Comparative Analysis of Fumonisin Contamination and Infant Dietary Exposure in Kongwa District, Tanzania: A Sub-Study Within a Two-Arm Cluster-Randomized Trial
1Department of Food Sciences and Biotechnology, Nelson Mandela African Institution of Science and Technology, Arusha, Tanzania.
2Division of Nutritional Sciences, Cornell University, Ithaca, USA
Corresponding Author Email: rosemarykayanda@gmail.com
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.3.12
ABSTRACT:In central Tanzania, maize is frequently consumed in many households as a staple food. Reliance on maize-based complementary foods for infants and young children puts them at risk of Fumonisin (FUM) exposure. This observational study explores whether the provision of low-aflatoxin porridge flour as part of an intervention within a larger cluster randomized control trial, Mycotoxin Mitigation Trial (MMT), had any effect on the intake and exposure to FUM in complementary foods consumed by children aged 12 to 13 months. Sampling was done for randomly selected 12-13-month-old children from 20 of the 52 trial clusters within the two arms of MMT. The households within the intervention arm of the MMT were provided with low aflatoxin blended flour, 4 parts maize, and 1 part ground nuts to feed the index child, every month for 12 months. The Standard of Care (SoC) households were educated on preparing the 4:1 blended flour from locally produced maize and groundnuts. Blended and maize flours consumed by the index child in the past 24 hours were collected for FUM analysis. The samples were analyzed using the Enzyme-Linked Immunosorbent Assay (ELISA) method. Data analysis was done through STATA version 16 software at a 5% significance level. The levels of FUM in blended flour samples were not significantly different between the intervention and the SoC arm p > 0.05. Similarly, differences existed in FUM intake and exposure through blended flour between arms (p>0.05). There were no significant differences in FUM intake and exposure between arms from maize (p>0.05). This study suggests that the provision of low-aflatoxin flour to households in the intervention arm did not impact differences in intake and exposure to FUM across the two arms.
KEYWORDS:Contamination; Exposure; Fumonisin; Infants’ Diets; Maize-Based Food
Introduction
Fumonisin is a type of mycotoxin produced by Fusarium verticillioides and Fusarium proliferatum fungi species.1,2 They are ubiquitous and contaminate several foods that pose detrimental health hazards to humans.3 Fumonisin consists of different forms, such as fumonisin B1 (FUM1), fumonisin B2 (FUM2), and fumonisin B3 (FUM3), with FUM1 being the most toxic.4 Maize is used as a staple food for the majority of the population in Tanzania who live in rural areas. Maize and maize-based products are susceptible to fumonisin contamination.5,6 Due to this fact, fumonisin has been mentioned as one of the challenges affecting foods in Tanzania. A study conducted in each of the four main maize-producing regions of Ruvuma, Kilimanjaro, Iringa, and Tabora estimated the total fumonisin(FUM1 + FUM2) and found contamination of up to 11,048 µg/kg (median: 363 µg/kg) from the samples collected from 120 households.7
Fumonisin is found globally, with particularly high concentrations reported in regions such as Southern Africa and China.8,9 Fumonisin concentrations in “home-grown” corn from Linxiang, China, and areas of the Transkei, southern Africa, are continuously high, and concentrations exceeding 100,000 µg/kg have been occasionally reported.10 The extremely high fumonisin produced in Benin, West Africa, by two species, Fusarium verticillioides (68%) and Fusarium proliferatum (31%), was most commonly isolated from maize with total fumonisin levels ranging from 8240 µg/kg to 16,690 µg/kg.11
Factors promoting the occurrence and growth of fumonisin species include higher temperatures and humid climates.12 Fumonisin climatic conditions regions experience extreme rainfall patterns, temperature, as well as longer durations of drought.5 Fumonisin toxins are predominant in temperate and Mediterranean climatic regions.13 Fumonisin becomes the predominant contaminant of maize and maize-based products.14 Fumonisins’ growth and accumulation are associated with drought and insect stress, and growing hybrids outside their areas of adaptation.15,16
Infants and young children who consume maize-based foods are at a high risk of exposure to fumonisin.17 Fumonisin exposure during the first trimester of pregnancy has been associated with an increased risk of neural tube defects in infants, particularly through the consumption of maize-based foods containing high levels of the toxin.18–21 Children are at greater risk of toxic effects as compared to adults because of their lower body mass and do not have a mature detoxification system.22 The first 1000 days of life, the period from the fetal stage to 2 years old, is characterized by accelerated growth and developmental plasticity.23 This early-life period is very sensitive to any event that alters the programming of the main body functions and represents a window for intervention to improve child health.24 In this regard, pregnant women, fetuses, and infants are particularly vulnerable to exposure to food contaminants.24 Fumonisin from contaminated food can cross the placental barrier and affect fetal health.25
Throughout the world, different countries set regulations to protect clients from fumonisin contamination. The Maximum Tolerable Limit (MTL) in developed countries, including Europe, is 1.0 µg kg-1 body weight per day.26 In developing countries, including the East African community, Tanzania, being one among them, has 2000 µg/kg.27,28 There are no established Maximum Tolerable Limits (MTLs) of fumonisin concentrations specifically for children in low- and middle-income countries.
To assess the causal evidence between aflatoxin exposure and linear growth faltering, a two-arm cluster randomized control trial (Mycotoxins Mitigation Trial; MMT) was conducted in Kongwa District, in Dodoma region, Tanzania. The intervention arm households were provided with low aflatoxin maize and groundnuts (4:1) blended and groundnut flour for the index child every month for 12 months. The comparison or standard of care (SoC) arm participants were educated on preparing the 4:1 blended flour from locally produced maize and groundnuts. During the preparation of low aflatoxin blended flour, rigorous sorting was done to reduce AF in maize and groundnuts. Sorting was also done to ensure that the blended flour provided to households in the intervention arm did not exceed the legal limit for fumonisin in Tanzania. Maize was not provided by the MMT project in all arms but study participants utilized maize acquired at household’s level.
The primary objectives of the sub-study were (i) to assess fumonisin contamination of foods fed to infants and the consequent dietary exposure of the infants based on body weight, and (ii) to compare the infant food contamination and infant dietary exposures among selected households from the two arms of the trial. This observational study aimed at exploring whether the provision of low aflatoxin flour led to differences in intake and exposure to fumonisin among children from households across the two arms of MMT.
Materials and Methods
This sub-study took place in the Kongwa District of Dodoma region among infants recruited in the Mycotoxin Mitigation Trial (Figure 1).
 |
Figure 1: A map of (a) Tanzania showing Dodoma region, (b) Dodoma region showing Kongwa DC, (c) Kongwa district showing wards |
Background and study population
Mycotoxin Mitigation Trial
Children were recruited into the MMT from health facilities. When the infant turned 6 months of age, the intervention was introduced to the mother. Pre-blended porridge flour made of low-aflatoxin maize and groundnut in the ratio of 4:1, and low-aflatoxin groundnut flour were provided to participants in the intervention arm along with the infant feeding education. The low-aflatoxin maize and groundnut flours were processed through a collaboration with a local food producer in Arusha, Tanzania, known as Halisi Products Limited. The threshold for low aflatoxin was 5 µg/kg29 and 2000 µg/kg for fumonisin. Participants in the SoC arm received the same infant feeding education along with the promotion of infant porridge made from household-acquired maize and groundnut flours in the same 4:1 ratio as the intervention arm.
Sub-Study Participants’ Selection
This sub-study was carried out in 20 of the 52 MMT clusters (10 matched pairs per arm). The 20 clusters were selected based on the accessibility of the health facilities. A random number generator was used when infants turned 12 months of age, and researchers randomly selected and recruited study participants. One hundred forty mothers in the intervention arm and 142 from the SoC from these clusters were selected randomly to participate in this sub-sample study. This sample size represented approximately 10% of the full trial sample planned for household visits and food sample collection (Figure 2).
Infants were qualified for the study if (i) they participated in the 6-month visit within the MMT, (ii) they were between 11 and 13 months of age, and (iii) mothers were willing to provide informed consent for data collection, in addition to the informed consent for the larger MMT trial. Mother-infant dyads were excluded from this study if they did not provide informed consent for additional data collection or dropped out of the MMT by the time they were visited. The age of 11 to 13 months was purposely targeted since the children had already started consuming porridge and other family foods, especially the stiff porridge, locally called ugali, made of maize and maize products.
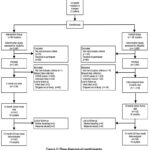 |
Figure 2: Flow diagram of participants. |
Assessment of food intake and collection of food samples
Pantry-level sample collection of fumonisin prone complementary food ingredients (maize and blended flours) from households in both arms was conducted during 24-hour dietary recalls. Maize (250 grams) and blended flour (100 grams) were sampled and weighed using a Pull-Out Display-5lb-(black)- food scale (OXO- model 1157100).
During food sampling, each batch of cereals or flour weighing less than 20 kg was mixed by flipping the storage bag or container up and down four times. Four random sub-samples (approximately 100-250 grams) were drawn from different sections of the stock bag/tin and carefully sealed in a food-grade, clean zip-lock bag. The air within the zip-lock bags was expelled during packing, and the bags were securely sealed and stored at -20 °C at Kongwa District Hospital. Subsequently, the samples were transported to the Nelson Mandela African Institution of Science and Technology (NM-AIST) laboratory in Arusha and kept at -20 oC until analysis.
Compensation ranging from US$0.50-0.65 was provided to households for each collected sample, depending on the type of food. Additionally, information regarding the source of food (market-sourced or home-grown) for the cereals and groundnuts was recorded.
Fumonisin detection by ELISA
Fumonisin concentrations were quantified using commercially available low-matrix enzyme-linked immunosorbent assay (ELISA) kits (Helica Biosystems Inc., USA), following the manufacturer’s protocol. Fumonisin was analyzed using total fumonisin ELISA (Catalogue number: 941AFL01M-96). Kit choices were determined by the manufacturer’s recommendations for specific food types. The assay standards provided a 1-20 µg/kg range, and fumonisin samples exceeding the upper limit were diluted and retested. In each assay, a blank sample and two Quality Control Reference Materials (QCRM).
Estimation of the daily flour intake
The estimates of dietary exposure of Infant Young Children (IYC) to fumonisin in the present study were based on flours consumed in stiff/thick porridge (ugali) or thin porridge only because they are not only susceptible to contamination with fumonisin, but also the most commonly and frequently consumed foods in the study area. The proportion of flour consumed through a thin or stiff porridge was computed30 estimated that the flour content of thin and stiff porridge consumed in Tanzania is 17 and 36% (w/w), respectively. The weight in grams of the thin or stiff porridge prepared from either cereal-only or cereal groundnut-based flour, as estimated from each of the two 24-hour recalls, was used to estimate the average flour intake per day per child. Calculating the amount of consumed flour and adjusting the volumes and weights of the thin porridge enabled us to estimate its specific gravity to be 1.1 g/mL. Therefore, the volumes (mL) of the thin porridge were multiplied by the specific gravity (g/mL) to obtain their consumed weight in grams.
Estimation of fumonisin dietary exposure
The estimation of dietary exposure to fumonisin was estimated by the deterministic approach (Equation (1)) (JECFA, 2010). The estimated exposure of a child to fumonisin (ng/kg bw/day) was calculated through different CF ingredients consumed by the child. Exposure through an ingredient was estimated by multiplying the daily intake of contaminated ingredients and the contamination of fumonisin level in the consumed food ingredient, and dividing the result by the child’s body weight.

Statistical analysis
Data analysis was conducted using STATA 16. A Chi-square test was used to compare differences in the distribution of fumonisin contamination for each type of food by study arm, using categories of fumonisin< 1 μg/kg (a value less than half of the legal limit), 1-2 μg/kg as intermediate, and >2 μg/kg (above the legal limit). An unpaired t-test was also conducted to compare mean fumonisin across arms; however, since fumonisin was not normally distributed, the data were additionally log transformed.
Ethical Clearance
Ethical approval for conducting this research was obtained from the Northern Tanzania Health Research Ethics Committee (KNCHREC) with registration number KNCHREC00041/02/2021.
Consent to participate in the study was obtained from mothers at the initial stages of study implementation.
Results
Demographic Information of Participants
Dietary recall and food sampling were conducted among 282 children aged 11 to 13 months (n=140 in the intervention arm and n=142 in the SoC arm). The demographic characteristics of the mothers were similar between arms. The average age of the recruited infants was 11.7 months, and 46.4% and 43.7% of the infants were female in the intervention and SoC arms, respectively. Most of the mothers were married and above the age of 18 years, with a mean of 27.8 and 27.9 in the intervention and SoC arms, respectively. 99.3% in the intervention arm and 97.2% were continuing breastfeeding while starting complementary feeding.30
Table 1: Participants and household characteristics
| Variable | Description | Intervention
(n = 140) |
Standard of Care
(n=142) |
| Infant age (months) | Mean (SD, range) | 11.7 (0.5, 11-13) | 11.7 (0.4, 11-13) |
| Infant gender n (%) | Males | 75 (53.6) | 80 (56.3) |
| Females | 65 (46.4) | 62 (43.7) | |
| Breastfeeding n (%) | Currently breastfed | 139 (99.3) | 138 (97.2) |
| No longer breastfed | 1 (0.7) | 4 (2.8) | |
| Infant appetite n (%) | Normal | 95 (67.9) | 91 (64.1) |
| Less than normal | 43 (30.7) | 49 (34.5) | |
| More than normal | 2 (1.4) | 2 (1.4) | |
| Mother’s age (years) | Mean (SD, range) | 27.8 (7.2, 16-47) | 27.9 (7.8, 16-47) |
| Married | 111 (79.3) | 112 (78.9) | |
| Marital status n (%) | Separated/Divorced | 13 (9.3) | 9 (6.3) |
| Single/Never married | 16 (11.4) | 20 (14.1) | |
| Widowed | 0 (0) | 1 (0.7) | |
| People in the house | Mean (SD, range) | 5.3 (1.9, 1-11) | 5.7 (1.9,1-11) |
| Pit latrine | 119 (85.0) | 98 (69.0) | |
| Type of toilet n (%) | Ventilated improved pit | 20 (14.3) | 37 (26.0) |
| None, Bush | 1 (0.7) | 5 (3.6) | |
| Others | 0 (0) | 2 (1.4) | |
| Water source n (%) | Protected source | 107 (76.4) | 108 (76.1) |
| 32 (23.6) | 34 (23.9) |
Types of complementary food flours and fumonisin results
Food samples collected covered maize and blended flour as follows;
Table 2: Food sample collection between the intervention and SoC
| Arm | Type of food | Sample collected | Samples analyzed |
| Intervention | Blended flour | 181 | 181 |
| SoC | Maize flour | 205 | 165 |
| Total | 386 | 346 | |
Table 3: Level of total fumonisin (μg/kg) in 4:1 blended flour sample between
| Arm | Intervention | SoC |
| Mean (SD) | 0.82 (0.74) | 1.01 (1.25) |
| Median (range) | 0.68 (0.0-4.81) | 0.58 (0-5.2) |
| <1 μg/kg (%) | 86 (80%) | 52 (71%) |
| <2 μg/kg (%) | 101 (94%) | 61 (84%) |
| ≥2 μg/kg (%) (range) | 7 (6%) | 12 (16%) |
| n | 108 | 73 |
Table 4: Level of total fumonisin (μg/g) in maize between arms
| Arm | Intervention | SoC |
| Mean (SD) | 0.92 (1.55) | 1.37 (1.56) |
| Median (range) | 0.56 (0.01- 12.63) | 0.78 (0.01- 6.9) |
| <1 μg/kg (%) | 64 (78%) | 47 (57%) |
| <2 μg/kg (%) | 73 (89%) | 64 (77%) |
| ≥2 μg/kg (%) (range) | 9 (11%) | 19 (23%) |
| n | 82 | 83 |
Table 5: Fumonisin intake and exposure levels between the intervention and SoC arms
| Food type | Arm | Fumonisin Intake (ng*g) | Intake mean | p-value | Fumonisin Range (ng*g/kg) | Fumonisin mean exposure | p-value |
| Blended flour | Intervention | 0.0 – 0.27 | 0.030 | 0.6 | 0 – 4.811 | 0 | 0.6 |
| SoC | 0.0 – 0.27 | 0.050 | 0- 5.202 | 0 | |||
| Maize | Intervention | 0.0 – 0.24 | 0.02 | 0.1 | 0 – 2.85 | 0.003 | 0.1 |
| SoC | 0.0 – 0.36 | 0.05 | 0 – 6.9 | 0.005 |
Discussion
Of great value, the MMT project ensures the safety of maize and blended flour from fumonisin during the process of reducing aflatoxin in maize and blended flour. Fumonisin contamination ranged from 0-4.8 µg/kg in the intervention and 0- 5.2 µg/kg in the SoC arm for blended flour; likewise, fumonisin levels in maize ranged from 0 -12.6 µg/kg in the intervention µg/kg and 0-3.9 µg/kg in the SoC arm. Fumonisin concentrations for blended flour samples were low in the intervention compared to SoC; this might be due to the pre-preparation of maize, including sorting and screening, which helped in lowering the concentration for blended flour. For maize samples, fumonisin concentration was higher in the intervention arm than in the SoC because no process was done on both the intervention and SoC. Maize samples were not provided by the MMT project in all study arms, but all study participants utilized maize acquired from households. Generally, in these two arms, fumonisin levels in maize and blended flow were quite low compared to other studies. For example, results reported by Kimanya, who conducted a study in Tabora, Ruvuma, Kilimanjaro, and Iringa regions, show the fumonisin ranges (71-2763 µg/kg, 62-3560 µg/kg, 65-11048 µg/kg, and 61-3353 µg/kg), respectively. Likewise, results from our study were low to total fumonisin of 10,140 µg/kg determined in maize from the former Transkei region in South Africa.9,32 On the other hand, blended flour produced by the project and distributed to the intervention arm had low fumonisin compared to maize samples, which were directly acquired at family levels. These results demonstrated that Tanzanian home-grown maize and maize products were contaminated with fumonisin.
Results from this study also show that the occurrence and level of fumonisin in maize and blended flour did not differ significantly between the two arms. Similarly, there are no differences in exposure across households from the two arms.
There was no association between the weight of babies and fumonisin intake and exposure in this study because the weight of babies was almost the same between the SoC and intervention. The mean Weight of babies who consumed blended flour was (8.04, 9.18) kg, and (8.48, 8.75) kg in the maize intervention and SoC, respectively.
Furthermore, this study demonstrated that there is no evidence that the frequency of consumption of fumonisin level for blended samples was significantly different between the two arms (p > 0.05).
Fumonisin consumption from maize samples from the two arms is significantly different (p = 0.05).
Variation of fumonisin levels between the intervention and SoC arms was due to sorting done to the maize samples used by MMT to make blended flour for the intervention arms. High fumonisin levels were noted in maize samples because there was no treatment done to reduce fumonisin in maize, which was acquired at the household level. Thus, sorting as practiced on blended flour was effective enough to reduce fumonisin contamination in maize and subsequently minimize fumonisin exposures in these communities; likewise, these levels were low compared to fumonisin concentration reported in other African countries.
Maize grown in Africa has been reported to contain fumonisin concentrations as high as 10000 µg/kg.32 Fumonisin exposure assessments conducted in communities dependent on maize in South Africa and Tanzania indicated that consuming maize with fumonisin levels exceeding 155 µg/kg can lead to exposure surpassing the provisional maximum tolerable daily intake (PMTDI) of 2 mg/kg body weight (BW).7
Fumonisin exposure through diet poses a serious risk to children’s health, as it can adversely affect growth and development. These mycotoxins are prevalent in staple foods and constitute a substantial portion of complementary foods for young children.33 In some regions of sub-Saharan Africa, the prevalence of child growth impairment rises as children get older. This trend has been associated with the introduction of complementary foods, which increases fumonisin exposure due to the higher intake of maize and maize-based products.34
Young children in rural areas of sub-Saharan Africa experience elevated levels of dietary fumonisin intake.33 Two epidemiological studies have been carried out to examine the link between fumonisin exposure and child growth. In a study by Chen et al. (2018), it was observed that infants in Tanzania with fumonisin intakes exceeding the JECFA’s PMTDI of 2 μg/kg bw/day, as estimated through caregivers’ dietary recall questionnaires, were notably shorter and lighter than those with lower fumonisin intake. The study, based on validated urinary biomarkers of fumonisin exposure, found a negative correlation between fumonisin exposure and child growth in children from four villages in Tanzania.
Shirima et al. (2015) found a negative association between fumonisin exposure and child growth in children from four villages in Tanzania, with fumonisin levels assessed using validated urinary biomarkers.30
This paper reports for the first time the level of fumonisin contamination in blended flour and maize samples after the MMT intervention implemented in central Tanzania. The study used an appropriate sample of 181 blended samples and 165 maize samples collected in the Kongwa District. The study aimed at identifying the extent of fumonisin levels in samples collected in the Dodoma region of central Tanzania, where the contamination is likely to be high and urgent intervention is needed. Additionally, the great value of this study was to examine whether the aflatoxin intervention did not impart fumonisin, and the study results confirmed that.
Based on the findings of this study, further research on fumonisin contamination in maize and maize-based products is essential, given that maize is a key component of complementary foods. Additional data will provide valuable insights for agricultural stakeholders working with rural communities, enabling them to offer informed guidance to farmers and households on strategies to minimize fumonisin contamination. Moreover, educating farmers on good agricultural and post-harvest management practices can play a crucial role in reducing mycotoxin exposure in rural populations.
Conclusion
Results from this sub-study confirmed that there were no significant differences in risks to fumonisin dietary exposure between selected households from the intervention and SoC arms. The results further confirmed that the process of low-aflatoxin blended flours contributed to lowering the levels of fumonisin in the food consumed by children in the household. This indicates that the production process, which involved procuring maize from different regions, did not raise fumonisin levels. Samples collected from this study had low fumonisin levels compared to other studies done in Tanzania. The preparation of low-aflatoxin complementary feeding flours also reduced fumonisin µg/kg levels in blended and maize flour consumed by study participants in the intervention households.
We recommend the development of guidelines and education materials based on lessons from the processing of low aflatoxin flours that could contribute to the reduction of fumonisin. Winnowing, rigorous maize sorting, and slight decortication can offer low-cost, cost-effective fumonisin reduction strategies, especially in regions prone to high fumonisin.
Acknowledgement
The authors extend their gratitude to the MMT leaders for project funding and their valuable contributions to the study’s design. They also appreciate the laboratory technical support by John Mshanga (NM-AIST Laboratory Scientist).
Funding Sources
This research is a component of the Mycotoxin Mitigation Trial (MMT) initiative, which receives financial support from the Bill & Melinda Gates Foundation (BMGF) through Grant Number OPP1155626 and is affiliated with Cornell University in the United States.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
The corresponding author will be happy to provide the data supporting the findings of this study upon request.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Materials from Other Sources
Not Applicable
Author Contributions
- Rosemary Alphonce Kayanda – conceptualization, and methodology, and performed data collection, analysis, interpretation, and writing of initial and consequent drafts of the manuscript.
- Francis Muigai Ngure – designing of the study and provided a critical review of the manuscript, also engaged in oversight on lab analysis, data analysis and interpreting the results. All authors have read and agreed to publish this manuscript.
Reference
- Anfossi L, Baggiani C, Giovannoli C, et al. Occurrence of aflatoxin M1 in dairy products. Aflatoxins detection, measurement and control. Published online 2011.
CrossRef - Fandohan P, Hell K, Marasas W, et al. Infection of maize by Fusarium species and contamination with fumonisin in Africa. African Journal of Biotechnology. 2003;2(12):570-579.
CrossRef - Kamle M, Mahato DK, Devi S, et al. Fumonisins: impact on agriculture, food, and human health and their management strategies. Toxins. 2019;11(6):328.
CrossRef - Doko MB, Canet C, Brown N, et al. Natural co-occurrence of fumonisins and zearalenone in cereals and cereal-based foods from Eastern and Southern Africa. Journal of Agricultural and Food Chemistry. 1996;44(10):3240-3243.
CrossRef - Cendoya E, Chiotta ML, Zachetti V, et al. Fumonisins and fumonisin-producing Fusarium occurrence in wheat and wheat by products: A review. Journal of cereal science. 2018;80:158-166.
CrossRef - Dall’Asta C, Battilani P. Fumonisins and their modified forms, a matter of concern in future scenario? World Mycotoxin Journal. 2016;9(5):727-739.
CrossRef - Kimanya M, De Meulenaer B, Tiisekwa B, et al. Human exposure to fumonisins from home grown maize in Tanzania. World Mycotoxin Journal. 2008;1(3):307-313.
CrossRef - Marasas WF. Fumonisins: their implications for human and animal health. Natural toxins. 1995;3(4):193-198.
CrossRef - Shephard GS, Thiel PG, Stockenström S, et al. Worldwide survey of fumonisin contamination of corn and corn-based products. Journal of AOAC International. 1996;79(3):671-687.
CrossRef - Shetty PH. Natural occurrence and management of Fumonisin mycotoxins. Published online 2011.
- Fandohan P, Gnonlonfin B, Hell K, et al. Natural occurrence of Fusarium and subsequent fumonisin contamination in preharvest and stored maize in Benin, West Africa. International Journal of Food Microbiology. 2005;99(2):173-183.
CrossRef - Van Zyl K. Genetics of fumonisin biosynthesis and resistance to Fusarium verticillioides in maize. Published online 2018.
- Cavaglieri L, Keller K, Pereyra C, et al. Fungi and natural incidence of selected mycotoxins in barley rootlets. Journal of Stored Products Research. 2009;45(3):147-150.
CrossRef - Magan N, Medina A, Aldred D. Possible climate‐change effects on mycotoxin contamination of food crops pre‐and postharvest. Plant pathology. 2011;60(1):150-163.
CrossRef - Balconi C, Berardo N, Locatelli S, et al. Evaluation of ear rot (Fusarium verticillioides) resistance and fumonisin accumulation in Italian maize inbred lines. Phytopathologia Mediterranea. Published online 2014:14-26.
- Miller JD. Factors that affect the occurrence of fumonisin. Environmental health perspectives. 2001;109(suppl 2):321-324.
CrossRef - Mollay C, Kassim N, Stoltzfus R, et al. Childhood dietary exposure of aflatoxins and fumonisins in Tanzania: A review. Cogent Food & Agriculture. 2020;6(1):1859047.
CrossRef - Alizadeh AM, Roshandel G, Roudbarmohammadi S, et al. Fumonisin B1 contamination of cereals and risk of esophageal cancer in a high risk area in northeastern Iran. Asian Pacific journal of cancer prevention. 2012;13(6):2625-2628.
CrossRef - Mutiga SK, Hoffmann V, Harvey JW, et al. Assessment of aflatoxin and fumonisin contamination of maize in western Kenya. Phytopathology. 2015;105(9):1250-1261.
CrossRef - Shephard GS. Impact of mycotoxins on human health in developing countries. Food Additives and contaminants. 2008;25(2):146-151.
CrossRef - Van der Westhuizen L, Shephard GS, Rheeder J, et al. Simple intervention method to reduce fumonisin exposure in a subsistence maize-farming community in South Africa. Food Additives and Contaminants. 2010;27(11):1582-1588.
CrossRef - Peraica M, Richter D, Rašić D. Mycotoxicoses in children. Archives of Industrial Hygiene and Toxicology. 2014;65(4):347-363.
CrossRef - Passarelli S, Bromage S, Darling AM, et al. Aflatoxin exposure in utero and birth and growth outcomes in Tanzania. Maternal & child nutrition. 2020;16(2):e12917.
CrossRef - Sarron E, Pérot M, Barbezier N, et al. Early exposure to food contaminants reshapes maturation of the human brain-gut-microbiota axis. World Journal of Gastroenterology. 2020;26(23):3145.
CrossRef - Wild C, Rasheed F, Jawla M, et al. In-utero exposure to aflatoxin in West Africa. The Lancet. 1991;337(8757):1602
CrossRef. - Stanciu O, Juan C, Miere D, et al. Presence of enniatins and beauvericin in Romanian wheat samples: From raw material to products for direct human consumption. Toxins. 2017;9(6):189.
CrossRef - Kimanya ME, Meulenaer BD, Baert K, et al. Exposure of infants to fumonisins in maize‐based complementary foods in rural Tanzania. Molecular nutrition & food research. 2009;53(5):667-674.
CrossRef - Nyangi C, Mugula J, Beed F, et al. Aflatoxins and fumonisin contamination of marketed maize, maize bran and maize used as animal feed in Northern Tanzania. African Journal of Food, Agriculture, Nutrition and Development. 2016;16(3):11054-11065.
CrossRef - Ngure FM, Makule E, Mgongo W, et al. Processing complementary foods to reduce mycotoxins in a medium scale Tanzanian mill: A hazard analysis critical control point (HACCP) approach. Food Control. Published online 2024:110463.
CrossRef - Kayanda RA, Kassim N, Ngure FM, et al. Nutrient intake and dietary adequacy among rural Tanzanian infants enrolled in the Mycotoxin Mitigation Trial. Nutrients. 2024;17(1):131.
CrossRef - Shephard GS, Marasas WF, Burger HM, et al. Exposure assessment for fumonisins in the former Transkei region of South Africa. Food additives and contaminants. 2007;24(6):621-629.
CrossRef - Fandohan P, Zoumenou D, Hounhouigan DJ, et al. Fate of aflatoxins and fumonisins during the processing of maize into food products in Benin. International Journal of Food Microbiology. 2005;98(3):249-259.
CrossRef - Chen C, Mitchell NJ, Gratz J, et al. Exposure to aflatoxin and fumonisin in children at risk for growth impairment in rural Tanzania. Environment international. 2018;115:29-37.
CrossRef - Kamala A, Kimanya M, Lachat C, et al. Risk of exposure to multiple mycotoxins from maize-based complementary foods in Tanzania. Journal of Agricultural and Food Chemistry. 2017;65(33):7106-7114.
CrossRef













