Ultrasound Pretreatment Effect on Mass Transfer in Osmotic Dehydration of Mammea Americana
1Faculty of Food Industry Engineering and Biotechnology, National University of Frontera, Sullana, Peru.
2Faculty of Economic and Environmental Sciences, National University of Frontera, Sullana, Peru.
3Faculty of Chemical Engineering and Food Industries, Pedro Ruiz Gallo National University, Lambayeque, Peru.
4Faculty of Social Sciences and Education, National University of Piura, Piura, Peru.
5Faculty of Veterinary Medicine and Animal Science, National University of San Luis Gonzaga, Ica, Peru.
Corresponding Author E-mail:msanchezch@unf.edu.pe
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.3.30
ABSTRACT:Ultrasound technology has gained increasing attention in the food sector as an effective tool to enhance product quality and process efficiency. In osmotic dehydration (OD), ultrasound acts as a pretreatment that accelerates mass transfer by inducing cavitation and microstructural changes in plant tissues. This study aimed to evaluate the effect of ultrasound pretreatment on mass transfer during the osmotic dehydration of mamey (Mammea americana), a tropical fruit with limited prior research in this context. Fruit samples were exposed to ultrasound frequencies of 28, 37, and 80 kHz for 20 and 30 minutes, followed by osmotic dehydration in 50 °Brix sucrose syrup at 35 ± 1 °C for 120 minutes. Statistical analysis (ANOVA, p < 0.05) showed that both frequency and the frequency–time interaction significantly affected water loss (WL) and weight reduction (WR), while pretreatment time alone was not significant. Treatments at 37 kHz (T2: 20 min and T5: 30 min) achieved the highest mass transfer values, with WR = 31.87%, WL = 40.19%, and solid gain (SG) = 6.08%. In contrast, samples treated at 80 kHz showed reduced dehydration efficiency. These findings demonstrate that applying ultrasound at 37 kHz for 20 minutes optimizes the osmotic dehydration of mamey by enhancing mass transfer efficiency. The results contribute to the understanding of ultrasound-assisted OD processes and support its application as a sustainable process intensification strategy for tropical fruit preservation.
KEYWORDS:Mamey; Mammea americana; Osmodehydration; Osmotic dehydration; Ultra-sound; Ultrasonic bath
Introduction
Currently there is a marked trend in the consumption of minimally processed foods preserving their nutritional content, a trend that is linked to the growing concern of consumers about the use of preservatives and additives in food. Regarding the processing of fruits, these are acquired in their natural state, with no additional processes except washing, however, they are highly perishable, due to their high-water content, which makes them susceptible to the proliferation of microorganisms and the activation of enzymes that accelerate their deterioration.1 Therefore, the search for a method to preserve fruits for long periods of time, without losing their nutrients and without the use of preservation additives, is necessary.
In this context, the different preservation methods come under consideration, with dehydration being the most suitable method according to the customer’s expectations. This method allows the shelf life of the food to be extended by reducing its moisture content, preventing microorganisms from using it for deterioration.2 Conventional drying techniques such as hot-air drying entail high energy consumption because they require elevated temperatures and long processing times. Moreover, the use of high temperatures diminishes the nutritional and sensory quality of foods.3 An alternative that reduces energy use and shortens drying time is osmotic dehydration, a partial dehydration process that operates without high temperatures and can lower moisture content by up to 50 % within two hours.4
The action of immersing food in hypertonic solutions is called osmotic dehydration (OD), and is characterized as a mass transfer process where there is a loss of water (WL) and a gain of solids (SG) from the solution.5 Regarding process time, the highest mass transfer rates occur during the first two hours of processing, however, it has been shown that the longer the immersion time, the higher the water loss and solids gain.6,7
For fruit processing the most commonly used osmotic agent is sucrose8 and it has been shown that increasing the concentration and temperature of syrups results in increased mass transfer.7 However, it should be noted that it is recommended to use temperatures for syrups below 50°C, because above this temperature certain nutrients such as vitamins are lost.9 On the other hand, fruit immersion in syrups must have a proportion that allows maintaining high osmotic concentrations during the whole process, so fruit: osmotic solution ratios between 1:4 and 1:25 have been studied; however, high ratios are not recommended since higher ratios tend to generate waste of the osmotic solution.10–12 An additional key factor in osmotic dehydration is the agitation of the solution, which prevents the development of diluted zones and helps to maintain a constant concentration throughout the process.13
In recent years, osmotic dehydration as a process has also developed to such an extent that several authors have proposed to use it together with methods such as ultrasound, vacuum pulses, high and low pressures, either before or during the process, in order to further improve mass transfer.14
The effect of ultrasound as a technique to assist the osmotic dehydration process has been studied in several fruits.15–18 This technology consists of the application of waves generated by mechanical vibrations with a frequency between 18 kHz and 100 kHz, which is the limit of sound that a human can perceive.19 These elastic waves generate cavitation when they propagate in the liquid medium in which the food to be treated is found.20 Cavitation refers to the generation and implosion of very small gas bubbles in the liquid medium when ultrasound waves impinge on it.21
On the other hand, ultrasound waves induce rapid alternating compressions and expansions in the treated material—an effect known as the “sponge” phenomenon; the stress generated by these waves can exceed the surface tension that retains water within the capillary structures of the food, thereby creating microchannels that facilitate moisture removal. 22
Studies on ultrasound assistance in osmotic dehydration have used distilled water as a medium to apply ultrasound directly to the feed and then carry out dehydration by osmosis.23–25 Other authors, proposed to perform the treatment with an ultrasonic bath, also with distilled water, but during osmotic dehydration, in this way the application is indirect, i.e., the ultrasound is generated in the water and transferred to the osmotic solution contained in beakers.26,27
Although osmotic dehydration is a well-established technique for reducing water activity and extending shelf life, the limitation in process speed, particularly in perishable fruits such as mamey (Mammea americana), remains a key challenge. While ultrasonic pretreatment has been shown to facilitate diffusion, the current literature has not quantified the influence of this treatment on the mass transfer coefficients (WL, WR, and SG) specific to mamey under osmotic dehydration conditions.
In this context, and considering the high perishability of mamey, the primary objective of this study is to rigorously evaluate the influence of ultrasonic pretreatment on improving mass transfer kinetics, quantifying Weight reduction (WR), Solid Gain (SG), and Water Loss (WL) during osmotic dehydration.
Materials and Methods
The experimental procedure is divided into two parts, the first part where ultrasound was applied to the mamey (Mammea americana) slices and the second part referred to the osmotic dehydration treatment. The methodology was obtained from the work of Quinde-Montero,28 with some modifications, obtaining the stages shown in Figure 1 and described below.
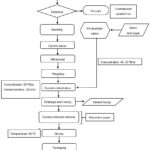 |
Figure 1: Process flowchart illustrating ultrasound as a pretreatment before osmotic dehydration of mamey (Mammea americana) slices. |
Sample Preparation
Fresh Mammea americana fruits were purchased from a local market in Sullana, Peru. Only fruits at commercial maturity and free from over-ripeness, bruising, or visible defects were selected. The fruits were washed under running water to remove surface impurities and disinfected in a 50-ppm chlorinated water solution for 10 minutes. After disinfection, the peel was manually removed, and the edible pulp was cut into uniform cubes of 10 × 10 × 10 mm using a stainless-steel dicer to ensure geometric consistency among samples.
Control sample
To ensure the validity and adequate interpretability of the experimental design, it was decided to include a Control Group (identified as T0). This group was established as the baseline reference for comparison, representing the conventional osmotic dehydration process without the application of any physical improvement techniques. The mamey samples intended for the control were prepared with the same geometric characteristics as the treatment groups and were osmotically dehydrated in a solution with a concentration of 50ºBrix for 120 minutes, strictly omitting the ultrasonic pretreatment.
The contrast between the kinetic results obtained by ultrasonic treatments and the T0 values was fundamental in quantifying the increase in process efficiency and conclusively attributing any observed improvement to the direct effect of pretreatment.
Ultrasound pre-treatment
Ultrasound pretreatment was carried out following the methodology described by Prithani and Dash,29 using distilled water as the ultrasound transmission medium. Mammea americana samples were immersed in 600 mL glass beakers filled with distilled water and treated in an ultrasonic bath (model P30H, manufacturer Elma Schmidbauer GmbH, Germany) operating at frequencies of 28, 37, and 80 kHz with an effective power of 90 W. Pretreatments were applied for 20 and 30 minutes at a constant temperature of 30 ± 1 °C, controlled by the bath’s built-in thermostat. The temperature of the samples was recorded at the end of each treatment to monitor potential thermal effects generated by the ultrasound waves.
Osmotic Dehydration
Immediately after pretreatment, the mamey cubes were transferred into 50 °Brix sucrose syrup maintained at 35 ± 1 °C using a fruit-to-syrup ratio of 1:4 (w/v). Osmotic dehydration was conducted for 120 minutes in sealed 300 mL glass jars, which were placed in a thermostatic water bath with gentle agitation every five minutes to minimize boundary layer resistance and prevent syrup dilution.
Mass transfer was evaluated based on water loss (WL), weight reduction (WR), and solid gain (SG), calculated according to Equations (1-3): 30
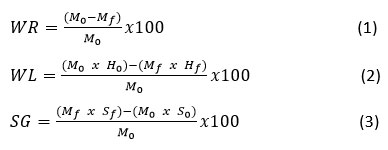
where and are the initial and final sample masses, and are the initial and final moisture contents, and and are the soluble solids (°Brix) before and after OD, respectively. Moisture content was determined using a moisture analyzer (model MX-50, A&D Co., Japan), and soluble solids were measured with a handheld digital refractometer (model HI96801, HANNA, Italia).
Hot air drying
Following osmotic dehydration, samples were subjected to convective drying in a hot air tray dryer at 50 °C until reaching a final moisture content below 15%, in order to prevent microbial or fungal proliferation. 31 The final moisture content was confirmed gravimetrically.
Statistical analysis
The experimental design (Table 1) followed a completely randomized design (CRD) with a 2 × 3 factorial arrangement, where ultrasound exposure time (20 and 30 min) and frequency (28, 37, and 80 kHz) were the independent factors. The response variables were WR, WL, and SG. Each treatment was performed in triplicate (n = 3). Data were analyzed using one-way ANOVA, and mean comparisons were performed using Tukey’s test (p < 0.05) with statistical software (SPSS version 26.0.0.0., IBM Corp., USA).
Table 1: 2×3 experimental design
| Treatment | Time (min) | Frequency (kHz) |
| T1 | 20 | 28 |
| T2 | 20 | 37 |
| T3 | 20 | 80 |
| T4 | 30 | 28 |
| T5 | 30 | 37 |
| T6 | 30 | 80 |
Results
Ultrasound Pretreatment
Mamey (Mammea americana) cubes were subjected to an ultrasonic bath at three ultrasound frequencies (28 kHz, 37 kHz and 80 kHz) and a power of 90 kW. It was observed that the use of ultrasound generated a weight gain between 2% – 6.1%, and thus a gain of water by the feed.
Figure 2 shows a comparison of the moisture content of fresh mamey (Mammea americana) and after being treated with ultrasound, where it can be observed that the first 12 samples from the treatments with 28 kHz and 37 kHz showed an increase in moisture compared to the fresh samples, while the last six samples from the 80 kHz treatment showed a slight decrease in moisture. Another behavior observed during ultrasound pretreatment was the increase in temperature, reaching up to 60ºC when frequencies of 80 kHz were used.
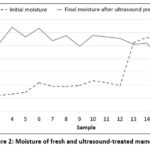 |
Figure 2: Moisture of fresh and ultrasound-treated mamey |
Osmotic Dehydration
Mamey samples without ultrasound pretreatment, were osmotically dehydrated at a concentration of 50 ºBrix for 120 minutes, generated weight reduction (WR), water loss (WL) and solids gain (SG) values of 28.66%, 31.12%, 2.74%.
Table 2. Summary of WR, WL, and SG values in the different proposed treatments
| Treatment | WR (%) | WL (%) | SG (%) |
| No US | 28.66 ± 0.50d | 31.12 ± 1.77b, c | 2.74 ± 1. 12a |
| T1 (20 min-28 kHz) | 26.32 ± 1.04c, d | 34.55 ± 0.51d | 4.64 ± 0.65a, b |
| T2 (20 min-37 kHz) | 31.87 ± 1.61e | 40.19 ± 0.70e | 6.08 ± 0.20b |
| T3 (20 min-80 kHz) | 18.03 ± 1. 29a | 26.51 ± 1. 53a | 6.69 ± 1.04b |
| T4 (30 min-28 kHz) | 25.45 ± 1.04b, c | 33.94 ± 1.05c, d | 9.74 ± 0.80c |
| T5 (30 min-37 kHz) | 29.32 ± 0.43d, e | 38.18 ± 0.35e | 4.89 ± 0.35b |
| T6 (30 min-80 kHz) | 23.30 ± 1.31b | 30.39 ± 1.05b | 5.28 ± 0.58b |
The analysis of variance (ANOVA) determined that the frequency factor and the Frequency-Time interaction were significant for weight reduction (WR) with p-values <0.05, while time, with a p-value of 0.313, was not significant. Additionally, no significant differences were found between osmodehydrated samples without pretreatment and those pretreated at a frequency of 37 kHz for 30 minutes.
Table 2 shows the average results obtained for the weight reduction (WR) variable during the osmotic dehydration process of mamey pretreated with ultrasound. The highest weight reduction of 31.87% were observed in the samples pretreated with ultrasonic baths at 37 kHz, while the lowest losses were obtained using the highest frequency of 80 kHz at 18.03%.
Regarding time, for samples pretreated at 28 kHz and 37 kHz, it was observed that increasing the immersion duration to 30 minutes resulted in lower weight reduction, whereas for the sample pretreated at 80 kHz, increasing the time caused an increase in weight reduction (Figure 3).
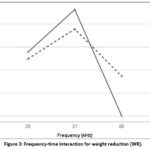 |
Figure 3: Frequency-time interaction for weight reduction (WR). |
The analysis of variance (ANOVA) for water loss (WL) determined that the frequency factor and the Frequency-Time interaction were significant for water loss (WL) with p-values <0.05, while the time, with a p-value of 0.365, was not significant. Furthermore, it was found that there are no significant differences between osmodehydrated samples without pretreatment and those pretreated at a frequency of 37 kHz for 30 minutes.
Table 2 shows the average results obtained for the water loss (WL) variable during the osmotic dehydration process of mamey pretreated with ultrasound. The highest water losses of 40.19% were observed in samples pretreated with ultrasonic baths at 37 kHz, while the lowest losses were obtained using the highest frequency of 80 kHz at 26.51%. Regarding time, for samples pretreated at 28 kHz and 37 kHz, it was observed that increasing the immersion duration to 30 minutes resulted in lower water losses, whereas for the sample pretreated at 80 kHz, increasing the time caused an increase in water loss.
Figure 4 illustrates the interaction between frequency and time factors on water loss (WL). It is evident that the highest water losses occurred in samples pretreated with an ultrasonic bath at 37 kHz. Additionally, for samples treated at 28 kHz and 37 kHz, increasing the ultrasound application time led to a decrease in the percentage of water loss. Conversely, for the 80 kHz treatment, increasing the pretreatment time caused an increase in the water loss percentage.
 |
Figure 4: Frequency-time interaction for Water Loss (WL). |
The analysis of variance (ANOVA) for solid gain (SG) revealed that the factors frequency, time, and their interaction were significant, with p-values less than 0.05. Table 2 shows the average results obtained for the solid gain (SG) variable during the osmotic dehydration process of mamey pretreated with ultrasound. The highest solid gains of 9.74% and the lowest of 4.64% were observed in samples pretreated with ultrasonic baths at 28 kHz. Regarding time, for samples pretreated at 28 kHz, increasing the immersion duration to 30 minutes resulted in greater solid gains, whereas for samples pretreated at frequencies of 37 kHz and 80 kHz, increasing the time led to a reduction in solid gain.
Figure 5 displays the graph illustrating the interaction between frequency and time factors on solid gain (SG). It shows that the highest solid gains were achieved in samples pretreated with an ultrasonic bath at 28 kHz. Conversely, for samples pretreated at 37 kHz and 80 kHz, increasing the ultrasound application time led to a decrease in the solid gain percentage, while for the 28 kHz treatment, increasing the pretreatment time resulted in an increase in solid gain.
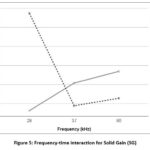 |
Figure 5: Frequency-time interaction for Solid Gain (SG). |
Discussion
Ultrasound pretreatment
The results obtained from Mammea americana samples pretreated with ultrasound at 28 and 37 kHz using distilled water as the medium showed an increase in water content and a decrease in solids. This behavior was also reported by Fernandes et al.32 and García-Noguera et al.33 in melon and strawberry treated at similar frequencies. This phenomenon can be explained by water diffusion into the tissue caused by the concentration gradient between the fruit and the aqueous medium,34 which favors water imbibition instead of dehydration during the initial stage.
In fruits with high initial moisture content (85–90%), ultrasound may cause water expulsion due to the so-called sponge effect, resulting from the cyclic compression and expansion of plant tissue.35 This explains the difference between compact fruits, where ultrasound promotes absorption, and highly porous fruits, where it induces water loss.36
The temperature rise observed after ultrasound treatment agrees with the findings of Fan et al.,37 Kek et al.38 and Pantelidou et al.39 According to Bermúdez-Aguirre and Barbosa-Cánovas40 and Luchese et al.,41 this thermal increase results from the conversion of acoustic energy into heat due to molecular friction caused by cavitation, which alters the diffusivity of water and solutes during osmotic dehydration.
Mass Transfer
Analysis of variance (ANOVA) showed that both frequency and the frequency–time interaction was significant for weight reduction and water loss (WR and WL) (p < 0.05), while treatment time alone was not significant (p > 0.05). This indicates that ultrasound frequency exerts a stronger influence than treatment duration, consistent with Luchese et al.,41 who observed similar behavior in cape gooseberry. Samples treated at 28 and 37 kHz exhibited higher WR and WL values compared with those treated at 80 kHz. This can be attributed to the greater cavitation intensity at low frequencies, where larger bubbles collapse releasing more energy and forming microchannels that facilitate water removal.14,42
At higher frequencies, cavitation becomes less intense and more energy is dissipated in the liquid medium, reducing process efficiency.43,44 Authors like Garcia-Noguera et al.33 reported a similar trend in strawberries, where ultrasound at 25 kHz for 10–20 minutes yielded higher WR and WL values, but prolonged exposure reduced these effects, likely due to tissue re-compaction. This behavior depends on the structural resistance of each fruit matrix.34,45
Regarding solid gain (SG), ultrasound-pretreated samples (4.64% < SG < 9.74%) showed a significant increase compared to non-pretreated samples (2.74%). This can be attributed to enhanced cell permeability induced by cavitation, which facilitates solute diffusion into the tissue.29,46 Similar findings were reported by Luchese et al.,41 Nowacka et al.,47 demonstrating that ultrasound accelerates osmotic impregnation. However, Kek et al.38 suggested applying ultrasound after osmotic dehydration to obtain products with lower sugar content, indicating that the timing of ultrasound application influences mass transfer behavior.
Under similar process conditions (50 °Brix, 120 min), other studies, such as Corzo et al.48 and Lech et al., 49 reported WR and WL values ranging from 20% to 62%, depending on the fruit type. These differences confirm that results cannot be extrapolated among different species, since mass transfer during osmotic dehydration strongly depends on cell microstructure and tissue composition.50,51
In conclusion, the improvement in mass transfer during ultrasound-assisted osmotic dehydration arises from a complex interplay of mechanical disruption, microstructural modification, and physicochemical enhancement of diffusion mechanisms. The technology provides a promising alternative to traditional osmotic treatments, offering higher efficiency, lower processing time, and the potential to preserve key sensory attributes.
Conclusion
The application of ultrasound as a pretreatment in the osmotic dehydration of Mammea americana significantly influenced the mass transfer behavior. Frequencies between 28 and 37 kHz enhanced both water loss and weight reduction, indicating that low-frequency ultrasound effectively promoted cellular disruption and improved diffusional exchange. In contrast, treatment at 80 kHz resulted in a decline in dehydration efficiency, likely due to reduced cavitation intensity at higher frequencies. Regarding solid gain, all ultrasound-pretreated samples exhibited higher solute incorporation compared to the untreated control, confirming the ability of ultrasound to facilitate bidirectional mass transfer. Overall, the optimal parameters for enhancing osmotic dehydration of mamey were 37 kHz and 20 minutes of ultrasound exposure, achieving a weight reduction (WR) of 31.87%, water loss (WL) of 40.19%, and solid gain (SG) of 6.08%.
These findings demonstrate that ultrasound pretreatment is an effective process intensification technique capable of improving osmotic dehydration efficiency while maintaining the structural and nutritional integrity of tropical fruits. The results contribute to a better understanding of how acoustic cavitation can be exploited to optimize food preservation methods, potentially reducing processing time and energy consumption in the food industry. Further studies are recommended to evaluate the combined effect of ultrasound with other pretreatments such as pulsed vacuum or mild heating, as well as to investigate the influence of ultrasonic power, duty cycle, and solvent composition on mass transfer kinetics. In addition, microstructural and sensory analyses should be performed to better correlate physical modifications with the quality attributes of the final product.
Acknowledgement
The authors would like to thank the Research, Development and Innovation Group in Food Industries (GIDIIA) of the National University of Frontera for their support with the use of the osmodehydration equipment for the study.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Milagros Jackeline Rosales-Ruiz: Conceptualization, Writing—original draft,
- Manuel Jesus Sanchez-Chero: Project administration-writing
- Lesly Carolina Flores- Mendoza: Writing—review and editing
- William Rolando Miranda-Zamora: Formal analysis- investigation
- Jose Antonio Sanchez-Chero: Review-data curation
- Abraham Guillermo Ygnacio Santa Cruz: Methodology-writing
- Isidora Concepción Zapata Periche: Methodology
- María Emilia Dávalos-Almeyda: Laboratory testing and safety performance
References
- Moreno J, Simpson R, Pizarro N, et al. Influence of ohmic heating/osmotic dehydration treatments on polyphenol oxidase inactivation, physical properties, and microbial stability of apples (cv. Granny Smith). Innov Food Sci Emerg Technol. 2013;20:198-207. doi:10.1016/j.ifset.2013.06.006.
CrossRef - Goyal MR, Veena N, Watharkar RB. Advances in Food Process Engineering: Novel Processing, Preservation, and Decontamination of Foods. CRC Press; 2023. doi:10.1201/9781003303848.
CrossRef - Atungulu GG, Teh HE, Wang T, et al. Infrared pre-drying and dry-dehulling of walnuts for improved processing efficiency and product quality. Appl Eng Agric. 2013;29(6):961-971. doi:10.13031/aea.29.10195.
CrossRef - Bekele Y, Ramaswamy H. Going beyond conventional osmotic dehydration for quality advantage and energy savings. Ethiop J Appl Sci Technol. 2010;1(1):1-15.
- Abrahão FR, Corrêa JLG. Osmotic dehydration: more than water loss and solid gain. Crit Rev Food Sci Nutr. 2023;63(17):2970-2989. doi:10.1080/10408398.2021.1983764.
CrossRef - Manzoor A, Khan MA, Mujeebu MA, et al. Comparative study of microwave-assisted and conventional osmotic dehydration of apple cubes at a constant temperature. J Agric Food Res. 2021;5:100176. doi:10.1016/j.jafr.2021.100176.
CrossRef - Ahmed I, Qazi IM, Jamal S. Developments in osmotic dehydration technique for the preservation of fruits and vegetables. Innov Food Sci Emerg Technol. 2016;34:29-43. doi:10.1016/j.ifset.2016.01.003.
CrossRef - Sravani DV, Saxena D. A mini review on osmotic dehydration of fruits and vegetables. Pharma Innov J. 2021;10(7).
- Cao H, Zhang M, Mujumdar AS, Du WH, et al. Optimization of osmotic dehydration of kiwifruit. Dry Technol. 2006;24(1):89-94. doi:10.1080/07373930500538741.
CrossRef - Rastogi NK, Raghavarao KSMS. Mass transfer during osmotic dehydration of pineapple: considering Fickian diffusion in cubical configuration. Food Sci Technol Int. 2004;37(1):43-47. doi:10.1016/S0023-6438(03)00131-2.
CrossRef - Rastogi NK, Niranjan K. Enhanced mass transfer during osmotic dehydration of high-pressure treated pineapple. J Food Sci. 1998;63(3):508-511. doi:10.1111/j.1365-2621.1998.tb15774.x.
CrossRef - Rastogi NK, Raghavarao KSMS. Effect of temperature and concentration on osmotic dehydration of coconut. Food Sci Technol Int. 1994;27(6):564-567. doi:10.1006/fstl.1994.1110.
CrossRef - Tortoe C. A review of osmodehydration for food industry. Afr J Food Sci. 2010;4(6):303-324.
- Nowacka M, Tylewicz U, Laghi L, et al. Effect of ultrasound treatment on the water state in kiwifruit during osmotic dehydration. Food Chem. 2014;144:18-25. doi:10.1016/j.foodchem.2013.05.129.
CrossRef - Nowacka M, Tylewicz U, Romani S, et al. Influence of ultrasound-assisted osmotic dehydration on the main quality parameters of kiwifruit. Innov Food Sci Emerg Technol. 2017;41:71-78. doi:10.1016/j.ifset.2017.02.002.
CrossRef - Fernandes FAN, Braga TR, Silva EO, et al. Use of ultrasound for dehydration of mangoes (Mangifera indica L.): kinetic modeling of ultrasound-assisted osmotic dehydration and convective air-drying. J Food Sci Technol. 2019;56(4):1793-1800. doi:10.1007/s13197-019-03622-y.
CrossRef - Salehi F, Cheraghi R, Rasouli M. Mass transfer kinetics (soluble solids gain and water loss) of ultrasound-assisted osmotic dehydration of apple slices. Sci Rep. 2022;12:15392. doi:10.1038/s41598-022-19826-w.
CrossRef - Amami E, Khezami W, Mezrigui S, et al. Effect of ultrasound-assisted osmotic dehydration pretreatment on the convective drying of strawberry. Ultrason Sonochem. 2017;36:286-300. doi:10.1016/j.ultsonch.2016.12.007.
CrossRef - Singla M, Sit N. Application of ultrasound in combination with other technologies in food processing: a review. Ultrason Sonochem. 2021;73:105506. doi:10.1016/j.ultsonch.2021.105506.
CrossRef - De Castro MDL, Capote FP. Introduction: fundamentals of ultrasound and basis of its analytical uses. Tech Instrum Anal Chem. 2007;26:1-34. doi:10.1016/S0167-9244(07)80017-5.
CrossRef - Chen F, Zhang M, Yang Ch. Application of ultrasound technology in processing of ready-to-eat fresh food: a review. Ultrason Sonochem. 2020;63:104953. doi:10.1016/j.ultsonch.2019.104953.
CrossRef - Luchese CL, Gurak PD, Marczak LDF. Osmotic dehydration of Physalis: influence of ultrasound pretreatment. Food Eng Rev. 2015;7(2):193-197. doi:10.1007/s12393-014-9086-7.
CrossRef - Bozkir H, Ergün AR. Effect of sonication and osmotic dehydration applications on the hot-air drying kinetics and quality of persimmon. LWT. 2020;131:109704. doi:10.1016/j.lwt.2020.109704.
CrossRef - Fernandes FAN, Gallão MI, Rodrigues S. Effect of osmotic dehydration and ultrasound pretreatment on cell structure: melon dehydration. LWT. 2008;41(4):604-610. doi:10.1016/j.lwt.2007.05.007.
CrossRef - Prithani R, Dash KK. Mass transfer modelling in ultrasound-assisted osmotic dehydration of kiwifruit. Innov Food Sci Emerg Technol. 2020;64:102407. doi:10.1016/j.ifset.2020.102407.
CrossRef - Kroehnke J, Szadzińska J, Radziejewska-Kubzdela E, et al. Osmotic dehydration and convective drying of kiwifruit (Actinidia deliciosa): influence of ultrasound on process kinetics and product quality. Ultrason Sonochem. 2021;71:105377. doi:10.1016/j.ultsonch.2020.105377.
CrossRef - Nowacka M, Tylewicz U, Tappi S, et al. Ultrasound-assisted osmotic dehydration of organic cranberries (Vaccinium oxycoccus): study on quality parameters evolution during storage. Food Control. 2018;93:40-47. doi:10.1016/j.foodcont.2018.05.005.
CrossRef - Quinde-Montero HJ, Sanchez-Chero MJ, Flores-Mendoza LC, et al. Osmotic dehydration pretreatment with panela for the production of apple snacks. Curr Res Nutr Food Sci J. 2025;13(2):969-983. doi:10.12944/crnfsj.13.2.30.
CrossRef - Prithani R, Dash KK. Mass transfer modelling in ultrasound-assisted osmotic dehydration of kiwifruit. Innov Food Sci Emerg Technol. 2020;64:102407. doi:10.1016/j.ifset.2020.102407.
CrossRef - Bozkir H, Ergün AR, Serdar E, et al. Influence of ultrasound and osmotic dehydration pretreatments on drying and quality properties of persimmon fruit. Ultrason Sonochem. 2019;54:135-141. doi:10.1016/j.ultsonch.2019.02.006.
CrossRef - Badui DS. Química de los Alimentos. 5th ed. Pearson Educación; 2013.
- Fernandes FAN, Gallão MI, Rodrigues S. Effect of osmotic dehydration and ultrasound pretreatment on cell structure: melon dehydration. LWT. 2008;41(4):604-610. doi:10.1016/j.lwt.2007.05.007.
CrossRef - Garcia-Noguera J, Oliveira FIP, Gallão MI, et al. Ultrasound-assisted osmotic dehydration of strawberries: effect of pretreatment time and ultrasonic frequency. Dry Technol. 2010;28(2):294-303. doi:10.1080/07373930903530402.
CrossRef - Rastogi N. Opportunities and challenges in application of ultrasound in food processing. Crit Rev Food Sci Nutr. 2011;51(8):705-722. doi:10.1080/10408391003770583.
CrossRef - Knorr D, Zenker M, Heinz V, et al. Applications and potential of ultrasonics in food processing. Trends Food Sci Technol. 2004;15(5):261-266. doi:10.1016/j.tifs.2003.12.001.
CrossRef - Fernandes FAN, Gallão MI, Rodrigues S. Effect of osmotic dehydration and ultrasound pretreatment on cell structure: melon dehydration. LWT. 2008;41(4):604-610. doi:10.1016/j.lwt.2007.05.007.
CrossRef - Fan K, Zhang M, Wang W, et al. Osmotic-dehydrofreezing with ultrasound enhancement to improve water status and physicochemical properties of kiwifruit. Int J Refrig. 2020;113:49-57. doi:10.1016/j.ijrefrig.2020.02.013.
CrossRef - Kek SP, Chin NL, Yusof YA. Direct and indirect power ultrasound-assisted pre-osmotic treatments in convective drying of guava slices. Food Bioprod Process. 2013;91(4):495-506. doi:10.1016/j.fbp.2013.05.003.
CrossRef - Pantelidou D, Gerogiannis K, Goula AM, et al. Ultrasound-assisted osmotic dehydration as a method for supplementing potato with unused chokeberry phenolics. Food Bioproc Technol. 2021;14(12):2231-2247. doi:10.1007/s11947-021-02720-0.
CrossRef - Bermúdez-Aguirre D, Mobbs T, Barbosa-Cánovas GV. Ultrasound applications in food processing. In: Food Engineering Series. Springer; 2011:65-105. doi:10.1007/978-1-4419-7472-3_3.
CrossRef - Luchese CL, Gurak PD, Marczak LDF. Osmotic dehydration of Physalis: influence of ultrasound pretreatment. Food Eng Rev. 2015;7(2):193-197. doi:10.1007/s12393-014-9086-7.
CrossRef - Jambrak AR, Nutrizio M, Djekić I, et al. Internet of nonthermal food processing technologies (IoNTP): food industry 4.0 and sustainability. Appl Sci. 2021;11(2):686. doi:10.3390/app11020686.
CrossRef - Shamaei S, Emam-Djomeh Z, Moini S. Ultrasound-assisted osmotic dehydration of cranberries: effect of finish-drying methods and ultrasonic frequency on textural properties. J Texture Stud. 2012;43(2):133-141. doi:10.1111/j.1745-4603.2011.00323.x.
CrossRef - Oladejo AO, Ma H. Optimisation of ultrasound-assisted osmotic dehydration of sweet potato (Ipomoea batatas) using response surface methodology. J Sci Food Agric. 2016;96(11):3688-3693. doi:10.1002/jsfa.7552.
CrossRef - Rastogi NK, Raghavarao KSMS, Niranjan K. Developments in osmotic dehydration. In: Emerging Technologies for Food Processing. 2004:259-263. doi:10.1016/B978-012676757-5/50011-6.
CrossRef - Chemat F, Rombaut N, Sicaire AG, et al. Ultrasound-assisted extraction of food and natural products: mechanisms, techniques, combinations, protocols, and applications—a review. Ultrason Sonochem. 2017;34:540-560. doi:10.1016/j.ultsonch.2016.06.035.
CrossRef - Nowacka M, Dadan M, Tylewicz U. Current applications of ultrasound in fruit and vegetable osmotic dehydration processes. Appl Sci. 2021;11(3):1269. doi:10.3390/app11031269.
CrossRef - Corzo O, Ramírez O, Bracho N. Aplicación del modelo de Peleg en el estudio de la transferencia de masa durante la deshidratación osmótica de láminas de mamey (Mammea americana L.). Saber. 2008;20(1):87-95.
- Lech K, Michalska A, Wojdyło A, et al. Influence of the osmotic dehydration process on physicochemical properties of osmotic solution. Molecules. 2017;22(12):2246. doi:10.3390/molecules22122246.
CrossRef - Seguí L, Fito PJ, Fito P. Analysis of structure-property relationships in isolated cells during osmotic dehydration treatments: effect of initial structure on cell behaviour. J Food Eng. 2010;99(4):417-423. doi:10.1016/j.jfoodeng.2009.05.019.
CrossRef - Nieto AB, Salvatori DM, Castro MA, et al. Structural changes in apple tissue during glucose and sucrose osmotic dehydration: shrinkage, porosity, density, and microscopic features. J Food Eng. 2004;61(2):269-278. doi:10.1016/S0260-8774(03)00108-0.
CrossRef
Abbreviations List
OD Osmotic dehydration, osmodehydration
SG Solid gain
WL Water loss
WR Weight reduction













