobacillus delbrueckii subsp. Bulgaricus
(107 CFU/mL) |
Prebiotic activity score (0.74) was significantly higher Higher consumption of prebiotic source by the lactic acid bacteria, indicated better prebiotic capacity 147
|
|
| Beetroot pomace (Beta vulgaris) | Beetroot pomace (BP) | Natural Colorants | – | Levilactobacillus brevis KKP 804, Limosilactobacillus fermentum KKP 811, Lactiplantibacillus
plantarum ATCC 4080 (1 × 109 CFU/mL) |
Dry matter indicated storage properties of dried substances and ranged from 2.9–12.1 for beetroot pomace
No changes in the red/violet pigment content of pomace |
Concentration of Levilactobacillus brevis, Limosilactobacillus fermentum and Lactiplantibacillus plantarum in fermented beetroot pomace was 2.93, 2.46, 2.82 log CFU/g 148 |
Critical considerations in technology adoption
The existence of beneficial components in fruit and vegetable waste has been recognized for some time, but their potential for prebiotic uses has only recently attracted interest. Notwithstanding this advancement, numerous facets of employing these substances remain unexamined. A comprehensive investigation and understanding are essential for the complete development and implementation of this approach. For instance, limited research has evaluated the long-term impacts of consuming such fractions. The information about the synergistic or antagonistic effects of dietary habits with these substances is likewise scarce. For example, particular bioactive chemicals or their combinations may be inappropriate for certain chronic illnesses, age groups, or demographics, hence increasing potential dangers for consumers.
A primary worry is the prevalent application of herbicides, insecticides, fungicides, and fertilizers in the cultivation of fruits and vegetables. These chemicals and their residues may be present in higher levels in such waste fractions. Typically, fruits and vegetables suffer lengthy handling and supply chain operations, sometimes leading to quality deterioration such as rotting, shriveling, over-ripening, and weight loss. To mitigate these effects, the fruit and vegetable sector predominantly uses synthetic chemicals such as chlorine dioxide, nitric oxide, 1-methylcyclopropane, and salicylic acid. These compounds and their residues may come along with waste fractions and then present heightened dangers to human health, especially for at-risk populations. Similarly, waste fractions may contain anti-nutritional components, including saponins, alkaloids, tannins, phytates, oxalates, and heavy metals (e.g., lead, chromium, cadmium), which can negatively impact human metabolism. Prevalent anti-nutrients comprise amino acids, alkaloids, cyanogenic glycosides, tannins, phytates, and inhibitory enzymes. Furthermore, natural toxins such as mycotoxins, aflatoxins, and fumonisins can build up in byproducts and pose a risk to human health if ingested. This is of particular concern when such waste fractions reach processing facilities after a significant time delay since their occurrence. Apart from biological and chemical hazards, waste fractions may also come along with physical hazards that require attention.
Consumer acceptance is another significant element, as the concept of utilizing waste or byproducts for prebiotic compounds may encounter reluctance. Sensory concerns, including alterations in color, flavor, aroma, texture, and mouthfeel, may further dissuade consumers. Preserving the sensory attributes of fortified or enhanced goods is crucial for the optimal integration of prebiotic chemicals sourced from waste.
Future directions
The future of turning fruit and vegetable processing wastes into prebiotics depends on increasing market potential, enhancing customer awareness, ensuring safety assurance, promoting technological acceptance, and fostering creative product development. To ensure broad adoption, the future direction of utilizing fruit and vegetable processing wastes as prebiotics heavily emphasizes the use of scalable and reasonably priced technologies. Through enhanced extraction and biotechnological methods, including fermentation and enzymatic treatment, prebiotic fibers derived from these wastes will exhibit improved yield, purity, and functional characteristics. To guarantee consumer trust and compliance, safety and legal requirements will be crucial. Consumer safety is still crucial, and strict guidelines are necessary to ensure the efficacy and safety of these prebiotic substances.
Market acceptability will be driven by increasing consumer awareness of sustainability, health benefits, and ethical food production practices. Additionally, by combining innovation, safety, and consumer involvement, prebiotics made from fruit and vegetable waste will be positioned as essential components of circular economy and sustainable nutrition models, which in turn increases the profitability of agricultural supply chains and creates employment. It is anticipated that functional foods, drinks, and supplements utilizing prebiotic-rich recycled ingredients would be included in new product formulations. The worldwide prebiotics market’s explosive growth and sustainability factors are driving its potential, which encompasses prospects in both developed and developing nations.
Significance in the context of the circular food economy
In line with the United Nations’ Sustainable Development Goals (SDGs), the circular economy model provides a sustainable way to handle produce waste by turning it into valuable products like prebiotics. Prebiotics can be made from the bioactive components found in fruit and vegetable waste, which is typically thrown out after harvesting, processing, or consumption. This waste is rich in fibers, polyphenols, and oligosaccharides. By utilizing techniques like enzymatic hydrolysis and microbial fermentation, this transformation produces functional components for use in food and nutraceuticals and helps decrease food waste. This method contributes to numerous SDGs by improving food systems and advocating for inexpensive, nutrient-rich meals, aligning with SDG 2 (Zero Hunger) and SDG 3 (Good Health and Well-being) by promoting gut health and decreasing the likelihood of chronic diseases. In addition, reducing landfill methane emissions and maximizing resource efficiency helps achieve SDG 12 (Responsible Consumption and Production) and SDG 13 (Climate Action). Reducing the likelihood of trash accumulation helps achieve SDG 15 (Life on Land). It is necessary to overcome problems such as increasing production scale, obtaining regulatory permissions, and gaining market acceptability. A resilient and sustainable bioeconomy that benefits public health, the environment, and global food systems can be achieved by repurposing fruit and vegetable waste into prebiotics, with the help of correct legislation, technical innovation, and stakeholder participation. In addition to the directly linked SDGs listed above, several other SDGs are indirectly associated.
Conclusion
It is evident that a lot of waste has been generated and thrown away from fruit and vegetable processing and contains valuable bioactive and fibre materials. Considering the circularity of resources and the thrust on sustainability, generated wastes can effectively be used for different food applications. By practicing this, a tremendous amount of waste can be converted to valuable materials and the impact on the environment can also be minimized. It is interesting to note that the inedible portions of fruits and vegetables can promote gut health, given their prebiotic capabilities. Overall, the approach of valorizing fruit and vegetable discards closely associates with several prominent global mandates: ‘zero discards’, ‘resource recovery’, ‘waste-to-wealth’, ‘upcycling’, ‘biorefinery’, ‘waste management’ and ‘sustainable food processing’. Challenges exist, but these can certainly be overcome, strengthening the focus towards effective valorization of such horticultural discards.
Acknowledgement
The authors wish to thank the National Institute of Food Technology, Entrepreneurship and Management, Thanjavur (NIFTEM-T) for all the support.
Funding Sources
This work was funded by the Department of Science and Technology, Government of India through the Scheme for Young Scientists and Technologists (SP/YO/2021/2220 (G)).
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This is a review article. All relevant information has been included in the manuscript itself.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Ravikrishnan Venugopalan: Writing – original draft.
- Aditi Negi: Writing – original draft, Methodology.
- Praveena Balasubramanian: Writing – original draft.
- Sruti Chandra: Writing – original draft.
- Santhoshkumar Paramasivam: Writing – review and editing, Methodology.
- Jeyan Arthur Moses: Writing – review and editing, Supervision, Methodology, Conceptualization.
References
- Agricultural Production Statistics 2000–2022. Rome, Italy; 2023.
- Cassani L, Gomez-Zavaglia A. Sustainable food systems in fruits and vegetables food supply chains. Front Nutr. 2022;9. doi:10.3389/fnut.2022.829061
CrossRef - FAO/WHO. Guidelines for the evaluation of probiotics in food. Food and Agriculture Organization of the United Nations and World Health Organization. 2002.
- Yoha KS, Nida S, Dutta S, Moses JA, et al. Targeted delivery of probiotics: perspectives on research and commercialization. Probiotics Antimicrob Proteins. 2022;14(1):15-48. doi:10.1007/s12602-021-09791-7
CrossRef - You S, Ma Y, Yan B, et al. The promotion mechanism of prebiotics for probiotics: A review. Front Nutr. 2022;9. doi:10.3389/fnut.2022.1000517
CrossRef - Dong C Di, Tsai ML, Nargotra P, et al. Bioprocess development for the production of xylooligosaccharide prebiotics from agro-industrial lignocellulosic waste. 2023;9(7). doi:10.1016/j.heliyon.2023.e18316
CrossRef - Davani-Davari D, Negahdaripour M, Karimzadeh I, et al. Prebiotics: Definition, types, sources, mechanisms, and clinical applications. Foods. 2019;8(3). doi:10.3390/foods8030092
CrossRef - Zhou X, Zhao J, Zhang X, et al. An eco-friendly biorefinery strategy for xylooligosaccharides production from sugarcane bagasse using cellulosic derived gluconic acid as efficient catalyst. Bioresour Technol. 2019;289. doi:10.1016/j.biortech.2019.121755
CrossRef - Rinninella E, Costantini L. Polyunsaturated fatty acids as prebiotics: Innovation or confirmation? 2022;11(2). doi:10.3390/foods11020146
CrossRef - de Araújo FF, de Paulo Farias D, Neri-Numa IA, et al. Polyphenols and their applications: An approach in food chemistry and innovation potential. Food Chem. 2021;338. doi:10.1016/j.foodchem.2020.127535
CrossRef - Singh AK, Cabral C, Kumar R, et al. Beneficial effects of dietary polyphenols on gut microbiota and strategies to improve delivery efficiency. Nutrients. 2019;11(9). doi:10.3390/nu11092216
CrossRef - Kothari D, Goyal A. Gentio-oligosaccharides from Leuconostoc mesenteroides NRRL B-1426 dextransucrase as prebiotics and as a supplement for functional foods with anti-cancer properties. Food Funct. 2015;6(2):604-611. doi:10.1039/c4fo00802b
CrossRef - Ashwini A, Ramya HN, Ramkumar C, et al. Reactive mechanism and the applications of bioactive prebiotics for human health: Review. J Microbiol Methods. 2019;159:128-137. doi:10.1016/j.mimet.2019.02.019
CrossRef - SNS Insider. Prebiotics Market Size, Share and Segmentation by Ingredients (Fructo-Oligosaccharide, Galacto-oligosaccharides), by Product (Dietary Supplements, Dairy Food), by end-use (foods & beverages, pharmaceutical, animal feed, and others.), by Regions and Global Market Forecast 2023-2030.https://www.snsinsider.com/ reports/prebiotics-market-1965. Published June 2022. Accessed Novemner 1, 2025.
- Deepa Pandey. Prebiotic Ingredients Market Size, Share, and Trends 2024 to 2034. Precedence Research. https://www.precedenceresearch.com/prebiotic-ingredients-market. Published December 19, 2024. Accessed November 1, 2025.
- Bunyatratchata A, Chumroenphat T, Saensouk S, et al. Bioactive compounds, amino acids, fatty acids, and prebiotics in the seed of mahuad (Lepisanthes rubiginosa (Roxb.) Leenh). Horticulturae. 2023;9(10):1159. doi:10.3390/horticulturae9101159
CrossRef - Zheng L, Yu P, Zhang Y, et al. Evaluating the bio-application of biomacromolecule of lignin-carbohydrate complexes (LCC) from wheat straw in bone metabolism via ROS scavenging. Int J Biol Macromol. 2021;176:13-25. doi:10.1016/j.ijbiomac.2021.01.103
CrossRef - You S, Ma Y, Yan B, et al. The promotion mechanism of prebiotics for probiotics: A review. Front Nutr. 2022;9. doi:10.3389/fnut.2022.1000517
CrossRef - Ruszkowski J, Witkowski JM. Lactulose: Patient- and dose-dependent prebiotic properties in humans. Anaerobe. 2019;59:100-106. doi:10.1016/j.anaerobe.2019.06.002
CrossRef - Bothe M, Maathuis A, Bellmann S, et al. Dose-dependent prebiotic effect of lactulose in a computer-controlled in vitro model of the human large intestine. Nutrients. 2017;9(7):767. doi:10.3390/nu9070767
CrossRef - Singh DP, Singh J, Boparai RK, et al. Isomalto-oligosaccharides, a prebiotic, functionally augment green tea effects against high fat diet-induced metabolic alterations via preventing gut dysbacteriosis in mice. Pharmacol Res. 2017;123:103-113. doi:10.1016/j.phrs.2017.06.015
CrossRef - Dhingra D, Michael M, Rajput H, et al. Dietary fibre in foods: A review. J Food Sci Technol. 2012;49(3):255-266. doi:10.1007/s13197-011-0365-5
CrossRef - Cook CM, Rains TM, Maki KC. Effects of oats on obesity, weight management, and satiety. In: Oats Nutrition and Technology. Wiley; 2013:265-279. doi:10.1002/9781118354100.ch12
CrossRef - Merino D, Quilez-Molina AI, Perotto G, et al. A second life for fruit and vegetable waste: a review on bioplastic films and coatings for potential food protection applications. Green Chemistry. 2022;24(12):4703-4727. doi:10.1039/d1gc03904k
CrossRef - Lim HY, Yusup S, Loy ACM, et al. Review on Conversion of Lignin Waste into Value-Added Resources in Tropical Countries. Waste Biomass Valorization. 2021;12(10):5285-5302. doi:10.1007/s12649-020-01307-8
CrossRef - Zdunek A, Pieczywek PM, Cybulska J. The primary, secondary, and structures of higher levels of pectin polysaccharides. Compr Rev Food Sci Food Saf. 2021;20(1):1101-1117. doi:10.1111/1541-4337.12689
CrossRef - Mellinas C, Ramos M, Jiménez A, et al. Recent trends in the use of pectin from agro-waste residues as a natural-based biopolymer for food packaging applications. Materials. 2020;13(3). doi:10.3390/ma13030673
CrossRef - Bojarczuk A, Skąpska S, Mousavi Khaneghah A, et al. Health benefits of resistant starch: A review of the literature. J Funct Foods. 2022;93. doi:10.1016/j.jff.2022.105094
CrossRef - Saini R, Patel AK, Saini JK, et al. Recent advancements in prebiotic oligomers synthesis via enzymatic hydrolysis of lignocellulosic biomass. Bioengineered. 2022;13(2):2139-2172. doi:10.1080/21655979.2021.2023801
CrossRef - Walton GE, van den Heuvel EG, Kosters MH, et al. A randomised crossover study investigating the effects of galacto-oligosaccharides on the faecal microbiota in men and women over 50 years of age. Br J Nutr. 2012;107(10):1466-1475. doi:10.1017/S0007114511004697
CrossRef - Caipang CMA, Lazado CC. Nutritional impacts on fish mucosa: immunostimulants, pre- and probiotics. In: Beck BH, Peatman E, eds. Mucosal Health in Aquaculture. San Diego: Academic Press; 2015:211-272. doi:10.1016/B978-0-12-417186-2.00009-1
CrossRef - Deshmukh N, Rao PS, Sharma H, et al. Waste to nutrition: The evolution of whey, a byproduct to galactooligosaccharides production. Food Chemistry Advances. 2024;4. doi:10.1016/j.focha.2024.100642
CrossRef - Mascco Rosales JL, Jorge-Montalvo P, Mansilla-Samaniego R, et al. Determination of fructooligosaccharides and inulin in yacon (Smallanthus sonchifolius) products from two Peruvian regions exhibiting genetic similarity. J Food Compos Anal. 2025;148(1):108231. doi:10.1016/j.jfca.2025.108231
CrossRef - Singh SP, Jadaun JS, Narnoliya LK, et al. Prebiotic oligosaccharides: Special focus on fructooligosaccharides, its biosynthesis and bioactivity. Appl Biochem Biotechnol. 2017;183(2):613-635. doi:10.1007/s12010-017-2605-2
CrossRef - Panesar PS, Kumari S, Panesar R. Biotechnological approaches for the production of prebiotics and their potential applications. Crit Rev Biotechnol. 2013;33(4):345-364. doi:10.3109/07388551.2012.709482
CrossRef - Scantlebury, Gibson GR. Prebiotics. Best Practice & Research: Clinical Gastroenterology. 2004;18(2):287-298. doi: 10.1016/j.bpg.2003.10.008.
CrossRef - Huang C, Wang X, Liang C, et al. A sustainable process for procuring biologically active fractions of high-purity xylooligosaccharides and water-soluble lignin from Moso bamboo prehydrolyzate. Biotechnol Biofuels. 2019;12(1):189. doi:10.1186/s13068-019-1527-3
CrossRef - Corim Marim AV, Gabardo S. Xylooligosaccharides: prebiotic potential from agro-industrial residue, production strategies and prospects. Biocatal Agric Biotechnol. 2021;37:102190. doi:https://doi.org/10.1016/j.bcab. 2021.102190
CrossRef - Baker JT, Duarte ME, Holanda DM, Kim SW. Friend or foe? Impacts of dietary xylans, xylooligosaccharides, and xylanases on intestinal health and growth performance of monogastric animals. Animals. 2021;11(3):1-28. doi:10.3390/ani11030609
CrossRef - Lin SH, Chou LM, Chien YW, et al. Prebiotic Effects of Xylooligosaccharides on the Improvement of Microbiota Balance in Human Subjects. Gastroenterol Res Pract. 2016;2016. doi:10.1155/2016/5789232
CrossRef - Scheller HV, Ulvskov P. Hemicelluloses. Annu Rev Plant Biol. 2010;61(1):263-289. doi:10.1146/annurev-arplant-042809-112315
CrossRef - Sanchez JI, Marzorati M, Grootaert C, et al. Arabinoxylan‐oligosaccharides (AXOS) affect the protein/carbohydrate fermentation balance and microbial population dynamics of the simulator of human intestinal microbial ecosystem. Microb Biotechnol. 2009;2(1):101-113. doi:10.1111/j.1751-7915.2008.00064.x
CrossRef - Santibáñez L, Henríquez C, Corro-Tejeda R, et al. Xylooligosaccharides from lignocellulosic biomass: A comprehensive review. Carbohydr Polym. 2021;251:117118. doi:10.1016/j.carbpol.2020.117118
CrossRef - Poletto P, Pereira GN, Monteiro CRM, et al. Xylooligosaccharides: Transforming the lignocellulosic biomasses into valuable 5-carbon sugar prebiotics. Process Biochemistry. 2020;91:352-363. doi:10.1016/j.procbio.2020.01.005
CrossRef - Abik F, Palasingh C, Bhattarai M, et al. Potential of Wood Hemicelluloses and Their Derivates as Food Ingredients. J Agric Food Chem. 2023;71(6):2667-2683. doi:10.1021/acs.jafc.2c06449
CrossRef - Barbosa FC, Kendrick E, Brenelli LB, et al. Optimization of cello-oligosaccharides production by enzymatic hydrolysis of hydrothermally pretreated sugarcane straw using cellulolytic and oxidative enzymes. Biomass Bioenergy. 2020;141. doi:10.1016/j.biombioe.2020.105697
CrossRef - Karnaouri A, Matsakas L, Krikigianni E, et al. Valorization of waste forest biomass toward the production of cello-oligosaccharides with potential prebiotic activity by utilizing customized enzyme cocktails. Biotechnol Biofuels. 2019;12(1). doi:10.1186/s13068-019-1628-z
CrossRef - Divyashri G, Sadanandan B, Chidambara Murthy KN, et al. Neuroprotective potential of non-digestible oligosaccharides: An overview of experimental evidence. Front Pharmacol. 2021;12. doi:10.3389/fphar.2021.712531
CrossRef - Lina BAR, Jonker D, Kozianowski G. Isomaltulose (Palatinose®): a review of biological and toxicological studies. Food and Chemical Toxicology. 2022;40(10):1375-1381. doi:10.1016/S0278-6915(02)00105-9.
CrossRef - Bhatia L, Sharma A, Bachheti RK, et al. Lignocellulose derived functional oligosaccharides: production, properties, and health benefits. Prep Biochem Biotechnol. 2019;49(8):744-758. doi:10.1080/10826068.2019.1608446
CrossRef - Hu Y, Winter V, Gänzle M. In vitro digestibility of commercial and experimental isomalto-oligosaccharides. Food Research International. 2020;134. doi:10.1016/j.foodres.2020.109250
CrossRef - Lam KL, Cheung PCK. Carbohydrate-based prebiotics in targeted modulation of gut microbiome. J Agric Food Chem. 2019;67(45):12335-12340. doi:10.1021/acs.jafc.9b04811
CrossRef - Shu B, Wu G, Wang Z, et al. The effect of microwave vacuum drying process on citrus: drying kinetics,
physicochemical composition and antioxidant activity of dried citrus (Citrus reticulata Blanco) peel. Journal of Food Measurement and Characterization. 2020;14(5):2443-2452. doi:10.1007/s11694-020-00492-3
CrossRef - Czech A, Malik A, Sosnowska B, et al. Bioactive substances, heavy metals, and antioxidant activity in whole fruit, peel, and pulp of citrus fruits. Int J Food Sci. 2021;2021. doi:10.1155/2021/6662259
CrossRef - Zahid HF, Ranadheera CS, Fang Z, Ajlouni S. Utilization of mango, apple and banana fruit peels as prebiotics and functional ingredients. Agriculture (Switzerland). 2021;11(7). doi:10.3390/agriculture11070584
CrossRef - Reißner AM, Al-Hamimi S, Quiles A, et al. Composition and physicochemical properties of dried berry pomace. J Sci Food Agric. 2019;99(3):1284-1293. doi:10.1002/jsfa.9302
CrossRef - Munekata PES, Pateiro M, Domínguez R, et al. Bioactive compounds from fruits as preservatives. Foods. 2023;12(2). doi:10.3390/foods12020343
CrossRef - Omer HAA, Abdel-Magid SS, Awadalla IM. Nutritional and chemical evaluation of dried pomegranate (Punica granatum L.) peels and studying the impact of level of inclusion in ration formulation on productive performance of growing Ossimi lambs. Bull Natl Res Cent. 2019;43(1). doi:10.1186/s42269-019-0245-0
CrossRef - Caballero V, Estévez M, Tomás-Barberán FA, et al. Biodegradation of punicalagin into ellagic acid by selected probiotic bacteria: a study of the underlying mechanisms by ms-based proteomics. J Agric Food Chem. 2022;70(51):16273-16285. doi:10.1021/acs.jafc.2c06585
CrossRef - Gulsunoglu Z, Karbancioglu-Guler F, Raes K, et al. Soluble and insoluble-bound phenolics and antioxidant activity of various industrial plant wastes. Int J Food Prop. 2019;22(1):1501-1510. doi:10.1080/10942912.2019.1656233
CrossRef - Kassim MA, Hussin AH, Meng TK, et al. Valorisation of watermelon (Citrullus lanatus) rind waste into bioethanol: an optimization and kinetic studies. International Journal of Environmental Science and Technology. 2022;19(4):2545-2558. doi:10.1007/s13762-021-03310-5
CrossRef - Toupal S, Coşansu S. Effects of freeze-dried banana and watermelon peel powders on bile salt resistance, growth kinetics, and survival of probiotic bacteria. Probiotics Antimicrob Proteins. 2023. doi:10.1007/s12602-023-10131-0
CrossRef - Hao CL, Esah EM, Tajarudin HA, et al. Effect of potential prebiotics from selected fruits peel on the growth of probiotics. J Food Process Preserv. 2021;45(6). doi:10.1111/jfpp.15581
CrossRef - Moysidou AM, Cheimpeloglou K, Koutra SI, et al. A comprehensive review on the antioxidant and anti-inflammatory bioactives of kiwi and its by-products for functional foods and cosmetics with health-promoting properties. Applied Sciences (Switzerland). 2024;14(14). doi:10.3390/app14145990
CrossRef - Alves-Santos AM, Sugizaki CSA, Lima GC, Naves MMV. Prebiotic effect of dietary polyphenols: A systematic review. J Funct Foods. 2020;74. doi:10.1016/j.jff.2020.104169
CrossRef - Krajewska A, Dziki D, Yilmaz MA, Özdemir FA. Physicochemical properties of dried and powdered pear pomace. 2024;29(3). doi:10.3390/molecules29030742
CrossRef - Brahem M, Renard CMGC, Eder S, et al. Characterization and quantification of fruit phenolic compounds of European and Tunisian pear cultivars. Food Research International. 2017;95:125-133. doi:10.1016/j.foodres.2017.03.002
CrossRef - Sinrod AJG, Shah IM, Surek E, Barile D. Uncovering the promising role of grape pomace as a modulator of the gut microbiome: An in-depth review. 2023;9(10):e20499. doi:10.1016/j.heliyon.2023.e20499
CrossRef - Parisi V, Vassallo A, Pisano C, et al. A herbal mixture from propolis, pomegranate, and grape pomace endowed with anti-inflammatory activity in an in vivo rheumatoid arthritis model. Molecules. 2020;25(9). doi:10.3390/molecules25092255
CrossRef - García-Mahecha M, Soto-Valdez H, Carvajal-Millan E, et al. Bioactive compounds in extracts from the agro-industrial waste of mango. Molecules. 2023;28(1):458. doi:10.3390/molecules28010458
CrossRef - Laophongphit A, Siripornadulsil S, Siripornadulsil W. Improvements in the functions of probiotic-based mango pulp rich in phenolic and proline antioxidants by treatment with pectinase and fermentation with lactic acid bacteria. LWT. 2023;181. doi:10.1016/j.lwt.2023.114756
CrossRef - Jakha KH, Goyal C, Dhyani P, et al. Characterization of langra mango peel powder and assessment of its prebiotic and antioxidant potential. J Food Biochem. 2024;2024:1-17. doi:10.1155/2024/9026617
CrossRef - Hsu CT, Chang YH, Shiau SY. Color, antioxidation, and texture of dough and Chinese steamed bread enriched with pitaya peel powder. Cereal Chem. 2019;96(1):76-85. doi:10.1002/cche.10097
CrossRef - Morais SGG, da Silva Campelo Borges G, dos Santos Lima M, et al. Effects of probiotics on the content and bioaccessibility of phenolic compounds in red pitaya pulp. Food Research International. 2019;126:108681. doi:10.1016/j.foodres.2019.108681
CrossRef - Islam MdR, Haque AR, Kabir MdR, et al. Fruit by-products: the potential natural sources of antioxidants and α-glucosidase inhibitors. J Food Sci Technol. 2021;58(5):1715-1726. doi:10.1007/s13197-020-04681-2
CrossRef - Dam SM, Nguyen NT. Production of fermented beverage from fruit rags of jackfruit (Artocarpus Heterophyllus). Acta Hortic. 2013;(989):285-292. doi:10.17660/ActaHortic.2013.989.37
CrossRef - Cruz-Casillas FC, García-Cayuela T, Rodriguez-Martinez V. Application of conventional and non-conventional extraction methods to obtain functional ingredients from jackfruit (Artocarpus heterophyllus lam.) tissues and by-products. Applied Sciences (Switzerland). 2021;11(16). doi:10.3390/app11167303
CrossRef - Bussolo de Souza C, Jonathan M, Isay Saad SM, Schols HA, Venema K. Degradation of fibres from fruit by-products allows selective modulation of the gut bacteria in an in vitro model of the proximal colon. J Funct Foods. 2019;57:275-285. doi:10.1016/j.jff.2019.04.026
CrossRef - Oh NS, Lee JY, Joung JY, et al. Microbiological characterization and functionality of set-type yogurt fermented with potential prebiotic substrates Cudrania tricuspidata and Morus alba L. leaf extracts. J Dairy Sci. 2016;99(8):6014-6025. doi:10.3168/jds.2015-10814
CrossRef - Afrazeh M, Tadayoni M, Abbasi H, et al. Extraction of dietary fibers from bagasse and date seed, and evaluation of their technological properties and antioxidant and prebiotic activity. Journal of Food Measurement and Characterization. 2021;15(2):1949-1959. doi:10.1007/s11694-020-00774-w
CrossRef - El-Kholy WM. Production of probiotic yoghurt fortified with date seeds (phoenix dactylifera l.) powder as prebiotic and natural stabilizer. Egyptian Journal of Agricultural Research. 2017;96(1):159-173. doi:10.21608/ejar.2018.132079
CrossRef - Pereira MAF, Cesca K, Poletto P, et al. New perspectives for banana peel polysaccharides and their conversion to oligosaccharides. Food Research International. 2021;149:110706. doi:10.1016/j.foodres.2021.110706
CrossRef - Lim SY, Tham PY, Lim HYL, et al. Potential Functional Byproducts from Guava Purée Processing. J Food Sci. 2018;83(6):1522-1532. doi:10.1111/1750-3841.14155
CrossRef - Jatav J, Tarafdar A, Saravanan C, et al. Assessment of antioxidant and antimicrobial property of polyphenol-rich chitosan-pineapple peel film. J Food Qual. 2022; 1(2022): 8064114.. doi:10.1155/2022/8064114
CrossRef - Mohd Zaini H, Roslan J, Saallah S, et al. Banana peels as a bioactive ingredient and its potential application in the food industry. J Funct Foods. 2022;92. doi:10.1016/j.jff.2022.105054
CrossRef - Leong SS, Abdul Rahim EF, Sarbini SR, et al. Banana (Musa acuminata), Orange (Citrus reticulata), and Watermelon (Citrullus lanatus) Peels as Prebiotic. Borneo Journal of Resource Science and Technology. 2022;12(1):81-94. doi:10.33736/bjrst.4528.2022
CrossRef - Brito TBN, R.S. Lima L, B. Santos MC, et al. Antimicrobial, antioxidant, volatile and phenolic profiles of cabbage-stalk and pineapple-crown flour revealed by GC-MS and UPLC-MSE. Food Chem. 2021;339. doi:10.1016/j.foodchem.2020.127882
CrossRef - Medhe S, Anand M, Anal AK. Dietary fibers, dietary peptides and dietary essential fatty acids from food processing by-products. In: Anil K Anal (eds.) Food Processing By‐Products and their Utilization. 2017:111-136. doi:10.1002/9781118432921.ch6
CrossRef - Dong R, Liu S, Xie J, et al. The recovery, catabolism and potential bioactivity of polyphenols from carrot subjected to in vitro simulated digestion and colonic fermentation. Food Research International. 2021;143:110263. doi:10.1016/j.foodres.2021.110263
CrossRef - Hussain S, Jõudu I, Bhat R. Dietary fiber from underutilized plant resources-A positive approach for valorization of fruit and vegetable wastes. Sustainability (Switzerland). 2020;12(13). doi:10.3390/su12135401
CrossRef - Riva A, Kolimár D, Spittler A, et al. Conversion of rutin, a prevalent dietary flavonol, by the human gut microbiota. Front Microbiol. 2020;11. doi:10.3389/fmicb.2020.585428
CrossRef - Rodríguez-Martínez B, Gullón B, Yáñez R. Identification and recovery of valuable bioactive compounds from potato peels: A comprehensive review. Antioxidants. 2021;10(10). doi:10.3390/antiox10101630
CrossRef - Kainat S, Arshad MS, Khalid W, et al. Sustainable novel extraction of bioactive compounds from fruits and vegetables waste for functional foods: a review. Int J Food Prop. 2022;25(1):2457-2476. doi:10.1080/10942912.2022.2144884
CrossRef - Plakantonaki S, Roussis I, Bilalis D, et al. Dietary Fiber from plant-based food wastes: A comprehensive approach to cereal, fruit, and vegetable waste valorization. Processes. 2023;11(5). doi:10.3390/pr11051580
CrossRef - Boubaker M, Omri AEL, Blecker C, et al. Fibre concentrate from artichoke (Cynara scolymus L.) stem by-products: Characterization and application as a bakery product ingredient. Food Science and Technology International. 2016;22(8):759-768. doi:10.1177/1082013216654598
CrossRef - Bharat Helkar P, Sahoo A. Review: Food industry by-products used as a functional food ingredients. Int J Waste Resour. 2016;6(3). doi:10.4172/2252-5211.1000248
CrossRef - Sasaki H, Lyu Y, Nakayama Y, et al. Combinatorial effects of soluble, insoluble, and organic extracts from jerusalem artichokes on gut microbiota in mice. Microorganisms. 2020;8(6):954. doi:10.3390/microorganisms8060954
CrossRef - Rivas MÁ, Benito MJ, Martín A, et al. Improve the functional properties of dietary fibre isolated from broccoli by-products by using different technologies. Innovative Food Science & Emerging Technologies. 2022;80:103075. doi:10.1016/j.ifset.2022.103075
CrossRef - Sikorska-Zimny K, Beneduce L. The glucosinolates and their bioactive derivatives in Brassica : a review on classification, biosynthesis and content in plant tissues, fate during and after processing, effect on the human organism and interaction with the gut microbiota. Crit Rev Food Sci Nutr. 2021;61(15):2544-2571. doi:10.1080/10408398.2020.1780193
CrossRef - Thavarajah D, Thavarajah P, Abare A, et al. Mineral micronutrient and prebiotic carbohydrate profiles of USA-grown kale (Brassica oleracea L. var. acephala). Journal of Food Composition and Analysis. 2016;52:9-15. doi:10.1016/j.jfca.2016.07.003
CrossRef - Jiménez-Moreno N, Esparza I, Bimbela F, et al. Valorization of selected fruit and vegetable wastes as bioactive compounds: Opportunities and challenges. Crit Rev Environ Sci Technol. 2020;50(20):2061-2108. doi:10.1080/10643389.2019.1694819
CrossRef - Sabater C, Calvete-Torre I, Villamiel M, et al. Vegetable waste and by-products to feed a healthy gut microbiota: Current evidence, machine learning and computational tools to design novel microbiome-targeted foods. Trends Food Sci Technol. 2021;118:399-417. doi:10.1016/j.tifs.2021.10.002
CrossRef - Aǧagündüz D, Şahin TÖ, Yilmaz B, Ekenci KD, Duyar Özer Ş, et al. Cruciferous Vegetables and Their Bioactive Metabolites: From Prevention to Novel Therapies of Colorectal Cancer. Evidence-based Complementary and Alternative Medicine. 2022;2022. doi:10.1155/2022/1534083
CrossRef - Vlad CC, Păcularu-Burada B, Vasile AM, et al. Upgrading the functional potential of apple pomace in value-added ingredients with probiotics. Antioxidants. 2022;11(10):2028. doi:10.3390/antiox11102028
CrossRef - Zahid HF, Ranadheera CS, Fang Z, Ajlouni S. Utilization of mango, apple and banana fruit peels as prebiotics and functional ingredients. Agriculture. 2021;11(7):584. doi:10.3390/agriculture11070584
CrossRef - Ahmad I, Khalique A, Shahid MQ, et al. Studying the influence of apple peel polyphenol extract fortification on the characteristics of probiotic yoghurt. Plants. 2020;9(1):77. doi:10.3390/plants9010077
CrossRef - Ahmad I, Khalique A, Junaid M, et al. Effect of polyphenol from apple peel extract on the survival of probiotics in yoghurt ice cream. Int J Food Sci Technol. 2020;55(6):2580-2588. doi:10.1111/ijfs.14511
CrossRef - Wang X, Kristo E, LaPointe G. Adding apple pomace as a functional ingredient in stirred-type yogurt and yogurt drinks. Food Hydrocoll. 2020;100:105453. doi:10.1016/j.foodhyd.2019.105453
CrossRef - Utpott M, Ramos de Araujo R, Galarza Vargas C, et al. Characterization and application of red pitaya (Hylocereus polyrhizus) peel powder as a fat replacer in ice cream. J Food Process Preserv. 2020;44(5). doi:10.1111/jfpp.14420
CrossRef - Yu B, Zeng X, Wang L, Regenstein JM. Preparation of nanofibrillated cellulose from grapefruit peel and its application as fat substitute in ice cream. Carbohydr Polym. 2021;254:117415. doi:10.1016/j.carbpol.2020.117415
CrossRef - Acan BG, Kilicli M, Bursa K, et al. Effect of grape pomace usage in chocolate spread formulation on textural, rheological and digestibility properties. LWT. 2021;138:110451. doi:10.1016/j.lwt.2020.110451
CrossRef - Gül H, Şen H. Effects of pomegranate seed flour on dough rheology and bread quality. CyTA – Journal of Food. 2017;15(4):622-628. doi:10.1080/19476337.2017.1327461
CrossRef - Li YF, Wu B, Chen J ping, et al. Prebiotic characteristics of added-value polysaccharides from jackfruit peel waste during in vitro digestion and fecal fermentation. LWT. 2023;187:115330. doi:10.1016/j.lwt.2023.115330
CrossRef - Doan CT, Chen CL, Nguyen VB, et al. Conversion of pectin-containing by-products to pectinases by bacillus amyloliquefaciens and its applications on hydrolyzing banana peels for prebiotics production. Polymers (Basel). 2021;13(9):1483. doi:10.3390/polym13091483
CrossRef - Maneerat N, Tangsuphoom N, Nitithamyong A. Effect of extraction condition on properties of pectin from banana peels and its function as fat replacer in salad cream. J Food Sci Technol. 2017;54(2):386-397. doi:10.1007/s13197-016-2475-6
CrossRef - Jakha KH, Goyal C, Dhyani P, et al. Characterization of langra mango peel powder and assessment of its prebiotic and antioxidant potential. Balti R, ed. J Food Biochem. 2024;2024:1-17. doi:10.1155/2024/9026617
CrossRef - Ahmed A. Utilization of mango peel in development of instant drink. Asian Journal of Agriculture and Biology. 2020;8(3):260-267. doi:10.35495/ajab.2020.02.094
CrossRef - Montalvo-González E, Aguilar-Hernández G, Hernández-Cázares AS, et al. Production, chemical, physical and technological properties of antioxidant dietary fiber from pineapple pomace and effect as ingredient in sausages. CyTA – Journal of Food. 2018;16(1):831-839. doi:10.1080/19476337.2018.1465125
CrossRef - Almeida J dos SO de, Dias CO, Arriola NDA, et al. Feijoa (Acca sellowiana) peel flours: A source of dietary fibers and bioactive compounds. Food Biosci. 2020;38:100789. doi:10.1016/j.fbio.2020.100789
CrossRef - Micheni B, Owaga E, Mugendi B. Physicochemical and sensory properties of fermented milk supplemented with sundried African horned melon. Applied Research. August 2024. doi:10.1002/appl.202400102
CrossRef - Gómez-García R, Vilas-Boas AA, Machado M, et al. Impact of simulated in vitro gastrointestinal digestion on bioactive compounds, bioactivity and cytotoxicity of melon (Cucumis melo L. inodorus) peel juice powder. Food Biosci. 2022;47:101726. doi:10.1016/j.fbio.2022.101726
CrossRef - Lal Bajya S, Shankar Bunkar D, Kumar Goyal S, et al. Foodomics-based metabolites profiling of the Greek yogurt incorporated with unripened papaya peel powder. Food Chemistry: Molecular Sciences. 2024;8:100199. doi:10.1016/j.fochms.2024.100199
CrossRef - Chaidech P, Suhem K, Kongchoosi N, et al. Prebiotic potential of rambutan peel with cardamom oil and its application to extend the shelf-life of plant-base rice milk. Bioactive Carbohydrates and Dietary Fibre. 2024;32:100438. doi:10.1016/j.bcdf.2024.100438
CrossRef - Suhem K, Chaidech P, Kongchoosi N, et al. Novel mangosteen peel prebiotic with cardamom oil supports probiotic growth and preservation of rice sausage. Food Sci Biotechnol. September 2024. doi:10.1007/s10068-024-01710-0
CrossRef - Koca I, Tekguler B, Yilmaz VA, Hasbay I, Koca AF. The use of grape, pomegranate and rosehip seed flours in Turkish noodle (erişte) production. J Food Process Preserv. 2018;42(1):e13343. doi:10.1111/jfpp.13343
CrossRef - Guerra L, Romanini D, López S, Castelli V, Clementz A. Upcycling of carrot discards into prebiotics (fructooligosaccharides) as high value food ingredientsod ingredients. Food and Bioproducts Processing. 2023;138:172-180. doi:10.1016/j.fbp.2023.01.011
CrossRef - Vénica CI, Spotti MJ, Pavón YL, et al. Influence of carrot fibre powder addition on rheological, microstructure and sensory characteristics of stirred‐type yogurt. Int J Food Sci Technol. 2020;55(5):1916-1923. doi:10.1111/ijfs.14415
CrossRef - Nouri M, Nasehi B, Samavati V, et al. Optimizing the effects of Persian gum and carrot pomace powder for development of low-fat donut with high fibre content. Bioactive Carbohydrates and Dietary Fibre. 2017;9:39-45. doi:10.1016/j.bcdf.2017.01.001
CrossRef - Admasu F, Fentie EG, Admassu H, et al. Functionalization of wheat bread with prebiotic dietary insoluble fiber from orange-fleshed sweet potato peel and haricot bean flours. 2024;200:116182. doi:10.1016/j.lwt.2024.116182
CrossRef - de Albuquerque TMR, Borges CWP, Cavalcanti MT, et al. Potential prebiotic properties of flours from different varieties of sweet potato (Ipomoea batatas L.) roots cultivated in Northeastern Brazil. Food Biosci. 2020;36:100614. doi:10.1016/j.fbio.2020.100614
CrossRef - Hussien H, Afify H, Shabib Z. Production and quality evaluation of cupcakes enriched with prebiotics. Food Technology Research Journal. 2023;2(3):55-67. doi:10.21608/ftrj.2023.328808
CrossRef - Gurbuz B, Demirci AŞ. Probiotic yoghurt fortified with rosehip seed powder: Viability, physicochemical, and sensorial properties. Int Food Res J. 2023;30(5):1187-1200. doi:10.47836/ifrj.30.5.08
CrossRef - Nilufa Yeasmin, Sarker BR, Begum A, et al. Fortification of yogurt with red dragon fruit’s (hylocereus polyrhizus) peel powder: Effects on comprehensive quality attributes and sensory properties. Turkish Journal of Agriculture – Food Science and Technology. 2023;11(12):2300-2307. doi:10.24925/turjaf.v11i12.2300-2307.6187
CrossRef - Fathy HM, Abd El-Maksoud AA, Cheng W, Elshaghabee FMF. Value-added utilization of citrus peels in improving functional properties and probiotic viability of acidophilus-bifidus-thermophilus (abt)-type synbiotic yoghurt during cold storage. Foods. 2022;11(17):2677. doi:10.3390/foods11172677
CrossRef - Byresh TS, Malini B, Meena L, et al. Effect of addition of pineapple peel powder on white finger millet vegan probiotic beverage. J Food Process Preserv. 2022;46(10). doi:10.1111/jfpp.16905
CrossRef - Hui CY, Lee KC, Chang YP. Cellulase-xylanase-treated guava purée by-products as prebiotics ingredients in yogurt. Plant Foods for Human Nutrition. 2022;77(2):299-306. doi:10.1007/s11130-022-00981-4
CrossRef - Zahid HF, Ranadheera CS, Fang Z, et al. Functional and healthy yogurts fortified with probiotics and fruit peel powders. Fermentation. 2022;8(9):469. doi:10.3390/fermentation8090469
CrossRef - Varnaitė L, Keršienė M, Šipailienė A, et al. Fiber-rich cranberry pomace as food ingredient with functional activity for yogurt production. Foods. 2022;11(5):758. doi:10.3390/foods11050758
CrossRef - Haghani S, Hadidi M, Pouramin S, et al. Application of cornelian cherry (cornus mas l.) peel in probiotic ice cream: functionality and viability during storage. Antioxidants. 2021;10(11):1777. doi:10.3390/antiox10111777
CrossRef - Hao CL, Esah EM, Tajarudin HA, et al. Effect of potential prebiotics from selected fruits peel on the growth of probiotics. J Food Process Preserv. 2021;45(6). doi:10.1111/jfpp.15581
CrossRef - Abd El-Sattar El, Ramadan M, Abd El-Wahed El. Production of probiotic low-fat yoghurt supplemented with guava seeds powder. Journal of Food and Dairy Sciences. 2020;11(4):91-96. doi:10.21608/jfds.2020.95596
CrossRef - Ahmad I, Khalique A, Shahid MQ, et al. Studying the influence of apple peel polyphenol extract fortification on the characteristics of probiotic yoghurt. Plants. 2020;9(1):77. doi:10.3390/plants9010077
CrossRef - Ibrahim A, Awad S, El-Sayed M. Impact of pomegranate peel as prebiotic in bio-yoghurt. British Food Journal. 2020;122(9):2911-2926. doi:10.1108/BFJ-04-2019-0296
CrossRef - Sah BNP, Vasiljevic T, McKechnie S, Donkor ON. Effect of pineapple waste powder on probiotic growth, antioxidant and antimutagenic activities of yogurt. J Food Sci Technol. 2016;53(3):1698-1708. doi:10.1007/s13197-015-2100-0
CrossRef - Gavril (Rațu) RN, Cârlescu PM, Veleșcu ID, et al. The development of value-added yogurt based on pumpkin peel powder as a bioactive powder. J Agric Food Res. 2024;16:101098. doi:10.1016/j.jafr.2024.101098
CrossRef - Salas-Millán JÁ, Conesa-Bueno A, Aguayo E. A novel antidiabetic lactofermented beverage from agro-industrial waste (broccoli leaves): Process optimisation, phytochemical characterisation, and shelf-life through thermal treatment and high hydrostatic pressure. Food Biosci. 2024;59:103999. doi:10.1016/j.fbio.2024.103999
CrossRef - Pérez-Chabela M de L, Cebollón-Juárez A, Bosquez-Molina E, et al. Mango peel flour and potato peel flour as bioactive ingredients in the formulation of functional yogurt. Food Science and Technology. 2022;42. doi:10.1590/fst.38220
CrossRef - Janiszewska-Turak E, Kołakowska W, Pobiega K, Gramza-Michałowska A. Influence of drying type of selected fermented vegetables pomace on the natural colorants and concentration of lactic acid bacteria. Applied Sciences. 2021;11(17):7864. doi:10.3390/app11177864
CrossRef
Abbreviations List
OS – Oligosaccharides
NDOs – Nondigestible Oligosaccharides
CAGR – Compound Annual Growth Rate
ROS – Reactive Oxygen Species
TCA – Tricarboxylic Acid cycle (or Krebs cycle)
GSTs – Glutathione S-transferases
SCFAs – Short-Chain Fatty Acids
GPR – G Protein-Coupled Receptor
IMO – Isomaltooligosaccharides
GalA – Galacturonic Acid
HG – Homogalacturonan
RG – Rhamnogalacturonan
DP – Degree of Polymerization
XOS – Xylose-oligosaccharides
AXOS – Arabinoxylo-oligosaccharides
MOS – Mannan-oligosaccharide
COS – Cello-oligosaccharides
WPP – Watermelon Peel Powder
BPP – Banana Peel Powder
RSP – Rosehip Seed Powder
PCP – Pineapple Crown Powder
TPC – Total Phenolic Content
PP – Pomegranate Peel
HHP – High Hydrostatic Pressure
LAB – Lactic Acid Bacteria
SDGs – Sustainable Development Goals













