Effect of Sour Creams Fermented by Limosilactobacillus fermentum AG8 and Lactiplantibacillus plantarum AG9 on Mice
Institution of Food Technology and Biotechnology, Kazan National Research Technological University, Kazan, Russia
Corresponding Author Email: ev-nikitina@inbox.ru
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.2.11
ABSTRACT:Sour cream is a traditional high-fat dairy product fermented by lactic acid bacteria (LAB) with potential health benefits when consumed in small amounts. This study investigated the effects of sour cream fermented with Limosilactobacillus fermentum AG8 and Lactiplantibacillus plantarum AG9 on physiological and biochemical parameters in mice. Twenty-eight mice were divided into four groups: a control group and three groups fed sour cream fermented with classical starter cultures, L.fermentum AG8 or L.plantarum AG9. Body weight, organ parameters, hematological and biochemical parameters, and gut microbiota were analyzed for 21 days. The results showed that sour cream with strains L.plantarum AG9, L.fermentum AG8 significantly improved lipid metabolism, reducing triglycerides, cholesterol and liver enzymes (alanine aminotransferase, aspartate aminotransferase) levels compared to the control group and the classic sour cream group with Lactococcus lactis subsp. lactis and L. lactis subsp. cremoris. These probiotic strains also improved immune function, as shown by increases in white blood cells, lymphocytes, and spleen counts. Gut microbiota analysis revealed a decrease in coliform bacteria in the L.fermentum AG8 and L.plantarum AG9 groups, indicating enhanced gut health. The study definitively shown that sour cream fermented with L. fermentum AG8 and L. plantarum AG9 can act as a functional food, providing cardiovascular and immune benefits without compromising traditional qualities. This highlights the potential of non-starter probiotic LAB in high-fat dairy products to promote metabolic and immune health.
KEYWORDS:Health; Immune System; Lipids; Mice; Probiotic; Sour cream
Introduction
Sour cream, also fermented cream, is a type of dairy product with a unique, slightly sour taste, which is produced from high-quality cream as a raw material and a complex of mesophilic lactic acid bacteria (LAB).1 Sour cream is a traditional and popular product in Russia and the other Slavic countries, it is produced in various fat content from 10 to 30%. Sour cream with 15% fat content is the most popular.1,2 In various countries, sour cream is used as a sauce for soups, vegetable salads, meat, fish, flour dishes such as ravioli, and as a base for cake cream or as an ingredient in baked products.2
Fermentation is an established technique that is utilized in the preservation of valuable dairy products, with the aim of prolonging shelf life. In addition to its function in prolonging the shelf life of products, fermentation can also contribute towards a distinctive flavour and texture.3 Cream is the fat fraction of all types of milk, can be obtained from different animals by separating from the water fraction, so the fat content of sour cream is higher than other dairy products. Sour cream is traditionally fermented using starter cultures composed mainly of lactic acid bacteria (LAB), such as Lactococcus lactis subsp. lactis and L. lactis subsp. Cremoris.4-6 These strains are characterized by their ability to synthesize elevated levels of aromatic compounds, which play a key role in developing the typical flavor and sensory properties of the final product.7 The process of forming low molecular weight substances such as aldehydes, alcohols, ketones is associated with the synthesis of enzymes for hydrolysing proteins, lipids and other organic substances during the fermentation of dairy raw materials. Besides lactococci, sour cream can be fermented by Leuconostoc spp. which, due to their heterofermentative nature, can release carbon dioxide, volatile compounds and organic acids that give fermented cream flavor and texture.8,9 These aroma and flavors of sour cream makes it popular as an appetizer, salad dressing, a base for sauces.10,11
However, the high fat content of sour cream limits its consumption in large quantities. The functional value of sour cream can be improved by using only probiotic bacteria12 or by incorporating non-starter probiotic bacteria into a traditional sour cream starter.13 A popular strategy today is the use of non-starter probiotic bacteria as part of fermentation-derived products.14 The researchers are considering lactobacilli as a bacterial culture that can be used as a probiotic culture for the production of sour cream.13 Lactobacilli of various species are being tested for use in dairy products: Lactobacillus acidophilus,15 Lactiplantibacillus plantarum,16,17 Lactobacillus rhamnosus,18 Lactobacillus helveticus.19
A considerable number of studies are available which address the use of probiotics in dairy products,20,21 including in products high in milk fat.22,23 However, the number of research papers focusing on sour cream is comparatively low. Researchers are primarily focused on studying the regularities of changes in rheology, texture, and the state of milk fat, as well as changes in the composition of aromatic compounds in sour cream.1,12,24 There are limited studies in existence that address the effects of sour cream or fermented cream containing probiotic bacteria on mammals.
In the previous section, the objective was to elucidate the distinctive effects of sour cream made with traditional starter cultures (Lactococcus lactis subsp. cremoris, Lactococcus lactis subsp. lactis biovar diacetilactis) and sour cream obtained by fermentation with probiotic lactic acid bacteria on mice. The selection of Limosilactobacillus fermentum AG8 and Lactiplantibacillus plantarum AG9 was made on the basis of their well-documented probiotic properties.25 These strains have probiotic properties, are capable of fermenting milk and metabolic products have antioxidant properties.26-28 In addition, L. fermentum strain AG8 was found to have high antibacterial activity.29
Material and Methods
Strains and Fermented Cream?
The experiment utilized cream with a fat content of 15% (Valio®, Russia). To prepare the fermented cream, commercial starter cultures were employed, including Lactococcus lactis subsp. cremoris, Lactococcus lactis subsp. lactis biovar diacetilactis (Lactosintez LLC, Moscow, Russia), strains Limosilactobacillus fermentum AG8 and Lactiplantibacillus plantarum AG9 were isolated from the silage and described earlier like probiotic strains.25 These strains were cultivated on de Man, Rogosa, and Sharpe (MRS) medium or MRS-agar (HiMedia, India). The following sour cream samples were prepared for in vivo experiments: Fer_cream – classic sour cream fermented using commercial L. cremoris and L. diacetilactis starter cultures; Fer.cream_AG8 – fermented cream obtained by fermentation with L. fermentum AG8; Fer.cream_AG9 – fermented cream obtained by fermentation with L.plantarum AG9.
The sour cream samples were obtained as follows. For sour cream production, lactic acid bacteria (LAB) precultures were grown in sterile skim milk at 36°C for 16 h. The resulting precultures, with a volume of 5 ml, were added to the 100 ml of cream containing 15% fat. The mixture was then subjected to a fermentation process at a temperature of 32°C for a period of 14 hours. Following this, the sample underwent a stabilization step at a temperature of 4°C for a duration of 16 hours. Subsequent to this stabilization phase, each sample was stored at a temperature of 4°C and utilized for the purpose of animal feeding over a 21-day period. A fresh batch of fermented cream was prepared every 5 days to ensure freshness.
Animals
White laboratory mice (male) were used in this study. Twenty-eight mice, 6-7 weeks old (the mice weighed in at 12-14 g), were obtained from the Kazan Federal University, Kazan, Russia. The study was approved by the local ethical committee of Kazan Federal University (protocol No. 40 of March 9, 2023), where the research was conducted. All mice were kept at 20-24 º C in a room with 60-65% humidity and a light/dark cycle of 12 h, given free access to standard food and tap water for one week.
The mice were randomly divided into four groups of 7 individuals each, the following experimental studies were performed while varying the food supplementation:
(Control) group with normal diet received the basic feed and sterile water. Commercial mouse feed (DeltaFids, BioPro Company, Novosibirsk, Russia) was used (metabolizable energy – 2500 kcal/kg, crude protein – 19 %. Composition: two-component grain mixture, high-protein components (vegetable and animal proteins), vegetable oil, amino acids, organic acids, vitamin and mineral complex, fiber);
(Fer_cream) normal diet + fermented cream (1 g/animal per day);
(Fer.cream_AG8) normal diet + fermentum AG8 fermented cream (1 g/animal);
(Fer.cream_AG9) normal diet + plantarum AG9 fermented cream (1 g/animal).
Throughout the study period, body weights were recorded on a weekly basis at 7:00 am. After 21 days, blood samples from the animals were collected into disposable tubes with separation gel. The tubes were then put into a centrifuge (DM0506, Dlab, China) at 3500 rpm for 10 minutes. The spleen, heart, and both kidneys together, as well as the liver, were weighed for each animal. The organ index was determined as a percentage of the animal’s weight. Blood samples for serum lipid and blood morphology analysis were taken from the coronary artery after 21 days. Blood analysis was performed using an Abacus Junior 5 VET haematology analyser (Diatron Messtechnik GmbH, Austria). Blood samples were stored in a refrigerator at 2-4°C for no more than 1-2 hours prior to analysis. Biochemical parameters, including total cholesterol (TC), high-density lipoprotein (HDLP), low-density lipoprotein (LDLP), triglycerides (TG), alanine aminotransferase (ALT), aspartate aminotransferase (AST), total protein and bilirubin, were determined using a ChemWell2902 device (Awareness Technology, USA) and kits or reagents from Spinreact S.A. (Spain).
Microbiological Analyses
The contents of the large intestine (faeces) were analysed in this study. For this purpose, after autopsy, faeces were scraped into sterile plastic tubes, placed on ice and seeded onto appropriate media within 6 h. Numbers of different groups of microorganisms were analyzed by serial dilutions, the following nutrient media were used: for LAB (lactobacilli) – MRS agar (Himedia, India), total bacteria – nutrient agar SPA (Microgen, Russia), coli-forming bacteria and E. coli – Endo medium (Microgen, Russia), yeast – Saboura medium (Microgen, Russia). Cultivation after sowing was carried out at 36 º C for 1-5 days, depending on the species of microorganisms.
Statistical Analysis
All analyses were performed in three repetitions. Statistical significance of the results was determined at P < 0.05. Visualization of principal component analysis (PCA) and correlation analysis was performed on a two-dimensional P1/P2 map using OriginPro software v.8, which allowed us to identify relationships between variables at a significance level of P < 0.05.
Results
Organ Indices and Hematologic Indices
The addition of a moderate amount of sour cream to the diet of mice resulted in a slight decrease in body weight compared to the control. This effect may be due to an adjustment in lipid metabolism (Table 1). Compared to the control group, the organ indices (heart, kidneys, liver) were lower in the experimental groups than in the control group. Spleen index was lower in sour cream than in control, but consumption of L.fermentum AG8 and L.plantarum AG9 cream resulted in an increase in spleen index.
Table 1: Absolute weight gains of mice, the organ indexes over 21 days of feeding in an experiment with the introduction of sour cream into a balanced diet (mean values ± SD, n = 7).
|
Parameters |
Control | Fermented cream | Fer.cream_AG8 | Fer.cream_AG9 |
| Average body weight, g | 23.6±2.4 | 21.4±2.1 | 21.7±2.8 |
22.3±1.7 |
|
Spleen, % |
0.45±0.11 | 0.36±0.13 | 0.51±0.15 | 0.54±0.12 |
| Heart, % | 0.69±0.09 | 0.52±0.10 | 0.51±0.05 a |
0.45±0.11 a |
|
Kidneys, % |
2.07±0.09 | 1.65±0.09 a | 1.71±0.11 a | 1.57±0.09a |
| Liver, % | 8.56±1.23 | 6.35±1.03 a | 6.12±0.88 a |
5.96±0.52 a |
With the exception of leukocytes and lymphocyte counts, analysis of the hematological parameters of the mice showed no significant differences between the experimental groups (Table 2). In the Fer.cream_AG8 and Fer.cream_AG9 groups, the amount of leukoytes increased, the increase being due to an increase in the absolute amount of lymphocytes. The relative proportion of lymphocytes in this group was also higher than in the control. Thrombocrit levels decreased in the same groups of animals. However, it should be noted that the hematological indices did not exceed the norms for mice.
Fer.cream_AG8 and Fer.cream_AG9
Biochemical Indices
When analyzing biochemical parameters, special attention was paid to lipid metabolism and liver health parameters, as the animals were fed a product with a high fat content of 15 %. The HDL index changed in the experimental groups compared to the control group. In particular, it increased in the sour cream group and decreased in the fermented cream AG8 and AG9 groups (Fig. 1A). The LDLP index increased in all experimental groups compared to the control. The very low density lipoprotein index in Fer.cream_AG8 and Fer.cream_AG9 groups was significantly lower than control.
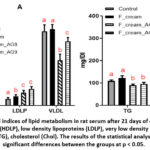 |
Figure 1: Biochemical indices of lipid metabolism in rat serum after 21 days of experiment: A – high density lipoproteins (HDLP), low density lipoproteins (LDLP), very low density lipoproteins (VLDL); B – triglycerides (TG), cholesterol (Chol). |
Table 2: Effect of sour cream on hematologic indices of blood of mice after 21 days of experiment. (mean values ± SD, n = 7).
|
Parameters |
Abbreviations | Control | Fermented cream |
Fer.cream_AG8 |
Fer.cream_AG9 |
|
leukocytes, 10 9/L |
WBC | 1.7712±0.71 | 1.8264±0.63 | 3.5016±0.6 a | 2.3256±0.85 |
| lymphocytes, 10 9/L | LYM | 0.8064±0.28 | 1.212±0.51 | 2.4456±0.41 a |
1.536±0.44a |
|
sum: neutrophils, |
MID | 0.0888±0.04 | 0.1296±0.06 | 0.2016±0.14 |
0.1392±0.06 |
|
granulocytes, 10 9/L |
GRA | 0.876±0.47 | 0.4944±0.12 | 0.8496±0.42 | 0.6312±0.27 |
| Lymphocytes, % | LY% | 46.82±12.87 | 63.78±10.05 | 68.46±7.86 a | 53.26±27.14 |
| sum: neutrophils, monocytes, eosinophils, basophils, % |
MI% | 5.14±2.18 | 7.1±2.26 | 5.54±3.47 |
5.8±1.8 |
|
granulocytes, % |
GR% | 48.08±13.29 | 29.1±9.47 | 24±6.02 a | 26.96±9.72 a |
| erythrocytes, 10 12/L | RBC | 7.9176±0.48 | 6.9072±1.62 | 6.2304±1.24 |
7.4688±0.54 |
|
hemoglobin, g/L |
HGB | 121.92±14.43 | 112.08±26.88 | 95.04±18.65 | 109.68±3.21 |
| hematocrit, % | HCT | 33.222±4.12 | 29.886±10.07 | 25.454±5.92 |
30.528±2.58 |
|
average red blood |
MCV | 60.24±3.27 | 61.92±3.51 | 59.28±3.05 | 59.28±3.21 |
| average hemoglobin concentration in blood, pg |
MCH | 28.48±15.13 | 19.44±0.23v | 18.288±0.68 a |
17.712±1.34 a |
|
average hemoglobin |
MCHC | 367.44±24.94 | 307.2±124.45 | 372.72±18.27 | 361.2±31.85 |
| erythrocyte population distribution latitude, % |
RDWc | 16.96±1.25 | 15.7±0.31 | 18±1.60 |
17.82±0.16 |
|
platelets, 10 9/L |
PLT | 381.36±67.47 | 261.6±65.09 | 281.76±85.67 | 281.52±38.07 |
| thrombocrit, % | PCT | 0.206±0.04 | 0.15±0.03 a | 0.126±0.09 a |
0.144±0.03 a |
|
average platelet volume, ft |
MPV | 7.848±0.54 | 7.944±0.39 | 8.136±0.45 | 7.296±0.28 |
| platelet population distribution latitude, % |
PDWc | 31.58±1.86 | 31.38±1.35 | 33.64±2.67 |
31.18±1.14 |
a The results of the statistical analysis indicate significant differences between the groups at p < 0.05.
The amount of triglycerides in the blood of animals in the Fer.cream_AG8 and Fer.cream_AG9 groups was lower than in the control and sour cream groups. It should be noted that the lowest cholesterol level was observed in the group that received fermented cream of strain AG8. (Fig. 1B).
Figure 2 shows the activities in blood of the liver enzymes ALT and AST. It was found that ALT activity was significantly decreased in the group with fermented cream AG9 compared with the control. In case of AST enzyme, a decrease in its activity was observed in the group with fermented cream L.fermentum AG8, although there was a tendency to decrease AST activity in the group with sour cream AG9.
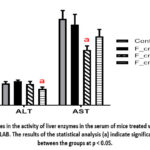 |
Figure 2: Changes in the activity of liver enzymes in the serum of mice treated with sour cream with different LAB. |
The addition of sour cream fermented by strains L.plantarum AG9 or L.fermentum AG8 to the diet slightly decreased the amount of total protein in the blood, but this indicator remained normal, although it differed from the control (Figure 3). When classical sour cream with L. cremoris and L. diacetilactis (F_cream group) was added to the diet, the amount of protein in the blood of mice increased slightly. Regular consumption of sour cream, regardless of the starter bacteria, led to an increase in bilirubin levels, but not above the normal level. It is important to note that the highest bilirubin elevation was in the classic sour cream group (F_cream group), while this index was lower in the groups with sour cream based on probiotic strains L.fermentum AG8 or L.plantarum AG9.
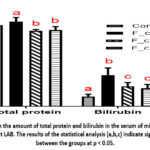 |
Figure 3: Variation in the amount of total protein and bilirubin in the serum of mice treated with sour cream with different LAB. |
Shifts in the abundance of a number of groups were observed when the gut microbial community was analyzed using classical microbiological methods (Fig. 4). Consumption of sour cream decreased the total number of heterotrophic facultative anaerobic and aerobic microorganisms in all experimental groups (Total bacteria). As for the LABs, their number increased in the F_cream group compared to the control and decreased in the Fer.cream_AG8 and Fer.cream_AG9 experimental groups. Unfortunately, it should be noted that the presence of sour cream in the daily diet led to an increase in the number of coliforms. However, in the variants with sour cream L.plantarum AG9 and especially L.fermentum AG8, the increase in the abundance of this group compared to the control was less intense. The comparable trend was found with the yeast population.
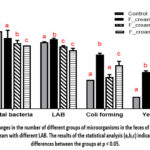 |
Figure 4: Changes in the number of different groups of microorganisms in the feces of mice treated with sour cream with different LAB. |
PCA and Correlation
PCA was utilized to investigate the associations between groups of mice fed diets fortified with sour cream and various starters (Fig. 5A). PCA was performed on two principal components, selected according to established criteria. The first principal component (PC1; X axis) had an explanation of 21.31% of the total variation in the data set, while the second principal component (PC2; Y axis) had an explanation of 15.29% of the total variation. The PCA values categorised the groups of mice into clusters. These clusters practically do not overlap. Their position in the coordinate system clearly indicates the essential role of the bacteria present in the sour cream. Depending on whether probiotic bacteria L.plantarum AG9 or L.fermentum AG8 were used instead of the classical starter, it is evident that the functional parameters of the mice were significantly improved.
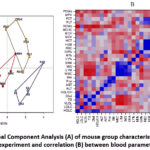 |
Figure 5: Principal Component Analysis (A) of mouse group characteristics after 21 days of experiment and correlation (B) between blood parameters. |
It is clear from the correlation analysis of hematological and biochemical parameters (Figure 5B) that there is a strong negative correlation. (a) Leucocytes, lymphocytes (WBC, LYM/TG, Chol, VLDL (TG, Chol, VLDL); (b) Erythrocyte population breadth/TG, Chol, VLDL, HDLC (TG, Chol, VLDL, HDLC); (c) AST/percentage of lymphocytes (LY %). Positive correlations were found only in similar groups of parameters.
Discussion
It is well known that lactic acid bacteria and products based on them have beneficial effects on macroorganisms, including humans.30 In this work, we demonstrated the probiotic potential of lactic acid bacteria at the level of fermented cream under in vivo conditions in a mouse model. To study the physiological effects of sour cream fermented with different isolates on mice, we evaluated changes in body weight, organ indices, lipid profile, liver enzymes and haematological markers.
Mice that were fed a diet comprising sour cream fermented with L. fermentum AG8 and L. plantarum AG9 exhibited a modest yet persistent decline in body mass in comparison to the control group. Haematological data showed an increase in leukocyte and lymphocyte counts, particularly in the L. fermentum AG8 group (p < 0.05), which may indicate an enhanced immune response. Many studies have reported the positive role of lactobacilli in forming immune status.31,32
Despite the diet including fatty sour cream, this decrease may indicate improved lipid utilisation or reduced fat accumulation. Analysis of organ indices revealed a significant decrease in liver and kidney weights in the AG8 and AG9 groups (p < 0.05), suggesting a possible reduction in metabolic burden. However, the spleen index increased in these groups, correlating with increased immune activity.
Introducing sour cream containing the L. fermentum AG8 and L. plantarum AG9 strains into the diet resulted in significantly lower triglyceride and total cholesterol levels than the control group and the group receiving classic sour cream containing L. cremoris and L. diacetilactis. Notably, mice receiving sour cream containing L. fermentum AG8 exhibited the lowest cholesterol levels (p < 0.05). While HDL levels increased in the classic sour cream group, they remained lower in the L. fermentum AG8 and L. plantarum groups. This suggests that these strains may regulate lipid transport and metabolism differently, possibly through the activity of bile salt hydrolases or by modulating the gut microbiota. In addition, there was evidence of the positive role of L. fermentum AG8 and L. plantarum AG9 strains in the regulation of mouse liver function in sour cream composition, as indicated by a significant decrease in the levels of ALT and AST activity.
The results, which included a reduction in triglycerides, cholesterol, bilirubin and liver enzymes, clearly indicate the feasibility of incorporating them into a high-fat product without compromising their probiotic functionality. These findings are particularly significant in the context of heightened interest in the use of non-fermented strains as probiotics, with the potential to enhance human health. There is an increasing focus on reducing elevated levels of lipids, low-density lipoproteins and triglycerides in the blood through dietary foods.
Some people may have a mild form of hypercholesterolaemia in which taking pharmaceuticals like statins is not justified given their side effects.33 Probiotics, defined as “live microorganisms that, when administered in sufficient quantity, provide health benefits to the host”.34 Probiotic LAB can be effective in lowering cholesterol levels even with a high-fat diet,35-37 these probiotics are used to ferment milk.38 Many authors studying Lactiplantibacillus plantarum have noted its ability to lower cholesterol, regulate gut flora and boost the immune system.39-41 Limosilactobacillus fermentum strains have now been identified as the best candidates for use as probiotic lactic acid bacteria (LAB) with hypocholesterolemic effects.38,42-44 In the present studies, the observed effect of reducing cholesterol and triglyceride levels when consuming sour cream fermented with Limosilactobacillus fermentum AG8 and Lactiplantibacillus plantarum AG9 may be due to the ability of these strains and their metabolites, particularly exopolysaccharides, to exert the lipase-inhibiting effect previously shown.28
It appears that the microbiota in the gut may play a role in the regulation of cholesterol levels and the body’s immune system.45-47 The addition of Lactococcus-based 15% fat sour cream to the diet of mice resulted in increased levels of coliform bacteria (e.g. Escherichia, Enterobacter, Klebsiella, Citrobacter) compared to the control, which was induced by an increase in the fat content of the diet. However, this increase was lower in mice receiving L.plantarum AG9 and especially AG8-based sour cream, which is a positive outcome. The observed effects of L.fermentum AG8 and L.plantarum AG9 sour cream are probably due to the antibacterial properties of the strains. This has been shown previously.25,29
The present study definitively shows that the use of classic sour cream did not affect the leucocyte count. The control group and the F_cream group both had a lower content of leucocytes and lymphocytes, which indicates a reduced immunological status. Whereas the presence of the product with L.fermentum AG8 and L.plantarum AG9 in the diet of mice increased the number of leucocytes and lymphocytes to normal. The increase in the leukocyte level within the normal range may indicate the stimulation of immunity, which correlates with the increase in the spleen index. Meta-analyses of data from many researchers clearly show that strains of Lactobacillus plantarum species can significantly affect the immune system48 and lipid profile.49 Researchers have noted the positive role of L. fermentum in promoting mammalian health, including through changes in the composition of the microbiome.50,51
In addition to the previously mentioned positive changes in the lipid profile and liver function indices, it is important to emphasize that the utilization of L. plantarum AG9 and L. fermentum AG8 in fermented sour cream may contribute to the improvement of the gut microbiota. This is because the gut microbiota plays a key role in lipid metabolism and immune homeostasis. It is hypothesized that alterations in the composition of intestinal flora may enhance gut barrier function, thereby reducing systemic inflammation and promoting metabolic normalization. Furthermore, exopolysaccharides, the production of which is characteristic of these strains, are likely to have a prebiotic effect, thereby stimulating the growth of beneficial bacteria and further supporting macro-organismal health.26,28 The long-term utilization of probiotics has the potential to prevent metabolic disorders, particularly in individuals with a genetic predisposition to hypercholesterolemia, thereby reducing the necessity for pharmacotherapy, which is associated with significant adverse effects.
Our in vivo studies definitively demonstrate that LAB strains isolated from silage are promising probiotics for use in functional foods. These strains can effectively modulate lipid metabolism and reduce cholesterol levels, enhance immune status and correct gut microbiota. Notably, we observe these effects in high-fat foods, highlighting their significance. In summary, the integration of specific probiotic strains into high-fat foods offers new opportunities for the rational and safe management of lipid metabolism and immune enhancement.
Conclusion
This study definitively investigated the effect of classical 15 % sour cream based on Lactococcus lactis subsp. cremoris, Lactococcus lactis subsp. lactis biovar diacetilactis, as well as sour cream fermented with probiotic Limosilactobacillus fermentum AG8 and Lactiplantibacillus plantarum AG9 on physiological and biochemical parameters of mice. The results show that there was a significant improvement in lipid metabolism, immune response and liver condition when sour cream fermented with these probiotic strains was included in the diet. Specifically, the consumption of sour cream fermented with L. plantarum AG9 and L. fermentum AG8 resulted in a decrease in triglycerides, cholesterol and liver enzymes (ALT and AST), indicating a potential protective effect against metabolic disorders induced by the high-fat product. Furthermore, an increase in the number of leukocytes and lymphocytes, as well as an increase in the spleen index, indicates an immunomodulatory effect of these probiotic strains.
The results clearly show that probiotic bacteria can be used in fermented dairy products to improve their functional properties without compromising traditional characteristics. There is no question that these probiotics promote cardiovascular health, as evidenced by the reduction in harmful lipid fractions (LDLP and VLDL) and the increase in beneficial HDLP levels. What is certain is that the changes observed in the composition of the gut microbiota, particularly the reduction in coliform bacteria, highlight the potential of these strains to improve gut health.
This study confirms that sour cream fermented with L. plantarum AG9 or L. fermentum AG8 is a functional food with significant health benefits, particularly for modulating lipid metabolism and enhancing immune function. These results are consistent with the growing interest in probiotics as a natural approach to treat metabolic and immune disorders. Further studies are needed to investigate the long-term effects and potency of these probiotic strains in the human diet, especially in the context of high-fat dairy products.
Acknowledgement
The Author would like to thank Kazan National Research Technological University and Kazan Federal University for the support.
Funding Sources
The work was funded by the grant of the Academy of Sciences of the Republic of Tatarstan, provided to young candidates of sciences (postdoctoral fellows) for the purpose of de-fending doctoral dissertation, performing research work, as well as performing labor functions in scientific and educational organizations of the Republic of Tatarstan within the framework of the State Program of the Republic of Tatarstan “Scientific and Technological Development of the Re-public of Tatarstan”, grant No. 117/2024-PD.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of Kazan Federal University (protocol No. 40 of March 9, 2023).
Informed Consent Statement
This study did not involve human participants, animals or any such material and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not applicable.
Author Contributions
The sole author was responsible for the conceptualization, methodology, data collection, analysis, writing, and final approval of the manuscript.
References
- Wang M.S., Fan M., Zheng A.R., et al. Characterization of a fermented dairy, sour cream: Lipolysis and the release profile of flavor compounds. Food Chem. 2023; 423:136299. doi.org/10.1016/j.foodchem.2023.136299.
CrossRef - Meunier-Goddik L. Sour cream and creme fraîche. In Y. H. Hui, E. O. Evranuz (Eds.), Handbook of animal-based fermented food and fermentation technology. Boca Raton, FL, USA: CRC Press. 2012.
- Born B. Cultured/Sour Cream, In Manufacturing Yogurt and Fermented Milks (Ramesh C. Chandan, ed.) Blackwell Publishing, USA. 2006; pp. 285–293.
CrossRef - Tamime A. Y., Skriver A., Nilsson L.-E. Starter cultures. In A. Y. Tamime (Ed.), Fermented milks. Oxford, UK: Blackwell Publishing. 2006. doi.org/10.1002/9780470995501.ch2
CrossRef - Kim Y., Yoon S., Shin H., et al. Isolation of Lactococcus lactis ssp. cremoris LRCC5306 and optimization of diacetyl production conditions for manufacturing sour cream. Food Science of Animal Resources. 2021; 41(3): 373–385. https://doi.org/10.5851/kosfa.2021.e3
CrossRef - Seo C. W. Physicochemical changes during cream fermentation and quality characteristics of lactic butter produced using cultured creams with different fermentation levels. International Dairy Journal. 2023; 145: 105730. doi.org/10.1016/j.idairyj.2023.105730
CrossRef - Lee H. W., Kim I. S., Kil B. J., et al. Investigation of flavor-forming starter Lactococcus lactis subsp. lactis LDTM6802 and Lactococcus lactis subsp. cremoris LDTM6803 in miniature gouda-type cheeses. Journal of Microbiology and Biotechnology. 2020; 30(9): 1404–1411. doi.org/10.4014/jmb.2004.04004
CrossRef - Flórez A. B., Campedelli I., Delgado S., et al. Antibiotic susceptibility profiles of dairy Leuconostoc, analysis of the genetic basis of atypical resistances and transfer of genes in vitro and in a food matrix. PLoS One. 2016; 11(1): 1–20. doi.org/ 10.1371/ journal. pone.0145203
CrossRef - Paula A. T., Jeronymo-Ceneviva A. B., Todorov S. D., et al. The two faces of Leuconostoc mesenteroides in food systems. Food Reviews International. 2015; 31: 147–171. doi.org/10.1080/87559129.2014.981825
CrossRef - Yilmaz-Ersan, L. Fatty acid composition of cream fermented by probiotic bacteria. Mljekarstvo. 2013; 63: 132–139.
- Zheng, J., Wittouck, S., Salvetti, E., et al. A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. J. Syst. Evol. Microbiol. 2020; 70: 2782–2858. doi.org/10.1099/ijsem.0.004107
CrossRef - Yilmaz-Ersan, L., Ozcan, T., Akpinar-Bayizit, A., et al. Probiotic cream: Viability of probiotic bacteria and chemical characterization. Journal of Food Processing and Preservation. 2017; 41(1): 1–9. doi.org/10.1111/jfpp.12797
CrossRef - Khademi F, Naghizadeh Raeisi S, Younesi M, et al. Effect of probiotic bacteria on physicochemical, microbiological, textural, sensory properties and fatty acid profile of sour cream. Food Chem Toxicol. 2022; 166: 113244. doi.org/10.1016/j.fct.2022.113244.
CrossRef - Granato, D., Branco, G.F., Nazzaro, F., et al. Functional foods and nondairy probiotic food development: trends, concepts, and products. Rev. Food Sci. Food Saf. 2010; 9(3): 292-302. doi.org/10.1111/j.1541-4337.2010.00110.x. PMID: 33467814.
CrossRef - Oh, J.K., Kim, Y.R., Lee, B., et al. Prevention of cholesterol gallstone formation by Lactobacillus acidophilus ATCC 43121 and Lactobacillus fermentum MF27 in lithogenic diet-induced mice. Food Science of Animal Resources. 2021; 41(2): 343. doi.org/10.5851/kosfa.2020.e93
CrossRef - Tian, L., Liu, R., Zhou, Z., et al. Probiotic characteristics of Lactiplantibacillus Plantarum N-1 and its cholesterol-lowering effect in Hypercholesterolemic rats. Probiotics and Antimicrobial Proteins. 2022; 14 (2): 337-348. doi.org/10.1007/s12602-021-09886-1.
CrossRef - Wong, W.-Y., Chan, B. D., Cho, P.-T., et al. Beneficial and immunomodulatory effects of heat-killed Lactobacillus plantarum L137 in normal and acute colitis mice. Journal of Functional Foods. 2024; 116: 106167. doi.org/10.1016/j.jff.2024.106167
CrossRef - Daniel, M., Szymanik-Grzelak, H., Turczyn, A., et al. Lactobacillus rhamnosus PL1 and Lactobacillus plantarum PM1 versus placebo as a prophylaxis for recurrence urinary tract infections in children: A study protocol for a randomised controlled trial. BMC Urology. 2020; 20 (1): 168. doi.org/10.1186/s12894-020-00723-1
CrossRef - Xin, Y., Hu, C., Li, Y., et al. Immunomodulatory potential of Lactobacillus helveticus KLDS 1.8701 postbiotics: by regulating the Th17/Treg balance. Food Bioscience. 2024; 104842. doi.org/10.1016/j.fbio.2024.104842
CrossRef - Mazinani, S., Motamedzadegan, A., Naghizadeh Raeisi, S., et al. Impact of pea protein isolate in partial substitution of milk protein concentrate on the microstructural, rheological, and sensory properties of bacteriologically acidified feta-type cheese. Food Process. Preserv. 2020; 44: e14448.
CrossRef - Farahmand, N., Ouoba, L.I., Naghizadeh Raeisi, S., et al. Probiotic lactobacilli in fermented dairy products: selective detection, enumeration and identification scheme. 2021; 9: 1600. doi.org/10.3390/microorganisms9081600
CrossRef - Ewe J.A., Loo, S.Y. Effect of cream fermentation on microbiological, physicochemical and rheological properties of L. helveticus-butter. Food Chemistry. 2016; 201: 29-36. doi.org/10.1016/j.foodchem.2016.01.049
CrossRef - Ferreira L., Borges A., Gomes D., et al. Adding value and innovation in dairy SMEs: From butter to probiotic butter and buttermilk. Journal of Food Processing and Preservation. 2022; 46: e14867. doi:10.1111/jfpp.14867
CrossRef - Seo C.W., Oh N.S. Rheological, physicochemical, microbiological, and aroma characteristics of sour creams supplemented with milk protein concentrate. Food Science of Animal Resources. 2023; 43: 540-551. doi.org/10.5851/kosfa.2023.e16
CrossRef - Gavrilova E., Anisimova E., Gabdelkhadieva A., et al. Newly isolated lactic acid bacteria from silage targeting biofilms of foodborne pathogens during milk fermentation. BMC Microbiology. 2019; 19: 248. doi.org/10.1186/s12866-019-1618-0
CrossRef - Nikitina E., Petrova T., Sungatullina A., et al. The Profile of Exopolysaccharides Produced by Various Lactobacillus Species from Silage during Not-Fat Milk Fermentation. Fermentation. 2023; 9: 1-19. doi.org/10.3390/fermentation9020197
CrossRef - Sungatullina A., Petrova T., Nikitina E. Investigation on Fermented Milk Quality after the Addition of Flaxseed Mucilage and the Use of Lactobacillus delbrueckii subsp. bulgaricus and Lactiplantibacillus plantarum AG9. Front Biosci (Elite Ed). 2024; 16(2): 11. doi.org/10.31083/j.fbe1602011
CrossRef - Mikshina P., Kharina M., Sungatullina A., et al. Influence of flaxseed mucilage on the formation, composition, and properties of exopolysaccharides produced by different strains of lactic acid bacteria. International Journal of Biological Macromolecules. 2024; 281: 136092. doi.org/10.1016/j.ijbiomac.2024.136092
CrossRef - Pavlova A., Ozhegov G., Yahia M.N., et al. Whole genome sequence data of Lactobacillus fermentum AG8, the producer of antibacterial peptides. IOP Conf. Series: Earth and Environmental Science. 2021; 715(1): 012071. doi.org/1010.1088/1755-1315/715/1/012071
CrossRef - Masood M. I., Qadir M. I., Shirazi J. H., et al. Beneficial effects of lactic acid bacteria on human beings. Critical Reviews in Microbiology. 2010; 37(1): 91–98. doi.org/10.3109/1040841X.2010.536522
CrossRef - Wan Z., Zhang J., Cui F., et al. The effect of Lacticaseibacillus rhamnosus LRa05 probiotic tablets on the immune function of mice and its safety. Journal of Functional Foods. 2025; 127: 106752, doi.org/10.1016/j.jff.2025.106752.
CrossRef - Wong W.-Y., Chan B. D., Cho P.-T., Leung T.-W., Tai W. Ch.-S. Beneficial and immunomodulatory effects of heat-killed Lactobacillus plantarum L137 in normal and acute colitis mice. Journal of Functional Foods. 2024; 116: 106167. org/10.1016/j.jff.2024.106167.
CrossRef - Ward N.C., Pang J., Ryan J.D.M., et al. Nutraceuticals in the management of patients with statin-associated muscle symptoms, with a note on real-world experience. Cardiol. 2018; 41: 159–165. doi.org/10.1002/clc.22862
CrossRef - Hill C., Guarner F., Reid G., et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014; 11: 506–514. doi.org/10.1038/nrgastro.2014.66
CrossRef - Liu D. M., Guo X. A., Zeng J., et al. The probiotic role of Lactobacillus plantarum in reducing risks associated with cardiovascular disease. J. Food Sci. Technol. 2016; 52: 127–136. doi .org/ 10 .1111/ ijfs .13234.
CrossRef - Liu Y., Zhao F., Liu J., et al. Selection of cholesterol-lowering lactic acid bacteria and its effects on rats fed with high-cholesterol diet. Microbiol. 2017; 74: 623–631. doi.org/10 .1007/ s00284 -017 -1230 -1.
CrossRef - Tian L., Liu R., Zhou Z., et al. Probiotic characteristics of Lactiplantibacillus Plantarum N-1 and its cholesterol-lowering effect in Hypercholesterolemic rats. Probiotics and Antimicrobial Proteins. 2022; 14 (2): 337-348. doi.org/10.1007/s12602-021-09886-1.
CrossRef - Zafar H., Ain N.U., Alshammari A., et al. Lacticaseibacillus rhamnosus FM9 and Limosilactobacillus fermentum Y57 Are as Effective as Statins at Improving Blood Lipid Profile in High Cholesterol, High-Fat Diet Model in Male Wistar Rats. 2022; 14(8): 1654. doi.org/10.3390/nu14081654.
CrossRef - Li X, Xiao Y, Song L, et al. Effect of Lactobacillus plantarum ht121 on serum lipid profile, gut microbiota, and liver transcriptome and metabolomics in a highcholesterol diet-induced hypercholesterolemia rat model. Nutrition (Burbank, Los Angeles County, Calif.). 2020; 79-80: 110966. doi.org/10.1016/j.nut.2020.110966
CrossRef - Keleszade E., Kolida S., Costabile A. The cholesterol lowering efficacy of Lactobacillus plantarum ECGC 13110402 in hypercholesterolemic adults: a double-blind, randomized, placebo controlled, pilot human intervention study. Journal of Functional 2022; 89: 104939, doi.org/10.1016/j.jff.2022.104939.
CrossRef - Yue F., Han H., Xu J., et al. Effects of exopolysaccharides form Lactobacillus plantarum KX041 on high fat diet-induced gut microbiota and inflammatory obesity. International Journal of Biological Macromolecules. 2025; 289: 138803, doi.org/10.1016/j.ijbiomac.2024.138803.
CrossRef - Thakkar P.N., Patel A., Modi H.A., et al. Hypocholesterolemic effect of potential probiotic Lactobacillus fermentum strains isolated from traditional fermented foods in wistar rats. Probiotics Antimicrob Proteins. 2020; 12(3): 1002-1011. doi.org/10.1007/s12602-019-09622-w
CrossRef - Palaniyandi S.A., Damodharan K., Suh J.W., et al. Probiotic characterization of Cholesterol-Lowering Lactobacillus fermentum MJM60397. Probiotics Antimicrob Proteins. 2020; 12(3): 1161-1172. doi.org/10.1007/s12602-019-09585-y
CrossRef - de Luna Freire M. O., Cruz Neto J. P. R., de Albuquerque Lemos D. E., et al. Limosilactobacillus fermentum strains as novel probiotic candidates to promote host health benefits and development of biotherapeutics: A comprehensive review. Probiotics Antimicrob Proteins. 2024; 16(4): 1483–1498. doi.org/10.1007/s12602-024-10235-1
CrossRef - Le Roy T., Lecuyer E., Chassaing B., et al. The intestinal microbiota regulates host cholesterol homeostasis. BMC Biol. 2019, 17, 94. doi.org/10.1186/s12915-019-0715-8.
CrossRef - Molinero, N., Ruiz, L., Sanchez, B., et al. Intestinal Bacteria Interplay with Bile and Cholesterol Metabolism: Implications on Host Physiology. Physiol. 2019; 10: 185. doi.org/10.3389/fphys.2019.00185
CrossRef - Molina-Tijeras J. A., Diez-Echave P., Vezza T., et al. Lactobacillus fermentum CECT5716 ameliorates high fat diet-induced obesity in mice through modulation of gut microbiota dysbiosis. Pharmacological research. 2021; 167: 105471. https://doi.org/10.1016/ j.phrs.2021.105471
CrossRef - Zhao W., Peng C., Sakandar H.A., et al. Meta-Analysis: Randomized Trials of Lactobacillus plantarum on Immune Regulation Over the Last Decades. Front Immunol. 2021; 12: 643420. doi: 10.3389/fimmu.2021.643420.
CrossRef - Wu Y., Zhang Q., Ren Y., et al. Effect of probiotic Lactobacillus on lipid profile: A systematic review and meta-analysis of randomized, controlled trials. PLoS ONE. 2017; 12: e0178868. doi.org/10.1371/journal.pone.0178868
CrossRef - Zhao Y., Hong K., Zhao J., et al. Lactobacillus fermentum and its potential immunomodulatory properties. Journal of Functional Foods. 2019; 56: 21-32. doi.org/10.1016/j.jff.2019.02.044.
CrossRef - Huang, J., Cai, X., Liu, X., et al. Lactobacillus fermentum B153 from human colostrum modulates intestinal immunity and gut microbiota in obese mice model. Journal of Functional Foods. 2025; 125: 106662, doi.org/10.1016/j.jff.2025.106662.
CrossRef
Abbreviations List
ALT – Alanine aminotransferase
AST – Aspartate aminotransferase
HDLP – High-density lipoprotein
LAB – Lactic acid bacteria
LDLP – Low-density lipoprotein
LY – Lymphocytes
LY % – Percentage of lymphocytes
PCA – Principal Component Analysis
TC – Total cholesterol
TG – Triglycerides













