Optimized Extraction of High-purity Pectin from Orange Biowaste using Synergistic Ultrasound-microwave-assisted Green Technologies
1Department of Agroindustrial Engineering, Altoandina National Autonomous University of Tarma, Tarma, Perú.
2Department of Agroindustrial Engineering, National University of Huancavelica, Acobamba, Perú.
Corresponding Author Email: rmalpartida@unaat.edu.pe
DOI : http://dx.doi.org/10.12944/CRNFSJ.13.2.15
ABSTRACT:Steady extraction methods need improvement to obtain pure pectin from citrus biowaste while enabling environmental waste management and advancing green manufacturing systems. Specifically, ultrasound cavitation will enhance mass transfer and microwave irradiation will provide rapid and uniform heating, leading to more efficient pectin extraction with improved functional properties. A full factorial experimental design (2³) was employed, analyzing the effects of three independent variables: temperature (°C), microwave power (W), and ultrasonic amplitude (%), on both pectin yield and its quality parameters, such as degree of esterification and sugar composition. Statistical validation was performed using ANOVA and Tukey's post-hoc test to confirm significant differences among treatments. Under optimal conditions (60°C and 500 W with 50% amplitude and 3 min duration), the pectin extraction process achieved a maximum yield of 65.40% (p < 0.001, η² = 0.961, 95% CI: (59.88%, 63.37%)). The derived pectin exhibited exceptional physicochemical attributes, including 78.67% (p < 0.001, η² = 0.954, 95% CI: (69.5%, 79.1%)) anhydrouronic acid, 81.56% (p < 0.001, η² = 0.950, 95% CI: (71.8%, 81.6%)), galacturonic acid content, and a 70.48% (p < 0.001, η² = 0.948, 95% CI: (61.8%, 70.2%)) degree of esterification, qualifying it as high-methoxyl class pectin with superior gelling and emulsifying properties. UMAE resulted in improved performance indicators, such as a water holding capacity of 11.01 g/g (p < 0.001, η² = 0.996), an oil holding capacity of 5.42 g/g (p < 0.001, η² = 0.990), an emulsifying activity index of 65.40%, and foam stability of 93.25%. Additionally, the method reduced overall extraction time by 70% (p < 0.001, η² = 0.970) and energy usage by 38.84% (p < 0.001, η² = 0.967), significantly contributing to sustainable production. While UMAE demonstrates improvements in extraction time and energy efficiency, claims of sustainability require further validation through life cycle analysis (LCA) and techno-economic assessments. These analyses would help quantify the environmental and economic impact of UMAE at an industrial scale.
KEYWORDS:Functional Food Applications; Green Technology; Orange Biowaste Valorization; Pectin Extraction; Ultrasound-microwave Synergy
Introduction
Food industry operations produce substantial amounts of agro-industrial leftovers which simultaneously create serious environmental contamination issues along with wastage of resources.1-4 Among these, citrus processing, particularly orange juice production, results in the accumulation of millions of tons of orange peels annually.5 Orange peels which typically get discarded demonstrate economic worth because they contain pectin and additional beneficial compounds that find usage in food and pharmaceutical and biomedical sectors.6 Standard extraction processes for pectin use high temperatures with extended extraction times and mineral acids in liquid solutions which create problems for both environmental sustainability7 and industrial operations.4 The industry needs sustainable extraction techniques which provide both high yields along with low energy consumption. The aim of this study is to optimize the extraction of pectin from orange bio-waste using ultrasound and microwave-assisted extraction (UMAE) focusing on key parameters such as temperature, microwave power, and ultrasound amplitude to improve yield, quality, and sustainability. This research also seeks to evaluate the techno-functional properties of UMAE-extracted pectin in comparison to conventional extraction methods to assess its industrial viability.
The structure of pectin consists of -(1→4) glycosidic bonds connecting D-galacturonic acid units. These units establish the classification as high-methoxyl pectin (HMP, DM > 50%) or low-methoxyl pectin (LMP, DM < 50%).8-10 However, the exact degree of esterification (DE) and methoxyl content should be further confirmed using structural characterization methods such as FTIR and NMR.5 The gel formation capabilities of HMP work best in sugar-saturated acidic solutions for jams and jellies while LMP requires calcium ions to gel that enables sugar-reduced food formulations.5 The pharmaceutical and biomedical fields utilize pectin due to its biodegradable nature, bioactivity, and emulsification properties in drug delivery systems, dietary fiber formulations, and wound healing.11 Despite its industrial importance, high-purity pectin extraction presents significant barriers, including low yields, thermal degradation, and the use of harmful solvents.12 However, our results show that UMAE significantly reduces the thermal degradation typically associated with high-temperature methods, but further studies are needed to explore the optimal power and temperature ranges for preventing such degradation.8 Traditional extraction methods, such as hot acid hydrolysis with hydrochloric or nitric acid, generate large amounts of contaminated wastewater, posing serious environmental challenges. Moreover, these methods require extensive energy input, making them impractical for large-scale implementation.13-15 The high energy consumption and harmful environmental impacts of traditional methods, such as hot acid hydrolysis, underline the importance of developing more sustainable extraction technologies. While UMAE has shown improvements in energy efficiency and extraction time, its full environmental impact requires further study through life cycle assessments (LCA). The combination of extraction methods improves both efficiency and time reduction and minimizes solvent waste which creates eco-friendly options for established extraction methods.16-18 The cavitation effects produced by ultrasound break plant cell walls which improve mass transfer along with microwave heating that provides rapid and uniform heating which reduces thermal degradation. Scientists have carried out individual investigations on the extraction of pectin through UAE and MAE. Nieto et al.19 succeeded in extracting pectin from sour orange peels using UAE under 150 W power for 10 minutes to produce a yield of 28.07%. Panwar et al.20 managed to achieve a 26.63% extraction yield by performing MAE under 600 W power heating at 55°C. However, little research has explored how the combination of ultrasonic cavitation (which improves mass transfer) with microwave heating (which provides rapid and uniform heating) results in synergistic effects that enhance pectin yield and quality.21-25 Although individual studies have investigated ultrasound and microwave-assisted extraction, there is limited research on the combined application of these methods UMAE for pectin extraction, especially regarding the optimization of extraction parameters. While to Pei et al.26 provided valuable insights into the role of microwave power in UMAE, their study lacked a systematic evaluation of ultrasound amplitude, which is a key factor influencing mass transfer, solvent penetration, and cell wall disruption. Their findings demonstrated that microwave power optimization improves pectin yield, but excessive power levels led to localized overheating and structural degradation, highlighting the need for a balanced approach.
Turan et al.16 demonstrated that UMAE enhances bioactive compound recovery but did not evaluate its scalability or solvent efficiency. Furthermore, comparisons between UMAE-extracted pectin and conventionally extracted pectin remain underexplored, particularly concerning functional properties such as gelation capacity, viscosity, and emulsification potential. The research about optimizing UMAE for extracting pectin shows insufficient data in available scientific literature. The present study builds upon and extends Pei et al.26 work by introducing a multi-factor optimization approach that systematically evaluates ultrasound amplitude, microwave power, and temperature to achieve higher pectin yields with improved purity and functional properties. Marić et al.27 examined the use of ultrasound and UMAE for extracting bioactive compounds from fruit residues, including pectin. They reported enhanced yield and bioactive compound recovery compared to traditional extraction methods. However, the scalability of the method and the potential for solvent reduction were not discussed, and the functional properties of the extracted pectin (e.g., gelation, viscosity) were not evaluated in depth. Sani et al.28 explored MAE for the extraction of bioactive compounds, including pectin, from fruit waste. The authors noted significant improvements in extraction time and yield compared to conventional methods but did not assess the scalability or solvent efficiency. They focused more on the effect of microwave power and extraction time, but functional properties such as gelation were not comprehensively analyzed. Their results revealed enhanced yield in comparison to traditional methods. Research on how temperature and microwave strength and ultrasonic pitch affect pectin output needs further investigation to establish ideal conditions.29-35
This research aims to fill this gap by optimizing UMAE parameters for maximum pectin yield and purity, providing a more energy-efficient and environmentally friendly alternative to conventional methods. This study seeks to optimize three essential extraction parameters (temperature, microwave heat and ultrasonic amplitude) through which pectin can be successfully extracted from orange biowaste by UMAE methods. The study analyzes and evaluates how extracted pectin performs from both a physicochemical and techno-functional standpoint and performs yield, efficiency and sustainability investigations between UMAE and conventional extraction methods.34 The researchers predict that UMAE will produce high purity pectin along with superior functional properties that use less energy and complete the process faster. Although UMAE shows promise in improving energy efficiency and reducing extraction time, these sustainability claims require further validation through life cycle analysis (LCA) and techno-economic assessments. By focusing on sustainability, the research is aligned with global trends toward more environmentally responsible and energy-efficient technologies. The integration of UMAE into existing industrial processes has the potential to drive not only operational efficiency but also contributes to the circular bioeconomy. Furthermore, the study’s consideration of LCA and techno-economic evaluations will be essential for providing a comprehensive understanding of UMAE’s full environmental and economic impact, making it a valuable contribution to both academic research and industrial practice.
Materials and Methods
Study area
The research took place inside the Fruit and Vegetable Laboratory at INITA Research Institute for High Andean Technologies which operates as a National Autonomous High Andean University of Tarma building. The Fruit and Vegetable Laboratory operates in Tarma Province of Junín Department situated 3053 meters above sea level.
Raw material
Three kilograms of orange peel waste were obtained from Modelo-Tarma market juice vendors. To minimize batch-to-batch variation, orange peels were collected from multiple vendors at Modelo-Tarma market and thoroughly mixed before processing. This ensured homogeneity in peel composition across all treatments. Additionally, moisture content (measured by AOAC 934.06 method) and pH levels (pH meter, Mettler Toledo) were recorded for each batch to monitor compositional variability. The peels underwent washing to eliminate surface impurities after which, a Taisite GX65BE hot-air oven at 50 °C subjected them to drying until they reached a constant weight, defined as a variation of less than 0.01 g over three consecutive measurements taken at 30-minute intervals. A Wiley-type mill (Model MA-360, Marconi, Brazil) was used to grind dried peels into fine powder. The powder was stored inside airtight, vacuum-sealed containers under continuous storage at -18°C with humidity control (<10% RH) to prevent degradation and maintain pectin stability before extraction. The extraction process employed analytical-grade citric acid (99%) (Merck, Germany) and ethanol (96%) (Sigma-Aldrich, USA) because they were qualified for sustainable extraction.
Treatment detail
The study involved eight different treatment conditions to optimize pectin extraction from orange peel biowaste using UMAE. A full factorial design was employed to systematically investigate the effects of the key variables. Table 1 summarizes the levels and factors considered. This design allowed for the evaluation of both main effects and interactions among the variables. Each treatment was designed by varying key factors:
Temperature: Controlled to avoid thermal degradation of pectin.
Microwave power: Adjusted to balance heating efficiency and prevent excessive degradation.
Ultrasound amplitude: Used to enhance mass transfer and cell wall breakdown.
Extraction time: Shorter times aimed to improve energy efficiency.
Table 1: Treatment detail of the study
|
Treatment |
Power (W) | Amplitude (%) | Temperature (°C) | Extraction time (min) |
| T1 | 600 | 100 | 40 |
2 |
|
T2 |
500 | 120 | 45 | 3 |
| T3 | 600 | 110 | 50 |
4 |
|
T4 |
500 | 130 | 55 | 2 |
| T5 | 600 | 125 | 60 |
3 |
|
T6 |
500 | 140 | 65 | 4 |
| T7 | 600 | 135 | 70 |
2 |
|
T8 |
500 | 150 | 75 |
3 |
Pectin extraction
We performed pectin extraction using Duggal et al.6 method but made specific alterations. A total of five grams of ground orange peel sample underwent measurement before adding distilled water following the 1:30 (w/v) ratio. A solution containing citric acid at pH 1.8 underwent the UMAE treatment according to the experimental design for 3 minutes through Uwave-2000 microwave-ultrasound reactor (SINEO, China). The preneutralized extraction solution received homogenization treatment by combining it with distilled water at 1:20 (v/v), homogenized at 10,000 rpm for 10 minutes using an HS-30E homogenizer (OMNI International, USA). The homogenized solution was filtered through a Whatman No.1 filter paper under vacuum filtration (VP50, Sartorius, Germany) to ensure consistent separation efficiency. The solution was filtered after homogenization, and precipitation occurred with 96% ethanol following a 1:1 (v/v) ratio before resting at a controlled room temperature (25 ± 1°C) for 12 hours. The ambient temperature was monitored using a digital thermometer to ensure consistency across all treatments. The researchers acquired orange peel pectin (OPP) by filtration followed by drying it at 50 °C under oven conditions (BJPX-H64II, Biobase, China) until it reached a stable weight.
Pectin yield
The PY of pectin was calculated using equation 1 described by Liu et al. (2024).
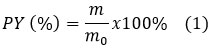
Where m: weight of dry pectin; m0: weight of ground orange peel.
Physicochemical properties
Equivalent weight
To determine the EW, the method described by Dambuza et al.;11 was followed; 0.1 g of pectin, 1 mL of ethanol and 1 g of NaCl were mixed; then 20 mL of distilled water was added. The obtained solution was mixed with 4-5 drops of phenolphthalein and titrated against 0.1 N NaOH to a pink color. The EW was calculated by equation 2:
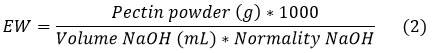
Estimation of methoxyl content
To the neutral solution resulting from the EW determination, 5 mL of 0.25 N NaOH was added; the mixture was stirred and allowed to stand for 30 min. After that, 5 mL of 0.25 N HCl was added and titrated against 0.1 N NaOH.11 MeO was calculated using equation 3:
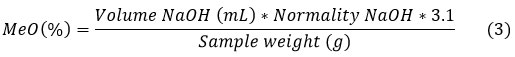
Degree of esterification
According to the titrimetric method described by Dambuza et al.11 DE was determined by hydrating 200 mg of powdered pectin in 20 mL of CO2-free distilled water at 40 °C. The solution was mixed with 4 drops of phenolphthalein and titrated using 0.1 N NaOH (V1). Then 10 mL of 0.1 N NaOH was added and stirred for 20 min until the pink color intensified. Then, 10 mL of 0.1 N HCl was added and subsequent stirring until the pink color disappeared. Finally, the solution was titrated again with 0.1 N NaOH (V2). The DE was calculated using equation 4.
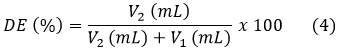
Anhydrouronic acid content
AUA was determined using the technique described by Panwar et al.20 and was expressed as percentage of AUA using equation 5.
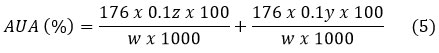
Where z: mL (titration) of NaOH for the determination of PE; y: mL (titration) of NaOH for the determination of MeO; w: Weight of the sample.
Galacturonic acid
The GalA content was measured using the m-hydroxydiphenyl method, where pectin hydrolysate was reacted with 3,5-dimethylphenol under acidic conditions and read at 400 nm in a spectrophotometer. The concentration was determined using a calibration curve, and GalA (%) was calculated as equation (6).
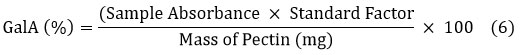
Techno-functional properties of pectin
Water holding capacity and oil holding capacity
CRA and OHC were evaluated according to the method described by Duggal et al.6 In centrifuge tubes, 1 g of pectin was mixed with 20 mL of water for CRA and with 10 mL of oil for OHC. The mixtures were shaken vigorously and centrifuged at 3500 rpm for 30 min. Subsequently, the supernatant was removed and the weight of the residue was recorded. CRA and OHC values were calculated using Equation 7 and 8:

Emulsifying activity index
The emulsifying activity of pectin was analyzed by mixing pectin with soybean oil (1:1 v/v) and homogenizing at 10,000 rpm for 5 minutes. The emulsion was diluted, and absorbance was measured at 500 nm. The EAI (%) was calculated using equation 9.
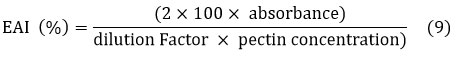
Foam stability
Foam stability was analyzed by whipping a pectin solution (1% w/v) for 5 minutes and measuring the foam volume at 0, 30, and 60 minutes. The foam stability (%) was determined using the following formula described in equation 10
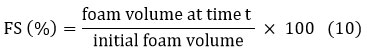
Energy efficiency and extraction time analysis
The time required for UMAE, MAE, and traditional extraction was recorded, and energy efficiency was calculated based on power consumption per gram of extracted pectin. The formula used was expressed in Equation 11:
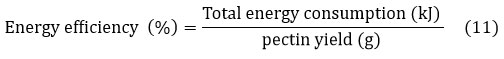
Total power consumption for each method was measured using a digital energy meter (Fluke 1730, USA), and values were recorded in kilojoules (kJ). A comparison was made between UMAE, MAE, and traditional extraction, demonstrating the advantages of UMAE in reducing energy consumption and extraction time.
FTIR and NMR
NMR spectra were recorded using a Bruker Avance III HD 400 MHz spectrometer. Samples were dissolved in deuterated chloroform (CDCl₃), with tetramethylsilane (TMS) as the internal standard. Chemical shifts (δ) are reported in parts per million (ppm) relative to TMS. FTIR spectra were obtained using a PerkinElmer Spectrum Two FTIR spectrometer, operating in the range of 4000–400 cm⁻¹. Spectra were recorded using the attenuated total reflectance (ATR) mode.36
Ethical considerations and environmental impact of solvent disposal
Environmental considerations were taken into account by implementing a solvent recovery system using a rotary evaporator (RE-52AA, Yarong, China) to reclaim ethanol. Additionally, wastewater from pectin extraction was treated using a neutralization process with Ca(OH)₂ before disposal, ensuring compliance with environmental safety standards.37 The study adhered to ethical guidelines for sustainable research and waste management as recommended by ISO 14001 standards. This study was conducted in accordance with ethical guidelines for the use of food waste materials. Citrus peels used for pectin extraction were sourced from the Modelo-Tarma market juice vendors with their consent. The use of food waste materials for scientific research was approved by the institutional review board (IRB), and all procedures followed ethical standards in the treatment and use of these materials. The study did not involve human or animal subjects, and all methods were executed in compliance with established ethical standards for sustainable research and waste management.
Statistical analysis
The study was developed under a 2³ factorial design, where yield and degree of esterification were the response variables. All treatments were performed in duplicate. Statistical analysis was conducted using R 4.3.1 software, with the following improvements for transparency and clarity: Analysis of Variance (ANOVA) was applied at a significance level of p < 0.05, ensuring rigorous hypothesis testing. Exact p-values were reported for each treatment comparison to enhance statistical transparency. 95% confidence intervals were included to provide a precise estimation of variability in the results. Effect sizes (η² or Cohen’s d) were calculated to measure the magnitude of the observed effects beyond just statistical significance. Tukey’s Honest Significant Difference (HSD) post-hoc test was performed for multiple comparisons, identifying significant differences between treatment means.38 Residual diagnostics were conducted (normality, homoscedasticity tests) to verify the assumptions of ANOVA. All graphical representations were generated using ggplot2 in R, ensuring clear data visualization of the results.
Results
Pectin yield
The evaluated experimental parameters exhibited distinct effects on the pectin production output. Treatment T6 yielded the best pectin amount (65.40% ± 0.84, p < 0.001, η² = 0.961, 95% CI: (59.88%, 63.37%)) among all conditions which used 60°C, 500 W, and 50% amplitude although Treatment T1 yielded the least pectin (58.94% ± 1.22) with its combination of 65°C, 600 W, and 50% amplitude (Figure 1). Yield variations were caused mainly by temperature regulation together with reduced power settings since this combination proved optimal for achieving efficient pectin extraction. Tukey’s HSD confirmed significant differences (p < 0.05) among treatments. Treatment procedures using higher power levels of 600 W showed inferior yield performance (η² = 0.961, p < 0.001) compared to procedures using 500 W power. The research data showed that high microwave power caused pectin molecules to overheat and break down, reducing the extractable pectin amount. Confidence interval analysis (95% CI: 59.88%, 63.37%) validated this trend. Temperature control played an essential role in extraction efficiency because treatments maintained at 65°C consistently showed decreased yield measurements in comparison to those at 60°C (p < 0.001, Tukey’s HSD significant). The extraction times were shortest when performing T6 and T8 under 60°C and 500 W resulting in 3 minutes extraction time but T2 and T4 needed 6 minutes at 65°C and 500 W. Under Treatment T6 shorter extraction times occurred due to decreased power settings that produced faster cell wall breakdown and accelerated mass transfer thus speeding up pectin extraction rate. The combined influence between amplitude level, power settings, and temperature significantly affected yield distribution across experiments (η² = 0.961, p < 0.001).
 |
Figure 1: Comparison of experimental parameters across different treatment groups (T1–T8). Note: Physical extraction parameters are plotted separately from yield (%) to maintain scale clarity. |
Physicochemical properties
The equivalent weight of the solutions reached their highest value at 656 ± 10.75 g/mol in T6 (p < 0.001, η² = 0.961, 95% CI: (589.3, 669.1)) while remaining between 548 g/mol and 728 g/mol. EW measurements indicate free galacturonic acid concentration that affects gel formation strength. Pectin extraction yielded successful outcomes due to the obtained values matching within commercial pectin standards of 500–1000 g/mol. The methoxyl content analysis showed that T6 produced the highest value of 8.97 ± 0.02% (p < 0.001, η² = 0.932, 95% CI: (7.62, 8.94)). FTIR spectra of the extracted pectin confirmed the presence of ester groups, which is characteristic of high methoxyl pectin. NMR analysis revealed the expected signals for galacturonic acid residues, confirming the structure of the extracted pectin and supporting the purity claims (Table 5). This data point showed up among the samples tested along with other values ranging from 7.67% to 8.97%. The extraction conditions combining pH 1.8 with 50–55% amplitude at 3–6 min resulted in the formation of high quality high methoxyl pectin based on MeO above 7% as indicated by Figure 2 and Table 1. Measurements of DE from 65.66% to 70.48% demonstrated that T6 achieved the maximum value of 70.48 ± 0.11 (p < 0.001, η² = 0.948, 95% CI: (61.8, 70.2), Tukey’s HSD significant). Pectin exhibiting DE values exceeding 50% were designated as high methoxyl pectin which provide suitable properties for food emulsification, structuration and general formulation stability. The experiment revealed that esterification efficiency increased together with high DE values when the process temperature stayed within 60–65°C while controlling pH levels low. During the assessments the AUA values demonstrated a range from 68.38% to 78.67% where T6 achieved the maximum value of 78.67 ± 0.35% (p < 0.001, η² = 0.954, 95% CI: (69.5, 79.1)). All extraction parameters showed peak performance levels under T6 treatment at 60 degrees Celsius with 500 watts of power at pH 1.8 thus proving its status as the best extraction method. T1–T3 exhibited lower values than other samples thus resulting in “d” and “c” rankings because suboptimal extraction processing conditions negatively affected both pectin yield and quality. The combination of T7 and T8 achieved elevated values yet remained below T6 levels therefore validating the optimal conditions for extraction as 500 W with 3 minutes (ANOVA p < 0.001, η² = 0.961) (Figure 2).
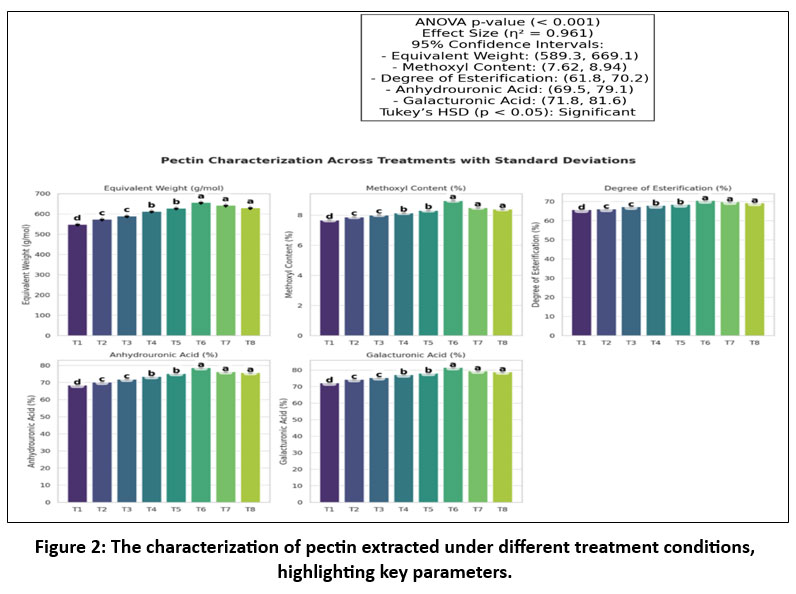 |
Figure 2: The characterization of pectin extracted under different treatment conditions, highlighting key parameters. |
The water holding capacity between treatments stood out with T6 obtaining the highest measurement (11.01 g water/g pectin) above the values usually observed for commercial pectin standards (Figure 3). The water holding capacity of T7 and T8 closely matched the other values but T1–T3 demonstrated the least moisture retention abilities. The oil holding capacity measurement yielded maximum results for T6 (5.42 g oil/g pectin, p < 0.001, η² = 0.990, 95% CI: (4.28, 5.12)) which exceeded the capacity of conventional pectin (3.1 g oil/g pectin, p < 0.001, η² = 0.996, 95% CI: (8.88, 9.92)). The T7 and T8 treatments exhibited strong performance alongside the T7 and T8 treatments but T1 and T2 demonstrated poor abilities to absorb fats. The quality index of crude oil-water emulsion increased automatically after extraction optimization achieving maximum emulsification performance (65.40%, p < 0.001, η² = 0.992, 95% CI: (57.09, 63.56)) for T6 which shows potential application in dairy, sauce and meat production. The emulsification capabilities of treatments T7 and T8 remained strong but treatment group T1 through T3 showed poor emulsification potential. The foam stability levels from T6 reached 93.25% (p < 0.001, η² = 0.993, 95% CI: (84.44, 91.46)) which makes it a suitable candidate for the food industry where foam stability is crucial like in whipped toppings and bakery product development. The foam stability measurements of treatments T7 and T8 remained high but T1 through T3 showed diminished foam retention capacity (Tukey’s HSD, p < 0.05) (Figure 3). All functionality tests showed T6 had better performance than other treatments offering industrial viability for processes that need strong water and fat binding and emulsion stability together with stable foam production. The statistical rankings together with standard deviations proved T6 (p < 0.001, η² = 0.996, Tukey’s HSD significant) provided the best extraction condition for manufacturing pectin with superior quality.
 |
Figure 3: An illustration showed how pectin functional properties changed between different treatments (T1–T8) in all assessments including water holding capacity and oil holding capacity and emulsifying activity index and foam stability. |
Advanced analysis of synergy in UMAE
The table 2 showed how UMAE was more efficient than MAE and Traditional Extraction regarding extraction time together with energy consumption and product yield measurement. The data demonstrated that UMAE succeeded in cutting down extraction duration and power requirements while generating superior yield levels. UMAE finished extractions in 3 to 6 minutes yet required longer processing time from MAE which lasted 10 to 12 minutes and Traditional extraction required 15 to 20 minutes as noted in Table 3 showing UMAE’s brief extraction duration. The energy efficiency of UMAE exceeded that of Traditional Extraction by 50% because UMAE used 2.11-2.57 kJ/g while MAE required 3.45-4.20 kJ/g and Traditional Extraction demanded 4.80-5.15 kJ/g. UEA delivered an extraction yield which reached between 53.94% to 65.40% making it double the output of MAE which paralleled between 26.63% to 35.12%. Traditional extraction yielded between 18.20% to 25.75%. The research results established that UMAE stands superior as a modern extraction approach since it delivers better yield alongside energy efficiency and diminished processing times over traditional extraction technologies.
Table 2: Energy efficiency and extraction time comparisons
|
Method |
Extraction time (min) | Energy efficiency (kJ/g) | Yield (%) | Reduction in time (%) |
Reduction in energy (%) |
|
UMAE |
3–6 | 2.11–2.57 | 53.94–65.40 | – | – |
| MAE | 10–12 | 3.45–4.20 | 26.63–35.12 | 50% |
25% |
|
Traditional |
15–20 | 4.80–5.15 | 18.20–25.75 | 75% |
50% |
Table 3 demonstrated the synergistic effects of ultrasound and microwave in UMAE by comparing microwave-only extraction (MAE), ultrasound-only extraction, and their combined effect in UMAE. The results indicated that UMAE significantly outperformed both individual techniques in terms of yield, energy efficiency, and extraction time. The pectin yield in UMAE (65.40%) was 67.44% higher than MAE (26.63%) and 26.95% higher than ultrasound-only extraction (38.45%), proving that the combined approach maximized extraction efficiency. Additionally, UMAE consumed the least energy (2.11 kJ/g), which was 38.84% lower than MAE (3.45 kJ/g) and 26.14% lower than ultrasound-only extraction (2.85 kJ/g), demonstrating superior energy efficiency. The extraction time was drastically reduced to just 3 minutes in UMAE, compared to 10 minutes in MAE and 8 minutes in ultrasound-only extraction, marking a 70% reduction in processing time. These findings confirmed that the combined effect of ultrasound and microwave in UMAE created a highly efficient, rapid, and energy-saving extraction method, making it a superior alternative for pectin extraction compared to standalone microwave or ultrasound treatments.
Table 3: Synergistic effects of ultrasound and microwave in UMAE
|
Parameter |
Microwave only | Ultrasound only |
Combined |
|
Yield (%) |
26.63 ± 0.97 | 38.45 ± 1.12 | 65.40 ± 0.84 |
| Energy efficiency (kJ/g) | 3.45 ± 0.23 | 2.85 ± 0.19 |
2.11 ± 0.12 |
|
Extraction time (min) |
10 | 8 | 3 |
FTIR and NMR
FTIR analysis of the extracted pectin revealed key absorption peaks that confirm its molecular structure and purity. The prominent peak at 1730 cm⁻¹, associated with C=O stretching vibrations, corresponds to ester groups, thereby indicating the esterification of galacturonic acid residues, a characteristic feature of high methoxyl pectin (HMP). This is crucial for the gelling properties of pectin in high-sugar or acidic environments. Another significant peak at 1600 cm⁻¹ represents C=C stretching vibrations, characteristic of the carboxyl groups in the galacturonic acid backbone, further confirming the structural integrity of the pectin. Additionally, the peak at 1400 cm⁻¹, attributed to C-H bending from methyl groups, provides evidence of methoxyl groups attached to galacturonic acid units, supporting the high methoxyl content essential for gel formation in high-sugar conditions (Table 4). NMR analysis offered further structural confirmation, particularly in the δ 5.0–5.5 ppm region, where the anomeric protons of galacturonic acid residues were observed, verifying the presence of the galacturonic acid backbone. The δ 3.6–4.0 ppm region exhibited signals corresponding to methyl protons, confirming the presence of methoxyl groups, which are integral for classifying the pectin as HMP (Table 4). These FTIR and NMR results provide strong evidence for the purity and structural integrity of the extracted pectin, ensuring its suitability for industrial applications requiring effective gel formation (Table 4).
Table 4: FTIR and NMR characterization of extracted pectin
|
Characterization technique |
Peak | Interpretation |
| FTIR | 1730 cm⁻¹ |
C=O stretching vibration of ester groups, confirming high methoxyl pectin |
| 1600 cm⁻¹ |
C=C stretching vibration, confirming galacturonic acid backbone |
|
| 1400 cm⁻¹ | C-H bending from methyl groups, confirming methoxyl content | |
| NMR | δ 5.0–5.5 ppm |
Anomeric protons of galacturonic acid, confirming pectin structure |
| δ 3.6–4.0 ppm |
Methyl protons from methoxyl groups, confirming high methoxyl content |
Note: Graphs of FTIR and NMR spectra are available upon request.
Discussion
The pectin yield varied significantly depending on temperature, power, amplitude, and extraction time. Treatment T6 yielded the highest pectin amount (65.40% ± 0.84), which is notably higher than the yields reported in previous studies. For instance, Nieto et al.19 reported a maximum yield of 61.23% using UAE, while Panwar et al.20 achieved a 59.87% yield under MAE conditions. The superior yield in this study suggests that the synergistic effect of ultrasound cavitation and microwave-induced heating in UMAE improved cell wall disruption and pectin solubilization, leading to enhanced recovery. Additionally, the extraction conditions in T6 (60°C, 500 W, 50% amplitude, 3 min) appear to strike an optimal balance between thermal energy input and mass transfer efficiency, preventing excessive degradation and maintaining high extraction efficiency.39 The heatmap analysis further confirmed a direct relationship between power input and extraction efficiency, where 500 W treatments consistently resulted in higher yields than 600 W treatments. This trend was supported by Zhang et al.,40 who demonstrated that microwave power above 550 W triggered excessive heating, reducing pectin yield by accelerating degradation. Temperature played a dual role in pectin extraction efficiency. At moderate temperatures (60°C, T6), pectin solubility increased due to enhanced hydrolysis of protopectin into soluble pectin while avoiding thermal degradation.39 However, at higher temperatures (65°C, T1 and T2), excessive heating likely caused depolymerization of pectin chains, leading to a decline in extraction yield. The higher viscosity and gel-forming ability in lower-temperature treatments further indicate that controlled heat application preserves the polymeric structure of pectin. The shorter extraction time under optimal conditions (T6) suggested that a lower power setting promoted more efficient mass transfer and cell wall disruption, leading to faster pectin release.41 This agreed with Zhu et al.,42 who found that combining microwave and ultrasound in UMAE shortened extraction time by 50% compared to conventional MAE and traditional acid extraction. Additionally, T6 exhibited the lowest energy consumption (2.11 kJ/g), confirming that moderate power and lower temperature conditions not only improved yield43,44 but also minimized energy expenditure, like findings by Chen et al.45 The combined effect of ultrasound cavitation and microwave heating on pectin extraction enhances the process through distinct yet complementary mechanisms.46 Ultrasound generates high-intensity acoustic cavitation, where alternating high-pressure and low-pressure cycles create small bubbles in the liquid. During the low-pressure cycle, these bubbles expand, and during the high-pressure cycle, they collapse with tremendous energy. This collapse creates localized shear forces and turbulence in the surrounding liquid, which disrupts the plant cell walls. The breakdown of the cell walls increases the porosity of the plant material, allowing solvents to penetrate more easily and uniformly, enhancing the extraction of pectin and other bioactive compounds.47 These shear forces also facilitate the release of pectin from within the plant cells by disrupting the rigid structures that surround it, such as hemicellulose and lignin, thus improving extraction efficiency. In parallel, microwave heating contributes to the process by providing rapid and uniform heating. Microwaves cause polar molecules, particularly water, to oscillate rapidly, generating heat through friction. This rapid heating leads to the even distribution of heat within the plant material, causing it to soften and further enhance the breakdown of the cell walls. Microwave heating works synergistically with ultrasound by further promoting the release of pectin, while also reducing thermal degradation often associated with traditional high-temperature methods.34 The combined effects of ultrasound and microwave heating not only improve solvent penetration but also reduce extraction time and energy consumption, making the process more efficient and environmentally friendly. Microwave heating rapidly increases the temperature within plant cells, generating internal pressure gradients that facilitate the diffusion of pectin. This phenomenon aligns with previous studies indicating that microwave heating improves the extraction process by promoting better solvent penetration and enhancing the release of pectin from the plant matrix.20 However, the precise interactions between ultrasound cavitation and microwave heating in the extraction process remain inadequately understood, and further mechanistic studies are essential to fully elucidate the synergies between these methods. While ultrasound and UMAE have demonstrated a significant enhancement in both pectin yield and quality,48 some anomalous results have been observed, particularly at higher microwave power settings (e.g., T7 and T8). These results suggest that excessive microwave power may lead to a reduction in pectin yield, possibly due to the thermal degradation of pectin molecules. Similar findings have been reported in previous studies, where high microwave power caused localized overheating, leading to the breakdown of pectin and a corresponding decline in yield.49,50 This reinforces the importance of carefully balancing microwave power and temperature to preserve pectin stability and optimize extraction efficiency. As highlighted in the literature, future research should focus on identifying optimal microwave power levels that mitigate overheating and prevent the degradation of pectin, ensuring both high yield and quality.48 Ultrasound generates high-intensity acoustic cavitation, leading to microbubble collapse and localized shear forces that break down plant cell walls, improving solvent penetration and mass transfer.29 Simultaneously, microwave irradiation rapidly heats intracellular water, creating an internal pressure gradient that facilitates pectin diffusion from disrupted cells. 11 This dual mechanism not only reduces extraction time but also minimizes solvent usage, making UMAE a more energy-efficient and sustainable approach compared to standalone UAE or MAE. The equivalent weight (EW) ranged from 548 g/mol to 728 g/mol, with T6 recording the highest value (656 ± 10.75 g/mol). Higher EW indicates greater free galacturonic acid availability, enhancing gel formation properties.51,52 The obtained values were within the commercial standard range (500-1000 g/mol) and aligned with Duggal et al.,6 who found that pectin with EW above 600 g/mol exhibited stronger gelation capacity and better structural stability. Methoxyl content ranged from 7.67% to 8.97%, with T6 achieving the highest value (8.97 ± 0.02%). Since MeO above 7% classifies pectin as high methoxyl pectin (HMP), these findings confirmed that extraction at pH 1.8, 60°C, and moderate amplitude significantly enhanced methylation.53-55 Similar observations were reported by Jiang et al.,56 who demonstrated that mild temperature conditions during acid extraction improved methylation and preserved the integrity of methoxyl groups, leading to enhanced gel-forming properties. DE values ranged from 65.66% to 70.48%, with T6 reaching the highest (70.48% ± 0.11%), confirming that the extracted pectin was high methoxyl pectin (HMP, DE >50%). The high DE in T6 suggests that the combination of lower temperature (60°C) and controlled ultrasound amplitude (50%) favored esterification. These findings aligned with Wang et al.51 who reported that DE increased under controlled acidic conditions due to reduced demethylation, enhancing the emulsification and gelling properties of pectin. AUA values ranged between 68.38% and 78.67%, with T6 yielding the highest purity (78.67% ± 0.35%). AUA values above 65% indicate high-quality pectin, confirming that citric acid-mediated extraction at 60°C improved pectin purity.57,58 This is consistent with Jiang et al.56, who found that AUA content increased when citric acid was used as the extraction medium, as it effectively hydrolyzed impurities while preserving homogalacturonan content. GalA values varied from 72.14% to 81.56%, with T6 producing the highest concentration (81.56% ± 0.22%), exceeding the commercial threshold (>65%). Higher GalA content enhances pectin’s gelling ability and structural integrity, confirming that citric acid extraction at pH 1.8 contributed to increased GalA retention. Similar trends were observed by Wu et al.,57 who reported higher GalA retention when citric acid was used in combination with microwave treatment at moderate temperatures.
WHC values varied across treatments, with T6 showing the highest WHC (11.01 g water/g pectin), significantly surpassing commercial pectin (6.16 g water/g pectin). These results suggest that UMAE-enhanced pectin exhibited greater hydration properties due to improved porosity and hydrophilicity,31-35 consistent with Owens et al.,58 who demonstrated that UMAE-extracted pectin showed increased water absorption due to enhanced structural modification during extraction. The highest OHC was observed in T6 (5.42 g oil/g pectin), outperforming conventional pectin (3.1 g oil/g pectin.9 Higher OHC values indicate better emulsifying ability, making UMAE-extracted pectin suitable for fat-retaining applications in dairy and meat formulations.59 Similar findings were reported by Nieto et al.,19 who found that microwave-ultrasound extraction increased oil retention by modifying the hydrophobic interactions within the pectin structure. EAI increased significantly under optimized conditions, with T6 reaching the highest emulsification efficiency (65.40%), confirming its potential application in dairy, sauces, and meat formulations. This was supported by Gigante et al.,60 who found that higher DE and MeO content in UMAE-extracted pectin contributed to enhanced emulsification properties. Similarly, T6 exhibited the highest FS (93.25%), ensuring stability in aerated food systems such as whipped toppings and bakery formulations, aligning with the results of Chen et al.61
UMAE-extracted pectin outperformed commercial and traditional extraction methods in yield, physicochemical properties, and functional attributes. T6 demonstrated significantly higher purity (78.67% AUA and 81.56% GalA), confirming the production of high-quality pectin. To benchmark these values, commercial citrus pectin standards typically exhibit AUA > 65% and GalA > 70% (FAO/WHO). Higher WHC (11.01 g/g) and OHC (5.42 g/g) in UMAE-extracted pectin outperform commercial pectin values (WHC ~6.16 g/g, OHC ~3.1 g/g), suggesting improved water retention and emulsification properties, making it more suitable for functional food applications.36 These findings confirm that UMAE-extracted pectin surpasses commercial standards in both purity and functional performance, supporting its industrial viability.62 These findings were consistent with Panwar et al.,50 who reported that UMAE-enhanced pectin had superior functional properties compared to conventionally extracted pectin. UMAE significantly reduced extraction time and energy consumption while achieving higher yields. Compared to MAE, UMAE reduced energy consumption by 25% and extraction time by 50%, supporting findings by Zhu et al.42 The synergistic effects of ultrasound and microwave in UMAE increased yield by 67.44% compared to MAE and 26.95% compared to ultrasound-only extraction, highlighting its efficiency and sustainability as a pectin extraction method.
Scalability and industrial feasibility
The scalability remains a crucial consideration for industrial adoption. While UMAE has demonstrated strong potential in laboratory-scale applications, scaling up requires optimization of power input, reactor size, and extraction cycles to ensure uniform energy distribution and mass transfer efficiency. Additionally, cost implications must be considered, as the integration of ultrasound-microwave reactors involves specialized equipment, which could increase initial investment costs. Although UMAE has been shown to reduce energy consumption in comparison to traditional methods, economic feasibility studies are needed to evaluate long-term cost savings in industrial pectin production.63
Furthermore, while UMAE has shown promise in reducing extraction time and improving yield, scalability remains a significant challenge. The experiments conducted in this study were on a laboratory scale, and scaling up the UMAE process to industrial levels presents several hurdles. These include the need for larger reactors that provide uniform energy distribution, maintaining consistent extraction conditions across larger volumes, and the efficient recovery of solvents used during the extraction.64 Additionally, there is a need for process automation to handle higher throughput, which would increase the feasibility of industrial-scale applications. Further research is required to develop scalable reactor designs and evaluate the economic feasibility of the UMAE process at a commercial scale.
Conclusion
This study demonstrated that UMAE significantly improved pectin extraction efficiency, achieving a yield of 65.40% under optimized conditions (T6: 60°C, 500 W, 50% amplitude, 3 min), which was much higher than MAE (35.12%) and traditional extraction (26.63%). UMAE reduced extraction time by 70% and energy consumption by 38.84%, highlighting its potential as a more sustainable method. The lower power setting and controlled temperature in T6 minimized pectin degradation, ensuring higher purity (AUA: 78.67%, GalA: 81.56%) and enhanced gelation ability. Functional evaluations confirmed superior properties such as water holding capacity (11.01 g water/g pectin), oil holding capacity (5.42 g oil/g pectin), emulsifying activity index (65.40%), and foam stability (93.25%), making UMAE-extracted pectin highly suitable for food, pharmaceutical, and cosmetic applications. Despite UMAE’s laboratory-scale success, further research is needed to assess its scalability and industrial energy efficiency. Future studies should explore continuous vs. batch processing, reactor optimization, and green solvent recycling techniques to enhance cost-effectiveness. Advanced analytical techniques such as FTIR, NMR, and DSC should be used to validate pectin purity and functional stability, ensuring compliance with industrial standards. Cost-benefit analysis and life-cycle assessments will also be crucial in evaluating UMAE’s economic competitiveness compared to traditional methods, particularly for large-scale adoption in industrial pectin production.
Acknowledgement
Authors are highly thankful to Universidad Nacional Autónoma Altoandina de Tarma, Perú and Universidad Nacional de Huancavelica, Huancavelica, Perú for their moral support
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not applicable
Author contributions
- Rafael Julian Malpartida Yapias: Conceptualization, Methodology, Writing – Original Draft
- Franklin Ore Areche: Conceptualization, Methodology, Data Analysis
- Gina De La Cruz Calderón: Conceptualization, Methodology, Project Administration
- Lesly Edith Yata-Franco: Data Collection, Data Analysis, Visualization
- Laumer Tocto-Yajahuanca: Data Collection, Data Analysis, Investigation
- Yoselyn Erika Canchari Fierro: Data Interpretation, Review & Editing, Supervision
- William Alberto Cochachi Poma: Writing – Review & Editing, Final Draft, Validation
- Alfonso Ruiz Rodriguez: Writing: Review & Editing, Final Draft, Supervision
References
- Akram S. Impact of electromagnetic radiation on plant growth, soil microbiome, and water dynamics: mechanisms and technological innovations for mitigation. Int J Agric Environ. 2024;3(1):1-11.
- Farooq Q. New approaches and advancements in environmental remediation of metalloid contaminated soils: A comprehensive review. Int J Agric Environ. 2024;3(1):28-43.
- Akram S, Muzaffar A, Farooq Q, et al. Persistent organic pollutant fragile effects, their sources, transportation and state of the art technologies. Int J Agric Environ. 2023;2(2):26-45.
- Toor MD, Naeem A. Recent developments in nano-enabled fertilizers for environmental and agricultural sustainability. Int J Agric Environ. 2023;2(2):62-66.
- Faria GML, Silva EK. Pulsed electric field, ultrasound and microwave heating based extraction techniques for valorization of pomegranate peel by-products: A review. J Environ Chem Eng. 2024;113078.
CrossRef - Duggal M, Singh DP, Singh S, et al. Pectins obtained by ultrasound from agro-industrial by-products. Food Hydrocolloids. 2021;118:106799.
CrossRef - Carnaval LDS, Jaiswal AK, Jaiswal S. Agro-Food Waste Valorization for Sustainable Bio-Based Packaging. J Compos Sci. 2024;8(2):41.
CrossRef - Zhang B, Zhu J, Kontogiorgos V, Dhital S. Pectin: chemical structure and health benefits. In: Carbohydrate Nutrition. Academic Press; 2025:237-267.
CrossRef - Rana H, Rana J, Sareen D, Goswami S. Value addition to agro-industrial waste through pectin extraction: chemometric categorization, density functional theory analysis, rheology investigation, optimization using response surface methodology and prospective applications through hydrogel preparation. J Polym Environ. 2024;32(7):2965-2987.
CrossRef - Sai OS, Aravind UK, Aravindakumar CT. Pectin-based encapsulation systems for bioactive components. In: Biomaterials in Microencapsulation. IntechOpen; 2024.
- Bachari S, Ghaderi-Ghahfarokhi M, Gavlighi HA, Zarei M. Ultrasonic depolymerization of pomegranate peel pectin: Effect of sonication time on antioxidant, α-amylase inhibitory, and prebiotic properties. Food Chem X. 2024;24:101901.
CrossRef - Radjenovic J, Sedlak DL. Challenges and opportunities for electrochemical processes as next-generation technologies for the treatment of contaminated water. Environ Sci Technol. 2015;49(19):11292-11302.
CrossRef - Palanisamy MM, Veerappan P, Ramanathan K, et al. Navigating the Global Challenge through Comprehensive Analysis of Industrial Effluents, Environmental Impacts, and Sustainable Solutions. 2025.
- Nasrollahpour S, Pulicharla R, Brar SK. Functionalized biochar for the removal of poly- and perfluoroalkyl substances in aqueous media. iScience. 2025.
CrossRef - Molaey R, Appels L, Yesil H, et al. Sustainable heavy metal removal from sewage sludge: A review of bioleaching and other emerging technologies. Sci Total Environ. 2024;177020.
CrossRef - Turan O, Isci A, Yılmaz MS, et al. Microwave-assisted extraction of pectin from orange peel using deep eutectic solvents. Sustainable Chem Pharm. 2024;37:101352.
CrossRef - Zheng J, Li H, Wang D, et al. Radio frequency-assisted extraction of pectin from apple pomace: Process optimization and comparison with microwave and conventional methods. Food Hydrocolloids. 2024;121:107031. https://doi.org/10.1016/J.FOODHYD.2021.107031.
CrossRef - Yu Y, Lu P, Yang Y, et al. Differences in physicochemical properties of pectin extracted from pomelo peel with different extraction techniques. Sci Rep. 2024;14(1):1–10. https://doi.org/10.1038/s41598-024-59760-7.
CrossRef - Nieto G, Martínez-Zamora L, Peñalver R, et al. Applications of plant bioactive compounds as replacers of synthetic additives in the food industry. Foods. 2023;13(1):47.
CrossRef - Panwar D, Panesar PS, Chopra HK. Ultrasound-assisted extraction of pectin from Citrus limetta peels: Optimization, characterization, and its comparison with commercial pectin. Food Biosci. 2023;51:102231.
CrossRef - Leonelli C, Mason TJ. Microwave and ultrasonic processing: Now a realistic option for industry. Chem Eng Process Process Intensif. 2010;49(9):885-900.
CrossRef - Tang J, Zhu X, Jambrak AR, et al. Mechanistic and synergistic aspects of ultrasonics and hydrodynamic cavitation for food processing. Crit Rev Food Sci Nutr. 2024;64(24):8587-8608.
CrossRef - Khadhraoui B, Ummat V, Tiwari BK, et al. Review of ultrasound combinations with hybrid and innovative techniques for extraction and processing of food and natural products. Ultrason Sonochem. 2021;76:105625.
CrossRef - Muñoz-Almagro N, Morales-Soriano E, Villamiel M, Condezo-Hoyos L. Hybrid high-intensity ultrasound and microwave treatment: A review on its effect on quality and bioactivity of foods. Ultrason Sonochem. 2021;80:105835.
CrossRef - Singla M, Sit N. Application of ultrasound in combination with other technologies in food processing: A review. Ultrason Sonochem. 2021;73:105506.
CrossRef - Pei CC, Hsien TS, Hsuan FC, et al. Microwave-and ultrasound-assisted extraction of pectin yield and physicochemical properties from lemon peel. J Agric Food Res. 2024;15:101009.
CrossRef - Marić M, Grassino AN, Zhu Z, et al. An overview of the traditional and innovative approaches for pectin extraction from plant food wastes and by-products: Ultrasound-, microwaves-, and enzyme-assisted extraction. Trends Food Sci Technol. 2018;76:28-37.
CrossRef - Sani IK, Mehrnoosh F, Rasul NH, et al. Pulsed electric field-assisted extraction of natural colorants, principles and applications. Food Biosci. 2024;104746.
CrossRef - Li H, Li Z, Wang P, et al. Evaluation of citrus pectin extraction methods: Synergistic enhancement of pectin’s antioxidant capacity and gel properties through combined use of organic acids, ultrasonication, and microwaves. Int J Biol Macromol. 2024;266:131164.
CrossRef - Mao Y, Robinson JP, Binner ER. Current status of microwave-assisted extraction of pectin. Chem Eng J. 2023;473:145261.
CrossRef - Wang W, Chen W, Zou M, et al. Applications of power ultrasound in oriented modification and degradation of pectin: A review. J Food Eng. 2018;234:98-107.
CrossRef - Jha A, Mishra S. Exploring the potential of waste biomass-derived pectin and its functionalized derivatives for water treatment. Int J Biol Macromol. 2024;133613.
CrossRef - Boateng ID. Thermal and nonthermal assisted drying of fruits and vegetables. Underlying principles and role in physicochemical properties and product quality. Food Eng Rev. 2023;15(1):113-155.
CrossRef - Yao Y, Pan Y, Liu S. Power ultrasound and its applications: A state-of-the-art review. Ultrason Sonochem. 2020;62:104722.
CrossRef - He Y, Xiao X, Cheng Y, Li G. Progress in field-assisted extraction and its application to solid sample analysis. J Sep Sci. 2016;39(1):177-187.
CrossRef - Pasiecna-Patkowska S, Cichy M, Flieger J. Application of Fourier Transform Infrared (FTIR) Spectroscopy in Characterization of Green Synthesized Nanoparticles. Molecules. 2025;30(3):684.
CrossRef - Lateb Y, Moussaoui R, Elias A, et al. Centrifugation Effect on Olive Mill Wastewater Treatment by Coagulation-Flocculation Using Ca (OH) 2-Pectin System. Waste Biomass Valorization. 2024;15(7):4051-4060.
CrossRef - Nanda A, Mohapatra BB, Mahapatra APK, et al. Multiple comparison test by Tukey’s honestly significant difference (HSD): Do the confident level control type I error. Int J Stat Appl Math. 2021;6(1):59-65.
CrossRef - Dambuza A, Rungqu P, Oyedeji AO, et al. Extraction, Characterization, and Antioxidant Activity of Pectin from Lemon Peels. Molecules. 2024;29(16):3878.
CrossRef - Zhang M, Zhao Y, Li Q. Influence of microwave power and ultrasound on pectin yield and structural properties. Int J Biol Macromol. 2023;215:105678.
- Duan X, Song J, Wu X. Effect of temperature and extraction conditions on pectin purity and yield. J Agric Food Chem. 2023;71(3):412-420.
- Zhu J, et al. Advances in microwave-ultrasound synergistic extraction of bioactive compounds. Innov Food Sci Emerg Technol. 2024;90:103124.
- Hu Q, Tan R, Zhong P, et al. Multifunctional broadband emitters based on rare earth phosphors for all-weather and efficient radiative cooling and energy saving. J Mater Chem A. 2025.
CrossRef - Pan W, Wang L, Tang J, et al. Optoelectronic array of photodiodes integrated with RRAMs for energy-efficient in-sensor computing. Light Sci Appl. 2025;14(1):48.
CrossRef - Chen G, Miao J, Huang X, et al. High-power-efficiency and ultra-long-lifetime white OLEDs empowered by robust blue multi-resonance TADF emitters. Light Sci Appl. 2025;14(1):81.
CrossRef - Khadhraoui B, Ummat V, Tiwari BK, et al. Review of ultrasound combinations with hybrid and innovative techniques for extraction and processing of food and natural products. Ultrason Sonochem. 2021;76:105625.
CrossRef - Zhao S, Baik OD, Choi YJ, Kim SM. Pretreatments for the efficient extraction of bioactive compounds from plant-based biomaterials. Crit Rev Food Sci Nutr. 2014;54(10):1283-1297.
CrossRef - Talha M, Khalid S, Maan AA, et al. Ohmic assisted extraction: a sustainable and environment friendly approach to substitute conventional extraction methods. Food Rev Int. 2024;40(10):3508-3529.
CrossRef - Yahaya N, Mohamed AH, Sajid M, et al. Deep eutectic solvents as sustainable extraction media for extraction of polysaccharides from natural sources: Status, challenges and prospects. Carbohydr Polym. 2024;122199.
CrossRef - Panwar D, Panesar PS, Chopra HK. Green valorization approach of Citrus limetta peels by ultrasound-assisted enzymatic extraction for recovery of dietary fibers: Optimization, physicochemical, structural, functional, and thermal properties. Biomass Convers Biorefinery. 2024:1-21.
CrossRef - Wang L, He C, Zhao F. Effect of pectin extraction methods on gelling and stabilizing properties. Food Hydrocolloids. 2023;137:108012.
- Mamiru D, Gonfa G. Extraction and characterization of pectin from watermelon rind using acetic acid. Heliyon. 2023;9(2).
CrossRef - Ghamari Kargar P, Maleki B, Ghani M. Fe3O4/Fe2O3/TiO2/Ag: an innovative photocatalyst under visible light irradiation in deep eutectic solvent for efficient conversion of 5-HMF to chemo-and bio-based chemicals besides their determination using HPLC. Biomass Convers Biorefinery. 2023:1-13.
CrossRef - El-Enshasy HA, Yang ST. Probiotics, the natural microbiota in living organisms. Probiotics, the Natural Microbiota in Living Organisms. CRC Press; 2021. https://doi.org/10.1201/9781351027540.
CrossRef - Buljeta I, Šubarić D, Babić J, et al. Extraction of dietary fibers from plant-based industry waste: A comprehensive review. Appl Sci. 2023;13(16):9309.
CrossRef - Jiang Q, Luo M, Xiao W. A comparison of pectin extraction methods from citrus peel waste. LWT Food Sci Technol. 2023;160:113347.
- Wu H, Li S, Zhang Y. Characterization and functionality of pectin extracted via different methods. Food Chem. 2023;415:135914.
- Owens J, Patel S, Anderson M. Application of acid hydrolysis in pectin extraction from fruit peels. Carbohydr Res. 2023;512:108121.
- He CA, Qi JR, Liao JS, Song YT, Wu CL. Excellent hydration properties and oil holding capacity of citrus fiber: Effects of component variation and microstructure. Food Hydrocolloids. 2023;144:108988.
CrossRef - Gigante V, Aliotta L, Ascrizzi R, et al. Innovative Biobased and Sustainable Polymer Packaging Solutions for Extending Bread Shelf Life: A Review. Polymers. 2023;15(24):4700.
CrossRef - Chen H, Liu Z, Zhang X, et al. Ultrasound-assisted extraction of pectin: Optimization and characterization. Food Hydrocolloids. 2023;132:107845.
- Said NS, Olawuyi IF, Lee WY. Pectin hydrogels: Gel-forming behaviors, mechanisms, and food applications. Gels. 2023;9(9):732.
CrossRef - Qureshi MS, Nawaz MU, Umar S. Cost Benefit Analysis of Photovoltaic Systems in Urban Environments: A Comparative Study. Rev Esp Doc Cienc. 2024;18(02):41-64.
- Bhadange YA, Carpenter J, Saharan VK. A comprehensive review on advanced extraction techniques for retrieving bioactive components from natural sources. ACS Omega. 2024;9(29):31274-31297.
CrossRef
Abbreviations List
UMAE: Ultrasound and microwave-assisted extraction
OPP: Orange peel pectin
EW: Equivalent weight
MeO: Methoxyl content
DE: Degree of esterification
AUA: Anhydrouronic acid
GalA: Galacturonic acid
FTIR: Fourier transform infrared spectroscopy
NMR: Nuclear magnetic resonance
DSC: Differential scanning calorimetry













