Impact of Drying Temperature and Duration on Nutritional, Functional, and Microbial Qualities of Almond Bagasse
Department of Food Technology, East Asia University of Technology, Hanoi, Vietnam
Corresponding Author Email: khuetm@eaut.edu.vn
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.3.20
ABSTRACT:Dehydration is an important post-processing approach that stabilizes plant-based food residues and supports their transformation into powdered ingredients with improved shelf life and preserved nutritional quality. Almond bagasse, a nutrient-rich by-product of almond milk production, shows promising potential for food applications, especially in powdered form for bakery and gluten-free formulations. This study attempted to assess the impacts of drying temperature and duration on the nutritional, physicochemical, and microbial qualities of almond bagasse, supporting the selection of suitable processing parameters in its valorization. Four drying treatments were applied: Three single-stage conditions (80 °C or 120 °C for 2-3 hours) and a two-stage step-down process (120 °C for two hours followed by 80 °C for one hour). Carbohydrates, fat, crude fiber, and protein contents were statistically comparable (p > 0.05) between treated and raw samples, indicating minimal nutritional loss during drying. Nevertheless, increasing drying temperature and extending duration resulted in significant loss of vitamin C (29.2% to 61.4%). Drying conditions had limited effects on solubility and pH but significantly influenced swelling power (p<0.05). Oleic acid remained dominant (up to 40.8%), and both its content and the oleic-to-linoleic acid ratio increased with thermal treatment, suggesting improved lipid stability and resistance to oxidative degradation. Drying conditions significantly reduced the initial microbial load and improved microbial stability during 15 days of storage, particularly under higher temperature and extended drying durations. Thus, two-stage step-down drying process was found to be the most effective pre-treatment for improving both the nutritional and microbiological quality of almond bagasse.
KEYWORDS:Almond bagasse; Drying conditions; Functional properties; Microbial stability; Nutritional composition; Thermal drying
Introduction
Almonds (Prunus dulcis) are widely recognized as a nutrient-dense food, containing high levels of lipids (approximately 50%), proteins, carbohydrates, fiber, and essential vitamins.1 Rising consumer demand has expanded the use of almonds in processed foods, with almond milk emerging as a leading plant-based dairy alternative owing to its favorable nutritional profile and associated health benefits.2 Large-scale almond milk production, typically involving blending almonds with water and filtering the mixture, generates considerable amounts of almond bagasse (also known as almond okara or almond cake).3,4 This by-product can account for up to 82% of the soaked almond kernel mass after extraction, while still retaining proteins, residual lipids, and dietary fiber.3-5 In the United States, almond milk is the dominant segment of the plant-based milk market, holding the largest market share of 59%.6 Consequently, the expansion of almond milk production is expected to generate greater quantities of almond bagasse, highlighting the need for sustainable valorization strategies. Nevertheless, in contrast to soy okara, almond bagasse remains underutilized and has received limited scientific attention.
Among post-processing strategies, dehydration plays an important role in stabilizing plant-based food residues and supporting their transformation into powdered ingredients with improved shelf life and preserved nutritional quality.7 Recent studies suggested that almond bagasse can be valorized as a functional food ingredient, particularly in powdered form for bakery and gluten-free formulations.8,9 Its incorporation into biscuits and breads has been shown to improve texture, antioxidant stability, and shelf life, effects that are strongly influenced by the drying methods.8,9 Despite its importance, research on almond bagasse remains scarce, with most studies limited to comparing drying techniques rather than evaluating how temperature and duration affect its nutritional composition, hydration properties, and microbial stability.
This study evaluated the impact of four thermal treatments, varying in duration (2 and 3 hours) and temperature (80 °C and 120 °C), on the quality and functionality of almond bagasse. Furthermore, the effects of both single-stage treatments and the two-stage step-down process were also evaluated.
Materials and Methods
Preparation of Almond Bagasse
Almond kernels were purchased from a local grocery store. The materials were roasted at 150oC for 5 minutes using a Sanaky 120L oven (No. VH-129S2D) before being soaked in water for 1 hour. The almond skins were then removed by hands to obtain the almond kernels for producing almond milk. A suspension of almond kernels and water (1:5, w/w) was prepared using a blender (MJ-CB800SRA, Panasonic, Japan) at speed level 2 for 1 minute. Filtration was carried out using a double-layered cheesecloth, and the solid byproduct (almond bagasse) was collected.
The almond bagasse samples were dried using drying oven (Fengling, China) at four different thermal treatments to evaluate the effects of temperature and heating duration on their physicochemical, functional, microbiological properties, and fatty acid composition. Four drying protocols were applied: T1 (80 °C, 2 hours), T3 (120 °C, 2 hours), and T4 (120 °C, 3 hours) as single-stage treatments, and T2 as a two-step process that initiated at 120 °C for two hours and then dropped to 80 °C for one hour. The dried samples were stored in sealed zipper bags at 25 °C and analyzed for total plate count and yeast-mold growth after 5, 10, and 15 days of storage.
Composition Analysis
Proximate composition was determined using AOAC official methods.10 The samples were dried at 105oC to a constant weight to determine the moisture content (Method No 925.10).10 Protein, crude fiber, fat, total carbohydrate, vitamin C were determined. Crude fiber was determined based on Method No 962.09 (AOAC, 2005), involving acid-alkaline digestion, drying at 130 °C, and ashing at 550 °C.10 Total nitrogen was measured using Kjeldahl method, and the crude protein content was calculated using a conversion factor of 5.18.11 Soxhlet extraction with petroleum ether for 16 hours, followed by drying and gravimetric quantification, was used to measure lipid content (Method 948.22).10
The Phenol-Sulfuric acid method was applied to measure total carbohydrate levels.12 A glucose standard curve (0.1 mg/mL) was constructed, and absorbance was recorded at 490 nm. Almond flour (100 mg) was heat-treated in boiling tubes for 3 hours, neutralized with sodium carbonate, diluted to 100 mL, and centrifuged. Analysis of the supernatant was performed using the same conditions as those used for the standards.
Vitamin C content was determined by iodine titration method.13 The sample (2 grams) was mixed with 10 mL of distilled water and centrifuged (3000 rpm, 20 minutes, 4oC). A mixture of oxalic acid (2 mL), distilled water (150 mL), and 1 mL starch indicator was added to 20 mL of supernatant, followed by titration with 0.005 mol/L iodine until a stable blue-black endpoint was reached.
pH Measurement
pH was measured as described in AOAC Official Method 943.02.10 A 0.25g sample was dispersed in 25 mL of distilled water and shaken for 3 minutes. The suspension was kept at room temperature for 1 hour before pH measurement using a digital pH meter (SI Analytics, German). The electrode was calibrated using certified buffer solutions at pH 7.00 and 4.00.
Determination of swelling power and water solubility
Swelling power refers to the ratio of hydrated sediment weight to insoluble dry matter, while water solubility is defined as the percentage of dissolved solids relative to the initial dry mass.14,15 The swelling power and water solubility were assessed according to the method described by Chen and colleagues,16 with modifications. 0.5 grams of sample were mixed with 10 mL of distilled water. The initial dry weight (Wd) was recorded after drying to a constant mass at 105 °C. The tubes were then incubated at 60 °C for 30 minutes, followed by rapid cooling to 25 °C and centrifugation at 3000 × g for 15 minutes. The supernatant was collected and dried to constant weight (W1) at 105oC and the sediment remaining in the tube was obtained and weighed (Ws). The following formulas were used to determine swelling power and water solubility:
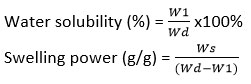
Microbial Analysis
Total bacterial counts, yeasts, and molds were determined on days 0, 5, 10, and 15 of storage. The microbial analysis method was described in a previous study.17 The initial dilution was obtained by mixing ten grams of sample with 100 mL of sterile 0.1% peptone water, and dilutions ranging from 10⁻¹-10⁻⁶ were used for analysis. Total bacterial counts were determined in PCA medium (5 g/L peptone, 1 g/L glucose, 2.5 g/L yeast extract, 16 g/L agar), aerobically incubated at 30 °C for two days. Yeasts and molds were counted in PDA (200 g/L potato infusion, 20 g/L dextrose, 20 g/L agar), aerobically incubated at 30 °C for five days.
Determination of fatty acids
Three major fatty acids – oleic acid (C18:1), linoleic acid (C18:2), and palmitic acid (C16:0) were determined according to AOAC Official Method 996.06.10 Samples were hydrolyzed with hydrochloric acid at 80 °C for 40 minutes, followed by lipid extraction using a mixture of ethyl ether and petroleum ether. Fatty acids were analyzed using a gas chromatograph (Agilent 7820A, Palo Alto, CA, USA) equipped with a flame ionization detector (FID) and a DB-WAX capillary column (60 m × 0.25 mm, 0.25 μm film thickness, Agilent Technologies, USA). Quantification was performed using triundecanoin (C11:0) as an internal standard.
Statistical analysis
All experimental data were obtained from three independent trials and expressed as mean values ± standard deviation (SD), except for fatty acid composition, which was presented as relative percentages. Data was analyzed using SPSS software version 22.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism version 8.1 (GraphPad Software Inc., San Diego, CA, USA) was used for data visualization. Statistical significance was determined at p-value < 0.05, based on ANOVA with Tukey’s post-hoc test.
Results
Impact of drying conditions on nutritional and physicochemical characteristics of almond bagasse
Table 1: Properties of almond bagasse under different thermal treatment
|
Sample |
Control | T1 | T2 | T3 |
T4 |
|
Nutrient composition (%)* |
|||||
|
Protein |
22.40 ± 0.23a | 20.64 ± 2.60a | 17.32 ± 0.01a | 18.05 ± 2.53a | 16.24 ± 0.07a |
| Crude Fiber | 2.48 ± 0.02a | 2.60 ± 1.19a | 2.55 ± 0.00a | 1.64 ± 0.23a |
2.56 ± 0.01a |
|
Fat |
58.58 ± 7.09a | 49.60 ± 0.96a | 54.45 ± 8.19a | 55.49 ± 7.88a | 56.14 ± 12.01a |
| Total Carbohydrate | 17.66 ± 0.71a | 15.72 ± 2.51a | 17.49 ± 3.67a | 18.03± 0.75a |
18.97 ± 2.64a |
|
Antioxidant Activity (%)* |
|||||
| Vitamin C | 0.44 ± 0,00a | 0.31 ± 0.04b | 0.17 ± 0,00c | 0.27 ± 0,04bc |
0.18 ± 0,00c |
|
Physicochemical Properties |
|||||
| pH | 7.37 ± 0.11 a | 7.02 ± 0.01b | 6.78 ± 0.04b | 7.03 ± 0.01b |
6.92 ± 0.04b |
|
Moisture (%) |
5.36 ± 0.09a | 66.26 ± 4.24b | 1.86 ± 0.01a | 52.84 ± 6.61b |
0.89±0.45a |
* Results are given on a dry matter basis. Data are presented as mean ± SD (n = 3). Different superscript letters (a-c) in same row present significant differences (p < 0.05). T1 (80 °C, 2 hours), T2 (120 °C in two hours, then 80 °C for one hour), T3 (120 °C, 2 hours), T4 (120 °C, 3 hours).
The physicochemical properties and key nutrient compositions of almond bagasse processed under different thermal drying conditions: T1 (80 °C, 2 hours), T2 (120 °C for two hours, followed by 80 °C for one hour), T3 (120 °C, 2 hours), and T4 (120 °C, 3 hours) with the control sample (raw almond kernels) were shown (Table 1). Moisture level was significantly reduced with increasing drying temperature and duration. Specifically, T1 and T3 samples contain higher moisture contents (66.26 ± 4.24% and 52.84 ± 6.61%, respectively), while considerably lower values were observed in the control (5.36 ± 0.09%), T2 (1.86 ± 0.01%), and T4 (0.89 ± 0.45%). A slight decrease in pH was observed in treated samples relative to the control; however, differences among treatments were not statistically significant. Carbohydrates, fat, crude fiber, and protein contents remained largely unchanged (p > 0.05) between treated and raw samples, suggesting minimal nutrient loss during thermal treatment. Fat remained the predominant macronutrient in all almond groups, ranging from 49.6% to 56.14% in treated samples (p > 0.05), compared with 58.58% in raw material. The total protein content of almond bagasse samples was 22.40 ± 0.23% in the control sample, 20.64 ± 2.60% in T1, 17.32 ± 0.01% in T2, 18.05 ± 2.53% in T3, and 16.24 ± 0.07% in T4 (Table 1), with no significant differences among treatments.
The crude fiber content of thermally treated almond bagasse samples varied between 1.64% to 2.60%, while the value for the control was 2.48%. Table 1 showed that the total carbohydrate content of thermally treated samples ranged from 15.72% to 18.97%, compared to 17.66% in the control sample. In addition to carbohydrates, bioactive compounds such as vitamin C also remain at retained levels in the residue, providing desirable antioxidant potential for this by-product. Thermal treatment significantly impacted the vitamin C content, with losses ranging from 29.2% to 61.4% compared to the untreated control. Specifically, vitamin C retention decreased from 4.37 mg/g in the control to 2.82 mg/g in T1 (~35.5% loss) and 2.42 mg/g in T3 (~44.6% loss). The most significant losses were observed in T2 and T4, with vitamin C levels decreasing by approximately 60% compared to the control. The vitamin C contents were 1.75 mg/g in T2 and 1.76 mg/g in T4, showing no significant difference (p > 0.05).
Effect of drying conditions on solubility and swelling power of almond bagasse
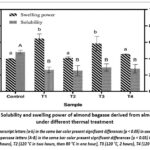 |
Figure 1: Solubility and swelling power of almond bagasse derived from almond milk under different thermal treatment |
The swelling power and solubility of almond bagasse samples derived from almond milk are shown in Fig. 1. All treated samples (T1-T4) were obtained from the solid residue of almond milk production, which involved soaking the almonds in water, followed by grinding and filtration. Solubility in the control sample was significantly higher than that observed in all thermally treated groups (p < 0.05). Solubility values for treated samples ranged from 25.4% to 29.2%, with no statistical differences among them (p > 0.05) (Fig. 1).
In contrast, thermal conditions had a major impact on swelling power. Interestingly, samples T1 and T3, which retained higher moisture levels, showed significantly higher swelling power (6.44 g/g and 5.85 g/g, respectively) compared to the control, T2, and T4 (3.96 g/g, 4.08 g/g and 4.53 g/g, respectively) (p < 0.05). However, these three samples showed no statistically significant differences (p>0.05) (Fig. 1).
Effect of thermal drying treatments on microbial stability of almond bagasse
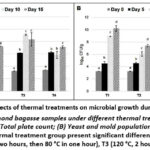 |
Figure 2: Effects of thermal treatments on microbial growth during storage. |
The changes in total plate count (TPC) and yeast, mold populations (YMP) under different thermal treatments (T1-T4) during 15 days of storage are shown in Fig. 2A and 2B. Although microbial counts significantly increased overtime in all treatments, the extent of microbial growth varied markedly among thermal processing conditions. Additionally, there were no significant differences in yeast and mold counts at day 0 among the three thermal treatments T1, T2, and T3, while T4 significantly reduced the initial yeast and mold population (p < 0.0001) (Table 2). At day 0, total plate counts were most effectively reduced by T2, with lower reductions observed under T3 and T4 when compared to the control treatment (T1) (Table 2).
Both T2 and T4 demonstrated a clear inhibitory effect on microbial proliferation, particularly in reducing TPC and YMP. T2 significantly lowered the initial microbial level and remained low TPC levels during the early storage period with TPC increasing only from 4.59 to 5.14 log₁₀ CFU/g from day 5 to day 10 (Data is not shown).
Table 2: Microbial Counts (log10 CFU/g) under different Thermal Treatment
|
Parameter |
Time | T1 | T2 | T3 | T4 |
| Total Plate Count | Day 0 | 3.56 ± 0.01 | 2.96 ± 0.00**** | 3.26 ± 0.05**** |
3.44 ± 0.00** |
|
Day 15 |
10.58 ± 0.06 | 7.26 ± 0.00*** | 8.33 ± 1.08** |
7.11 ± 0.21*** |
|
|
Yeast and Mold |
Day 0 | 3.26 ± 0.05 | 3.26 ± 0.01ns | 3.26 ± 0.00ns | 2.96 ± 0.00**** |
| Day 15 | 10.23 ± 0.46 | 7.41 ± 0.21*** | 8.51 ± 0.78** |
7.36 ± 0.00*** |
Microbial counts are presented in log CFU/g. Statistical significance between day 0 and day 15 within each row is indicated by symbols as follows: * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001, ns: not significant. T1 (80 °C, 2 hours), T2 (120 °C in two hours, then 80 °C in one hour), T3 (120 °C, 2 hours), T4 (120 °C, 3 hours).
By comparison, T4 showed a greatest ability to control microbial growth over the longer term of storage, effectively reducing the development of both TPC and yeast-mold levels. Between day 0 and day 15, TPC levels in T4 increased from 3.44 to 7.11 log₁₀ CFU/g and yeast-mold counts in the same conditions raised from 2.96 to 7.36 log₁₀ CFU/g, representing the slowest rate of increase among all treatments (Table 2). The mild control T1 showed the least effective microbial control, with both TPC and YMP rising rapidly throughout the storage period. In particular, TPC and YMP increased from 3.56 to 10.58 log₁₀ CFU/g and 3.26 to 10.23 log₁₀ CFU/g between day 0 and day 15, respectively (Table 2).
Effect of drying temperature and duration on major fatty acids in almond bagasse
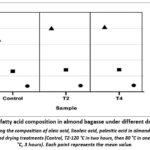 |
Figure 3: Major fatty acid composition in almond bagasse under different drying conditions |
This study analyzed the contents of three predominant fatty acids in almond, which are oleic acid (C18:1), linoleic acid (C18:2), and palmitic acid (C16:0) in the Control, T2, and T4 samples (Fig. 3). Oleic acid was present in the highest level, ranging from 33% in the untreated sample to over 40% after thermal treatment. Linoleic and palmitic acids were present at lower levels, accounting for approximately 12.9%-13.9% and 3.6%-4.0%, respectively.
All three major fatty acids-oleic, linoleic, and palmitic- showed noticeable increases in proportion between the untreated and heat-treated samples. However, only oleic acid content was significantly affected by the heating intensity, while changes in linoleic and palmitic acids between the two thermal treatments were not clearly observed. The oleic acid level increased with higher temperature, reaching 33.0%, 40.1%, and 40.8% in the control, T2, and T4 samples, respectively.
 |
Figure 4: Chromatogram of fatty acids in almond bagasse of the Control and T4 samples |
Discussion
Although a previous study on thermally treated okara (a by-product of soy milk) reported a slight decrease in protein content from 29.43% in the fresh sample to 28.14% after drying at 80 °C and 27.59% at 200 °C,17 our results did not show a similar trend in almond bagasse. Oven drying is widely recognized as an efficient technique for removing moisture in high-moisture matrices.18 Previous studies have demonstrated that prolonged drying at low temperatures can effectively reduce water content while preserving nutrients.18 In our study, samples dried at 80 °C for 2 hours and 120 °C for 2 hours retained relatively high residual moisture, indicating that this temperature range, when applied for short durations, is insufficient for complete dehydration. This finding is consistent with previous reports showing that extended drying times at 80 °C are required to achieve substantial moisture reduction and slight change in proximate composition.18,19 Extending the drying time by just one additional hour in our study markedly improved water removal, while proximate composition remained unaffected. Such results are reasonable given that the applied temperatures are still below the threshold at which reactions such as non-enzymatic browning have significant impact (≥140 °C).20
In the present study, the crude fiber values were considerably lower than those observed in other plant-based beverage by-products of interest to food industry, such as coconut cake (11.2-12.2%),21,22 cashew apple bagasse (~11%),23 and okara (20-50%).24,25 Almond kernels are widely recognized as a valuable source of carbohydrates and vitamin.26,27 While both components retained at appreciable levels in almond bagasse, vitamin C was significantly affected by thermal treatment, showing a noticeable decrease with increasing drying time and temperature. Similar results have been reported in previous researches, which reported that vitamin C loss during thermal processing is primarily due to its water solubility and thermal instability.28,29 Increasing heating temperature and prolonged heating duration also led to greater loss of vitamin C content.28,29 Regarding the observed decrease in pH, the changes appeared to be primarily driven by pre-processing, with soaking considered a key factor. Recent studies on almonds have reported that soaking whole nuts increases phytate content – naturally abundant in nuts such as almonds and walnuts – which may contribute to the reduction in pH.30,31
Regarding the functionality of almond bagasse, the results (Fig. 1) also indicate that the reduction in solubility primarily resulted from the milk extraction process, rather than from differences in drying conditions. Soaking and grinding have been reported to facilitate the migration of soluble proteins into the liquid phase in soybean.32 In soybeans, soaking and grinding enhance protein loss into the milk, decreasing solubility in the residue – a mechanism likely shared in almond milk production.32 A reduction in almond protein solubility due to the combination of heat and water was also reported.33 It was explained that protein structure changed during thermal treatment, in which structural unfolding and the formation of numerous molecular interactions occurred, leading to aggregation and precipitation within the protein matrix.34,35 Additionally, Tang et al35 demonstrated that mild heating (60-100 °C) induced protein unfolding, promoted thiol-disulfide exchange and hydrophobic interactions, resulting in the formation of soluble aggregates and altered protein functionality in pea and soy. In contrast, swelling power was markedly influenced by different thermal conditions. Similar observations have been reported in starches from white and red cocoyam as well as sweet potato.36,37 This phenomenon is primarily observed in starch-based products, where swelling power is strongly influenced by amylose-based interactions and heat-induced structural transitions.37,38 However, almond flour exhibits different composition, lacking both of starch and amylose.1 Therefore, the observed swelling in almond-based matrices is unlikely to result from starch-related mechanisms. It is suggested that both matrix hydration and fiber preservation result in enhancing swelling power in high-moisture samples. Moisture helps preserve porous microstructure and prevents shrinkage or densification of the fibrous matrix during thermal treatment.39,40 This allows insoluble dietary fiber (IDF), mainly cellulose and hemicellulose in almond bagasse sample, to retain its structure and remain accessible to water during heating stage.8 As a result, water can penetrate more effectively, resulting in greater expansion and swelling. In contrast, dried samples are more prone to structural shrinkage, limiting their ability to reabsorb water and swell.
In terms of microbial stability, our results (Table 2) indicates that the intensity of thermal processing, either by extending drying time, higher temperature or a combination of both, influenced both the initial microbial counts and its subsequent growth during storage. Our results are also supported by Duarte et al,4 who demonstrated that elevating the drying temperature from 60 °C to 70 °C improved the microbial stability of almond bagasse during a 180-day storage period. Although T2 was slightly less effective than T4 in maintaining the lowest microbial levels by the end of storage, it still demonstrated notable microbiological advantages. Interestingly, our results suggest that applying a step-down two-stage thermal treatment (120oC in 2 hours followed by 80oC in 1 hour) may offer double benefits: an initial aerobic microbial load reduction and extended inhibit of microbial growth during storage. This observation is further supported by previous studies,41,42 which have shown that step-down hot-air drying enhanced microbial inactivation efficiency by combining an initial high-temperature phase with a subsequent lower-temperature phase with minimal impact on the nutritional quality of almonds and walnut.41,42 Microbial load and stability can be significantly influenced by the dehydration process, which reduces the water activity for microbial growth.4,43 Although heat treatment reduced initial microbial contamination, retained moisture in the dried samples still allowed microbial growth during storage. A similar observation was reported in dried okara byproducts study, where microbial counts remained relevant due to the significant remaining moisture content after drying.17
Three major fatty acid values are lower than the typical concentrations found in almond kernels, which contain approximately 50-70% oleic acid, 10-26% linoleic acid, and 5-9% palmitic acid, suggesting that lipid composition could be significantly influenced by multiple factors including cultivar, soil characteristics, and climatic conditions.44 Notably, our results are agree with previous studies, which have highlighted that different drying temperature and thermal treatment conditions can influence the fatty acid composition, particularly the unsaturated fatty acids.45,46 The increase in fatty acid content may result from structural damage of nut cells and tissues during heating, which enables the release of chemical components and activates hydrolytic enzymes, thereby enhancing fatty acid levels.46,47 In this study, oleic-to-linoleic ratio in almond bagasse sample was increased from 2.6 in the raw sample to 2.9 in T4 sample, suggesting a potential for improving resistance to lipid oxidation and rancidity.46,48
Conclusion
In our study, thermal drying did not significantly affect the macronutrient contents of almond bagasse, whereas vitamin C showed notable degradation with increasing temperature and time. While drying treatments had minimal effects on solubility and pH, swelling power was notably influenced. Thermal processing also had impact on the fatty acid profile, with oleic acid identified as the predominant component and exhibiting the greatest increase under thermal treatment. Under intensive drying conditions, microbial loads were effectively reduced, thereby improving storage stability over 15 days. Step-down hot-air drying demonstrated efficiency in both preserving nutritional quality and controlling microbial growth. The optimized condition of 120 °C for two hours followed by 80 °C for one hour offers an effective strategy for thermal drying, enhancing the potential of almond bagasse as a functional ingredient in bakery and other food formulations. Further investigations should focus on the drying kinetics of almond bagasse to improve its preservation stability and functional valorization, especially for incorporation into plant-based and gluten-free food products.
Acknowledgement
The authors acknowledge the Faculty of Food Technology, East Asia University of Technology, for providing research facilities.
Funding Sources
The authors received no financial support for the research, authorship, and publication of the article
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other Sources
Not applicable.
Author Contributions
- Tran Minh Khue: Conceptualization, Data collection, Data Curation, Formal Analysis, Visualization, Writing – Original Draft, Writing – Review and Editing.
- Hoang Thi Oanh: Data collection, Data Curation, Writing – Review and Editing.
References
- Barreca D, Nabavi SM, Sureda A, et al. Almonds (Prunus Dulcis D. A. Webb): A Source of Nutrients and Health-Promoting Compounds. Nutrients. 2020;12(3):672. DOI: https://doi.org/10.3390/nu12030672
CrossRef - Ramsing R, Santo R, Kim BF, et al. Dairy and Plant-Based Milks: Implications for Nutrition and Planetary Health. Curr Environ Health Rep. 2023;10(3):291-302. DOI: https://doi.org/10.1007/s40572-023-00400-z
CrossRef - De Angelis D, Pasqualone A, Squeo G, et al. Almond okara as a valuable ingredient in biscuit preparation. J Sci Food Agric. 2023;103(4):1676-1683. DOI: https://doi.org/10.1002/jsfa.12286
CrossRef - Duarte S, Betoret E, Betoret N. Shelf Life and Functional Quality of Almond Bagasse Powders as Influenced by Dehydration and Storing Conditions. Foods. 2024;13(5):744. DOI: https://doi.org/10.3390/foods13050744
CrossRef - Duarte S, Betoret E, Barrera C, et al. Integral Recovery of Almond Bagasse through Dehydration: Physico-Chemical and Technological Properties and Hot Air-Drying Modelling. Sustainability. 2023;15(13):10704. DOI: https://doi.org/10.3390/su151310704
CrossRef - Abrieux A, Avilla M, Bagla A, et al. The evolving market for plant-based milk: alfalfa and other potential sources. Davis: Food and Health Institute, University of California, Davis; 2022.1-35.
- Ramírez-Pulido B, Bas-Bellver C, Betoret N, et al. Valorization of Vegetable Fresh-Processing Residues as Functional Powdered Ingredients: A Review on the Potential Impact of Pretreatments and Drying Methods on Bioactive Compounds and Their Bioaccessibility. Front Sustain Food Syst. 2021;5:654313. DOI: https://doi.org/10.3389/fsufs.2021.654313
CrossRef - Duarte S, Harasym J, Sánchez-García J, et al. Suitability of Almond Bagasse Powder as a Wheat Flour Substitute in Biscuit Formulation. J Food Qual. 2024;2024(1):7152554. DOI: https://doi.org/10.1155/2024/7152554
CrossRef - Duarte S, Sánchez-García J, Harasym J, et al. Enrichment of Rice Flour with Almond Bagasse Powder: The Impact on the Physicochemical and Functional Properties of Gluten-Free Bread. Foods. 2025;14(13):2382. DOI: https://doi.org/10.3390/foods14132382
CrossRef - Horwitz W, Latimer GW. Official Methods of Analysis of AOAC INTERNATIONAL. 18th ed. Maryland, USA: AOAC International; 2005.
- House JD, Hill K, Neufeld J, et al. Determination of the protein quality of almonds (Prunus dulcis ) as assessed by in vitro and in vivo methodologies. Food Sci Nutr. 2019;7(9):2932-2938. DOI: https://doi.org/10.1002/fsn3.1146
CrossRef - BeMiller JN. Carbohydrate analysis. In: Nielsen SS. Food Analysis. 5th ed. Cham: Springer; 2017:333-360.
CrossRef - Satpathy L, Pradhan N, Dash D, et al. Quantitative Determination of Vitamin C Concentration of Common Edible Food Sources by Redox Titration Using Iodine Solution. Lett Appl NanoBioSci. 2021;10(3):2361-2369. DOI: https://doi.org/10.33263/LIANBS103.23612369
CrossRef - Ramos AH, Rockenbach BA, Ferreira CD, et al. Characteristics of Flour and Starch Isolated from Red Rice Subjected to Different Drying Conditions. Starch – Stärke. 2019;71(7-8):1800257. DOI: https://doi.org/10.1002/star.201800257
CrossRef - Xu A, Guo K, Liu T, et al. Effects of Different Isolation Media on Structural and Functional Properties of Starches from Root Tubers of Purple, Yellow and White Sweet Potatoes. Molecules. 2018;23(9):2135. DOI: https://doi.org/10.3390/molecules23092135
CrossRef - Chen CJ, Shen YC, Yeh AI. Physico-Chemical Characteristics of Media-Milled Corn Starch. J Agric Food Chem. 2010;58(16):9083-9091. DOI: https://doi.org/10.1021/jf1020945
CrossRef - Voss GB, Rodríguez-Alcalá LM, Valente LMP, et al. Impact of different thermal treatments and storage conditions on the stability of soybean byproduct (okara). J Food Meas Charact. 2018;12(3):1981-1996. DOI: https://doi.org/10.1007/s11694-018-9813-5
CrossRef - Dhakal S, Pandey D, Van Der Heide ME, et al. Effect of different drying methods on the nutritional composition and phenolic compounds of the brown macroalga, Fucus vesiculosus (Fucales, Phaeophyceae). J Appl Phycol. 2024;36(6):3649-3663. DOI: https://doi.org/10.1007/s10811-024-03343-6
CrossRef - Puente L, Vega-Gálvez A, Fuentes I, et al. Effects of drying methods on the characterization of fatty acids, bioactive compounds and antioxidant capacity in a thin layer of physalis (Physalis peruviana) pulp. J Food Sci Technol. 2021;58(4):1470-1479. DOI: https://doi.org/10.1007/s13197-020-04659-0
CrossRef - El Hosry L, Elias V, Chamoun V, et al. Maillard Reaction: Mechanism, Influencing Parameters, Advantages, Disadvantages, and Food Industrial Applications: A Review. Foods. 2025;14(11):1881. DOI: https://doi.org/10.3390/foods14111881
CrossRef - Moraes S, Pereira A, Almeida V, et al. Coconut Cake in Diets for Quail in the Laying Phase. Braz J Poult Sci. 2020;22(1):eRBCA-2018-0970. DOI: https://doi.org/10.1590/1806-9061-2018-0970
CrossRef - Afoakwah N.A, Owusu J, Owusu V. Characteristics of Coconut (Cocos nucifera) Flour and Its Application in Cake. Asian Food Sci J. 2019;13(1):1-11. DOI: https://doi.org/10.9734/afsj/2019/v13i130095
CrossRef - Osei ED, Amotoe‐Bondzie A, Ataa Pokuah A, et al. Cashew Apple Pomace: Chemical Composition and Applications in Functional Food Product Development—A Review. Food Sci Nutr. 2025;13(4):e70185. DOI: https://doi.org/10.1002/fsn3.70185
CrossRef - Helstad A, Marefati A, Ahlström C, et al. High-Pressure Pasteurization of Oat Okara. Foods. 2023;12(22):4070. DOI: https://doi.org/10.3390/foods12224070
CrossRef - Li B, Qiao M, Lu F. Composition, Nutrition, and Utilization of Okara (Soybean Residue). Food Rev Int. 2012;28(3):231-252. DOI: https://doi.org/10.1080/87559129.2011.595023
CrossRef - Roncero JM, Álvarez-Ortí M, Pardo-Giménez A, et al. Review about Non-Lipid Components and Minor Fat-Soluble Bioactive Compounds of Almond Kernel. Foods. 2020;9(11):1646. DOI: https://doi.org/10.3390/foods9111646
CrossRef - Sanahuja AB, Maestre Pérez SE, Grané Teruel N, et al. Variability of Chemical Profile in Almonds (Prunus dulcis) of Different Cultivars and Origins. Foods. 2021;10(1):153. DOI: https://doi.org/10.3390/foods10010153
CrossRef - Lee S, Choi Y, Jeong HS, et al. Effect of different cooking methods on the content of vitamins and true retention in selected vegetables. Food Sci Biotechnol. 2018;27(2):333-342. DOI: https://doi.org/10.1007/s10068-017-0281-1
CrossRef - Kinyi HW, Tirwomwe M, Ninsiima HI, et al. Effect of Cooking Method on Vitamin C Loses and Antioxidant Activity of Indigenous Green Leafy Vegetables Consumed in Western Uganda. Int J Food Sci. 2022;2022:2088034. DOI: https://doi.org/10.1155/2022/2088034
CrossRef - Taylor H, Webster K, Gray AR, et al. The effects of ‘activating’ almonds on consumer acceptance and gastrointestinal tolerance. Eur J Nutr. 2018;57(8):2771-2783. DOI: https://doi.org/10.1007/s00394-017-1543-7
CrossRef - Kumari S, Gray AR, Webster K, et al. Does ‘activating’ nuts affect nutrient bioavailability? Food Chem. 2020;319:126529. DOI: https://doi.org/10.1016/j.foodchem.2020.126529
CrossRef - Nowshin H, Devnath K, Begum AA, et al. Effects of soaking and grinding conditions on anti-nutrient and nutrient contents of soy milk. J Bangladesh Agric Univ. 2018;16(1):158-163. DOI: https://doi.org/10.3329/jbau.v16i1.36497
CrossRef - De Angelis E, Bavaro SL, Forte G, et al. Heat and Pressure Treatments on Almond Protein Stability and Change in Immunoreactivity after Simulated Human Digestion. Nutrients. 2018;10(11):1679. DOI: https://doi.org/10.3390/nu10111679
CrossRef - Cuadrado C, Sanchiz Á, Linacero R. Nut Allergenicity: Effect of Food Processing. Allergies. 2021;1(3):150-162. DOI: https://doi.org/10.3390/allergies1030014
CrossRef - Tang J, Wichers HJ, Hettinga KA. Heat-induced unfolding facilitates plant protein digestibility during in vitro static infant digestion. Food Chem. 2022;375:131878. DOI: https://doi.org/10.1016/j.foodchem.2021.131878
CrossRef - Okunade OA, Arinola O. Physicochemical Properties of Native and Heat Moisture Treated Starches of White and Red Cocoyam (Colocasia esculenta) Varieties. Turk J Agric – Food Sci Technol. 2021;9(6):1195-1200. DOI: https://doi.org/10.24925/turjaf.v9i6.1195-1200.2701
CrossRef - Fonseca LM, Halal SLME, Dias ARG, et al. Physical modification of starch by heat-moisture treatment and annealing and their applications: A review. Carbohydr Polym. 2021;274:118665. DOI: https://doi.org/10.1016/j.carbpol.2021.118665
CrossRef - Schmidt C, Brunner M, Berger C, et al. Solubility and swelling of soils from native starch. Int J Food Sci Technol. 2022;57(10):6755-6762. DOI: https://doi.org/10.1111/ijfs.15911
CrossRef - Li J, Huang Y, Gao M, et al. Shrinkage properties of porous materials during drying: a review. Front Mater. 2024;11:1330599. DOI: https://doi.org/10.3389/fmats.2024.1330599
CrossRef - Tejeda-Miramontes JP, Espinoza-Paredes BC, Zatarain-Palffy A, et al. Process Modeling and Convective Drying Optimization of Raspberry Pomace as a Fiber-Rich Functional Ingredient: Effect on Techno-Functional and Bioactive Properties. Foods. 2024;13(22):3597. DOI: https://doi.org/10.3390/foods13223597
CrossRef - Chen C, Liao C, Wongso I, et al. Drying and disinfection of off-ground harvested almonds using step-down temperature hot air heating. LWT. 2021;152:112282. DOI: https://doi.org/10.1016/j.lwt.2021.112282
CrossRef - Chen C, Venkitasamy C, Zhang W, et al. Effect of step-down temperature drying on energy consumption and product quality of walnuts. J Food Eng. 2020;285:110105. DOI: https://doi.org/10.1016/j.jfoodeng.2020.110105
CrossRef - Pérez‐Rodríguez F, Carrasco E, Valero A. Impact of dehydration and drying operations on the microbial ecology of foods. In: Souza Sant’Ana A. Quantitative Microbiology in Food Processing: Modeling the Microbial Ecology. Wiley; 2017:160-175.
CrossRef - Roncero JM, Álvarez-Ortí M, Pardo-Giménez A, et al. Virgin almond oil: Extraction methods and composition. Grasas Aceites. 2016;67(3):e143. DOI: http://dx.doi.org/10.3989/gya.0993152
CrossRef - Han Y, Zheng Y, Li S, et al. Effects of Drying Process with Different Temperature on the Nutritional Qualities of Walnut (Juglans regia ). Food Sci Technol Res. 2019;25(2):167-177. DOI: https://doi.org/10.3136/fstr.25.167
CrossRef
- Özcan MM. A review on some properties of almond: ımpact of processing, fatty acids, polyphenols, nutrients, bioactive properties, and health aspects. J Food Sci Technol. 2023;60(5):1493-1504. DOI: https://doi.org/10.1007/s13197-022-05398-0
CrossRef - Capuano E, Pellegrini N, Ntone E, et al. In vitro lipid digestion in raw and roasted hazelnut particles and oil bodies. Food Funct. 2018;9(4):2508-2516. DOI: https://doi.org/10.1039/c8fo00389k
CrossRef - Csakvari AC, Lupitu A, Bungău S, et al. Fatty acids profile and antioxidant activity of Almond oils obtained from six Romanian varieties. Farmacia. 2019;67(5):882-887. DOI: https://doi.org/10.31925/farmacia.2019.5.19
CrossRef
Abbreviations List
AOAC – Association of Official Analytical Chemists
FAMEs – Fatty acid methyl esters
GC-FID – Gas chromatography flame ionization detector
IDF – Insoluble dietary fiber
LDL – Low-density lipoprotein
MUFA – Monounsaturated fatty acids
PCA – Plate count agar
PDA – Potato dextrose agar
SD – Standard deviation
TPC – Total plate count
W1 – Supernatant dried weight
Wd – Dry weight
Ws – Sediment weight
YMP – Yeast, mold populations













